Abstract
Cinnarizine (CIN) is an anti-histaminic drug which is mainly used for the control of vomiting due to motion sickness. The thermal behavior of CIN was investigated using thermogravimetry/derivative thermogravimetry and differential thermal analysis. The kinetic parameters were evaluated by isothermal and non-isothermal conditions including Ozawa’s conventional method, Ozawa–Flynn–Wall and Friedman isoconversional methods. The kinetic parameters were determined using the thermogravimetric curves of the decomposition process. The activation energy values obtained were 88.38 and 90.12 kJ mol−1 for the isothermal and non-isothermal conditions, respectively, with the conventional method. The activation energy values obtained by isoconversional method were practically equal; hence, the reaction involves a single step. Finally, the values of entropy (∆S), enthalpy (∆H), and Gibbs free energy change (∆G) of decomposition reaction were calculated and compared.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
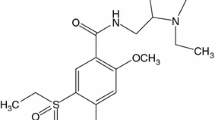
Cinnarizine (CIN) is (E)-(diphenylmethyl)-4-(3-phenylprop-2-enyl) piparizine (Fig. 1). It improves blood flow and is widely used for the treatment of cerebral apoplexy, cerebral arteriosclerosis, post-traumatic cerebral symptoms, and nausea and vomiting [1].
Literature review showed several methods have been reported for the analysis of CIN in pure form, dosage forms, and biological fluids or in combination with other drugs. These methods include gas chromatography [2], high-performance liquid chromatography [3–6], spectrophotometric methods [7–9], fluorescence spectroscopy [10], and electrochemical methods [11–13]. Also chemiluminesence method for flow injection analysis of CIN [14] and a capillary electrophoresis method [15] were also reported.
Thermal analysis techniques can provide important data regarding storage and stability of drugs [16, 17]. The most widely used thermal analysis techniques are TG, DTG, and DTA [18, 19]. These techniques are widely used in the pharmaceutical sciences for the characterization of solid drugs and excipients. The application of thermoanalytical methods may provide new information about the temperature and energy associated with events, such as melting, oxidation and reduction reactions, glass transition, boiling, sublimation, decomposition, and crystallization [20–22].
The thermal decomposition of drugs is interesting to predict the degradation rates at temperatures from data collected on accelerated processes that are studied at elevated temperatures. The temperature may increase the chemical reactions, providing sufficient energy (activation energy) required to break chemical bonds and starts the decomposition process [23, 24].
Solid-state kinetic studies have increasing importance in thermal analysis, in which the main purposes are to calculate the parameters of the Arrhenius equation and to determine the mechanism of pyrolysis reaction. These data can provide valuable information about time and condition of storage [25, 26]. The knowledge of such parameters for pure drugs and for drug–excipient mixtures is also meaningful to elucidate compatibility/incompatibility and its effects on thermal stability.
Physicochemical characterization and dissolution properties of CIN solid dispersions were studied using IR spectroscopy, X-ray diffractometry, and differential scanning calorimetry (DSC) [27]. To the best our knowledge, there is no report on the thermal behavior and decomposition kinetic of this drug alone.
The aim of this paper is to study the thermal behavior and the kinetic parameters of CIN obtained from thermogravimetric data under isothermal and non-isothermal conditions.
Experimental
Materials
CIN used in this study was provided by El Nasr Pharmaceutical Chemicals Company, Egypt. The sample was characterized using infrared absorption spectrophotometry, melting point, and assay according to the official European Pharmacopoeia [28], and it is suitable for submitting to thermal analysis experiments.
Methods
Thermogravimetric analysis (TG)
Thermogravimetry and differential thermal analysis were carried out using simultaneous Shimadzu thermogravimetric analyzer TGA-60H with TA 60 software in dry nitrogen atmosphere at a flow rate of 30 mL min−1 in platinum pan. The experiments were performed in room temperature up to 600 °C at different heating rates (5, 10, 15, and 20 °C min−1). The sample mass was about 5 mg of the drug. The kinetic parameters of decomposition such as, activation energy (E), frequency factor (A), and reaction order (n) were calculated. The kinetic parameter and the order of reaction for Ozawa’s method were obtained with TA 60 software. The Kinetics Committee of the ICTAC published a review of the existing methods for helping to perform reliable kinetic parameters through a combination of isothermal and non-isothermal experiments as the best way to establish kinetic models [29].
