Abstract
The radiolabeling of trovafloxacin dithiocarbamate (TVND) with technetium-99m using [99mTc-N]2+ core was investigated and biologically assessed as prospective infection imaging agent. The achievability of the 99mTcN-TVND complex as a future MRSA infection radiotracer was investigated in artificially methicillin-resistant Staphylococcus aureus (MRSA) infected male Sprague–Dawley rats (MSDR). Radiochemically the 99mTcN-TVND complex was characterized in terms of radiochemical purity (RCP) in saline, in vitro permanence in serum, in vitro binding with MRSA and biodistribution in living and heat killed MRSA infected MSDR. Radiochemically the complex showed stability in saline with a 97.90 ± 0.22% yield and serum at 37 °C up to 4 h. The 99mTcN-TVND complex showed saturated in vitro binding with MRSA. Normal in vivo uptake in the MRSA infected MDRS was observed with a five fold uptake in the infected muscle as compared to inflamed and normal muscles. The high RCP values, in vitro permanence in serum, better in vitro binding with MRSA, biodistribution behavior and the target to non-target (infected to inflamed muscle) ratios posed the 99mTcN-TVND complex as a promising MRSA infection radiotracer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The recognition of acute, subacute and chronic infection and its discrimination from inflammation for appropriate management of patients is highly significant for clinicians. The functional imaging modality of nuclear medicine outweigh the latest structural imaging techniques like high resolution computerized tomography (CT) and magnetic resonance imaging (MRI) by its capability of detecting early disease with accurate discrimination from non-infective pathology. However, selection of the right technique is always a serious matter of concern for the clinicians with special reference to the fever of unknown origin [1, 2].
The available infection imaging agents, developed in the last two decades, have facilitated the clinician in the early diagnoses of infection and its appropriate treatment [3–13]. We have also reported a number of novel technetium-99m (99mTc) labeled infection radiotracers [14–19]. The radiochemical purity (RCP), in vitro stability in saline and serum, in vitro binding with pathogens and biodistribution results forced us to keep on searching for newer and better infection imaging agents.
Trovafloxacin (TVN), {1,5,6-α-7-(6-amino-3-azabicyclo[3.1.0]hex-3-yl)-1-(2,4-difluorophenyl)-6-fluoro-1,4-dihydro-4-oxo-1,8-naphthyridine-3-carboxylic acid} (Fig. 1a) is a new synthetic fluoroquinolone broad spectrum antibiotic which is found to be more effective against both Gram-positive (G+ive) and Gram-negative (G−ive) bacteria. The TVN has proven very persuasive against Cocci (G+ive) and especially against methicillin-resistant Staphylococcus aureus (MRSA). The TVN through inhibition of the bacterial DNA gyrase terminate the growth of MRSA [20, 21].
The 99mTc-nitridio complexes played a vital role in the radiosynthesis of newer and promising agents intended for various nuclear medicine diagnostic tests [3]. In continuation to our ongoing research, now we are reporting the radiolabeling of TVN with 99mTc using [99mTcN]2+ core for more stable complexation. In the current investigation, achievability of the 99mTcN-TVND radiocomplex to map out the deep tissue infections in artificially MRSA infected MSDR was evaluate. The aptness of the 99mTcN-TVND radiocomplex was also observed in terms of in vitro eternalness in normal saline at room temperature and in serum at 37 °C, in vitro binding with MRSA, biodistribution in artificially MRSA infected MSDR.
Experimental
Materials
Trovafloxacin (TVN) (Pfizer Inc. New York, USA), TLC (Merk), succinic dihydrazide (SDH), propylenediamine tetra-acetic acid (PDTA) and all the other chemicals and solvents of analytical grade (Sigma). RP-HPLC (Shimadzu, Japan), well counter, scalar count rate meter (Ludlum, USA), Dose calibrator (Capintech, USA) and Gamma camera GKS-1000 (GEADE Nuclearmedizine system, Germany).
Method
Preparation and tricarbonyl complexation of trovafloxacin dithiocarbamate
Trovafloxacin (TVN) was converted to trovafloxacin dithiocarbamate (TVND) using the reported method [19]. Thereafter, 0.5 mL sodium pertechnetate (Na99mTcO4 −) freshly eluted was mixed with 0.10 mg SnCl2·2H2O, 5.0 mg propylenediamine tetra-acetic acid (PDTA), 5.0 mg succinic dihydrazide (SDH) in a sterilized vial. The preparation was incubated at room temperature for 10 min and 2 mg of TVND was added after gentle swirling.
