Abstract
Cerambycid beetles of the subfamily Lamiinae use male-produced aggregation-sex pheromones that are attractive to both sexes. Terpenoid pheromones have been identified from species in the tribes Acanthoderini and Acanthocinini native to North and South America, comprised of (E)-6,10-dimethyl-5,9-undecadien-2-one (geranylacetone), the structurally related 6-methylhept-5-en-2-one (sulcatone), and/or specific enantiomers or nonracemic ratios of enantiomers of the related compounds (E)-6,10-dimethyl-5,9-undecadien-2-ol (fuscumol), its acetate ester, (E)-6,10-dimethyl-5,9-undecadien-2-yl acetate (fuscumol acetate), and 6-methylhept-5-en-2-ol (sulcatol). Here, we present new information about the chemical ecology of six acanthoderine and acanthocinine species native to the eastern USA. The pheromone of Astyleiopus variegatus (Haldeman) previously was identified as a blend of (S)-fuscumol and (S)-fuscumol acetate, and we report here that geranylacetone is a synergistic component. Males of Aegomorphus modestus (Gyllenhal), Lepturges angulatus (LeConte), and Lepturges confluens (Haldeman) were found to produce similar blends composed of the enantiomers of fuscumol acetate and geranylacetone, whereas males of Astylidius parvus (LeConte) and Sternidius alpha (Say) produced both enantiomers of fuscumol together with (R)-fuscumol acetate and geranylacetone. Field experiments with synthesized chemicals revealed that species with similar pheromone composition nevertheless differed in their responses to individual components, and to various blends of components, and in how attraction was influenced by chemicals that were pheromone components of other species. Sulcatone and/or sulcatol antagonized attraction of some species to pheromones of the geranylacetone class, suggesting that there is an adaptive advantage in an ability to detect these heterospecific compounds, such as in avoiding cross attraction to other cerambycid species, as yet unknown, that use pheromones composed of both chemical classes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cerambycid beetles of the largest subfamily, the Lamiinae (Švácha and Lawrence 2014), are known to use aggregation-sex pheromones, produced by males and attractive to both sexes, that are either hydroxyethers or terpenoids (reviewed by Hanks and Millar 2016). An example of the hydroxyether pheromone class is 2-(undecyloxy)ethanol (common name monochamol), the pheromone for several species in the genus Monochamus (tribe Monochamini) that are native to North America and Eurasia (e.g., Ryall et al. 2015). Terpenoid pheromones of lamiines were first reported for the South American Hedypathes betulinus (Klug) (tribe Acanthoderini) whose male-produced pheromone is composed of (E)-6,10-dimethyl-5,9-undecadien-2-one (geranylacetone) and enantiomers of the corresponding alcohol, (E)-6,10-dimethyl-5,9-undecadien-2-ol (fuscumol), and acetate, (E)-6,10-dimethyl-5,9-undecadien-2-yl acetate (fuscumol acetate; Fonseca et al. 2010; Vidal et al. 2010). (S)-Fuscumol is also a pheromone component of cerambycid species in the subfamily Spondylidinae (Silk et al. 2007; Sweeney et al. 2010). Recent research has shown that pheromones of several North American species in the tribes Acanthoderini and Acanthocinini are composed of compounds of the geranylacetone class (Meier et al. 2016), or of (S)-6-methylhept-5-en-2-ol ([S]-sulcatol), an analog of the structurally related 6-methylhept-5-en-2-one (sulcatone; Meier et al. 2019).
Here, we provide new information about the chemical ecology of six lamiine species native to the eastern USA, including the acanthoderine Aegomorphus modestus (Gyllenhal), and the acanthocinines Astyleiopus variegatus (Haldeman), Astylidius parvus (LeConte), Lepturges angulatus (LeConte), Lepturges confluens (Haldeman), and Sternidius alpha (Say). All six species are broadly distributed in the eastern USA (Lingafelter 2007) and are among the most abundant lamiines in the area of our studies, east-central Illinois (Hanks et al. 2014). The adult beetles are active during summer (Hanks et al. 2014) and crepuscular to nocturnal (LMH, unpub. data). Larvae of all six species develop in woody tissues of hardwood trees and shrubs of many families (Lingafelter 2007).
Pheromone components of some of the study species have been identified previously. That is, males of A. parvus have been reported to produce geranylacetone and both enantiomers of fuscumol and (R)-fuscumol acetate, whereas males of L. angulatus produce a blend of geranylacetone with both enantiomers of fuscumol acetate (Meier et al. 2016). The pheromone of A. variegatus originally was identified as a blend of (S)-fuscumol and (S)-fuscumol acetate (Hughes et al. 2013), but we report here that geranylacetone is a synergistic pheromone component. Pheromones of A. modestus, L. confluens, and S. alpha are identified here for the first time.
