Abstract
Cruciferous plants (Brassicaceae) are characterized by the accumulation of a group of secondary metabolites known as glucosinolates that, following attack by pathogens or herbivores, may be hydrolyzed to one of a number of products including isothiocyanates and nitriles. Despite the range of hydrolysis products that may be produced, the toxicity of glucosinolates to pathogens and herbivores may be explained largely by the production of isothiocyanates. Isothiocyanates are also known to provide an indirect defense by acting as host finding cues for parasitoids of insect herbivores that attack crucifers. It has been speculated that nitriles may provide a similar indirect defense. Here, we investigate the olfactory perception and orientation behavior of the aphid parasitoid Diaeretiella rapae, to a range of alkenylglucosinolate hydrolysis products, including isothiocyanates, nitriles, and epithionitriles. Electroantennogram responses indicated peripheral odor perception in D. rapae females to all 3-butenylglucosinolate hydrolysis products tested. By contrast, of the 2-propenylglucosinolate hydrolysis products tested, only the isothiocyanate elicited significant responses. Despite showing peripheral olfactory detection of a range of 3-butenylglucosinolate hydrolysis products, naïve females oriented only to the isothiocyanate. Similarly, parasitoids oriented to 3-isothiocyanatoprop-1-ene, but not to the corresponding nitrile or epithionitrile. However, by rearing D. rapae either on Brassica nigra, characterized by the accumulation of 2-propenylglucosinolate, or Brassica rapa var rapifera, characterized by the accumulation of 3-butenylglucosinolate, altered the innate response of parasitoids to 3-isothiocyanatoprop-1-ene and 4-isothiocyanatobut-1-ene. These results are discussed in relation to the defensive roles of glucosinolate hydrolysis products and the influence of the host plant on aphid parasitoid behavior.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Secondary metabolism plays a role in a plant’s survival in the environment, providing a defense against insect herbivores and plant pathogens, while also aiding reproduction through the attraction of pollinators (Kliebenstein 2004). In cruciferous plants (Brassicaceae), glucosinolates, a group of amino acid-derived compounds that consist of a β-thioglucose moiety, a sulfonated oxime, and a variable side chain, are characteristically accumulated as the major secondary metabolite. Together with endogenous myrosinase (β-thioglucoside glucohydrolase, 3.2.1.147) glucosinolates serve a central role in defense against herbivores and pathogens (Wink 1988; Jander et al. 2001; Kliebenstein et al. 2005a, b). Tissue damage brings together myrosinase and glucosinolates, which are otherwise spatially separated (Kelly et al. 1998; Koroleva et al. 2000) yielding a variety of hydrolysis products such as isothiocyanates, epithionitriles, thiocyanates, and nitriles (Bones and Rossiter 1996, 2006; Halkier and Gershenzon 2006).
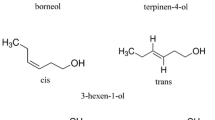
The final outcome of myrosinase-catalyzed hydrolysis of glucosinolates is determined by the epithiospecifier protein (ESP) and related proteins. The most common products are isothiocyanates, which are formed after myrosinase-catalyzed glucosinolate hydrolysis in the absence of ESP, by a spontaneous ‘Lossen’ like rearrangement (Bones and Rossiter 2006). By contrast, alkenylglucosinolates are converted to thiiranylalkylnitriles, commonly known as epithionitriles, and nitriles when ESP is present together with ferrous/ferric ions, while non-alkenylglucosinolates and indoleglucosinolates form simple nitriles. While epithionitrile formation is dependent on the presence of both ESP and iron, formation of nitriles may occur in the absence of ESP if iron is present (Zabala et al. 2005). Figure 1 shows the structures of the alkenylglucosinolate-derived isothiocyanates, epithionitriles, and nitriles. Despite this range of hydrolysis products, the toxicity of glucosinolates to pathogens and herbivores can be attributed mainly to the isothiocyanates and compounds derived from them (Halkier and Gershenzon 2006). Indeed, in studies of insect herbivory, Arabidopsis thaliana (L.) plants that express ESP and, therefore produce nitriles, were apparently less well defended against two species of generalist insect herbivore compared with plants that produced isothiocyanates (Lambrix et al. 2001; Burow et al. 2006). However, as short chain nitriles are volatile compounds, it has been speculated that they may serve as signals in indirect defense responses (Wittstock and Burow 2007). By contrast, little is known about the function of epithionitriles, as few bioassays have been carried out with epithionitriles derived from glucosinolates. This is, in part, due to their perceived instability, but also because they require either chemical synthesis or preparative isolation.
