Abstract
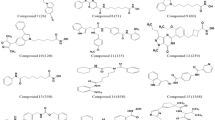
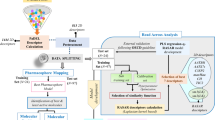
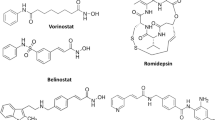
As an effort to develop therapeutics for cancer treatments, a number of effective histone deacetylase inhibitors with structural diversity have been discovered. To gain insight into optimizing the activity of an identified lead compound, a computational protocol sequentially involving homology modeling, docking experiments, molecular dynamics simulation, and free energy perturbation calculations was applied for rationalizing the relative activities of known histone deacetylase inhibitors. With the newly developed force field parameters for the coordination environment of the catalytic zinc ion in hand, the computational strategy proved to be successful in predicting the rank orders for 12 derivatives of three hydroxamate-based inhibitor scaffolds with indole amide, pyrrole, and sulfonamide moieties. The results showed that the free energy of an inhibitor in aqueous solution should be an important factor in determining the binding free energy. Hence, in order to enhance the inhibitory activity by adding or substituting a chemical group, the increased stabilization in solution due to the structural changes must be overcome by a stronger enzyme-inhibitor interaction. It was also found that to optimize inhibitor potency, the hydrophobic head of an inhibitor should be elongated or enlarged so that it can interact with Pro29 and His28 that are components of the flexible loop at the top of the active site.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jenuwein, T. and Allis, C.D., Science, 293 (2001) 1074.
Johnstone, R.W., Nat. Rev. Drug Discov., 1 (2002) 287.
Marks, P.A., Rifkind, R.A., Richon, V.M., Breslow, R., Miller, T. and Kelly, W.K., Nat. Rev. Cancer, 1 (2001) 194.
Gray, S.G. and Ekstrom, T.J., Exp. Cell Res., 262 (2001) 75.
Imai, S., Armstrong, C.M., Kaeberlein, M. and Guarente, L., Nature, 403 (2000) 795.
Finnin, M.S., Donigian, J.R. and Pavletich, N.P., Nat. Struct. Biol., 8 (2001) 621.
Finnin, M.S., Donigian, J.R., Cohen, A., Richon, V.M., Rifkind, R.A., Marks, P.A., Breslow, R. and Pavletich, N.P., Nature, 401 (1999) 188.
Lipscomb, W.N. and Sträter, N., Chem. Rev., 96 (1996) 2375.
Yoshida, M., Kijima, M., Akita, M. and Beppu, T., J. Biol. Chem., 265 (1990) 17174.
Kijima, M., Yoshida, M., Sugita, K., Horinouchi, S. and Beppu, T., J. Biol. Chem., 268 (1993) 22429.
Nakajima, H., Kim, Y.B., Terano, H., Yoshida, M. and Horinouchi, S., Exp. Cell. Res., 241 (1988) 126.
Darkin-Rattray, S., Gurnett, A.M., Myres, R.W., Dulski, P.M., Crumely, K.M., Allocco, J.J., Cannova, C., Meinke, P.T., Colletti, S.L., Bednarek, M.A., Singh, S.B., Goetz, M.A., Dombrowski, A.W., Polishook, J.D. and Schimatz, D.M., Proc. Natl. Acad. Sci. USA, 93 (1996) 13143.
Kelly, W.K., O'Connor, O.A. and Marks, P.A., Exp. Opin. Investig. Drugs, 11 (2001) 1695.
Dai, Y., Guo, Y., Guo, J., Pease, L.J., Li, J., Marcotte, P.A., Glaser, K.B., Tapang, P., Albert, D.H., Richardson, P.L., Davidsen, S.K. and Michaelides, M.R., Bioorg. Med. Chem. Lett., 13 (2003) 1897.
Bouchain, G., Leit, S., Frechette, S., Khalil, E.A., Lavoie, R., Moradei, O., Woo, S.H., Fournel, M., Yan, P.T., Kalita, A., Trachy-Bourget, M.-C., Beaulieu, C., Li, Z., Robert, M.F., MacLeod, A.R., Besterman, J.M. and Delorme, D., J. Med. Chem., 46 (2003) 820.
