Abstract
Studying the hygroscopic behavior of bamboo is important for industrial applications because it influences the dimensional stability and mechanical properties of the final bamboo products. In this study, the water vapor sorption behavior of 14 bamboo species was investigated using a dynamic vapor sorption apparatus and the results were analyzed using the Guggenheim–Anderson–de Boer (GAB) and Hailwood–Horrobin (H–H) models. The different bamboo species exhibited varying sorption isotherms and degrees of hysteresis. The reasons for this may be related to the different sorption isotherms of the two main cell units of bamboo, namely the fiber and parenchyma cells, and the chemical composition of bamboo. The GAB and H–H models provided good fits to the experimental data and meaningful physical parameters regarding the monolayer capacity, especially the water accessible specific surface area can be obtained from the GAB parameters. The fiber saturation point values were also determined using the GAB and H–H models, which ranged from 16.37 to 27.91% for the different bamboo species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bamboo is a grass that is abundant in tropical, subtropical and mild temperate regions and is one of the fastest growing plants on earth [1, 2]. Considering its rapid growth rate, low cost, high strength and stiffness, bamboo has been widely used in a variety of applications such as house construction, furniture, handicrafts and paper production, demonstrating its potential as a sustainable wood substitute [3, 4]. As a natural and heterogeneous material (like wood), bamboo exhibits certain deleterious properties when subject to varying moisture levels, such as dimensional instability, mold growth, fungal decay and loss of strength [5, 6]. Consequently, it is important to understand the hygroscopic response of bamboo to its surrounding environment, in order to develop optimal drying processes and safe storage in varying conditions [7].
A sorption isotherm is often used to describe the relationship between the equilibrium moisture states of bamboo and its surrounding environment at constant temperature and relative humidity (RH), and this includes the adsorption isotherm measured from an initial dry condition and the desorption one measured from an initially water-saturated condition [8, 9]. Although the water vapor sorption behavior of wood or chemically modified wood has been well documented, the investigations on bamboo were very limited [10, 11]. Therefore, there is a need to build knowledge on bamboo’s water vapor sorption behavior to support and improve the processing and manufacturing of bamboo products.
The purpose of this study is to establish a statistically significant water vapor sorption database of bamboo species, as there are few studies in the literature that contains data for a wide range of bamboo species in one given study. Such information would be particularly useful in predicting the properties of bamboo in service and for modeling and optimizing the drying process. In this study, a dynamic vapor sorption (DVS) apparatus was used instead of saturated salt solutions as the former is more capable of providing highly reproducible data over a wide RH range [12].
Materials and methods
Sample preparations
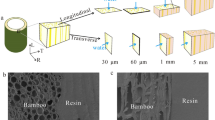
Fourteen two-to-three-year bamboo species (Table 1) were obtained from the Hua’an arboretum in Fujian Province, China, for this study. All bamboo culms were selected randomly. Small bamboo blocks were cut from the middle portion of the culm with a dimensions of 10 × 20 × T mm (tangential × longitudinal × culm wall thickness) (Fig. 1a), and their waxy epidermises and pith rings were remained in order to gain the comprehensive hygroscopic behavior of raw bamboo samples. To limit bamboo variability caused by the hierarchical architecture with vascular bundles non-uniformly distributed along the radial direction (taking M species as an example, see Fig. 1a), small bamboo block (around 10 mg) containing the whole radial structure was cut from bamboo block of the same type, with a final dimensions of 3 × 1 × T mm (tangential × longitudinal × culm wall thickness, Fig. 1b).
Dynamic water vapor sorption
The water vapor sorption behavior of the bamboo samples was determined using a dynamic vapor sorption apparatus (DVS Intrinsic, Surface Measurement Systems Ltd., UK). The samples were placed on a holder connected to a microbalance (± 0.1 μg) using hanging wires, and this was located in a thermostatically controlled chamber where a constant flow of dry nitrogen gas into which another flow of nitrogen containing a preset amount of water vapor was mixed to maintain a given RH [5]. The measurements were taken at a constant temperature of 25 ± 0.1 °C for approximately 3 days, starting at 0% RH and increasing in increments of 10% RH up to 98% RH, then decreasing back to 0% RH also in 10% RH decrements. Samples were then maintained at a constant RH, until the weight change was less than 0.002% per 10 min. Data on weight change were acquired every 20 s. As the DVS technique has been reported to yield highly reproducible data [13], in this study only one measurement is reported for each bamboo species.