The braconid wasp, Diaeretiella rapae (McIntosh), is a solitary endoparasitoid that characteristically attacks aphids that feed on crucifers, including crucifer specialists Brevicoryne brassicae (L.) and Lipaphis pseudobrassicae (= L. erysimi) (Kaltenbach). In several olfactometry studies, female D. rapae discriminated between aphid infested and uninfested crucifers of agricultural importance (Read et al. 1970; Reed et al. 1995; Blande et al. 2007), as well as the model crucifer, A. thaliana (Girling et al. 2006). Air entrainments completed by Blande (2004) confirmed that higher levels of isothiocyanates were produced by aphid infested turnip plants compared with uninfested plants. In addition, isothiocyanates also have been indicated as signaling chemicals by a number of olfactometry studies in which female D. rapae orient toward pure compounds, which were presented in place of aphid-infested plants (Read et al. 1970; Vaughn et al. 1996; Blande et al. 2007).
A number of studies demonstrated that host finding behavior of some species of aphid parasitoids may be determined by the plant on which the insect develops (Wickremasinghe and van Emden 1992; Storeck et al. 2000). From these studies, it is suggested that conditioning of the parasitoid occurs as it chews its way out of the aphid mummy. Blande et al. (2007) similarly suggest that D. rapae acquires chemical cues, probably glucosinolates, as it emerges from the mummy case, although it was proposed that these cues play a role in host recognition rather than host-finding behavior. In an olfactometry study by Bradburne and Mithen (2000), experienced D. rapae oriented into the air stream containing the odor from ground plant material of one of two near-isogenic Brassica oleracea L. lines, one of which primarily produced 3-isothiocyanatoprop-1-ene (3-PROP-ITC) and the other 4-isothiocyanatobut-1-ene, (4-BUT-ITC) when presented opposite a blank control. However, when the two lines were presented opposite each other, significantly more parasitoids oriented to the air stream containing the odor of the 3-butenylglucosinolate-producing line. This response was recorded regardless of the line on which the insects were reared and, therefore, suggests an innate preference for 4-BUT-ITC over 3-PROP-ITC, although the role of other plant volatiles in this study cannot be excluded.
Given that the chemical functionalities of the epithionitriles, isothiocyanates, and nitriles are different, it might be expected that the electrophysiological and behavioral responses of D. rapae would be affected. Thus, we used naïve D. rapae females to test their responses to a range of 2-propenyl and 3-butenylglucosinolate hydrolysis products.
Methods and Materials
Insects
Two cultures of the crucifer specialist aphid B. brassicae (L.) were maintained at 18°C, with a 16/8 hr, L/D photoperiod. Aphids were cultured either on individual 4 wk-old B. nigra (L.) plants or similar age B. rapa L. var rapifera plants, each plant being enclosed within a perforated bread bag. Aphids were transferred to fresh plants to establish new colonies every 1–2 wk.
The aphid parasitoid D. rapae (obtained from Rothamsted Research) was maintained at 21°C and 18°C (16:8 hr, L/D). Standardized cohorts were produced by allowing mated 2- to 3-d-old adults to parasitize mixed-age B. brassicae in a Petri dish for approximately 2 hr. Parasitized aphids were transferred to a fresh B. nigra or B. rapa var rapifera plant on which they continued to feed. Aphid mummies were removed approximately 8 d later. Adult parasitoids were provided with a dilute honey solution on emergence.
Synthesis of Isothiocyanates and Epithionitriles
Hydrolysis products derived from 2-propenylglucosinolate and 3-butenylglucosinolate were prepared for use in this study. For 2-propenylglucosinolate, 3-isothiocyanatoprop-1-ene (3-PROP-ITC) and but-3-enenitrile (3-BUT-NIT) were purchased from Sigma-Aldrich (>95% purity), while 2-(thiiran-2-yl)acetonitrile (2-ACETO-ETN) was synthesized according to the method of Luthy and Benn (1979). For 3-butenylglucosinolate, 4-isothiocyanatobut-1-ene (4-BUT-ITC) was purchased from Sigma-Aldrich (>95% purity), pent-4-enenitrile (4-PENT-NIT) was synthesized from 3-butenylbromide and sodium cyanide, and purified by distillation and 3-(thiiran-2-yl)propanenitrile (3-PROP-ETN) again synthesized according to the method of Luthy and Benn (1979).
Electroantennogram (EAG) Recordings
To prepare D. rapae females for EAG recordings, insects were anaesthetized with CO2 before the head was separated from the thorax and the distal portion of the last antennal segment of one antenna removed. Glass electrodes (borosilicate glass capillary, 1.5 mm O.D. × 1.17 mm I.D. with filament; Clark Electromedical Instruments®, UK) were filled with 0.1 M KCl. The base of the head was mounted on the reference electrode, while the cut end of the antenna was inserted a short way into the recording electrode. Chlorided-silver wire connected the recording electrode to a probe with an internal amplifier (Syntech®, the Netherlands). Responses were recorded by using a PC fitted with an IDAC interface board (Syntech®, the Netherlands) and running EAG software (Syntech®, the Netherlands).