Methods and Materials
Sources of Chemicals
Compounds from commercial sources included racemic (E)-fuscumol and (E)-fuscumol acetate (Bedoukian Research, Danbury, CT, USA), (E)-geranylacetone (Sigma-Aldrich, St. Louis, MO, USA), sulcatone and racemic sulcatol (Alfa Aesar, Haverhill, MA, USA). (S)-Fuscumol (98% e.e.), (R)-fuscumol acetate (96.6% e.e.), and (S)-fuscumol acetate (98% e.e.) were synthesized as described in Hughes et al. (2013).
Study Sites
Field bioassays were conducted at six study sites in eastern Illinois (Table 1), most of which were mature second-growth or successional forests dominated by species of deciduous trees, including species of hickory (Carya), oak (Quercus), maple (Acer), and ash (Fraxinus). The private residence was in a suburban urban forest (~60 years old) of mature deciduous and coniferous trees of many native and exotic species.
Identification of Pheromones
Live adults of A. modestus, A. variegatus, L. confluens, and S. alpha, for collection of headspace volatiles, were caught with cross-vane panel traps (black corrugated plastic, Alpha Scents, Portland, OR, USA) with interior surfaces coated with the lubricant Fluon® (fluoropolymer dispersion, 10% aqueous dilution; Northern Products, Woonsocket, RI, USA). The trap collection buckets were replaced with plastic jars having aluminum screen bottoms that allowed rainwater to drain. Traps were suspended ~1 m above ground from inverted L-shaped frames of polyvinylchloride pipe mounted on steel reinforcing bar posts. Lures were polyethylene sachets (5.1 × 7.6 cm, Bagettes® model 14,770, Cousin Corp., Largo, FL, USA) loaded with 50 mg of racemic fuscumol and/or fuscumol acetate dissolved in 1 ml isopropanol. Lures contained a cotton roll (1 × 4 cm dental wick, Patterson Dental Supply, Inc., St. Paul, MN, USA) that absorbed the solutions and minimized leakage. Lures were replaced every 10–14 d. Single traps were deployed during summer months of 2013 to 2017 at all six study sites, and beetles were collected every 1–2 d.
Captured beetles were sexed by the morphology of the fifth abdominal sternite (longer and more emarginate in females; Linsley and Chemsak 1995). Males and females were caged separately in the laboratory (ambient conditions: ~12:12 h L:D, ~20 °C). Because the adult beetles may feed on the tender bark of oak or maple branchlets (pers. obs.), they were provided pieces of twigs freshly cut from oak or maple trees (Quercus alba L., Q. rubra L., Acer saccharum Marshall) at the same field sites where beetles had been trapped. Beetles also were provided sugar water (10% aqueous sucrose solution in a glass vial with a cotton wick) as a source of moisture. Beetles were allowed to acclimate for 24 h prior to the first headspace collections, and between subsequent collections.
Volatiles produced by beetles were collected from beetles held in glass Mason-style canning jars near closed exterior windows (natural photoperiod, ~14:10 h L:D, ~20 °C) using previously described methods (Meier et al. 2016, see Supplementary online information). Chemicals were tentatively identified by gas chromatography-mass spectrometry and structures were confirmed by comparing retention times and mass spectra with those of authentic standards (for details, see Supplementary online information). Extracts of headspace volatiles were prepared from 14 males and 5 females of A. modestus, 15 males and 5 females of A. variegatus, 5 males and 0 females of L. confluens, and 13 males and 5 females of S. alpha.
Field Bioassays
Attraction of beetles to synthesized pheromone components, presented individually and in blends, was tested with field bioassays using cross-vane panel traps as described above, but with the supplied trap basins partly filled with saturated aqueous NaCl solution to kill and preserve captured beetles. Traps were positioned 10 m apart in linear transects, and treatments were randomly assigned to traps on the day of setup. Traps were serviced every 1–3 d, at which time treatments were shifted one position along transects to compensate for small-scale positional effects.