Sorption models
Several mathematical models have been developed to describe the water vapor sorption isotherms of hygroscopic materials [14]. The experimental sorption data of 14 bamboo species were fitted with two multilayer sorption models most frequently used for hygroscopic materials, namely the GAB and H–H models (Table 2). The GAB and H–H models represent two different theoretical concepts: the multi-molecular layer formation and water–polymer ideal solution assumption [11, 15]. The Origin 8.0 software (OriginLab Corporation, Northampton, MA, USA) was used for obtaining the parameters of the models. In addition, the fiber saturation point (FSP) values of the 14 bamboo species were calculated using the two isotherm models.
SEM observation
Sections of samples along the transverse and longitudinal directions were cut with blades and mounted on a sample holder with carbon conductive tape and then coated with a thin layer of platinum using a sputter coater (Leica EMSCD050). The morphology of samples was observed under a Field Emission Scanning Electron Microscope (FE-SEM, XL30, FEI, USA) operating at 7.0 kV.
Results and discussion
Adsorption and desorption isotherms
The water vapor adsorption and desorption isotherms of the 14 bamboo species are reported as the EMC against RH, presented in Fig. 2. All of the isotherms exhibited a sigmoidal shape typical of bamboo [10] and can be classified as type II according to the classification scheme of Brunauer et al. [16]. The adsorption isotherms of all bamboo species exhibited an upward bend at around 60–70% RH, whereas desorption isotherms were more linear at similar RH levels. Similar results were also reported in other studies on lignocellulosic materials [17, 18]. However, there were differences in the EMC values of the various bamboo species at certain RH levels, especially above 75% RH. At the highest RH (98%), Ma species reached an EMC of 14.81%, whereas the EMC of L species (belong to the same genus as Ma species) reached as high as 24.98%.
In addition to the difference in EMC values, there were also differences in the degree of hysteresis. Absolute hysteresis (obtained by subtracting the EMC of adsorption from the EMC of desorption isotherm at constant RH) and the hysteresis coefficient (a ratio of the EMC for adsorption to EMC for desorption at constant RH) are two common means to characterize sorption hysteresis [11, 13]. The changes in the absolute hysteresis and hysteresis coefficient are shown in Fig. 3. As shown in Fig. 3a, all bamboo species exhibited the hysteresis phenomenon over the entire RH region, but the degree of hysteresis varied. Hysteresis for all bamboo species increased first and then decreased with increasing RH, but the highest hysteresis values varied from 2.65 to 4.42% at 50–80% RH, depending on the species. The variation in the change in the hysteresis coefficient was remarkable (range 0.53–0.96) for the different bamboo species (Fig. 3b).
The water sorption and extent of hysteresis may be influenced by the structure and chemical composition of bamboo. On the one hand, the fine structure of different cell walls types plays a role in the hygroscopic behavior of bamboo [10]. Bamboo can be considered as a 2-phase composites consisting of fibers that are almost completely solid with insignificant lumen (Fig. 4c) embedded in parenchyma cells that are thin-walled and have large lumens (Fig. 4d) [19]. Adsorption/desorption isotherms and the hysteresis for the fiber and parenchyma cells of M species are shown in Fig. 5, where it can be clearly seen that the parenchyma cells exhibited higher EMC values but lower hysteresis capacity at higher RH, indicating a difference in hygroscopic capacity from bamboo fibers. The reason for this may be related to the vast differences in the geometry of the cell walls as shown in Fig. 4. Therefore, the water vapor adsorption behavior of bamboo rested on the properties of the fibers and parenchyma cells and on the distribution of fibers.