Test Compounds and Stimulation
Each compound tested was dissolved in paraffin liquid (Fisher Scientific, UK, density 0.83–0.86 g/ml) to make a 1% (v/v) solution. Twenty-five microliters of each solution were applied to a piece (8 × 60 mm) of filter paper (Whatman®, No. 2 ashless) immediately before each insect preparation was tested. The filter paper was inserted into a glass Pasteur pipette. The same amount of paraffin liquid was used as a blank control. The tip of the glass Pasteur pipette was inserted about 3 mm into a small hole of a glass tube (9 mm diam., 120 mm long) directed at the antennal preparation. An air stimulus controller (model CS-05, Syntech®, the Netherlands) was used for air and odor delivery. A constant flow (1.8 l/min) of charcoal-filtered and humidified air passed over the antenna through the open end of the glass tube, positioned 15 mm from the antenna. During odor stimulation, 60 ml/min of air was applied through the Pasteur pipette into the main air flow for 2 sec, with a 1-min interval between stimulations. In each case, EAG peak amplitude in response to a compound was determined and then normalized against the mean of two responses to (E)-2-hexenal (the standard stimulus, purchased from Sigma-Aldrich) presented before and after the test stimulus.
The EAG responses to 2-propenyl- and 3-butenylglucosinolate hydrolysis products were recorded from naïve female D. rapae. Eight insect preparations (replicates) were tested against each of the compounds, which were presented in random order. Differences between mean EAG responses to each compound were assessed by Fisher’s least significant difference tests (LSDs). Additional analysis of recovery rates of antennal receptors after maximum depolarization was completed for each compound tested by using the method of Baker & Roelofs (1976).
Olfactometry
A glass Y-tube olfactometer, of the design previously described by Du et al. (1996), was used to record behavioral responses of female D. rapae to pure compounds (glucosinolate hydrolysis products). The olfactometer had 12 mm internal diam, 100 mm stem, and 100 mm arms at 60° angle. Air was pumped through Teflon tubing by a Dymax30 pump (Charles Austen Pumps Ltd., Byfleet, Surrey, UK) through an activated charcoal filter before being regulated by a flowmeter to 800 ml/min. The airflow was split by a brass T-junction (Swagelok, OH, USA), each flow of 400 ml/min then passing into an airtight glass chamber (T. J. Adams—Artistic & Scientific Glassblower, Kidlington, Oxfordshire, UK) into which the volatile source was placed. From the two glass chambers, air flowed through additional Teflon tubing into the arms of the olfactometer via modified glass quick fit sockets. A white cardboard screen was placed around the olfactometer to exclude visual cues, and diffused lighting was provided by a fluorescent strip held 300 mm above and 100 mm in front of the branches of the olfactometer. A single female was introduced into the stem of the olfactometer and then given 5 min to make a choice. During this period, if the parasitoid failed to move more than 50 mm up the stem of the olfactometer, it was excluded from the experiment. Insects were recorded as having selected an odor if they moved more than 50 mm up one of the arms and remained beyond this point for more than 30 sec. Insects that moved more than 50 mm up the stem but did not select an arm of the olfactometer were recorded as not having selected an odor. After each individual was tested, the position of the odor sources was swapped in order to account for any directional bias by the parasitoids. These criteria are similar to those previously described (Girling et al. 2006; Blande et al. 2007). Between bioassay sets, glassware was washed with acetone and distilled water and then baked overnight at 200°C.
Pure compounds were prepared as 1 mg/ml solutions in diethyl ether. Test solutions were presented in 50-µl aliquots applied to a piece of filter paper (Whatman No. 1), which was changed after every fifth individual. Naïve female parasitoids were used in bioassays. Aphid mummies were removed from plants so that on emergence parasitoids were not provided with cues that might influence behavioral responses. Adult male and female D. rapae were kept together, and mating was assumed. Parasitoids were provided with a dilute honey solution, and females were used in the bioassays approximately 72 hr after emergence.
The following olfactometry experiments were completed with D. rapae females. In each case, responses of 30 naïve wasps were recorded:
-
1.
Responses of naïve D. rapae females to (a) 2-propenylglucosinolate hydrolysis products, and (b) 3-butenylglucosinolate hydrolysis products. Naïve D. rapae were reared on B. brassicae, which in turn were reared on B. nigra.
-
2.
Responses of naïve D. rapae females to 3-PROP-ITC and 4-BUT-ITC when parasitoids were reared on B. brassicae, which in turn were reared on (a) B. nigra or on (b) B. rapa var rapifera.
Results
EAG Responses
EAG responses of D. rapae females to 2-propenyl- and 3-butenylglucosinolate hydrolysis products were recorded (Fig. 2). Responses are presented normalized against a standard stimulus, (E)-2-hexenal. However, mean absolute responses recorded to isothiocyanates, 3-PROP-ITC and 4-BUT-ITC, were 0.53 and 0.57 mV, respectively.
