Abstract
The pathophysiology of sepsis is extremely complex. During this disease, the exacerbation of the inflammatory response causes oxidative stress, alterations in mitochondrial energy dynamics, and multiple organ failure. Some studies have highlighted the important role of the NLRP3 inflammasome in sepsis. This inflammasome is a macromolecular protein complex that finely regulates the activation of caspase-1 and the production and secretion of potent pro-inflammatory cytokines such as IL-1β and IL-18. In this review, we elucidate evidences to understand the connection between sepsis development and the NLRP3 inflammasome, the most widely investigated member of this class of receptor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Sepsis is a syndrome characterized by pathophysiologic and biochemical alterations caused by an infectious insult. These abnormalities are related to an exacerbated systemic inflammation, and they manifest as life-threatening clinical conditions [1] that may lead to multiple organ dysfunction [2] and a mortality rate of approximately 60% [3].
The initial stage of sepsis pathophysiology involves the production of signaling molecules responsible for recruiting immune cells, such as the pathogen-associated molecular patterns (PAMPs) and the damage-associated molecular patterns (DAMPs) and their interaction with germline-encoded receptors called pattern recognition receptors (PRRs) [4]. The involvement of Toll-like receptor-4 (TLR-4) in sepsis is well described [5,6,7]; however, another class of receptor has drawn attention in recent years: the nucleotide-binding and oligomerization domain (NOD)-like receptor (NLR) class, specifically the pyrin domain-containing 3 (NLRP3) member. NLRP3, along with other structures, generate a complex called inflammasome that can convert pro-inflammatory pro-caspases into their mature form, thus inducing the release of important pro-inflammatory cytokines [8, 9].
The NLRP3/caspase-1/IL-1 axis has emerged as a critical signaling pathway of the innate immune system and the progression of inflammation. There are important evidences regarding the involvement of NLRP3 inflammasome in different inflammatory diseases, such as cerebral ischemia [10], Parkinson’s and Alzheimer’s disease [11], inflammatory bowel disease [12], and atherosclerosis [13].
The connection of these diseases with bacterial sepsis occurs as follows: activation of Toll-like receptors by the presence of DAMPs and PAMPs, characterizing step 1 of the inflammasome activation and generation of ROS by phagocytic cells and mitochondrial damage activate the step 2 [14]. However, the connection of NLRP3 to the development of sepsis and organ dysfunction requires further investigation, and in this review, we elucidate evidences that lead the connection between sepsis development and the NLRP3 inflammasome, the most widely investigated member of this class of receptor.
NLRP3 INFLAMMASOME
The innate immune system has been recognized as the primary line of defense to fight back invading pathogens and maintain homeostasis [15]. Components of pathogens, injured tissue, and cells may release pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPS) that will be recognized by pattern recognition receptors (PRRs) and stimulate immune responses, and among these receptors, pyrin domain-containing 3 (NLRP3) is under intense investigation [16] as NLRs may form protein complexes called inflammasomes [17].
The NLRP3 is a group of high-molecular–weight cytosolic protein complex formed to mediate host immune responses to several DAMPs and PAMPs. This complex consists of three main parts: (a) a sensor/receptor protein of cytosolic location that serves as a platform for complex formation, for example, NLRP3; (b) an ASC (apoptosis-associated speck-like protein containing a CARD) adapter protein; and (c) an effector protein, pro-caspase-1 [9, 18, 19].
NLRP3 inflammasome activation requires two steps. The first one is called priming and consists in the expression of NLRP3 and pro-IL-1b through the stimulation of NF-kB in response to microbial molecules, such as TLR ligands or endogenous cytokines, e.g., tumor necrosis factor alpha (TNF-a). The second step is the activation phase in which an increase in extracellular adenosine triphosphate (ATP), extracellular osmolarity or pH alterations, β-amyloid fibers and degradation of extracellular matrix components, increase in potassium efflux, generation of mitochondrial reactive species, and lysosomal destabilization and leakage of cathepsin B trigger NLRP3 assembly and activation [20,21,22].
After conformational activation, NLRP3 interacts with the ASC adapter protein and induces the aggregation of ASC into a large cytosolic protein speck. Then, ASC specks generate a platform for recruitment of pro-caspase-1 monomers, which promotes its self-cleavage and activation. Consequently, active caspase-1 stimulates the cleavage of pro-IL-1b and pro-IL-18 into mature IL-1b and IL-18, thus promoting inflammatory responses [23, 24]. Active caspase-1 also cleaves gasdermin D to free its N-terminal domain and induce the formation of pores at the membrane, leading to a pro-inflammatory form of cell death called pyroptosis [19], and this process is called canonical NLPR3 inflammasome activation. In addition, recent evidences point to another process named noncanonical inflammasome activation, in which LPS derived from Gram-negative bacteria stimulates caspase-11, and this induces pyroptosis as well as caspase-1-dependent maturation and production of IL-1β and IL-18 [25, 26].