In the isothermal condition, the temperatures were 100, 110, 120, and 130 °C, with 10 °C temperature increments, under dynamic nitrogen atmosphere with the flow rate of 30 mL min−1 using aluminum oxide as reference standard in a platinum pan. The isothermal holding was monitored based on the time to a mass loss of 5 % decomposition.
Ozawa’s conventional method
The kinetic parameters and the order of reaction for Ozawa’s conventional method [30] were obtained with software TA 60 software.
Isoconversional method
Kinetic methods propose that the isothermal rate of conversion (dα/dt) is a linear function and is a function of temperature (T) and extent of conversion (α), as in Eq. 1 [29].
Kinetic methods suppose that dα/dt is a linear function of the temperature-dependent rate constant, k(T), and a temperature-independent function of conversion, f(α), which depends on the mechanism of the reaction [29]. Under non-isothermal conditions, Eq. 1 becomes:
where, β = dT/dt, is the heating rate, A is the pre-exponential factor, E is the activation energy, and R is the gas constant.
Ozawa–Flynn–Wall and Friedman isoconversional methods can be used to calculate E. These methods consider that for all values of α, f(α) does not change with different heating rates; therefore, measurements of temperature, corresponding to fixed values of α at different heating rates, are required [31]. Under these conditions Eq. 2 turns into Eqs. 3 and 4:
The plots of ln β and \(\ln (\beta \cdot {\text{d}}\alpha /{\text{d}}T)\) versus 1/T should give straight lines with slopes of −E/R.
If the values of E determined are almost constant for different values of α, then the decomposition reaction occurs in a single step; on the other hand, a change in E with increasing degree of conversion is an indication of a complex reaction mechanism. The results obtained by the isoconversional method may corroborate the results of activation energy using Ozawa’s method in the non-isothermal conditions. It is known that the most reliable kinetic methods are the isoconversional ones [29, 31–34].
Arrhenius parameters and the reaction mechanisms were determined through isothermal and non-isothermal kinetic analysis [32, 35, 36]. The activation energy can be obtained using several thermogravimetric curves at different heating rates by the non-isothermal method.
Melting point measurement was carried out using OptiMelt automated melting point instrument by the American Stanford Research System (SRS). FTIR of CIN was carried out using FTIR-8400S Fourier transform infrared spectrophotometer by Shimadzu, Japan.
Results and discussion
The melting point of CIN is 121.35 °C, the assay based on non-aqueous titration using 0.1 mol l−1 perchloric acid refers to high purity of the used drug (purity ≈ 99.34 %).
Figure 2 shows characteristic peaks of cinnarizine as aromatic C–H band at 3040 cm−1, aliphatic C–H bands at 2950, 2900, 2809, 2760 cm−1, C=C aromatic at 1600 cm−1, piperazine ring N,N disubstituted at 1495 cm−1, alkene C=C bands at 960, 750 cm−1 and alkene C–H band at 690 cm−1 [27, 37]. Therefore, CIN is suitable for thermal analysis experiments.
Thermal characterization of CIN raw material (Fig. 3) represents the TG/DTG and DTA curves of the CIN in nitrogen atmosphere of 30 mL min−1 at a heating rate of 10 °C min−1. The decomposition occurs in liquid medium consisting of pure melted CIN, and the thermal degradation process occurs in one well-defined step. The DTA curve shows a sharp and strong endothermic peak at 121.78 °C attributed to the melting of CIN which is acceptable and close to the values of the reported melting temperature using DSC method 120.66 [27], 119 [38], and 121.35 °C using melting point apparatus. The melting point values of CIN are within the range of 118–122 °C as in European Pharmacopoeia [28]. Also an exothermic peak appears at 327.51 °C indicating to the thermal degradation process of CIN.