EAG responses of Diaeretiella rapae females to 2-propenyl- and 3-butenylglucosinolate hydrolysis products (mean ± SE, N = 8). Stimuli: 1 = 3-isothiocyanatoprop-1-ene; 2 = but-3-enenitrile; 3 = 2-(thiiran-2-yl)acetonitrile; 4 = 4-isothiocyanatobut-1-ene; 5 = pent-4-enenitrile; 6 = 3-(thiiran-2-yl)propanenitrile. Different letters indicate significant differences (Fisher’s LSD, P < 0.05)
Diaeretiella rapae females responded significantly to all 3-butenylglucosinolate hydrolysis compounds tested, with normalized EAG responses 3.2, 2.0, and 1.9 times larger than that of the paraffin control for the isothiocyanate, nitrile, and epithionitrile, respectively (Fig. 2). By contrast, when compounds derived from 2-propenylglucosinolate were tested, females responded significantly only to 3-PROP-ITC, with a normalized EAG response 2.7 times larger than that of the paraffin control. Similarly, EAG responses to the 3-butenylglucosinolate derived 4-PENT-NIT and 3-PROP-ETN were significantly larger than responses to the corresponding 2-propenylglucosinolate derived nitrile and epithionitrile. There was no significant difference in EAG response size to 4-BUT-ITC and 3-PROP-ITC. Although D. rapae responded significantly to all 3-butenylglucosinolate hydrolysis products, the EAG response to 4-BUT-ITC was significantly larger than to the nitrile or to the epithionitrile. EAG response size was, however, similar for 4-PENT-NIT compared with 3-PROP-ETN (Fig. 2).
Receptor recovery rates were affected by the compound with which they were stimulated (F = 2.64, P = 0.02). Individual contrasts revealed that the recovery rate following stimulation by (E)-2-hexenal (x = 0.27 ± 0.06) was faster, between 1.7 and 2.9 times, than recovery rates following stimulation by the other compounds tested. Recovery rates following stimulation by the glucosinolate hydrolysis products were similar.
Olfactometry
Naïve D. rapae females reared on B. brassicae in turn feeding on B. nigra were presented with 3-butenylglucosinolate hydrolysis products. When each compound was presented opposite a solvent control (diethyl ether), more parasitoids oriented toward 4-BUT-ITC (χ 2 = 4.17, P < 0.05), but the insects did not respond to either the corresponding nitrile or epithionitrile (Fig. 3a). In comparisons between these compounds, more females selected the arm containing 4-BUT-ITC than either the nitrile or epithionitrile. However, in both cases differences were not significant. The comparison between the 3-butenylglucosinolate derived 4-PENT-NIT and 3-PROP-ETN was also not significant. A second experiment was completed in which similarly reared naïve D. rapae females were presented with a range of 2-propenylglucosinolate hydrolysis products. More parasitoids responded to the isothiocyanate, in this case 3-PROP-ITC (χ 2 = 4.48, P < 0.05) when presented opposite a solvent control (Fig. 3b). In contrast, although more D. rapae females oriented toward 3-BUT-NIT compared to the control, this difference was not significant. Similar numbers of insects moved toward 2-ACETO-ETN and the solvent control. From comparisons between 2-propenylglucosinolate derived hydrolysis products, more D. rapae responded to 3-PROP-ITC compared with either the corresponding nitrile (χ 2 = 6.00, P < 0.05) or epithionitrile (χ 2 = 5.54, P < 0.05). Similar numbers of insects moved toward 3-BUT-NIT and 2-ACETO-ETN when they were presented opposite each other.
Responses of naïve Diaeretiella rapae females in a Y-tube olfactometer to (a) 3-butenylglucosinolate hydrolysis products, where 4-BUT-ITC = 4-isothiocyanatobut-1-ene; 4-PENT-NIT = pent-4-enenitrile; 3-PROP-ETN = 3-(thiiran-2-yl)propanenitrile and (b) 2-propenylglucosinolate hydrolysis products, where 3-PROP-ITC = 3-isothiocyanatoprop-1-ene; 3-BUT-NIT = but-3-enenitrile; 2-ACETO-ETN = 2-(thiiran-2-yl)acetonitrile. A total of N = 30 naïve D. rapae females were tested for each comparison
Naïve D. rapae females reared on B. brassicae, which in turn were feeding on B. nigra, were presented with 4-BUT-ITC or 3-PROP-ITC. Each compound was presented opposite a solvent control and also opposite each other. More parasitoids moved into the air stream that contained the 4-BUT-ITC (χ 2 = 9.85, P < 0.01) or 3-PROP-ITC (χ 2 = 7.76, P < 0.05) compared with the control (Fig. 4a). When 4-BUT-ITC was presented opposite 3-PROP-ITC, more D. rapae responded to 3-PROP-ITC (χ 2 = 4.48, P < 0.05). A second similarly designed experiment was completed that used naïve D. rapae females reared on B. brassicae, which in turn were feeding on B. rapa var rapifera. Again, more parasitoids moved into the air stream containing the 4-BUT-ITC (χ 2 = 5.14, P < 0.05) or 3-PROP-ITC (χ 2 = 10.80, P < 0.01) compared with the control (Fig. 4b). However, when 4-BUT-ITC was presented opposite 3-PROP-ITC similar numbers of insects responded to each compound.