Experiments 1–4 tested 19 unique combinations of racemic and chiral compounds of the geranylacetone and sulcatone classes (Table 2), including several combinations of compounds that had not been tested in earlier experiments (see Hughes et al. 2016; Meier et al. 2016, 2019; Mitchell et al. 2011). Some treatments were included specifically to confirm attraction of targeted species to synthesized reconstructions of their pheromones, others assessed the contribution of individual pheromone components to attraction, while still others tested for antagonistic effects by compounds that were not pheromone components of a particular species, but were pheromone components of other sympatric species (hereafter referred to as “heterospecific” compounds), as follows:
- 1)
Experiment 1 was a follow-up to the field screening trials of Mitchell et al. (2011), but with treatments added to assess the influence of geranylacetone (Table 2). Lures were sachets made from heat-sealed polyethylene tubing sections (5 × 4 cm; Associated Bag, Milwaukee, WI, USA) loaded with 1 ml of undiluted chemical. Release rates were standardized to ~20 mg/d by using tubing of different wall thicknesses and adding a cotton roll to some lures, as follows: fuscumol (1.5 mil wall thickness, with cotton roll); fuscumol acetate (3 mil wall thickness, with cotton roll), geranylacetone (3 mil wall thickness, without cotton roll) (authors’ unpub. data). Separate lures containing different chemicals were grouped on traps to produce blends. Empty sachets (3 mil wall thickness) served as blank controls. Lures were replaced every ~14 d. The experiment was conducted during three years at four study sites (Table 3).
- 2)
Experiment 2 assessed the role of geranylacetone in attraction of A. variegatus with the treatments including a blend of (S)-fuscumol and (S)-fuscumol acetate, alone and combined with geranylacetone, as well as blends with (R)-enantiomers to test for antagonistic effects (Table 2). Lures were the same polyethylene sachets as were used to collect beetles for headspace sampling, containing a cotton wick loaded with individual compounds diluted in isopropanol in doses of 50 mg for racemic fuscumol and fuscumol acetate (i.e., 25 mg per enantiomer) or 25 mg of geranylacetone and specific enantiomers of fuscumol and fuscumol acetate. The experiment was conducted during three years at five study sites (Table 3).
- 3)
Experiment 3 tested for attraction of unspecified species to various combinations of geranylacetone-type compounds with sulcatone (Table 2), under the assumption that certain species might have pheromones comprised of both basic structures. Trap lures consisted of the same polyethylene sachets mentioned above, loaded with 50 mg of racemic fuscumol or fuscumol acetate and 25 mg of geranylacetone or sulcatone, diluted in 1 ml isopropanol. The experiment was conducted during 2017 at two study sites (Table 3).
- 4)
Experiment 4 tested further unique combinations of compounds of the geranylacetone and sulcatone classes (Table 2). Lures were constructed of heat-sealed polyethylene tubing (1.5 mil), loaded with 50 mg of racemic sulcatol, fuscumol, or fuscumol acetate, or with 25 mg of sulcatone, diluted in 1 ml of isopropanol. The experiment was conducted during two years at two study sites (Table 3).
Taxonomy of captured beetles follows Monné and Hovore (2005). Representative specimens are available from the laboratory collection of LMH, and voucher specimens have been placed in the collection of the Illinois Natural History Survey, Champaign, IL.
Statistical Analysis
Data from the four field experiments were analyzed separately for each of the study species that were represented by at least 10 specimens. The data violated homoscedasticity assumptions of ANOVA (Sokal and Rohlf 1995), so differences between treatment means, blocked by site and date, were tested using the nonparametric Friedman’s Test (PROC FREQ, option CMH; SAS Institute 2011). Replicates with zero specimens of the species of interest, for example as a consequence of inclement weather, were dropped from analyses. For each analysis, greater weight was given to replicates with the largest numbers of specimens (i.e., replicates that represented the independent responses of multiple beetles to experimental treatments) by dropping those having fewer than a threshold number of beetles. Threshold numbers were chosen so as to maximize the number of beetles captured per replicate while maintaining a robust statistical test (at least 12 replicates; range of threshold numbers 2 to 5). Type I errors were controlled by setting the significance level of P to 0.0025, so as to not be excessively conservative (Quinn and Keough 2002). Assuming a significant overall statistical test, pairs of treatment means were compared using the Ryan-Einot-Gabriel-Welsch Q multiple comparison test (REGWQ; SAS Institute 2011).
Results
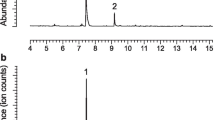
Extracts of volatiles emitted by males of A. modestus, A. variegatus, L. confluens, and S. alpha contained compounds of the geranylacetone class in significant amounts, as confirmed by matching mass spectra and retention times with those of authentic standards. There were detectable quantities of these compounds in 10 of 14 extracts (~71%) from males of A. modestus, 11 of 15 extracts (~73%) from males of A. variegatus, 4 of 5 extracts (~80%) from males of L. confluens, and 4 of 13 extracts (~31%) from males of S. alpha. None of these compounds were detected in headspace extracts from females nor in system controls.