On the other hand, hygroscopic behavior has also shown to differ in the main components of the cell wall, as hemicelluloses provide the highest number of OH groups available for water sorption, followed by cellulose and lignin [20]. Differences in the contents of these components clearly have an effect on the water vapor sorption across bamboo species. Therefore, the variation in water sorption behavior of different bamboo species seems to be a very complex phenomenon and a more detailed study should be conducted to explain the difference in the water sorption behavior of different bamboo species.
Fitting the GAB model to the data
The results of fitting the GAB model to the data of the 14 bamboo species are presented in Table 3. The fits were considered to be valid as all the R2 values above 99.0% [21]. GAB parameters can be used to compare the hygroscopic properties of different bamboo species. Among them, the X m (GAB) obtained from the adsorption branch refers to the moisture content when the monolayer is full and can be used to estimate the internal specific surface area corresponding to the monolayer capacity. The internal specific surface area (SGAB) is defined as
where ρ is water density, L is the Avogadro number, σ is the average area where water occupies the complete monolayer (0.114 nm2 was used in this study), and M is the molar mass of water [22].
The obtained X m value ranged from 3.26% for Ma species to 5.27% for HM species, and the corresponding SGAB increased from 124 to 201 m2/g, which represented the lowest and the highest monolayer capacity. This is due to their differences in cell wall ultrastructure and chemical composition, as mentioned above. The C value in the GAB model is the total heat of sorption of the monolayer and is always positive due to the strong exothermic interaction between the water vapor and adsorption sites [21]. Also, the K value is a factor that corrects the properties of the multilayer molecules with respect to the bulk liquid, which is unsound when higher than one as sorption becomes infinite at water activity values lower than one [23]. Moreover, the value of K given in Table 3 of ca. 0.8 (range: 0.74–0.83) can be taken as the characteristic of a non-porous sorbent [24].
The hygroscopic difference between bamboo and wood species was further compared. Here, the average GAB constants with corresponding uncertainties were obtained by fitting the entire sets of adsorption data to all 14 bamboo species. The average GAB constants of 21 wood species were also obtained by Bratasz et al. [22]. The average GAB constants of bamboo and wood species are given in Table 3, and the two general isotherms are shown in Fig. 6. It can be clearly observed that bamboo generally exhibits less hygroscopicity than wood during the both adsorption and desorption process.
Fitting data to the H–H model
The results of fitting experimental data to the H–H model are shown in Table 4. Like the GAB model, the H–H model also demonstrated satisfactory fitting to the experimental data (R2 ≥ 99%). The W value represents the apparent molecular mass of the dry bamboo per sorption, and K1 and K2 are equilibrium constants related to the formation of hydrates from dissolved water and the dry bamboo material, and the formation between dissolved water and external water vapor [18]. The value of W for Ma species (552.14) was remarkably higher than that of other species, indicating that it had less available sites than others, which agreed with the lower EMC of Ma species observed in Fig. 2a. The value of K2 ranged from 0.74 to 0.83 which indicates that the dissolved water had lower activity than the liquid water, which was close to the value of K2 (0.72) for wood [25] and a little higher than the value of K2 (0.619–0.682) for paper materials [18].
According to the H–H model, the sigmoidal isotherms can be divided into monolayer (M h ) and multilayer (M s ) water, referring, respectively, to hydrates and dissolved water. As an example, Fig. 7 demonstrates that the fit was generally satisfactory over the whole RH range for M species. The results showed that monolayer water increased at the low RH region, indicating that the water that is directly bound to active sites is mainly with hydroxyl groups, and with increasing RH, water preferentially adsorbs already hydrogen-bonded molecules and forms clusters to form polylayer water.
Comparison of models
Knowledge of the monolayer moisture content of lignocellulosic materials is important for industrial applications, as it corresponds to ensuring the stability of the material to minimize spoilage [26]. The comparison between the GAB and H–H models on determining monolayer moisture content is shown in Table 5. The results showed that the GAB model exhibited higher monolayer moisture content than the H–H model, and this is due to their difference in mathematical expression and physical concepts on monolayer sorption. Higher monolayer moisture contents obtained from GAB model than H–H model were also reported for paper samples [18].