Responses of naïve Diaeretiella rapae females to 3-PROP-ITC = 3-isothiocyanatoprop-1-ene and 4-BUT-ITC = 4-isothiocyanatobut-1-ene, having previously been reared on Brevicoryne brassicae feeding either on (a) Brassica nigra or (b) Brassica rapa var rapifera. A total of N = 30 naïve D. rapae females were tested for each comparison
In each of these experiments, the number of insects not responding was low. In the majority of cases, less than 10% of parasitoids did not respond with a maximum of 20%.
Discussion
Research that has investigated the importance of the myrosinase–glucosinolate defense against insect herbivores has so far largely focused on the role of isothiocyanates (Halkier and Gershenzon 2006; Wittstock and Burow 2007). Many studies have investigated the direct effects of isothiocyanates against herbivores, while others have focused on indirect effects of isothiocyanates that act as cues for parasitoids, including D. rapae. However, little is currently known about the peripheral odor perception of D. rapae to glucosinolate hydrolysis products. Indeed, D. rapae has previously been tested only against, and shown to respond to, 3-PROP-ITC (Vaughn et al. 1996).
Here, we confirmed this earlier result while also recording EAGs from naïve D. rapae females in response to 3-butenyl- and 2-propenylglucosinolate hydrolysis products, including isothiocyanates and (for the first time) nitriles and epithionitriles. Although significant EAG responses were recorded to 3-PROP-ITC, parasitoids did not respond to the corresponding nitrile or epithionitrile, reflecting either the lower volatility of these compounds or a lack of olfactory receptors. By contrast, D. rapae females showed peripheral odor perception to all of the 3-butenylglucosinolate hydrolysis products tested, including 4-BUT-ITC, 4-PENT-NIT, and 3-PROP-ETN, although again the largest response was to the isothiocyanate. Large EAG responses were also recorded in response to (E)-2-hexenal, and D. rapae females have previously been shown to respond to another green leaf volatile, (Z)-3-hexenol (Vaughn et al. 1996).
Vaughn et al. (1996) noted that although D. rapae females responded to both (Z)-3-hexenol and to 3-PROP-ITC; there were clear differences in the recovery rates following stimulation by each compound, suggesting possible differences in receptor type. Similarly, in the present study, antennal response recovery rates were significantly faster for (E)-2-hexenal than to the alkenylglucosinolate hydrolysis products tested. This result further supports the idea that, like its aphid hosts, L. pseudobrassicae and B. brassicae, female D. rapae possess more than one receptor type (Dawson et al. 1987; Nottingham et al. 1991). Slower recovery rates previously have been attributed to chemical-specific receptors (Baker and Roelofs 1976). Results presented here suggest that D. rapae females possess receptors specific for isothiocyanates, as previously identified by (Vaughn et al. 1996), and in addition for certain nitriles and epithionitriles. The physico-chemical properties of the isothiocyanates are quite different from that of the nitrile group, being more polar and electrophilic and with more potential for interaction with proteins. The thiirane ring of the epithionitriles, while less polar than that of the isothiocyanate group, is also electrophilic, and has the potential for ring opening with biological nucleophiles to form conjugates. Although no specific work has been carried out on isothiocyanate receptors in insects, a recent report describes the effect of mustard oil (3-PROP-ITC) on endogenous porcine transient receptor potential V1 (pTRPV1) channels (Ohta et al. 2007). It was found that 3-PROP-ITC acts as an agonist for porcine TRPV1. Other notable receptors of this type are the vanilloid and capsaicin receptors present in Drosophila that have been well characterized (Caterina et al. 1997, 1999). Another class of receptors is the odorant receptors (OR), which are undergoing characterization in Drosophila and are potential candidates for interactions with glucosinolate hydrolysis products (Benton 2006). It would be of interest to explore the action of glucosinolate-derived isothiocyanates as chemical cues and repellents at a molecular level in order to understand at the physiological level the behavior of insects.