Mai, A., Massa, S., Ragno, R., Cerbara, I., Jesacher, F., Loidl, P. and Brosch, G., J. Med. Chem., 46 (2003) 512.
Uesato, S., Kitagawa, M., Nagaoka, Y., Maeda, T., Kuwajima, H. and Yamori, T., Bioorg. Med. Chem. Lett., 12 (2002) 1347.
Remiszewski, S.W., Sambucetti, L.C., Atadja, P., Bair, K.W., Cornell, W.D., Green, M.A., Howell, K.L., Jung, M., Kwon, P., Trogani, N. and Walker, H., J. Med. Chem., 45 (2002) 753.
Mai, A., Massa, S., Ragno, R., Esposito, M., Sbardella, G., Nocca, G., Scatena, R., Jesacher, F., Loidl, P. and Brosch, G., J. Med. Chem., 45 (2002) 1778.
Wittich, S., Scherf, H., Xie, C., Brosch, G., Loidl, P., Gerhauser, C. and Jung, M., J. Med. Chem., 45 (2002) 3296.
Phiel, C.J., Zhang, F., Huang, E.Y., Guenther, M.G., Lazar, M.A. and Klein, P.S., J. Biol. Chem., 276 (2001) 36734.
Göttlicher, M., Minucci, S., Zhu, P., Krämer, O.H., Schimpf, A., Giavara, S., Sleeman, J.P., Coco, F.L., Nervi, C., Pelicci, P.G. and Heinzel, T., EMBO J., 20 (2001) 6969.
Saito, A., Yamashita, T., Mariko, Y., Nosaka, Y., Tsuchiya, K., Ando, T., Suzuki, T., Tsuruo, T. and Nakanishi, O., Proc. Natl. Acad. Sci. USA, 96 (1999) 4592.
Frey, R.R., Wada, C.K., Garland, R.B., Curtin, M.L., Michaelides, M.R., Li, J., Pease, L.J., Glaser, K.B., Marcotte, P.A., Bouska, J.J., Murphy, S.S. and Davison, S.K., Bioorg. Med. Chem. Lett., 12 (2002) 3443.
Kapustin, G.V., Fejer, G., Gronlund, J.L., McCafferty, D.G., Seto, E. and Etzkorn, F.A., Org. Lett., 5 (2003) 3053.
Pina, I.C., Gautschi, J.T., Wang, G.-Y.-S., Sanders, M.L., Schmitz, F.J., France, D., Cornell-Kennon, S., Sambucetti, L.C., Remiszewski, S.W., Perez, L.B., Bair, K.W. and Crews, P., J. Org. Chem., 68 (2003) 3866.
Wada, C.K., Frey, R.R., Ji, Z., Curtin, M.L., Garland, R.B., Holms, J.H., Li, J., Pease, L.J., Guo, J., Glaser, K.B., Marcotte, P.A., Richardson, P.L., Murphy, S.S., Bouska, J.J., Tapang, P., Magoc, T.J., Albert, D.H., Davidsen, S.K. and Michaelides, M.R., Bioorg. Med. Chem. Lett., 13 (2003) 3331.
Baker, D. and Sali, A., Science, 294 (2001) 93.
Evers, A. and Klebe, G., Angew. Chem. Int. Ed. Engl., 43 (2004) 248.
Bairoch, A. and Apweiler, R., Nucleic Acids Res., 27 (1999)
Sonnhammer, E.L.L., Eddy, S.R. and Durbin, R., Proteins, 28 (1997) 405.
Thompson, J.D., Higgins, D.G. and Gibson, T.J., Nucleic Acids Res., 22 (1994) 4673.
Sali, A. and Blundell, T.L., J. Mol. Biol., 234 (1993) 779.
Fiser, A., Do, R.K.G. and Sali, A., Protein Sci., 9 (2000) 1753.