Headspace extracts from adult males of A. variegatus contained traces of geranylacetone (Table 4), and analysis of extracts on a chiral stationary phase Cyclodex B GC column revealed that males also produce the (S)-enantiomers of fuscumol and fuscumol acetate, as reported previously (Hughes et al. 2013). Males of A. modestus, L. confluens, and S. alpha produced greater amounts of geranylacetone and specific enantiomers of fuscumol and fuscumol acetate in combinations apparently identical to those produced by other species. That is, males of A. modestus and L. confluens appeared to produce the same blend as males of L. angulatus, consisting of both enantiomers of fuscumol acetate + geranylacetone, whereas males of S. alpha produced the same blend as males of A. parvus, i.e., both enantiomers of fuscumol + (R)-fuscumol acetate + geranylacetone. Within species, ratios of enantiomers were in some cases difficult to estimate because compounds were present only in trace quantities, for which peak area could not be measured reliably. Nevertheless, there did appear to be some variability in the ratios of fuscumol or fuscumol acetate enantiomers produced by individual males of A. modestus, L. confluens, and S. alpha.
Field Bioassays
A total of 1484 beetles of the six species were captured during the field experiments (Table 5). During experiment 1, adults of A. variegatus were significantly attracted only to the complete blend of racemic fuscumol, fuscumol acetate, and geranylacetone (overall Friedman’s Q7,72 = 42.2, P < 0.001), with the blend attracting a mean (± SE) of 3.9 ± 0.8 beetles per replicate compared to means of fewer than 1.1 beetles for traps baited with the individual components, binary combinations, and the control (REGWQ, P < 0.05). This finding suggested that all the components were necessary for optimal attraction and that the non-natural (R)-enantiomers were not strongly antagonistic. However, previous studies have shown that adults of this species respond to racemic fuscumol acetate alone (i.e., attraction to [S]-fuscumol acetate in the racemate) or blended with fuscumol, in the absence of geranylacetone (e.g., Hanks and Millar 2013; Millar et al. 2018; Mitchell et al. 2011). Other species in the present study also were attracted by such incomplete reconstructions of pheromone blends when the optimal blend was not included as a treatment in a particular bioassay, as explained below.
We next consider the species whose pheromones were composed of both enantiomers of fuscumol acetate + geranylacetone: A. modestus, L. angulatus, and L. confluens. Adults of A. modestus were attracted only to the blend of racemic fuscumol acetate + geranylacetone during experiment 1 (Fig. 1A), i.e., the complete reconstruction of the pheromone, but not to either component alone, and attraction was antagonized by the heterospecific compound fuscumol. However, in experiment 3, beetles of this species were attracted to fuscumol acetate alone (Fig. 3A), and attraction was not significantly influenced by either geranylacetone or the heterospecific compound sulcatone. In experiment 4 the only significant attractant was the blend of fuscumol acetate + sulcatone + sulcatol (Fig. 4A), an unexpected enhancing effect by the latter two heterospecific compounds. Addition of fuscumol to this blend again resulted in antagonism. Earlier research had confirmed attraction of A. modestus to (S)-fuscumol acetate alone, and not to the (R)-enantiomer, but also to racemic fuscumol acetate, and racemic fuscumol acetate + fuscumol, despite the antagonistic properties of the latter compound (Hanks et al. 2012, 2018; Hughes et al. 2016; Millar et al. 2018; Mitchell et al. 2011).
Mean (± SE) number of beetles (sexes combined) per replicate captured during experiment 1 of the species: A) Aegomorphus modestus (Friedman’s Q7,64 = 36.0, P < 0.0001), B) Lepturges confluens (Friedman’s Q7,72 = 24.9, P = 0.0008), and C) Sternidius alpha (Q7,80 = 58.8, P < 0.0001). Chemical abbreviations: F = racemic fuscumol, Fa = racemic fuscumol acetate, Ga = geranylacetone (see Table 2 for complete list of treatments). Means within figures with different letters are significantly different (REGWQ test, P < 0.05)
Adults of L. angulatus were attracted to traps baited with fuscumol + fuscumol acetate in experiment 2 (Fig. 2A) which lacked a treatment with fuscumol acetate alone, and geranylacetone was not an attractant, but rather antagonized attraction. The lack of attraction to racemic fuscumol + (R)-fuscumol acetate, and to (S)-fuscumol + (S)-fuscumol acetate, suggested that the individual enantiomers of fuscumol acetate were insufficient for attraction. In experiment 3, beetles were attracted by fuscumol acetate alone (Fig. 3B), and apparently not influenced by either geranylacetone or the heterospecific compound sulcatone, but antagonized by the blend of these two compounds. The inconsistent effect of geranylacetone between experiments 2 and 3 suggests that it is antagonistic only when paired with the heterospecific compounds fuscumol or sulcatone. Lastly, experiment 4 again showed significant attraction to fuscumol acetate alone (Fig. 4B), and antagonism by sulcatone + sulcatol. There is a wealth of information about the chemical ecology of this common species, with several earlier studies having demonstrated attraction to fuscumol acetate, that both enantiomers are necessary, and that attraction is not influenced by the heterospecific compound fuscumol (e.g., Hanks and Millar 2013; Hanks et al. 2018; Meier et al. 2016; Mitchell et al. 2011).