There is considerable evidence that isothiocyanates function as signals in indirect plant defense responses, thus enhancing attraction of beneficial parasitoids (Read et al. 1970; Vaughn et al. 1996; Bradburne and Mithen 2000; Blande et al. 2007), and it has been speculated that simple nitriles may serve also as similar signals induced by herbivory (Wittstock and Burow 2007). Indeed, female Cotesia rubecula Marshall, a specialist parasitoid of Pieris rapae L. caterpillars, may use nitriles to discriminate between infested and uninfested A. thaliana plants (van Poecke et al. 2001). Air entrainments of these plants implicated two nitriles, 5-(methylthio) pentanenitrile and 6,7-dithiooctanenitrile, along with the monoterpene myrcene and higher levels of methyl salicylate, as potential cues used by this parasitoid. In the present study, there was a lack of orientation behavior to the 2-propenylglucosinolate-derived nitrile and epithionitrile, which supports the apparent lack of peripheral perception of these compounds indicated by the EAG results. By contrast, although EAG results indicated peripheral odor perception to all of the 3-butenylglucosinolate hydrolysis products tested, olfactometry results indicated only a significant behavioral response to 4-BUT-ITC. However, when 4-BUT-ITC was presented opposite either the corresponding nitrile or epithionitrile, there was no significant difference in the numbers of D. rapae orienting to each compound. This result suggests either a weak response to the 3-butenylglucosinolate-derived 4-PENT-NIT and 3-PROP-ETN or that these compounds in some way disrupt the response to 4-BUT-ITC. A similar result was recorded by Blande et al. (2007) who noted that there was no difference in the number of D. rapae females orienting to 4-BUT-ITC and 5-isothiocyanatopent-1-ene when the compounds were presented opposite each other. This result was recorded despite the fact that when each compound was presented opposite a solvent control, parasitoids oriented toward the 4-BUT-ITC but not toward the 5-isothiocyanatopent-1-ene.
The results from this study provide no evidence for naïve D. rapae females responding behaviorally to either the nitriles or epithionitriles tested. However, subtle effects such as potential interactions between compounds cannot be excluded. Indeed, the nitriles implicated as host-finding cues utilized by C. rubecula were present against a background of plant odors (van Poecke et al. 2001). In contrast with the lack of behavioral response to either the nitriles or epithionitriles, results from the olfactometry experiment indicated that naïve D. rapae females did orient toward both 3-PROP-ITC and 4-BUT-ITC. This result supports a number of earlier studies in suggesting that these two isothiocyanates provide an indirect defense by acting as host finding cues for D. rapae, a natural enemy of aphids feeding on crucifers (Read et al. 1970; Vaughn et al. 1996; Bradburne and Mithen 2000; Blande et al. 2007). In the present study, naïve D. rapae females responded positively to both 3-PROP-ITC and 4-BUT-ITC, whether the parasitoids were reared on B. nigra or B. rapa var rapifera. This is despite the fact that B. nigra is characterized by the accumulation of 2-propenylglucosinolate (Cole 1976), while B. rapa var rapifera accumulates 3-butenylglucosinolate (Carlson et al. 1981). In both cases, the glucosinolates are hydrolyzed to the corresponding isothiocyanates (unpublished data).
When the two compounds were presented opposite each other in the olfactometer, the responses of naïve D. rapae females were, however, affected by the host plant on which the insects were reared. When reared on B. nigra, more parasitoids oriented toward 3-PROP-ITC, but when reared on B. rapa var rapifera there was no difference in the numbers of females responding to the two isothiocyanates. This result contrasts with that of Bradburne and Mithen (2000) who also used a Y-tube olfactometer to record D. rapae behavior but utilized two near-isogenic B. oleracea lines. Hydrolysis analysis, rather than air entrainments of aphid-infested plants such as that used by Blande et al. (2007), was used to confirm that one of these isogenic lines produced predominantly 4-BUT-ITC and the other mainly 3-PROP-ITC. Bradburne and Mithen (2000) recorded more D. rapae orienting toward the 4-BUT-ITC line as opposed to the 3-PROP-ITC producing line, regardless of the line on which the insects were reared. It is unclear why these two studies produced contrasting results, although this may reflect differences in the way that the parasitoids interpret individual volatiles as opposed to a bouquet of different compounds. This point is particularly relevant, as Bradburne and Mithen (2000) presented hydrated ground plant material, which is likely to have presented the insects with many volatile compounds that may or may not be associated with aphid infested plants. In contrast, here, individual compounds were presented as cues in the olfactometry experiments.
Host plant chemistry plays a role in both host-finding and host recognition in other species of aphid parasitoids (Wickremasinghe and van Emden 1992; Storeck et al. 2000). The cereal aphid specialist Aphidius rhopalosiphi De Stefani Perez discriminated between different cultivated varieties of winter wheat in a Y-tube olfactometer, but showed a preference for the cultivar on which it was reared (Wickremasinghe and van Emden 1992). Similarly, the generalist aphid parasitoid Aphidius colemani Viereck also showed a preference for the host-plant on which it was reared (Storeck et al. 2000). In both cases, this preference was associated with chemical cues present on the mummy case, which the adult parasitoid comes into contact with at the time of emergence.
