Abstract
Males of several species in the large cerambycid beetle subfamily Lamiine, native to South and North America, are known to produce aggregation-sex pheromones composed of three related structures: the sesquiterpene catabolic product geranylacetone, its corresponding alcohol (known as fuscumol), and the acetate ester of the alcohol (fuscumol acetate). Here, we show that males of two lamiine species native to the eastern USA, Astylopsis macula (Say) and Leptostylus transversus (Gyllenhal) (both tribe Acanthocinini), produce pheromones composed of the structurally related (S)-6-methylhept-5-en-2-ol ([S]-sulcatol). Males of both species also produced 6-methylhept-5-en-2-one (sulcatone), and males of A. macula produced trace amounts of (R)-fuscumol acetate. Field experiments demonstrated that both species were attracted by (S)-sulcatol. Sulcatone was not attractive as a single component, nor did it influence attraction of A. macula to (S)-sulcatol, while its effect on L. transversus was unclear. Data from the field experiments, and a separate experiment using traps fitted with a timer mechanism that rotated trap jars, revealed that the two species overlap only slightly in both seasonal and daily flight period, thus minimizing interspecific attraction. Racemic fuscumol acetate and fuscumol antagonized attraction of both species to sulcatol. The identification of sulcatol as a cerambycid pheromone extends the known range of pheromone components in the subfamily Lamiinae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Research on the pheromone chemistry of cerambycid beetles has suggested that certain compounds have been evolutionarily conserved, with a number of examples of species on different continents sharing major pheromone components, or at least compounds of the same structural class (reviewed by Millar and Hanks 2017). Species in the subfamilies Cerambycinae, Lamiinae, and Spondylidinae all produce aggregation-sex pheromones, which are produced by males but attract both sexes (sensu Cardé 2014). Among species of the large subfamily Cerambycinae, interspecific attraction among species that share pheromone components may be averted by differences between species in seasonal or daily activity periods of the adults, or by minor pheromone components that synergize attraction of conspecifics while antagonizing attraction of heterospecifics (Hanks et al. 2019; Mitchell et al. 2015).
Males of several species in the large subfamily Lamiinae, native to South and North America, are known to produce aggregation-sex pheromones composed of three related structures (Fig. 1): the sesquiterpene catabolic product geranylacetone, its corresponding alcohol (known as fuscumol; Sweeney et al. 2010), and the acetate ester of the alcohol (fuscumol acetate; Hanks and Millar 2016; Hughes et al. 2013, 2016; Meier et al. 2016; Millar et al. 2018; Mitchell et al. 2011; Vidal et al. 2010). All of these species are in the tribes Acanthocinini or Acanthoderini. In an earlier study, we demonstrated that synergism among enantiomers of fuscumol and fuscumol acetate imparted species specificity to the pheromones of the acanthocinines Astylidius parvus (LeConte) and Lepturges angulatus (LeConte), which are sympatric and synchronic (Meier et al. 2016). That is, males of A. parvus produce both (R)- and (S)-fuscumol + (R)-fuscumol acetate whereas males of L. angulatus produce (R)- and (S)-fuscumol acetate, and adults of both species respond only to the combinations of enantiomers that constitute their pheromones.
Chemical structures of pheromone components produced by males of several species of cerambycid beetles in the subfamily Lamiinae of the tribes Acanthocinini and Acanthoderini (Hanks and Millar 2016)
Here, we show that (S)-6-methylhept-5-en-2-ol ([S]-sulcatol), a pheromone component of a related structural class based on the terpene catabolic product 6-methyl-5-hepten-2-one (sulcatone; Fig. 1), is a pheromone component of two lamiine species native to eastern North America, Astylopsis macula (Say) and Leptostylus transversus (Gyllenhal) (both Acanthocinini). Larvae of both species develop in the decaying wood of hardwood tree species of many families (Linsley and Chemsak 1995), and may even feed within the same individual hosts (Beutenmiller 1896; Craighead 1923). An earlier study suggested that the two species overlap in seasonal activity period, L. transversus being the earliest lamiine to fly in Illinois (active from May through July) whereas adults of A. macula fly from mid-June through early August (Hanks et al. 2014). However, that study summarized activity periods from data collected in different years and across different field sites, and the resulting variation may have obscured evidence of temporal segregation.