Mean (± SE) number of beetles (sexes combined) per replicate captured during experiment 2 of the species: A) Lepturges angulatus (Q7,112 = 46.3, P < 0.0001), B) Lepturges confluens (Q7,78 = 19.0, P = 0.008), and C) Astylidius parvus (Q7,88 = 33.7, P < 0.0001). Chemical abbreviations: F = racemic fuscumol, Fa = racemic fuscumol acetate, Ga = geranylacetone; R and S indicate individual enantiomers of fuscumol and fuscumol acetate (see Table 2 for complete list of treatments). Means within figures with different letters are significantly different (REGWQ test, P < 0.05)
Mean (± SE) number of beetles (sexes combined) per replicate captured during experiment 3 of the species: A) Aegomorphus modestus (Q7,96, = 38.2, P < 0.0001), and B) Lepturges angulatus (Q7,96 = 47.7, P < 0.0001). Chemical abbreviations: Fa = racemic fuscumol acetate, Ga = geranylacetone, Sone = sulcatone (see Table 2 for complete list of treatments). Means within figures with different letters are significantly different (REGWQ test, P < 0.05)
Mean (± SE) number of beetles (sexes combined) per replicate captured during experiment 4 of the species: A) Aegomorphus modestus (Q7,64 = 30.4, P < 0.0001). B) Lepturges angulatus (Q7,96 = 32.7, P < 0.0001), C) Astylidius parvus (Q7,88 = 37.8, P < 0.0001), and D) Sternidius alpha (Q7,103 = 57.3, P < 0.0001). Chemical abbreviations: F = racemic fuscumol, Fa = racemic fuscumol acetate, SoneSol = sulcatone + racemic sulcatol (see Table 2 for complete list of treatments). Means within figures with different letters are significantly different (REGWQ test, P < 0.05)
Adults of L. confluens were significantly attracted only to the blend of racemic fuscumol + fuscumol acetate + geranylacetone in experiment 1 (Fig. 1B), although weak statistical power limited interpretation of other treatment effects. Beetles were not significantly attracted to the same blend in experiment 2 (Fig. 2B), but rather only to (S)-fuscumol + (S)-fuscumol acetate + geranylacetone, suggesting that (S)-fuscumol acetate + geranylacetone was a sufficient attractant given that this species does not produce either enantiomer of fuscumol, and that (R)-fuscumol was antagonistic. Earlier studies have shown attraction of L. confluens to racemic fuscumol acetate alone, or combined with racemic fuscumol (Hanks and Millar 2013; Millar et al. 2018). This prior attraction to racemic fuscumol acetate, but only to the blend containing (S)-fuscumol acetate in experiment 2 of the present article, suggests that (R)-fuscumol acetate is not necessary for attraction, even though it was present in the insect-produced volatiles.
Despite apparently having pheromones of similar composition, A. modestus, L. angulatus, and L. confluens showed quite different responses to the same experimental treatments. For example, there were marked differences between A. modestus and L. confluens in their responses during experiment 1 (Fig. 1A,B), between L. angulatus and L. confluens during experiment 2 (Fig. 2A,B), and between A. modestus and L. angulatus during experiment 3 (Fig. 3A,B) and experiment 4 (Fig. 4A,B).
Males of both A. parvus and S. alpha produced both enantiomers of fuscumol, (R)-fuscumol acetate, and geranylacetone, and adults of A. parvus were significantly attracted to traps baited with the complete reconstruction of this blend during experiment 2 (Fig. 2C). However, significant numbers of beetles also were attracted to traps baited with racemic fuscumol + fuscumol acetate without geranylacetone. These findings suggest that the latter compound was at most a weak synergist, and that the non-natural (S)-fuscumol acetate was not antagonistic. Experiment 4 showed that this species was attracted by racemic fuscumol alone (Fig. 4C), a treatment missing in the previous experiment, and attraction was not influenced by the heterospecific compounds sulcatone + sulcatol. Racemic fuscumol acetate was not inherently attractive, further suggesting that (R)-fuscumol acetate was not likely to be attractive as a single component. Previous reports have shown that adults of A. parvus were attracted by racemic fuscumol, but not to the individual enantiomers, further supporting the case for synergism between enantiomers, and were not influenced by (R)-fuscumol acetate even though it was present in headspace volatiles from males (e.g., Meier et al. 2016; Millar et al. 2018).