Interestingly, Blande et al. (2007) also suggest that D. rapae encounters chemical cues, probably glucosinolates, as the wasp emerges from the aphid mummy. Indeed, when naïve D. rapae females were presented with B. rapa var rapifera plants infested either with L. pseudobrassicae (which characteristically accumulates glucosinolates) or Myzus persicae (Sulzer) (not known to accumulate glucosinolates), parasitoids attacked a higher proportion of the L. pseudobrassicae regardless of the aphid species on which the wasps were reared (Blande et al. 2004). However, subsequent olfactometry experiments, again using experienced D. rapae reared on L. pseudobrassicae or M. persicae feeding on B. rapa var rapifera, found no evidence for parasitoids preferentially orienting toward either plant/aphid complex (Blande et al. 2007). From these results, the authors suggest that glucosinolate cues accumulated as the parasitoid emerges from the aphid mummy are important in host acceptance but that similar cues appear to be utilized for locating L. pseudobrassicae or M. persicae feeding on B. rapa var rapifera. Here, by using one species of aphid and altering the species of host plant rather than, in the case of Blande et al. (2007), using two aphid species reared on the same species of host plant, we recorded changes in orientation behavior. This result raises interesting questions about the type of chemical cue that may be acquired as the wasp emerges from the aphid mummy and in particular whether these results are explained by non-volatile glucosinolates and/or the volatile isothiocyanates.
References
Baker, T. C., and Roelofs, W. L. 1976. Electroantennogram responses of the male moth, Argyrotaenia velutinana to mixtures of sex pheromone components of the female. J. Insect Physiol. 22:1357–1364.
Benton, R. 2006. On the origin of smell: odorant receptors in insects. Cell. Mol. Life Sci. 63:1579–1585.
Blande, J. D. 2004. Differential signaling from specialist and generalist Brassica feeding aphids to differentially adapted aphid parasitoids. Ph.D. thesis, University of Southampton, UK.
Blande, J. D., Pickett, J. A., and Poppy, G. M. 2004. Attack rate and success of the parasitoid Diaeretiella rapae on specialist and generalist feeding aphids. J. Chem. Ecol. 30:1781–1795.
Blande, J. D., Pickett, J. A., and Poppy, G. M. 2007. A comparison of semiochemically mediated interactions involving specialist and generalist Brassica-feeding aphids and the braconid parasitoid Diaeretiella rapae. J. Chem. Ecol. 33:767–779.
Bones, A. M., and Rossiter, J. T. 1996. The myrosinase-glucosinolate system, its organisation and biochemistry. Physiol. Plant. 97:194–208.
Bones, A. M., and Rossiter, J. T. 2006. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 67:1053–1067.
Bradburne, R. P., and Mithen, R. 2000. Glucosinolate genetics and the attraction of the aphid parasitoid Diaeretiella rapae to Brassica. Proc. R. Soc. Lond., Ser. B Biol. Sci. 267:89–95.
Burow, M., Muller, R., Gershenzon, J., and Wittstock, U. 2006. Altered glucosinolate hydrolysis in genetically engineered Arabidopsis thaliana and its influence on the larval development of Spodoptera littoralis. J. Chem. Ecol. 32:2333–2349.
Carlson, D. G., Daxenbichler, M. E., Vanetten, C. H., and Tookey, H. L. 1981. Glucosinolates in crucifer vegetables—turnips and rutabagas. J. Agric. Food Chem. 29:1235–1239.
Caterina, M. J., Schumacher, M. A., Tominaga, M., Rosen, T. A., Levine, J. D., and Julius, D. 1997. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824.
Caterina, M. J., Rosen, T. A., Tominaga, M., Brake, A. J., and Julius, D. 1999. A capsaicin-receptor homologue with a high threshold for noxious heat. Nature 398:436–441.
Cole, R. 1976. Isothiocyanates, nitriles and thiocyanates as products of autolysis of glucosinolates in Cruciferae. Phytochemistry 15:759–762.
Dawson, G. W., Griffiths, D. C., Pickett, J. A., Wadhams, L. J., and Woodcock, C. M. 1987. Plant-derived synergists of alarm pheromone from turnip aphid, Lipaphis (Hyadaphis) erysimi (Homoptera, Aphididae). J. Chem. Ecol. 13:1663–1671.
Du, Y. J., Poppy, G. M., and Powell, W. 1996. Relative importance of semiochemicals from first and second trophic levels in host foraging behavior of Aphidius ervi. J. Chem. Ecol. 22:1591–1605.
Girling, R. D., Hassall, M., Turner, J. G., and Poppy, G. M. 2006. Behavioural responses of the aphid parasitoid Diaeretiella rapae to volatiles from Arabidopsis thaliana induced by Myzus persicae. Entomol. Exp. Appl. 120:1–9.
Halkier, B. A., and Gershenzon, J. 2006. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 57:303–333.
Jander, G., Cui, J. P., Nhan, B., Pierce, N. E., and Ausubel, F. M. 2001. The TASTY locus on chromosome 1 of Arabidopsis affects feeding of the insect herbivore Trichoplusia ni. Plant Physiol. 126:890–898.