We provide evidence that males of A. macula and L. transversus produce volatiles of similar composition, consisting of a blend of sulcatone and (S)-sulcatol, and that differences in flight phenology likely minimizes interspecific attraction. Males of A. macula also produced small amounts of (R)-fuscumol acetate. The functional role of (S)-sulcatol as an aggregation-sex pheromone component was confirmed with field bioassays.
Methods and Materials
Sources of Chemicals
Sulcatone, racemic sulcatol, (R)-sulcatol, and (S)-sulcatol were purchased from Alfa Aesar (Haverhill, MA, USA), and racemic (E)-fuscumol and (E)-fuscumol acetate from Bedoukian Research (Danbury, CN, USA).
Study Sites
Field work to identify pheromones of A. macula and L. transversus, and to confirm attraction to synthesized pheromones, was conducted at two study sites in east-central Illinois: Brownfield Woods (Champaign Co.; 40.145 lat., −88.165 long.; ~25 ha; property of the University of Illinois) and Forest Glen Preserve (Vermilion Co., 40.015 lat., −87.568 long., ~730 ha; Vermilion Co. Conservation District). Both sites were wooded with mature second-growth and successional hardwoods and dominated by species of oak (Quercus), hickory (Carya), maple (Acer), and ash (Fraxinus).
Identification of Pheromones
Evidence that sulcatone and/or sulcatol were pheromone components of A. macula and L. transversus came from headspace sampling (methods below) of individual adult males that had been captured during earlier field experiments that used flight intercept traps (black corrugated plastic, cross-vane, Alpha Scents, Portland, OR) coated with Fluon® (10% aqueous dilution; e.g., Meier et al. 2016). Traps were modified to keep captured beetles alive by replacing the basins with 2-l plastic jars with the bottoms removed and covered with aluminum screen to permit drainage of rainwater. Traps were suspended from inverted L-shaped frames of polyvinylchloride irrigation pipe. More beetles of both species were captured to confirm pheromone composition using traps of the same design baited with polyethylene sachets (5.1 × 7.6 cm, Bagettes® model 14,770, Cousin Corp., Largo, FL, USA) loaded with 25 mg of sulcatone blended with 50 mg of racemic sulcatol (i.e., 25 mg per enantiomer) dissolved in 1 ml isopropanol. This solvent was selected because it is not inherently attractive to cerambycid beetles (Wong et al. 2017), and it is not a byproduct of decay or fermentation, unlike ethanol, a known attractant for some species of cerambycids (Millar and Hanks 2017). Single traps were deployed from May to August 2015 at the two study sites, and serviced every 1–2 d. Trap lures were replaced as needed, usually every 10–14 d.
Captured adults of A. macula and L. transversus were sexed by the morphology of the fifth abdominal sternite (Linsley and Chemsak 1995), or by caging pairs of beetles and observing mating behavior (i.e., males mount females). Beetles were held in single-sex groups, under ambient laboratory conditions (~12:12 h L:D, ~20 °C). Preliminary observations indicated that adults of both species will feed on the tender bark of branches from trees of host species of the larvae. Therefore caged adults of A. macula were provided freshly-cut branch segments (2–5 cm in diameter, ~8 cm long) of maple (Acer saccharum Marshall) and L. angulatus with branches of shagbark hickory (Carya ovata [Mill.] K. Koch; see Linsley and Chemsak 1995). Beetles also were provided 10% aqueous sucrose solution (in a glass vial with a cotton wick) as a water and nutrition source. They were allowed at least 24 h to acclimate prior to collection of headspace volatiles, and between subsequent collections.