Morris, G.M., Goodsell, D.S., Halliday, R.S., Huey, R., Hart, W.E., Belew, R.K. and Olson, A.J., J. Comput. Chem., 19 (1998) 1639.
Hoops, S.C., Anderson, K.W. and Merz, K.M., Jr., J. Am. Chem. Soc., 113 (1991) 8262.
Ryde, U., Proteins, 21 (1995) 40.
Stote, R.H. and Karplus, M., Proteins, 23 (1995) 12.
Fox, T. and Kollman, P.A., Phys. Chem. B, 102 (1998) 8070.
Cornell, W.D., Cieplak, P., Bayly, C.I., Gould, I.R., Merz, K.M., Jr., Ferguson, D.M., Spellmeyer, D.C., Fox, T., Cald-well, J.W. and Kollman, P.A., J. Am. Chem. Soc., 117 (1995) 5179.
Bayly, C.A., Cieplak, P., Cornell, W.D. and Kollman, P.A., J. Phys. Chem., 97 (1993) 10269.
Vanommeslaeghe, K., Alsenoy, C.V., Proft, F.D., Martins, J.C., Tourwe, D. and Geerlings, P., Org. Biol. Chem., 1 (2003) 2951.
Case, D.A., Pearlman, D.A., Caldwell, J.W., Cheatham, T.E., Ross, W.S., Simmerling, C., Darden, T., Merz, K.M., Jr., Stanton, R.V., Cheng, A., Vincent, J.J., Crowley, M., Tsui, V., Radmer, R., Duan, Y., Pitera, J., Massova, I., Seibel, G.L., Singh, U.C., Weiner, P. and Kollman, P.A., AMBER 7, University of California, San Francisco, 2002.
Zwanzig, R.J., J. Chem. Phys., 22 (1954) 1420.
Beveridge, D.L. and DiCapua, F.M., Annu. Rev. Biophys. Biophys. Chem., 18 (1989) 431.
Bash, P.A., Singh, U.C., Brown, F.K., Langridge, R. and Kollman, P.A., Science, 235 (1987) 574.
Merz, K.M., Jr. and Kollman, P.A., J. Am. Chem. Soc., 111 (1989) 5649.
Rao, B.G., Tilton, R.F. and Singh, U.C., J. Am. Chem. Soc., 114 (1992) 4447.
Rastelli, G., Thomas, B., Kollman, P.A. and Santi, D.V., J. Am. Chem. Soc., 117 (1995) 7213.
Essex, J.W., Severance, D.L., Tirado-Rives, J. and Jorgensen, W.L., J. Phys. Chem. B., 101 (1997) 9663.
Reddy, M.R. and Erion, M.D., J. Am. Chem. Soc., 123 (2001) 6246.
Guimaraes, C.R.W. and Bicca de Alencastro, R., J. Med. Chem., 45 (2002) 4995.
Berendsen, H.C., Postma, J.P.M., van Gunsteren, W.F., DiNola, A. and Haak, J.R., J. Chem. Phys., 81 (1984) 3684.
Ryckaert, J.P., Ciccotti, G. and Berendsen, H.C., J. Comput. Phys., 23 (1977) 327.
Laskowski, R.A., MacArthur, M.W., Moss, D.S. and Thornton, J.M., J. Appl. Crystallogr., 26 (1993) 283.
Munagala, N., Basus, V.J. and Wang, C.C., Biochemistry, 40 (2001) 4303.
Gill, H.S., Pfluegl, G.M.U. and Eisenberg, D., Biochemistry, 41 (2002) 9863.
Huntley, J.J.A., Scrofani, S.D.B., Osborne, M.J., Wright, P.E. and Dyson, H.J., Biochemistry, 39 (2000) 13356.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Park, H., Lee, S. Homology modeling, force field design, and free energy simulation studies to optimize the activities of histone deacetylase inhibitors. J Comput Aided Mol Des 18, 375–388 (2004). https://doi.org/10.1007/s10822-004-2283-3
Issue Date:
DOI: https://doi.org/10.1007/s10822-004-2283-3