Partition coefficient of the 99mTcN-TVND complex
The partition coefficient of the 99mTcN-TVND complex was determined using the reported method [22]. Briefly, equivalent volume of the 99mTcN-TVND complex, octanol and phosphate buffer (PB) was vortexed for 5 min at room temperature. The preparation was then centrifuged at 5000 rpm/min for 15 min. Subsequently, at different intervals aliquots were haggard and counted for activity using well counter interface with scalar count rate meter. Using the following formula partition coefficient was calculated.
HPLC analysis of the 99mTcN-TVND complex
Shimadzu SCL-10 AVP system equipped with SDP-10 AVP UV detector operating at 254 nm, Packard 500 TR series flow scintillation analyzer, binary pump and online degasser was used to characterized the 99mTcN-TVND complex using C-18 column (4.6 × 150 mm) as a stationary phase and water (W)/methanol (M) as mobile phase. Freshly prepared 99mTcN-TVND complex 10 μL was injected into the C-18 column and 1 mL/min mobile phase was applied for 15 min [for 0–2 min (100% W), 2–5 min (100–75% W), 5–7 min (75–66% W), 7–10 min (34–100 M), 10–12 min (100% M) and 12–15 min (100% M to 100% W)]. The radio-fractions collected at different intervals were counted for activity using single well counter interface with scalar count rate meter.
In vitro stability of the 99mTcN-TVND complex in serum
The 99mTcN-TVND radiocomplex was investigated for its in vitro stability in serum using TLC method. 1.8 mL serum was incubated with 0.2 mL of the 99mTcN-TVND radiocomplex at 37 °C for 16 h. At different intervals during incubation aliquots were drawn and applied on a TLC strip. Thereafter, the strip was developed in saline and CH2Cl2:CH3OH (9:1) (v/v). Thereafter, The TLC strips were divided into two at Rf 5 and measured for percent in vitro stability using single well counter interface with scalar count rate meter.
MRSA in vitro uptake
In vitro binding of the 99mTcN-TVND radiocomplex with MRSA was investigated using the reported method [23]. Briefly, the 99mTcN-TVND complex, 0.2 mL (10 MBq) was moved through a sterilized syringe to a tube containing 0.1 mL sodium phosphate buffer (Na-PB). Thereafter, 0.8 mL (50%, v/v) 0.01 M acetic acid (AA) containing approximately 1 × 108 colony forming units (CFU) of MRSA were added to the tube followed by incubation at 4 °C for 1 h. The pH of the preparation was adjusted to pH 5 and the preparation was centrifuged at 2000 rpm for 15 min. The supernatant was discarded and the pellets were resuspended in 2 mL of Na-PB and repeated the centrifugation at 2000 rpm for 15 min. Finally, the pellets were counted for activity with single well counter interface with scalar count rate meter.
Biodistribution
Male Sprague–Dawley rats (MSDR) (weight range, 180–220 g) were used for the determination of the percent absorbed dose of the 99mTcN-TVND radiocomplex. Twelve healthy MSDR were selected and divided into two groups (A and B) of six rats each. MSDR (group A and B) were intramuscularly (I.M.) administered 0.2 mL sterile turpentine oil to the left thigh. 0.2 mL of living MRSA in saline containing approximately 1 × 108 colony forming units (cfu) were injected to the right thigh of the group A and similarly heat killed MRSA to the group B MSDR, respectively. After 24 h 0.5 mL (37 MBq) of the freshly prepared radiotracer was intravenously (I.V.) injected to the MSDR of group A and B. Subsequently, the MSDR of group A and B animals were sacrificed as per rules of the Nuclear Medicine Research Laboratory Part-I and II. The absorbed dose per gram in blood, liver, spleen, stomach, intestine, kidney, infected muscle, inflamed muscle and normal muscle was calculated using single well counter interface with scalar count rate meter.
Results and discussion
Radiocomplexation and characterization of the 99mTcN-TVND complex
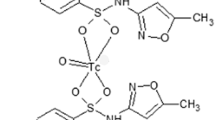
Trovafloxacin (Fig. 1a) was converted to its dithiocarbamate derivative (Fig. 1b) and tagged by treatment with an intermediate [99mTcN]2+ core using the method described earlier [19]. The proposed structure of 99mTcN-TVND radiocomplex (Fig. 1c) with the tetra-dentate ligand will have square planner pyramidal geometry with TcN: Ligand ratio of 1:1.