Insect-produced volatiles were collected from beetles held in glass Mason-style canning jars that were positioned near closed exterior windows (natural photoperiod, ~14:10 h L:D, ~20 °C). Volatiles usually were collected from individual beetles, but in some cases collections were made from groups of two or three beetles of the same sex to improve the chances of collecting pheromones. Once it was confirmed that only the males produced volatile chemicals (see Results), additional collections were made from mixed-sex groups in case the presence of females encouraged males to call. Beetles often were held with small sections of branches from their host plants, in which case we also collected volatiles from these materials without beetles as system controls. Additional controls were used to detect plant volatiles released by feeding beetles, by headspace collections from twigs that had been damaged mechanically by scraping or gouging the bark with a metal probe.
Collection jars were swept with clean air (1 l/min) for 24 h, with headspace volatiles being collected on a trap of the adsorbent polymer HayeSep® Q (150 mg; Sigma-Aldrich, St. Louis, MO, USA). Traps were extracted with 1.5 ml of dichloromethane into silanized glass vials that were stored at −20 °C. Collections were made from 37 males and 2 females for A. macula, and 35 males and 4 females for L. transversus.
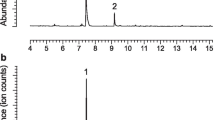
Headspace extracts from adult beetles were analyzed with a gas chromatograph interfaced to a mass selective detector (Models 6890 and 5973, Hewlett-Packard, Palo Alto, CA, USA; AT-5 ms column, 30 m × 0.25 mm i.d., 0.25 μm film, Alltech Associates Inc., Deerfield, IL, USA). The GC oven was programmed from 35 °C/1 min, 10 °C/min to 210 °C, hold 3 min. Injections were made in splitless mode, with an injector temperature of 250 °C, and helium carrier gas. Tentative identifications of sex-specific compounds were made by comparison of mass spectra with database spectra, and then were confirmed by comparing their GC retention times and mass spectra with those of authentic standards.
The absolute configurations of insect-produced chiral chemicals were determined by analyzing aliquots of extracts with an HP 5890 gas chromatograph equipped with a chiral stationary phase Cyclodex B column (30 m × 0.25 mm i.d., 0.25 μm film; Agilent Technologies Inc., Santa Clara, CA, USA) with the oven temperature programmed from 50 °C/1 min, 2.5 °C/min to 200 °C, hold 3 min, and an injector temperature of 210 °C. Absolute configurations of sulcatol and fuscumol acetate were confirmed by co-injection of an aliquot of aeration extract with the mixture of both enantiomers (see Millar et al. 2009). Resolution of (S)- and (R)-enantiomers was to baseline (retention times 26.10 and 26.38 min for sulcatol, 51.33 and 51.72 min for fuscumol acetate, respectively).
Field Bioassays of Synthesized Pheromones
Attraction of A. macula and L. transversus to synthesized candidate pheromones was tested with four independent field bioassays. Synthesized compounds were dispensed from sachets made from heat-sealed polyethylene tubing (5 × 7.5 cm, 1.5 mil, 38 μm wall thickness; catalog #27–3-02, Associated Bag Co., Milwaukee, WI, USA Associated Bag, Milwaukee, WI, USA). Lures were loaded with individual compounds (diluted in 1 ml isopropanol) in the following amounts: 25 mg of the achiral sulcatone, 50 mg of racemic sulcatol, fuscumol, or fuscumol acetate (i.e., 25 mg per enantiomer), and 25 mg of the individual enantiomers of sulcatol. All lures also contained a cotton roll (1 × 4 cm dental wick, Patterson Dental Supply, Inc., St. Paul, MN, USA) which minimized leakage and stabilized release rates (unpub. data). Blends of compounds were created by combining lures on the same trap. Control traps were baited with sachets of the same design, but containing 1 ml neat isopropanol. Traps were deployed ~10 m apart in linear transects with one trap per treatment, and treatments were assigned randomly to trap positions on the day of setup. They were serviced at intervals of 2–3 d, at which time treatments were shifted one position down the transect to control for location effects on trap catch. Lures were replaced every ~14 d.