Effect of heating rate
Figure 4 shows the DTA curves for the decomposition of CIN at several heating rates. It was found that by increasing the heating rate, the melting endothermic peaks of CIN are shifted to higher temperatures.
Kinetic studies
α–T curves for the non-isothermal decomposition of CIN at different heating rates are illustrated in Fig. 5. The values of E according to thermal decomposition of CIN are listed in Table 1. These values were calculated using the Ozawa–Flynn–Wall and Friedman differential methods by fitting the plots of ln β versus 1/T [39] and ln β(dα/dT) versus 1/T [40] for Ozawa–Flynn–Wall and Friedman differential methods, respectively. The results in Table 1 showed the variations between the values of E obtained using the two isoconversional methods. These variations may be due to the temperature integral approximation used in the derivations of the relations of the kinetic methods.
The activation energy values obtained for CIN by Ozawa–Flynn–Wall isoconversional method in the non-isothermal conditions showed practically constant, relative standard deviation of 0.63 %. The results suggest that the reaction occurs in a single step.
Figure 6 shows the plot of mass loss versus temperature. The thermogravimetric curves obtained at different heating rates are shifted to higher temperatures when heating rates increase. E, A, and n were determined by Ozawa’s conventional method.
The activation energy was obtained from a plot of logarithms of heating rates as a function of the inverse of temperature (1/T). The kinetic parameters E, A, and n were 90.12 kJ mol−1 and 2.76 × 107 min−1, and zero-order reaction, respectively.
The isothermal curves obtained are demonstrated in Fig. 7 and show mass loss rate as a function of time. The isothermal studies were investigated using four temperatures at 100, 110, 120, and 130 °C for 90, 50, 19, and 12 min, respectively. The natural logarithm of time (ln t) corresponding to a certain mass loss (α = 5 %) is linearly dependent on the reciprocal of temperature T.
Figure 8 shows the linear relation between ln t and 1/T. The equation obtained from this linear regression method was y = 10.63x − 24.44 with r = 0.992, and it showed that the order of reaction remains constant (n = 0) within the temperature and mass loss interval under consideration.
The activation energy was calculated from the slope of the line, from linear regression by the product of 10.63 with the molar gas constant (R = 8.314 J K−1 mol−1). The activation energy value was 88.38 kJ mol−1. The decomposition kinetics for isothermal conditions occurs in constant rate, zero order, and is independent from the concentration of the reactants.
Both values of E obtained by Ozawa’s conventional method and isothermal and conditions were similar. The combined experiments using isothermal and non-isothermal conditions are the best way to properly determine kinetic parameters [29].
The values of ∆S, ∆H, and ∆G of decomposition reaction were calculated using isothermal and non-isothermal methods. All data are listed in Table 2. Comparing the results of the application of the two methods, we observe that the calculated values are almost similar.
Conclusions
The thermal analysis behavior of CIN showed melting point at 119.75 °C, then it decomposes in liquid medium consisting in pure CIN melted in a single step. The isoconversional methods are used to determine the dependence of E on α. In these methods, the reaction model is independent of temperature or heating rate and confirms that the decomposition of CIN through a single step. The obtained E values using Flynn–Wall–Ozawa and Friedman isoconversional methods showed variations. The variation of E may be attributed to the temperature integral approximation used in the derivations of the relations of the kinetic methods. The activation energy values for CIN by Ozawa–Flynn–Wall isoconversional method in the non-isothermal conditions appeared practically constant. Comparing the results of kinetic parameters (E, ∆S, ∆H, ∆G, and A) indicates that these values are almost similar using Ozawa’s non- isothermal method and isothermal method.
References
Sweetman SC. Martindale: the complete drug references. 36th ed. London: Pharmaceutical Press; 2009.
Woestenborghs R, Michielsen L, Lorreyne W, Heykants J. Sensitive gas chromatographic method for the determination of cinnarizine and flunarizine in biological samples. J Chromatogr B. 1982;232(1):85–91.