The HPLC radiochromatogram of the 99mTcN-TVND complex is shown in Fig. 2 giving radio-peaks at retention time (RT) 3.9 and at 12.2 min. The peak at 3.9 min of RT correspond to [99mTcN]2+ intermediate and the one at 12.2 min to the 99mTcN-TVND complex.
Radiochemically the 99mTcN-TVND complex showed stable behaviour in normal saline as shown in Fig. 3. The radiochemical purity values of the 99mTcN-TVND complex went down with the time but not less than 90% up to 240 min after reconstitution. The 99mTcN-TVND complex showed maximum RCP value (97.90 ± 0.22%) at 30 min after reconstitution.
Lipophilicity of the 99mTcN-TVND complex
The participation coefficient values calculated for 99mTcN-TVND complex is 1.02 ± 0.04. The P value for 99mTcN-TVND complex suggested that the complex is lipophilic.
In vitro stability of 99mTcN-TVND complex in serum
In vitro stability of the 99mTcN-TVND complex in serum at 37 °C up to 16 h is given in Fig. 4. In serum the 99mTcN-TVND complex showed stable profile up to 4 h. The stability went down from 96.50 ± 0.24 to 80.25 ± 0.20% within 16 h.
In vitro MRSA uptake
The in vitro binding of the 99mTcN-TVND complex with MRSA is shown in Fig. 5. Maximum and saturated in vitro binding was observed at 90 min of the incubation.
Biodistribution
The absorbed dose per gram in blood, liver, spleen, stomach, intestine, kidney, infected muscle, inflamed muscle and normal muscle of MSDR artificially infected with living (group A) and heat killed MRSA (group B) after I.V. administration of the 99mTcN-TVND complex is given in Table 1. High percentage of the radioactivity was noted in blood after 30 min of the I.V. administration of the 99mTcN-TVND in group A and B, MSDR. However, this high uptake was found temporary and it went down from 20.50 ± 0.18 to 3.50 ± 0.15% within 120 min. Similar behaviour was observed in case of liver, spleen, stomach and intestines. However, in case of kidneys a reverse behaviour was noted where the activity of the complex was initially low which went up with time from 10.50 ± 0.16 to 23.25 ± 0.16%. Approximately, five fold higher activity of the tagged complex was noted in infected muscle of group A, MDSR as compared to inflamed and normal muscles as shown in Fig. 6. However, in group B, MDRS no such distinct difference in the infected muscle was seen as compared to the inflamed and normal muscles. The disappearance of the radiocomplex from circulatory system and appearance in the urinary system suggest the normal route of excretion in group A and B MDRS. The five fold uptake of the radiocomplex in the infected part of the group A, MDRS as compared to inflamed and normal muscles established the achievability of the 99mTcN-TVND complex as prospective MRSA infection radiotracer.
Conclusion
Radiolabeling of trovafloxacin dithiocarbamate (TVND) with technetium-99m using [99mTc-N]2+ core was studied and its efficacy as a prospective MRSA infection radiotracer was evaluated in MDRS. The high RCP values, in vitro permanence in serum, better in vitro binding with bacteria, better biodistribution behavior with high target to non-target ratios posed the 99mTcN-TVND complex as a promising MRSA infection radiotracer.