Adults of S. alpha were significantly attracted by racemic fuscumol in experiment 1 (Fig. 1C), and attraction was not influenced by geranylacetone, but was antagonized by racemic fuscumol acetate, even though males produce (R)-fuscumol acetate (see above). The latter finding suggests that the heterospecific (S)-fuscumol acetate is antagonistic. Attraction to racemic fuscumol again was evident in experiment 4 (Fig. 4D), as was antagonism by racemic fuscumol acetate, and also by the heterospecific compounds sulcatone + sulcatol. Earlier studies have shown attraction of this species to racemic fuscumol, but not to fuscumol acetate (Hanks et al. 2018; Millar et al. 2018; Mitchell et al. 2011).
As with the above mentioned three species with similar pheromones, adults of S. alpha and A. parvus differed in their responses to treatments in the one experiment they had in common, experiment 4 (compare Fig. 4C,D), despite their apparently having pheromones of similar composition.
Discussion
Our research revealed a third compound in the blend produced by males of A. variegatus, geranylacetone, which synergized attraction to the two components identified previously, (S)-fuscumol and (S)-fuscumol acetate (Hughes et al. 2013). Geranylacetone also was detected in headspace extracts from males of the other five species, and in fact is a pheromone component of the first lamiine species reported to have pheromones of the geranylacetone class, H. betulinus (Vidal et al. 2010). However, among the other five study species, geranylacetone appeared to play a variable role, enhancing attraction of L. confluens, having little if any effect on attraction of A. modestus, A. parvus, and S. alpha, but apparently antagonizing attraction of L. angulatus when in the presence of the heterospecific compounds fuscumol or sulcatone.
The varying role of geranylacetone is consistent with earlier research which has shown that some compounds released by male cerambycids appear not to be involved in attraction of conspecifics (Hanks et al. 2019). For example, the pheromone of the South American lamiine Psapharochrus maculatissimus (Bates) is composed of (S)-fuscumol acetate with a small proportion of the (R)-enantiomer, and the minor component appears to contribute nothing to attraction (Silva et al. 2019). However, such apparently inactive compounds may play a role in mediating interactions among sympatric species, such as by antagonizing attraction to a shared pheromone component. For example, the pheromone of the North American Xylotrechus colonus (F.) (subfamily Cerambycinae) is composed primarily of (R)-3-hydroxyhexan-2-one, a necessary and sufficient attractant, but enantiomers of the related 2,3-hexanediols also are produced in trace quantities. The diols apparently have no effect on attraction of conspecifics, but rather antagonize attraction of the sympatric Neoclytus mucronatus mucronatus (F.) to the shared ketol component (Hanks et al. 2019). Thus, at least some male cerambycids appear to produce pheromone blends containing components which affect the behavior of both conspecifics and heterospecifics.
In the latter example, adults of N. m. mucronatus clearly are capable of detecting the antagonistic 2,3-hexanediols of X. colonus, even though these compounds were not produced by males of N. m. mucronatus. Antagonism by compounds produced by heterospecifics provides further evidence of behavioral mechanisms that have evolved to limit interspecific attraction. As a further example, males of A. variegatus produced (S)-fuscumol + (S)-fuscumol acetate + geranylacetone, but attraction of A. modestus to the fuscumol acetate component would be antagonized by the fuscumol component, and attraction of S. alpha to fuscumol would be antagonized by the fuscumol acetate component.
The interplay of attractants and antagonists for the six species in our study is summarized in Table 6. In addition to the species specificity imparted by antagonistic compounds produced by heterospecifics, interspecific attraction between several other pairs of the study species is likely to be averted due to the absence of critical components, as follows:
- 1)
Adults of A. variegatus, A. modestus, L. angulatus, and L. confluens all require (S)-fuscumol acetate, which is absent in the pheromones of A. parvus and S. alpha;
- 2)
Adults of L. angulatus require both enantiomers of fuscumol acetate, but males of A. variegatus only produce the (S)-enantiomer;
- 3)
Adults of A. parvus and S. alpha require enantiomers of fuscumol that are absent in the pheromones of the other four species.
It also should be noted from these examples that the mechanisms that prevent interspecific attraction can be complimentary, for example preventing attraction between A. variegatus and A. parvus in both directions. There also is evidence that these mechanisms can be redundant. For example, attraction of A. modestus to calling males of A. parvus would be averted both due to antagonism by fuscumol and the absence of the critical attractant (S)-fuscumol acetate.