Argekar A, Shah S. Simultaneous determination of cinnarizine and domepiridone maleate from tablet dosage form by reverse phase ion pair high performance liquid chromatography. J Pharm Biomed Anal. 1999;19(6):813–7.
Hundt H, Brown L, Clark E. Determination of cinnarizine in plasma by high-performance liquid chromatography. J Chromatogr B. 1980;183(3):378–82.
Metwally FH, Elzeany B, Darwish H. New methods for determination of cinnarizine in mixture with piracetam by spectrodensitometry, spectrophotometry, and liquid chromatography. J AOAC Int. 2005;88(6):1666–76.
Van De Steene JC, Lambert WE. Validation of a solid-phase extraction and liquid chromatography–electrospray tandem mass spectrometric method for the determination of nine basic pharmaceuticals in wastewater and surface water samples. J Chromatogr A. 2008;1182(2):153–60.
Abdine H, Belal F, Zoman N. Simple spectrophotometric determination of cinnarizine in its dosage forms. Il Farmaco. 2002;57(4):267–71.
Devagondanahalli MH, Shaikh SMT, Jaldappagari S, Ramanaboyina SK, Kasalanti H. Determination of cinnarizine in pure and pharmaceutical formulations. J Chin Chem Soc. 2007;54(1):63–8.
Neagu A, Constantinescu I, Nedelcu A, Florea M. Spectrophotometric determination of some basic drugs in bulk and dosage forms using azo dyes. Farmacia. 2008;56(1):42–9.
Walash M, Belal F, El-Enany N, Abdelal A. Second-derivative synchronous fluorometric method for the simultaneous determination of cinnarizine and domperidone in pharmaceutical preparations. Application to biological fluids. J Fluoresc. 2008;18(1):61–74.
Hassan SS, Abdel-Aziz RM, Abbas AB. Potentiometric membrane sensors for the selective determination of cinnarizine in pharmaceutical preparations. Anal Chim Acta. 1996;321(1):47–52.
Hegde RN, Hosamani RR, Nandibewoor ST. Voltammetric oxidation and determination of cinnarizine at glassy carbon electrode modified with multi-walled carbon nanotubes. Colloids Surf B. 2009;72(2):259–65.
El-Sayed G, Yasin S, El Badawy A. Voltammetric behavior and determination of cinnarizine in pharmaceutical formulations and serum. Anal Lett. 2008;41(17):3021–33.
Townshend A, Youngvises N, Wheatley RA, Liawruangrath S. Flow-injection determination of cinnarizine using surfactant-enhanced permanganate chemiluminesence. Anal Chim Acta. 2003;499(1):223–33.
Abdelal A, Kitagawa S, Ohtani H, El-Enany N, Belal F, Walash M. Method development and validation for the simultaneous determination of cinnarizine and co-formulated drugs in pharmaceutical preparations by capillary electrophoresis. J Pharm Biomed Anal. 2008;46(3):491–7.
Gaisford S, Buanz ABM. Pharmaceutical physical form characterisation with fast (>200 oC min−1) DSC heating rates. J Therm Anal Calorim. 2011;106(1):221–6.
Neto HS, Novák C, Matos J. Thermal analysis and compatibility studies of prednicarbate with excipients used in semi solid pharmaceutical form. J Therm Anal Calorim. 2009;97(1):367–74.
Maximiano FP, Novack KM, Bahia MT, de Sá-Barreto LL, da Cunha-Filho MSS. Polymorphic screen and drug-excipient compatibility studies of the antichagasic benznidazole. J Therm Anal Calorim. 2011;106(3):819–24.
Salama NN, Mohammad MA, Fattah TA. Thermal behavior study and decomposition kinetics of amisulpride under non-isothermal and isothermal conditions. J Therm Anal Calorim. 2015;120(1):953–8.
Brown ME, Glass BD. Decomposition of solids accompanied by melting—Bawn kinetics. Int J Pharm. 2003;254(2):255–61.