The goal of Experiment 1 was to confirm attraction of A. macula and L. transversus to the candidate pheromone components they have in common (sulcatone + [S]-sulcatol), and the possible effect of the non-natural (R)-sulcatol enantiomer. Thus, traps were baited with synthetic sulcatone, racemic sulcatol, and (R)- and (S)-sulcatol individually and in binary blends (Table 1). The experiment was conducted at both study sites during 22 April to 29 July 2016 with one transect of traps per site (N = 51 replicates over time).
The remaining three experiments also tested the roles of sulcatone and sulcatol in attracting the two species (Table 1), and how attraction was influenced by fuscumol and fuscumol acetate (the [R]-enantiomer of the latter being present in trace amounts in headspace volatiles from A. macula; see Results). Thus, racemic fuscumol and fuscumol acetate, alone and blended, were combined with racemic sulcatol (experiment 2), sulcatone (experiment 3), and sulcatone + racemic sulcatol (experiment 4). Experiments 2 and 3 were conducted during 22 April to 12 August 2016 at Forest Glen (N = 33 replicates each), while experiment 4 was run during 15 July to 16 September 2015 at Forest Glen and 22 April to 4 August 2016 at Brownfield Woods (N = 49 replicates).
Captured beetles were identified to species using the key in Lingafelter (2007). Specimens are available from the laboratory collection of LMH and voucher specimens have been deposited with the collection of the Illinois Natural History Survey, Champaign, IL, USA.
Statistical Analyses of Bioassay Data
For each experiment, differences between treatment means were tested separately for each species using the nonparametric Friedman’s Test (PROC FREQ, option CMH; SAS Institute 2011) because data violated homoscedasticity assumptions of ANOVA (Sokal and Rohlf 1995). Thus, the analyses were blocked by site and date, excluding replicates that contained no specimens of the species in question due, for example, to inclement weather. Replicates with the greatest numbers of beetles (i.e., that represented the independent responses of multiple beetles to bioassay treatments) were lent greater weight by dropping from analyses those replicates having fewer than a threshold number of beetles. These threshold numbers were selected, separately for each analysis, so as to maximize the number of beetles captured while maintaining sufficient replication for a robust statistical test (at least 12 replicates; range of threshold numbers 2 to 5). Comparisons of treatment means were protected (i.e., assuming a significant overall Friedman’s test) and tested using the Ryan-Einot-Gabriel-Welsch Q multiple comparison test (REGWQ, SAS Institute 2011).
Flight Phenology of Adult Beetles
Seasonal flight period of the two beetle species was assessed using trap capture data from the field bioassays conducted during 2016: experiment 1 (both sites), experiment 2 (Forest Glen), and experiment 3 (Brownfield Woods). The numbers of beetles captured of each species were summed for 10-d intervals beginning 25 April (ordinal date 115) to 13 August (225), encompassing the entire flight periods of both species. Overlap in phenology was further characterized as the percentage of beetles of each species that were captured by the same traps as beetles of the other species.
Daily phenology of the two species was compared by estimating the time of day that beetles were captured using the same type of trap described above, but fitted with a mechanism that rotated eight collecting jars at programmable intervals (model #2850, BioQuip Products, Rancho Dominguez, CA, USA). The timer mechanism was set to rotate jars at seven 1-h intervals, beginning at 19:00 h and ending at 2:00 h, in order to encompass the crepuscular to nocturnal activity periods of the beetle species (LRM, pers. obs.). The eighth jar was not rotated for 17 h (2:00–19:00), to confirm that beetles were not active during the remainder of each 24-h cycle. The hour that beetles were caught was estimated as the median time that their trap jar was positioned under the trap. Timer traps were run between 15 May and 5 July 2017 at the Brownfield Woods study site.