FSP is defined as the moisture content corresponding to the saturation of the cell wall with no free water in the cell lumen [27]. Knowledge of the FSP values of lignocellulosic materials is necessary because it influences the dimensional changes and mechanical strength of materials [28]. The EMC obtained in adsorption isotherms at 100% RH is considered as the nominal FSP value. In contrast to the difference in monolayer moisture content, the nominal FSP value determined from the GAB and H–H models was the same given that their mathematical structures are similar [11]. As shown in Table 5, the FSP value varied from 16.36% for Ma species and 27.91% for L species, and the differences are mainly seen at high RH levels. However, it has been argued whether FSP value can be accurately extrapolated using sorption isotherms, because the adsorption isotherm increases sharply in the vicinity of 100% RH and large errors may arise from slight errors [29]. It is best to determine FSP value using other appropriate tests, such as volumetric swelling intersection [28] and pressure plate methods [30].
Conclusions
In this study, the hygroscopic behavior of 14 bamboo species was determined from their water vapor sorption isotherms at 25 °C in the range of 0–98% RH. The different bamboo species exhibited variable water vapor sorption isotherm as the EMC values and the degree of hysteresis observed varied throughout all RH levels. All sorption isotherms exhibited typical type II isotherms and could be predicted agreeably with GAB and H–H models. The values of monolayer moisture content for 14 bamboo species as estimated by the GAB and H–H models ranged from 3.26 to 5.27%, 2.49 to 3.83%, respectively. In particular, the internal specific surface area of bamboo was calculated from the GAB monolayer parameter, varying from 124 to 201 m2/g. Moreover, the ‘average’ sorption isotherm of bamboo can be obtained from the sorption data, which exhibited less hygroscopicity than wood. The information generated in this study can provide useful information in modeling the hygroscopic responses of bamboo in environments with changing moisture levels.
References
Brito BSL, Pereira FV, Putaux JL, Jean B (2012) Preparation, morphology and structure of cellulose nanocrystals from bamboo fibers. Cellulose 19(5):1527–1536
Ohmae Y, Saito Y, Inoue M, Nakano T (2009) Mechanism of water adsorption capacity change of bamboo by heating. Eur J Wood Wood Prod 67(1):13–18
Ahmad M, Kamke FA (2005) Analysis of Calcutta bamboo for structural composite materials: physical and mechanical properties. Wood Sci Technol 39(6):448–459
Wang YY, Yin H (2014) Analysis on significance of bamboo products based on sustainable development. Adv Mater Res 1010–1012:1944–1948
Čermák P, Vahtikari K, Rautkari L, Laine K, Horáček P, Baar J (2016) The effect of wetting cycles on moisture behaviour of thermally modified Scots pine (Pinus sylvestris L.) wood. J Mater Sci 51(3):1–8. https://doi.org/10.1007/s10853-015-9471-5
Popescu CM, Hill CAS, Curling S, Ormondroyd G, Xie Y (2014) The water vapour sorption behaviour of acetylated birch wood: how acetylation affects the sorption isotherm and accessible hydroxyl content. J Mater Sci 49(5):2362–2371. https://doi.org/10.1007/s10853-013-7937-x
Rhim JW, Lee JH (2009) Thermodynamic analysis of water vapor sorption isotherms and mechanical properties of selected paper-based food packaging materials. J Food Sci 74(9):E502–E511
Zelinka SL, Mitchell MR, Link RE, Glass SV (2010) Water vapor sorption isotherms for southern pine treated with several waterborne preservatives. J Test Eva 38(4):521–525
Engelund ET, Thygesen LG, Svensson S, Hill CAS (2013) A critical discussion of the physics of wood–water interactions. Wood Sci Technol 47(1):141–161