Rodomonte A, Antoniella E, Bertocchi P, Gaudiano MC, Manna L, Bartolomei M. Different crystal morphologies arising from different preparation methods of a same polymorphic form may result in different properties of the final materials: the case of diclofenac sodium trihydrate. J Pharm Biomed Anal. 2008;48(2):477–81.
Salvio Neto H, Matos JR. Compatibility and decomposition kinetics studies of prednicarbate alone and associated with glyceryl stearate. J Therm Anal Calorim. 2011;103(1):393–9.
Burnham L, Dollimore D, Alexander KS. Kinetic study of the drug acetazolamide using thermogravimetry. Thermochim Acta. 2002;392:127–33.
Felix FS, da Silva LC, Angnes L, Matos J. Thermal behavior study and decomposition kinetics of salbutamol under isothermal and non-isothermal conditions. J Therm Anal Calorim. 2009;95(3):877–80.
Oliveira MA, Yoshida MI, Gomes ECL, Mussel WN, Vianna-Soares CD, Pianetti GA. Thermal analysis applied to simvastatin characterization in pharmaceutical formulations. Quim Nova. 2010;33(8):1653–7.
Sovizi M. Thermal behavior of drugs: investigation on decomposition kinetic of naproxen and celecoxib. J Therm Anal Calorim. 2010;102(1):285–9.
Kalava BS, Demirel M, Yazan Y. Physicochemical characterization and dissolution properties of cinnarizine solid dispersions. Turkish J Pharm Sci. 2005;2(2):51–62.
The European Pharmacopoeia VII. Strasbourg: European Directorate for the Quality of Medicines and Heathcare; 2005.
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520(1):1–19.
Vyazovkin S. Thermal analysis. Anal Chem. 2002;74(12):2749–62.
Muraleedharan K, Kannan M, Devi TG. Thermal decomposition kinetics of potassium iodate. J Therm Anal Calorim. 2011;103(3):943–55.
Mothé CG, de Miranda IC. Study of kinetic parameters of thermal decomposition of bagasse and sugarcane straw using Friedman and Ozawa–Flynn–Wall isoconversional methods. J Therm Anal Calorim. 2013;113(2):497–505.
Málek J, Koga N, Pérez-Maqueda LA, Criado JM. The Ozawa’s generalized time concept and YZ-master plots as a convenient tool for kinetic analysis of complex processes. J Therm Anal Calorim. 2013;113(3):1437–46.
Fandaruff C, Araya-Sibaja A, Pereira R, Hoffmeister C, Rocha H, Silva M. Thermal behavior and decomposition kinetics of efavirenz under isothermal and non-isothermal conditions. J Therm Anal Calorim. 2014;115(3):2351–6.
Cides LC, Araújo AA, Santos-Filho M, Matos J. Thermal behaviour, compatibility study and decomposition kinetics of glimepiride under isothermal and non-isothermal conditions. J Therm Anal Calorim. 2006;84(2):441–5.
Han Y, Li T, Saito K. A modified Ortega method to evaluate the activation energies of solid state reactions. J Therm Anal Calorim. 2013;112(2):683–7.
Anthony CM, Osselton MD, Widdop B. Clark’s Analysis of Drugs and Poisons. 3rd ed. London: Pharmaceutical Press; 2004.
Persson LC, Porter CJH, Charman WN, Bergström CAS. Computational prediction of drug solubility in lipid based formulation excipients. Pharm Res. 2013;30:3225–37.
Tita D, Fulias A, Tita B. Thermal stability of ketoprofen. J Therm Anal Calorim. 2013;111(3):1979–85.
Fulias A, Vlase G, Grigorie C, Ledet I, Albu P, Bilanin M, Vlase T. Thermal behaviour studies of procaine and benzocaine. J Therm Anal Calorim. 2013;113:265–71.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohamed, M.A., Attia, A.K. Thermal behavior and decomposition kinetics of cinnarizine under isothermal and non-isothermal conditions. J Therm Anal Calorim 127, 1751–1756 (2017). https://doi.org/10.1007/s10973-016-5551-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5551-2