Results
Identification of Pheromones
Extracts of volatiles emitted by males of both species contained two compounds in significant amounts, which were tentatively identified as sulcatone and sulcatol by matching their spectra to those in the NIST mass spectral database. The identifications were then confirmed by matching their mass spectra and retention times with those of authentic standards. There were detectable quantities of the two compounds in 18 of 37 extracts (~49%) from males of A. macula and 24 of 35 extracts (~69%) from males of L. transversus. Of the 18 males of A. macula, five also produced trace amounts of fuscumol acetate (< 1% of the dominant peaks). None of these compounds were detected in any headspace extracts from females or in system controls. Analysis of extracts on a chiral stationary phase Cyclodex B GC column revealed that males of both species produced only the (S)-enantiomer of sulcatol, and the average percentages of sulcatone to (S)-sulcatol (± SD) were 61.5 ± 26.3 and 23.8 ± 10.7 for A. macula and L. transversus, respectively. Similarly, the fuscumol acetate found in trace amounts in extracts from males of A. macula was determined to be the (R)-enantiomer.
Field Bioassays of Synthesized Pheromones
A total of 287 adults of A. macula and 186 adults of L. transversus were trapped during the four field experiments. Both sexes were attracted, for example with sex ratios of 63% females for A. macula and 59% females for L. transversus in experiment 4.
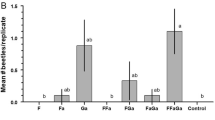
Adults of both species were caught in sufficient numbers for statistical analysis in all of the experiments except for experiment 3, in which none of the treatments contained sulcatol, the only compound that attracted either species (see below). Adult beetles of the two species responded similarly to trap treatments in the remaining three experiments (Fig. 2). In experiment 1, there was no significant difference in attraction of adults of A. macula to traps baited with racemic sulcatol or (S)-sulcatol (Fig. 2a), suggesting that the (R)-enantiomer was not antagonistic. Sulcatone was not inherently attractive, nor did it significantly influence attraction to (S)- or racemic sulcatol. Adults of L. transversus were significantly attracted by both (S)- and racemic sulcatol, again indicating no inhibition by (R)-sulcatol, whereas sulcatone appeared to antagonize attraction to (S)-sulcatol but did not influence attraction to racemic sulcatol.
Mean (± SE) number of adults of Astylopsis macula and Leptostylus transversus caught per replicate during a) experiment 1 (Friedman’s Q7,88 = 43.4, P < 0.0001; Q7,64 = 37.8, P < 0.0001), b experiment 2 (Q7,72 = 33.5, P < 0.0001; Q7,72 = 25.7, P = 0.0006), and c) experiment 4 (Q7,72 = 31.4, P < 0.0001; Q7,88 = 25.1, P = 0.0007). Chemical abbreviations: S-ol = (S)-sulcatol, R-ol = (R)-sulcatol, ol = racemic sulcatol, one = sulcatone, F = racemic fuscumol, Fa = racemic fuscumol acetate. Means within experiments and species with different letters are significantly different (REGWQ test, P < 0.05)
Both species were significantly attracted by racemic sulcatol during experiment 2 (Fig. 2b), and attraction was antagonized by fuscumol acetate, despite the presence of the (R)-enantiomer in some headspace extracts from males of A. macula. In experiment 3, no treatment contained sulcatol, and no treatments were significantly different than controls (data not shown), corroborating the lack of attraction to sulcatone seen in experiment 1. In experiment 4, adults of both species were attracted to the blend of racemic sulcatol and sulcatone (Fig. 2c), consistent with experiment 1, and attraction was antagonized by both fuscumol and fuscumol acetate.
Flight Phenology of Adult Beetles
The two species clearly differed in seasonal flight period at the Brownfield Woods and Forest Glen study sites (Fig. 3a), with adults of L. transversus beginning to emerge at least a month earlier, and peaking in numbers before A. macula began to emerge. Of the 178 adults of L. transversus that were captured, ~72% flew before the other species first appeared. Similarly, of 261 adults of A. macula, ~75% flew during the period that trap captures of the other species had reached low numbers. Across the two study sites, of 210 cases in which at least one beetle of either species was captured, only 17 (~8%) contained beetles of both species.