Ohmae Y, Nakano T (2009) Water adsorption properties of bamboo in the longitudinal direction. Wood Sci Technol 43(5–6):415–422
Olek W, Majka J, Czajkowski Ł (2013) Sorption isotherms of thermally modified wood. Holzforschung 67(2):183–191
Rautkari L, Hill CAS, Curling S, Jalaludin Z, Ormondroyd G (2013) What is the role of the accessibility of wood hydroxyl groups in controlling moisture content? J Mater Sci 48(18):6352–6356. https://doi.org/10.1007/s10853-013-7434-2
Xie Y, Hill CAS, Jalaludin Z, Curling SF, Anandjiwala RD, Norton AJ, Newman G (2011) The dynamic water vapour sorption behaviour of natural fibres and kinetic analysis using the parallel exponential kinetics model. J Mater Sci 46(2):479–489. https://doi.org/10.1007/s10853-010-4935-0
Simpson W (1980) Sorption theories applied to wood. Wood Fiber 12(3):183–195
Papadopoulos AN, Avramidis S, Elustondo D (2005) The sorption of water vapour by chemically modified softwood: analysis using various sorption models. Wood Sci Technol 39(2):99–111
Brunauer S, Deming LS, Deming WE, Teller E (1940) On a theory of the van der Waals adsorption of gases. J Am Chem Soc 62(7):1723–1732
Hill CAS, Norton A, Newman G (2009) The water vapor sorption behavior of natural fibers. J Appl Polym S 112(3):1524–1537
Bedane AH, Xiao H, Eić M (2014) Water vapor adsorption equilibria and mass transport in unmodified and modified cellulose fiber-based materials. Adsorption 20(7):863–874
Zhang X, Li J, Yu Z, Yu Y, Wang H (2017) Compressive failure mechanism and buckling analysis of the graded hierarchical bamboo structure. J Mater Sci 52(12):6999–7007. https://doi.org/10.1007/s10853-017-0933-9
Christensen GN, Kelsey KE (1959) The rate of sorption of water vapor by wood. Holz als Roh- und Werkstoff 17(5):178–188
Esteban LG, Simón C, Fernández FG, Palacios PD, Martín-Sampedro R, Eugenio ME, Hosseinpourpia R (2015) Juvenile and mature wood of Abies pinsapo Boissier: sorption and thermodynamic properties. Wood Sci Technol 49(4):725–738
Bratasz Ł, Kozłowska A, Kozłowski R (2012) Analysis of water adsorption by wood using the Guggenheim–Anderson–de Boer equation. Eur J Wood Wood Prod 70(4):445–451
Maskan M, Göǧüş F (1997) The fitting of various models to water sorption isotherms of pistachio nut paste. J Food Eng 33(3–4):227–237
Timmermann EO (2003) Multilayer sorption parameters: BET or GAB values? Colloids Surf A 220(1–3):235–260
Mantanis GI, Papadopoulos AN (2010) The sorption of water vapour of wood treated with a nanotechnology compound. Wood Sci Technol 44(3):515–522
Moreira R, Chenlo F, Torres MD, Vallejo N (2008) Thermodynamic analysis of experimental sorption isotherms of loquat and quince fruits. J Food Eng 88(4):514–521
Tiemann H (1906) Effect of moisture upon the strength and stiffness of wood, vol 63. US Dept. of Agriculture, Forest Service, Washington, D.C.
Hernández R (2007) Effects of extraneous substances, wood density and interlocked grain on fiber saturation point of hardwoods. Wood Mater Sci Eng 2(1):45–53
Hill CAS (2008) The reduction in the fibre saturation point of wood due to chemical modification using anhydride reagents: a reappraisal. Holzforschung 62(4):423–428
Hernandez RE, Pontin M (2006) Shrinkage of three tropical hardwoods below and above the fiber saturation point. Wood Fiber Sci 38(3):474–483
Acknowledgements
We would like to thank the National Science Foundation of China (31400519) and 12th Five Years Key Technology R&D Program (2015BAD04B03) for their financial support for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, X., Li, J., Yu, Y. et al. Investigating the water vapor sorption behavior of bamboo with two sorption models. J Mater Sci 53, 8241–8249 (2018). https://doi.org/10.1007/s10853-018-2166-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-018-2166-y