The research presented here has not identified factors that would prevent cross attraction between species that apparently have pheromones composed of the same components. Specifically, males of A. modestus, L. angulatus, and L. confluens produce blends consisting of both enantiomers of fuscumol + geranylacetone, while males of A. parvus and S. alpha produce both enantiomers of fuscumol + (R)-fuscumol acetate + geranylacetone. For other types of insects in which multiple related species have similar pheromones, in particular moths and bark beetles (Curculionidae, Scolytinae), species specificity often is imparted by relative ratios of components (e.g., Borden et al. 1980; Heath et al. 1990). However, previous research has suggested that conspecific males of lamiines may vary considerably in the ratios of pheromone components they produce (Meier et al. 2016, 2019), suggesting that ratios of components may not be reliable indicators of species specificity, and that other mechanisms, such as differing diel activity periods, may also play a role in reproductive isolation (e.g., Meier et al. 2019). Other factors that could serve to segregate the species include varying responses to particular host plant volatiles or preferences for different vertical strata within forests (reviewed by Hanks and Millar 2016; Millar and Hanks 2017).
The pheromone of Astylopsis macula (Say) provided the first evidence that pheromones of lamiines may be composed of compounds of both the geranylacetone and sulcatone classes (Meier et al. 2019). Although none of the six study species in the present article produced compounds of the sulcatone class, the field experiments nevertheless revealed that sulcatone and/or sulcatol influenced attraction of L. angulatus and S. alpha to their reconstructed pheromones, indicating that these two compounds are indeed perceived by these species. This in turn suggests that these species may be sympatric with as yet unknown species which produce blends of geranylacetone- and sulcatone-type compounds.
References
Borden JH, Handley JR, McLean JA, Silverstein RM, Chong L, Slessor KN, Johnston BD, Schuler HR (1980) Enantiomer-based specificity in pheromone communication by two sympatric Gnathotrichus species (Coleoptera: Scolytidae). J Chem Ecol 2:445–456
Fonseca MG, Vidal DM, Zarbin PHG (2010) Male-produced sex pheromone of the cerambycid beetle Hedypathes betulinus: chemical identification and biological activity. J Chem Ecol 36:1132–1139
Hanks LM, Millar JG (2013) Field bioassays of cerambycid pheromones reveal widespread parsimony of pheromone structures, enhancement by host plant volatiles, and antagonism by components from heterospecifics. Chemoecol 23:21–44
Hanks LM, Millar JG (2016) Sex and aggregation-sex pheromones of cerambycid beetles: basic science and practical applications. J Chem Ecol 42:631–654
Hanks LM, Millar JG, Mongold-Diers JA, Wong JCH, Meier LR, Reagel PF, Mitchell RF (2012) Using blends of cerambycid beetle pheromones and host plant volatiles to simultaneously attract a diversity of cerambycid species. Can J For Res 42:1050–1059
Hanks LM, Reagel PF, Mitchell RF, Wong JCH, Meier LR, Silliman CA, Graham EE, Striman BL, Robinson KP, Mongold-Diers JA, Millar JG (2014) Seasonal phenology of the cerambycid beetles of east-Central Illinois. Ann Entomol Soc Am 107:211–226
Hanks LM, Mongold-Diers JA, Atkinson TH, Fierke MK, Ginzel MD, Graham EE, Poland TM, Richards AB, Richardson ML, Millar JG (2018) Blends of pheromones, with and without host plant volatiles, can attract multiple species of cerambycid beetles simultaneously. J Econ Entomol 111:716–724
Hanks LM, Mongold-Diers JA, Mitchell RF, Zou Y, Wong JCH, Meier LR, Johnson TD, Millar JG (2019) The role of minor pheromone components in segregating 14 species of longhorned beetles (Coleoptera: Cerambycidae) of the subfamily Cerambycinae. J Econ Entomol 112:2236–2252
Heath RR, Mitchell ER, Tovar JC (1990) Effect of release rate and ratio of (Z)-11-hexadecen-1-ol from synthetic pheromone blends on trap capture of Heliothis subflexa (Lepidoptera: Noctuidae). J Chem Ecol 16:1259–1268
Hughes GP, Zou Y, Millar JG, Ginzel MD (2013) (S)-fuscumol and (S)-fuscumol acetate produced by a male Astyleiopus variegatus (Coleoptera: Cerambycidae). Can Entomol 145:1–6
Hughes GP, Meier LR, Zou Y, Millar JG, Hanks LM, Ginzel MD (2016) Stereochemistry of fuscumol and fuscumol acetate influences attraction of longhorned beetles (Coleoptera: Cerambycidae) of the subfamily Lamiinae. Environ Entomol 45:1271–1275