Flight phenology of adult beetles of Astylopsis macula and Leptostylus transversus: a seasonal flight period as determined by capture of beetles during field bioassays conducted during 2016 at the Brownfield Woods (thin lines) and Forest Glen (thick lines) study sites. Numbers of beetles were summed for 10-d intervals between 25 April (ordinal date 115) and 13 August (225; N = 261 adults of A. maculata and 178 adults of L. transversus); b daily flight period as determined with pheromone-baited traps equipped with a timer mechanism that rotated trap jars at intervals of 1-h (N = 8 adults of A. macula, 19 adults of L. transversus)
Timer traps captured only eight adults of A. macula (N = 5 dates) and 19 adults of L. transversus (14 dates), primarily due to inclement weather during the sampling period. Nevertheless, the data suggest that the species differed in the timing of peak activity (Fig. 3b). That is, most adults of A. macula were captured in early evening, suggesting that this species takes flight at dusk, while most adults of L. transversus were captured well after dark. Although the two species appeared to overlap slightly in daily flight period, none of the 19 trap samples contained beetles of both species.
Discussion
Variation among conspecific males of A. macula and L. transversus in whether they produced odors or not during headspace sampling appears to be common in cerambycids, and has been reported for species of lamiines (Meier et al. 2016) and cerambycines (Hanks et al. 2019). These findings suggest that emission of pheromone varies with the physiological condition of the field-captured insects (e.g., related to age, vigor, mating history), which may be further influenced by the unnatural conditions in which they were held in the laboratory. The same earlier studies also had shown that conspecific males vary in the composition of the odors they produce, with inconsistent ratios of components, and even variable blends of components. Other species of beetles show similar variation among males in the amounts of aggregation pheromone they produce and the ratios of pheromone components (e.g., Hodges et al. 2002).
Among cerambycine species that have pheromones composed of multiple components, males most consistently produce those which are inherently attractive, and sporadically produce components that are neither inherently attractive nor do they synergise attraction to other male-produced components (Hanks et al. 2019). The latter components may play a role in segregating species, such as by discouraging attraction of related species to shared pheromone components (Hanks et al. 2019). The pheromone chemistry of A. macula and L. transversus does not appear to be consistent with these patterns. That is, males of both species consistently produced both sulcatone and (S)-sulcatol, if they produced any compounds at all, but only the latter component was inherently attractive. Sulcatone appeared not to influence attraction of A. macula, and its effect on L. transversus remains unclear. Sulcatone may be present in part as a biosynthetic precursor to (S)-sulcatol, because the analogous geranylacetone has been shown to be a precursor to fuscumol and fuscumol acetate in the lamiine species Hedypathes betulinus (Klug) (Zarbin et al. 2013). In contrast, racemic fuscumol acetate, which contained the (R)-fuscumol acetate sporadically produced in trace amounts by males of A. macula, antagonized attraction of both species to (S)-sulcatol.
Sulcatone also has been detected in headspace extracts from males of two species in different tribes of the Cerambycinae, Brothylus gemmulatus LeConte (tribe Hesperophanini) and Phymatodes obliquus Casey (Callidiini; JGM, unpub. data), but its role as a possible pheromone component for these two species has not yet been tested. Both sulcatone and sulcatol are common pheromone components of insects in many orders, including wood-boring beetles of the Platypodidae and the Scolytinae of the Curculionidae (see El-Sayed 2018).
The similar attraction of A. macula and L. transversus to their shared pheromone component (S)-sulcatol raises the question of how these sympatric species maintain reproductive isolation. Although data from the field experiments suggests that the two species overlap to some degree in seasonal flight period, the rarity with which they were actually trapped together is evidence that they nevertheless rarely fly synchronously. When combined with the apparent differences between the species in daily flight period, it appears that interspecific attraction is unlikely.