Kelly, P. J., Bones, A., and Rossiter, J. T. 1998. Sub-cellular immunolocalization of the glucosinolate sinigrin in seedlings of Brassica juncea. Planta 206:370–377.
Kliebenstein, D. J. 2004. Secondary metabolites and plant/environment interactions: a view through Arabidopsis thaliana tinged glasses. Plant Cell Environ. 27:675–684.
Kliebenstein, D. J., Kroymann, J., and Mitchell-Olds, T. 2005a. The glucosinolate-myrosinase system in an ecological and evolutionary context. Curr. Opin. Plant Biol. 8:264–271.
Kliebenstein, D. J., Rowe, H. C., and Denby, K. J. 2005b. Secondary metabolites influence Arabidopsis/Botrytis interactions: variation in host production and pathogen sensitivity. Plant J. 44:25–36.
Koroleva, O. A., Davies, A., Deeken, R., Thorpe, M. R., Tomos, A. D., and Hedrich, R. 2000. Identification of a new glucosinolate-rich cell type in Arabidopsis flower stalk. Plant Physiol. 124:599–608.
Lambrix, V., Reichelt, M., Mitchell-Olds, T., Kliebenstein, D. J., and Gershenzon, J. 2001. The Arabidopsis epithiospecifier protein promotes the hydrolysis of glucosinolates to nitriles and influences Trichoplusia ni herbivory. Plant Cell 13:2793–2807.
Luthy, J., and Benn, M. 1979. Conversion of potassium allylglucosinolate to 3,4-epithiobutanenitrile by crambe-abyssinica seed flour. Phytochemistry 18:2028–2029.
Nottingham, S. F., Hardie, J., Dawson, G. W., Hick, A. J., Pickett, J. A., Wadhams, L. J., and Woodcock, C. M. 1991. Behavioral and electrophysiological responses of aphids to host and nonhost plant volatiles. J. Chem. Ecol. 17:1231–1242.
Ohta, T., Imagawa, T., and Ito, S. 2007. Novel agonistic action of mustard oil on recombinant and endogenous porcine transient receptor potential V1 (pTRPV1) channels. Biochem. Pharmacol. 73:1646–1656.
Read, D. P., Feeny, P. P., and Root, R. B. 1970. Habitat selection by the aphid parasite Diaeretiella rapae (Hymenoptera: Braconidae) and hyperparasite Charips brassicae (Hymenoptera: Cynipidae). Can. Entomol. 102:1567–&.
Reed, H. C., Tan, S. H., Haapanen, K., Killmon, M., Reed, D. K., and Elliott, N. C. 1995. Olfactory responses of the parasitoid Diaeretiella rapae (Hymenoptera: Aphidiidae) to odor of plants, aphids, and plant–aphid complexes. J. Chem. Ecol. 21:407–418.
Storeck, A., Poppy G. M., van Emden, H. F., and Powell, W. 2000. The role of plant chemical cues in determining host preference in the generalist aphid parasitoid Aphidius colemani. Entomol. Exp. Appl. 97:41–46.
Van, Poecke, R. M. P., Posthumus, M. A., and Dicke, M. 2001. Herbivore-induced volatile production by Arabidopsis thaliana leads to attraction of the parasitoid Cotesia rubecula: chemical, behavioral, and gene-expression analysis. J. Chem. Ecol. 27:1911–1928.
Vaughn, T. T., Antolin, M. F., and Bjostad, L. B. 1996. Behavioral and physiological responses of Diaeretiella rapae to semiochemicals. Entomol. Exp. Appl. 78:187–196.
Wickremasinghe, M. G. V., and van, Emden, H. F. 1992. Reactions of adult female parasitoids, particularly Aphidius rhopalosiphi, to volatile chemical cues from the host plants of their aphid prey. Physiol. Entomol. 17:297–304.
Wink, M. 1988. Plant breeding—importance of plant secondary metabolites for protection against pathogens and herbivores. Theor. Appl. Gen. 75:225–233.
Wittstock, U., and Burow, M. 2007. Tipping the scales—Specifier proteins in glucosinolate hydrolysis. IUBMB Life 59:744–751.
Zabala, M. D., Grant, M. Bones, A. M., Bennett, R., Lim, Y. S., Kissen, R., and Rossiter, J. T. 2005. Characterisation of recombinant epithiospecifier protein and its over-expression in Arabidopsis thaliana. Phytochemistry 66:859–867.
Acknowledgments
We thank Valerie Elliot for technical assistance and the BBSRC for providing financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pope, T.W., Kissen, R., Grant, M. et al. Comparative Innate Responses of the Aphid Parasitoid Diaeretiella rapae to Alkenyl Glucosinolate Derived Isothiocyanates, Nitriles, and Epithionitriles. J Chem Ecol 34, 1302–1310 (2008). https://doi.org/10.1007/s10886-008-9531-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9531-2