References
Gallagher H, Ramsay SC, Barnes J, Maggs J, Cassidy N, Ketheesan N (2006) Neutrophil labeling with [99mTc]-technetium stannous colloid is complement receptor 3-mediated and increases the neutrophil priming response to lipopolysaccharide. Nucl Med Biol 33:433
Lahiri S, Sarkar S (2007) Studies on 66,67Ga- and 199Tl-poly(N-vinylpyrrolidone) complexes. Appl Radiat Isot 65:309
Zhang J, Wang X, Tian C (2006) Synthesis of a bis-(N-butyl-dithiocarbamto)-nitrido 99mTc complex: a potential new brain imaging agent. J Radioanal Nucl Chem 273:15
Zhang J, Wang X (2000) Synthesis of 99mTcN(IPDTC)2 and its biodistribution in mice. J Radioanal Nucl Chem 249:573
Hong Z, Ningyi J, Lin Z (2009) Experimental studies on imaging of infected site with 99mTc-labeled ciprofloxacin in mice. Chin Med J 122:1907
Oh SJ, Ryu J, Shin JW, Yoon EJ, Ha H, Cheon JH, Lee HK (2002) Synthesis of 99mTc-ciprofloxacin by different methods and its biodistribution. Appl Radiat Isot 57:193
Zhang J, Guo H, Zhang S, Lin Y, Wang X (2008) Synthesis and biodistribution of a novel 99mTcN complex of ciprofloxacin dithiocarbamate as a potential agent for infection imaging. Bioorg Med Chem Lett 18:51
Motaleb MA (2007) Preparation of 99mTc-cefoperazone complex, a novel agent for detecting sites of infection. J Radioanal Nucl Chem 272:167
Motaleb MA (2007) Preparation and biodistribution of 99mTc-lomefloxacin and 99mTc-olfloxacin complex. J Radioanal Nucl Chem 272:295
EL-Gany EA, EL-Kolaly MT, Amine AM, EL-Sayed AS, Abdel-Gelil F (2005) Synthesis of 99mTc-pefloxacin: a new targeting agent for infectious foci. J Radioanal Nucl Chem 266:131
Roohi S, Mushtaq A, Jehangir M, Ashfaq MS (2006) Synthesis, quality control and biodistribution of 99mTc-Kanamycin. J Radioanal Nucl Chem 267:561
Motaleb MA (2009) Preparation, quality control and stability of 99mTc-sparafloxacin complex, a novel agent for detecting sites of infection. J Label Compd Radiopharm 52:415
Chattopadhyay S, Das SS, Chandra S, De K, Mishra M, Sarkar BR, Sinha S, Ganguly S (2010) Synthesis and evaluation of 99mTc-moxifloxacin, a potential infection specific imaging agent. Appl Radiat Isot 68:314
Qaiser SS, Khan AU, Khan MR (2010) Synthesis, biodistribution and evaluation of 99mTc-Sitafloxacin kit: a novel infection imaging agent. J Radioanal Nucl Chem 284:189
Shah SQ, Khan AU, Khan MR (2010) Radiosynthesis of 99mTc-nitrifuratonin a novel radiotracer for in vivo imaging of Escherichia coli infection. J Radioanal Nucl Chem (in press)
Shah SQ, Khan AU, Khan MR (2010) Radiosynthesis and biodistribution of 99mTc-rifampicin: a novel radiotracer for in vivo infection imaging. Appl Radiat Isot 68:2255
Shah SQ, Khan AU, Khan MR (2010) 99mTc-Novobiocin: a novel radiotracer for infection imaging. Radiochimica acta (in press)
Shah SQ, Khan AU, Khan MR (2010) Radiosynthesis, biodistribution and scintigraphy of the 99mTc-Teicoplanin complex in artificially infected animal models. J Label Compd Radiopharm (in press)
Shah SQ, Khan AU, Khan MR (2010) Radiosynthesis and biological evaluation of 99mTcN-sitafloxacin dithiocarbamate as a potential radiotracer for Staphylococcus aureus infection. J Radioanal Nucl Chem (in press)
Ocaña JA, Callejón M, Barragán FJ (2001) Determination of trovafloxacin in human serum by time resolved terbium-sensitised luminescence. Eur J Pharm Sci 13:297
Hsiao CJ, Younis H, Boelsterli UA (2010) Trovafloxacin, a fluoroquinolone antibiotic with hepatotoxic potential, causes mitochondrial peroxynitrite stress in a mouse model of underlying mitochondrial dysfunction. Chem Biol Interact 188:204
Shah SQ, Khan AU, Khan MR (2010) 99mTc(CO)3-Garenoxacin dithiocarbamate synthesis and biological evolution in rats infected with multiresistant Staphylococcus aureus and penicillin-resistant Streptococci. J Radioanal Nucl Chem. (in press)
Welling MM, Paulusma-Annema A, Batler HS, Pauwels EKJ, Nibbering PH (2000) Technetium-99m labelled antimicrobial peptides discriminate between bacterial infections and sterile inflammations. Eur J Nucl Med 27:292
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shah, S.Q., Khan, M.R. Radiocomplexation and biological characterization of the 99mTcN-trovafloxacin dithiocarbamate: a novel methicillin-resistant Staphylococcus aureus infection imaging agent. J Radioanal Nucl Chem 288, 215–220 (2011). https://doi.org/10.1007/s10967-010-0903-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-010-0903-z