Earlier field experiments that tested attraction of cerambycids to chemicals of the fuscumol class resulted in captures of sufficient numbers of A. macula for statistical analysis of treatments. For example, experiments conducted in different areas of the eastern USA have demonstrated attraction of this species to (S)-fuscumol alone (Hughes et al. 2016), both racemic fuscumol and fuscumol acetate (Millar et al. 2018), and to the blend of both racemic compounds (Hanks and Millar 2013). It is difficult to account for these responses to treatments in light of the lack of attraction of A. macula to these compounds, and their antagonistic effect on attraction reported in the present article.
Production of traces of (R)-fuscumol acetate, along with sulcatone and (S)-sulcatol, by males of A. macula suggests that pheromones of some lamiines could possibly be comprised of both classes of compounds. Our results clearly indicate that compounds of both classes can be perceived by at least some species, given the attraction to sulcatol and conversely, the antagonistic effects of fuscumol and fuscumol acetate on attraction to sulcatol in both A. macula and L. transversus. The addition of sulcatol and possibly sulcatone to the pheromone repertoire of lamiine species increases the seemingly limited number of components available for producing species-specific pheromones by members of this large cerambycid subfamily.
References
Beutenmiller W (1896) Food-habits of north American Cerambycidae. J N Y Entomol Soc 4:73–81
Cardé RT (2014) Defining attraction and aggregation pheromones: teleological versus functional perspectives. J Chem Ecol 40:519–520
Craighead FC (1923) North American cerambycid larvae: a classification and the biology of north American cerambycid larvae. Canadian Dept Ag Bull 27 – New series (Technical):1–239
El-Sayed AM (2018) The pherobase: database of pheromones and semiochemicals. http://www.pherobase.com. Accessed 7 Dec 2018
Hanks LM, Millar JG (2013) Field bioassays of cerambycid pheromones reveal widespread parsimony of pheromone structures, enhancement by host plant volatiles, and antagonism by components from heterospecifics. Chemoecology 23:21–44
Hanks LM, Millar JG (2016) Sex and aggregation-sex pheromones of cerambycid beetles: basic science and practical applications. J Chem Ecol 42:631–654
Hanks LM, Reagel PF, Mitchell RF, Wong JCH, Meier LR, Silliman CA, Graham EE, Striman BL, Robinson KP, Mongold-Diers JA, Millar JG (2014) Seasonal phenology of the cerambycid beetles of east-Central Illinois. Ann Entomol Soc Am 107:211–226
Hanks LM, Mongold-Diers JA, Mitchell RF, Zou Y, Wong JCH, Meier LR, Johnson TD, Millar JG (2019) The role of minor pheromone components in segregating 14 species of cerambycid beetles of the subfamily Cerambycinae. J Econ Entomol (in press)
Hodges RJ, Birkinshaw LA, Farman DI, Hall DR (2002) Intermale variation in aggregation pheromone release in Prostephanus truncatus. J Chem Ecol 28:1665–1674
Hughes GP, Zou Y, Millar JG, Ginzel MD (2013) (S)-Fuscumol and (S)-fuscumol acetate produced by a male Astyleiopus variegatus (Coleoptera: Cerambycidae). Can Entomol 145:327–332
Hughes GP, Meier LR, Zou Y, Millar JG, Hanks LM, Ginzel MD (2016) Stereochemistry of fuscumol and fuscumol acetate influences attraction of longhorned beetles (Coleoptera: Cerambycidae) of the subfamily Lamiinae. Environ Entomol 45:1271–1275
Lingafelter SW (2007) Illustrated key to the longhorned wood-boring beetles of the eastern United States. Special publication No. 3, Coleopterists Society, North Potomac, pp 206