Lingafelter SW (2007) Illustrated key to the longhorned wood-boring beetles of the eastern United States. Special publication no. 3. Coleopterists Society, North Potomac, p 206
Linsley EG, Chemsak JA (1995) The Cerambycidae of North America, part VII, no. 2: taxonomy and classification of the subfamily Lamiinae, tribes Acanthocinini through Hemilophini. Univ Calif Publ Entomol 114:1–292
Meier LR, Zou Y, Millar JG, Mongold-Diers JA, Hanks LM (2016) Synergism between enantiomers creates species-specific pheromone blends and minimizes cross-attraction for two species of cerambycid beetles. J Chem Ecol 42:1181–1192
Meier LR, Millar JG, Mongold-Diers JA, Hanks LM (2019) (S)-Sulcatol is a pheromone component for two species of cerambycid beetles in the subfamily Lamiinae. J Chem Ecol 45:447–454
Millar JG, Hanks LM (2017) Chemical ecology of cerambycids. In: Wang Q (ed) Cerambycidae of the world: biology and pest management. CRC Press/Taylor & Francis, Boca Raton, pp 161–208
Millar JG, Mitchell RF, Mongold-Diers JA, Zou Y, Bográn CE, Fierke MK, Ginzel MD, Johnson CW, Meeker JR, Poland TM, Ragenovich IR, Hanks LM (2018) Identifying possible pheromones of cerambycid beetles by field testing known pheromone components in four widely separated regions of the United States. J Econ Entomol 111:252–259
Mitchell RF, Graham EE, Wong JCH, Reagel PF, Striman BL, Hughes GP, Paschen MA, Ginzel MD, Millar JG, Hanks LM (2011) Fuscumol and fuscumol acetate are general attractants for many species of cerambycid beetles in the subfamily Lamiinae. Entomol Exp Appl 141:71–77
Monné MA, Hovore FT (2005) Checklist of the Cerambycidae (Coleoptera) of the Western hemisphere. BioQuip, Rancho Dominguez
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge, UK
Ryall K, Silk P, Webster RP, Gutowski JM, Meng Q, Li Y, Gao W, Fidgen J, Kimoto T, Scarr T, Mastro V, Sweeney JD (2015) Further evidence that monochamol is attractive to Monochamus (Coleoptera: Cerambycidae) species, with attraction synergised by host plant volatiles and bark beetle (Coleoptera: Curculionidae) pheromones. Can Entomol 147:564–579
SAS Institute (2011) SAS/STAT 9.3 user's guide. SAS Institute Inc., Cary
Silk PJ, Sweeney J, Wu J, Price J, Gutowski JM, Kettela EG (2007) Evidence for a male-produced pheromone in Tetropium fuscum (F.) and Tetropium cinnamopterum (Kirby) (Coleoptera: Cerambycidae). Naturwissenschaften 94:697–701
Silva WD, Zou Y, Hanks LM, Bento JMS, Millar JG (2019) Enantiomers of fuscumol acetate comprise the aggregation-sex pheromone of the south American cerambycid beetle Psapharochrus maculatissimus, and likely pheromones of the cerambycids Eupromerella plaumanni and Hylettus seniculus. Entomol Exp Appl 167:915–921
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. WH Freeman and Co, New York
Švácha P, Lawrence JF (2014) Chapter 2.4 Cerambycidae Latreille, 1802, pp. 77–177. In: Leschen RAB, Beutel RG (eds) handbook of zoology: Arthropoda: Insecta: Coleoptera, beetles. Vol. 3: morphology and systematics (Phytophaga), Walter de Gruyter, Berlin/Boston
Sweeney JD, Silk PJ, Gutowski JM, Wu J, Lemay MA, Mayo PD, Magee DI (2010) Effect of chirality, release rate, and host volatiles on response of Tetropium fuscum (F.), Tetropium cinnamopterum Kirby, and Tetropium castaneum (L.) to the aggregation pheromone, fuscumol. J Chem Ecol 36:1309–1321
Vidal DM, Fonseca MG, Zarbin PH (2010) Enantioselective synthesis and absolute configuration of the sex pheromone of Hedypathes betulinus (Coleoptera: Cerambycidae). Tetrahedron Lett 51:6704–6706
Acknowledgements
We thank Jodie A. Ellis for assistance with laboratory work. We also thank S. Buck and the University of Illinois Committee on Natural Areas for access to field sites. This research was supported by a grant to L.M.H. from The Alphawood Foundation of Chicago, and grants to J.G.M. and L.M.H. from the United Stated Department of Food and Agriculture, National Institute of Food and Agriculture (USDA-NIFA, grant numbers 2012-67013-19303 and 2015-67013-23173), and USDA-Animal and Plant Health Inspection Service grant #15-8130-1422-CA.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Meier, L.R., Zou, Y., Mongold-Diers, J.A. et al. Pheromone Composition and Chemical Ecology of Six Species of Cerambycid Beetles in the Subfamily Lamiinae. J Chem Ecol 46, 30–39 (2020). https://doi.org/10.1007/s10886-019-01128-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01128-7