Linsley EG, Chemsak JA (1995) The Cerambycidae of North America, part VII, no. 2: taxonomy and classification of the subfamily Lamiinae, tribes Acanthocinini through Hemilophini. Univ Calif Pub Entomol 114:1–292
Meier LR, Zou Y, Millar JG, Mongold-Diers JA, Hanks LM (2016) Synergism between enantiomers creates species-specific pheromone blends and minimizes cross-attraction for two species of cerambycid beetles. J Chem Ecol 42:1181–1192
Millar JG, Hanks LM (2017) Chemical ecology of cerambycids. In: Wang Q (ed) Cerambycidae of the world: biology and pest management. CRC Press/Taylor & Francis, Boca Raton, pp 161–208
Millar JG, Hanks LM, Moreira JA, Barbour JD, Lacey ES (2009) Pheromone chemistry of cerambycid beetles. In: Nakamuta K, Millar JG (eds) Chemical ecology of wood-boring insects. Forestry and Forest Products Research Institute, Ibaraki, pp 52–79
Millar JG, Mitchell RF, Mongold-Diers JA, Zou Y, Bográn CE, Fierke MK, Ginzel MD, Johnson CW, Meeker JR, Poland TM, Ragenovich IR, Hanks LM (2018) Identifying possible pheromones of cerambycid beetles by field testing known pheromone components in four widely separated regions of the United States. J Econ Entomol 111:252–259
Mitchell RF, Graham EE, Wong JCH, Reagel PF, Striman BL, Hughes GP, Paschen MA, Ginzel MD, Millar JG, Hanks LM (2011) Fuscumol and fuscumol acetate are general attractants for many species of cerambycid beetles in the subfamily Lamiinae. Entomol Exp Appl 141:71–77
Mitchell RF, Reagel PF, Wong JCH, Meier LR, Silva WD, Mongold-Diers J, Millar JG, Hanks LM (2015) Cerambycid beetle species with similar pheromones are segregated by phenology and minor pheromone components. J Chem Ecol 41:431–440
SAS Institute (2011) SAS/STAT 9.3 user's guide. SAS Institute Inc., Cary
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. WH Freeman and Co., New York
Sweeney J, Silk PJ, Gutowski JM, Wu J, Lemay MA, Mayo PD, Magee DI (2010) Effect of chirality, release rate, and host volatiles on response of Tetropium fuscum (F.), Tetropium cinnamopterum Kirby, and Tetropium castaneum (L.) to the aggregation pheromone, fuscumol. J Chem Ecol 36:1309–1321
Vidal DM, Fonseca MG, Zarbin PH (2010) Enantioselective synthesis and absolute configuration of the sex pheromone of Hedypathes betulinus (Coleoptera: Cerambycidae). Tetrahedron Lett 51:6704–6706
Wong JCH, Meier LR, Zou Y, Mongold-Diers JA, Hanks LM (2017) Evaluation of methods used in testing attraction of cerambycid beetles to pheromone-baited traps. J Econ Entomol 110:2269–2274
Zarbin PH, Fonseca MG, Szczerbowski D, Oliveira AR (2013) Biosynthesis and site of production of sex pheromone components of the cerambycid beetle, Hedypathes betulinus. J Chem Ecol 39:358–363
Acknowledgements
We thank Suzanne Vachula and Alexander Hazel for assistance with field work. For access to field sites, we thank S. Buck and the University of Illinois Committee on Natural Areas and the Vermilion County Conservation District. This research was supported by grants from The Alphawood Foundation of Chicago to LMH, and grants from USDA National Institute of Food and Agriculture (numbers 2012-67013-19303 and 2015-67013-23173) and USDA-APHIS (grant number 15-8130-1422-CA) to LMH and JGM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meier, L.R., Millar, J.G., Mongold-Diers, J.A. et al. (S)-Sulcatol Is a Pheromone Component for Two Species of Cerambycid Beetles in the Subfamily Lamiinae. J Chem Ecol 45, 447–454 (2019). https://doi.org/10.1007/s10886-019-01071-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01071-7