NLRP3 AND SEPSIS: ROLE ON INFLAMMATION
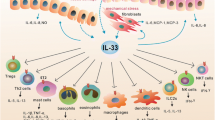
Considering that the immune system is the first one> affected in sepsis and the key signaling role played by NLRP3, different approaches have been studied to modulate this inflammasome in sepsis (Fig. 1) [27]. Biglycan is a stationary component of the extracellular matrix (ECM) found in most tissues under normal conditions, but it can become an endogenous ligand for TLR4 in macrophages after releasing from activated macrophages or the ECM during tissue damage. In this sense, biglycan can activate the NLRP3 inflammasome and elevate IL-1β levels in macrophages, without the need for costimulatory factors [28].
Effects of different substances on the immune system after sepsis. Several events and triggers are required for the activation of NLRP3. Like PAMPs, biglycan is an endogenous ligand for TLR4 and P2X7 receptor, activating the transcription of genes encoding NLRP3-forming proteins, or potassium efflux, one of the signals required for NLRP3 activation. Alpha-linolenic acid can reduce the expression of NLRP3, caspase-1, pro-IL-1β, and pro-IL-18. Dimethyl sulfoxide (DMSO) blocks ASC formation and caspase-1 formation. Follistatin-like protein 1 (FSTL-1) can activate the conversion of pro-caspase-1 into its mature form. Reactive species generated by mitochondria are triggers that activate NLRP3 inflammasome, whereas nitric oxide (NO) and sestrins seem to reduce this activation signal. Once activated, the NLRP3 inflammasome leads to the cleavage of pro-IL-1β and pro-IL-18 to their active form and causes pyroptosis.
Follistatin-like protein 1 (FSTL-1) is a secreted glycoprotein formed mainly by mesenchymal cells, and it is overexpressed in several inflammatory conditions characterized by elevated IL-1β. Chaly and colleagues (2014) proposed that FSTL-1 can access the intracellular space of cells that do not normally express this protein, such as monocytes/macrophages, and then FSTL-1 enters in the mitochondria, where it may improve ATP production, which is an important stimulus to activate NLRP3 inflammasome. Also, FSTL-1 can increase NLRP3 and pro-caspase-1 expression [29].
Sestrins (Sesn) are proteins that shield cells exposed to a variety of environmental stresses, including oxidative stress and DNA damage, maintain metabolic homeostasis, and induce autophagy under conditions of genotoxic stress [30]. Bone marrow-derived macrophages (BMDMs) of Sesn2 knockout mice exhibited defective mitophagy after immune stimulation, leading to hyperactivation of the NLRP3 inflammasome and increased mortality after sepsis [31].
Animals with genetic suppression for NLRP3 showed decreased autophagy in neutrophils along with increased phagocytosis and clearance of bacteria and improved survival [32]. In addition, genetic deficiency of NLRP3 induced the synthesis of lipoxin B4, a lipid mediator typically involved in the resolution of inflammation, in septic mice, and in macrophages stimulated with LPS and ATP [33]. On the other hand, in neonatal sepsis, the inhibition of caspase-1 and caspase-11 rather than NLRP3 blockade increased survival and decreased cytokine levels, as well as elevated the recruitment of macrophages and peritoneal neutrophils, associated with increased phagocytosis [34].
NLRP3 inflammasome seems to be inhibited by some substances. The administration of nitric oxide (NO) derived from iNOS may reduce the activation of NLRP3 due to a mitochondrial stabilization process, whereas it does not impact the TLR/NF-κB signaling pathways [35]. Dimethyl sulfoxide (DMSO) displayed ability to attenuate IL-1 maturation, caspase-1 activity, and ASC pyroptosome formation via NLRP3 inflammasome activator [36].
The extract of Syneilesis palmata (SP), a traditional Korean therapeutic herb widely used to treat pain or arthritis, also attenuated IL-1β secretion via the inhibition of NLRP3 inflammasome activation induced by monosodium urate, ATP, and nigericin. Further, SP ameliorated the severity of NLRP3 inflammasome-mediated symptoms in LPS-induced endotoxin and Escherichia coli-induced sepsis mouse models [37].
Alpha-linoleic acid metabolites also seemed to exert anti-inflammatory role by preventing the activation of NLRP3 inflammasome both in LPS-challenged RAW 264.7 cells and mouse peritoneal macrophages, as well as by the induction of apoptosis and inhibition of autophagy in the LPS-challenged macrophage [38]. In vitro tests with different macrophage lineages proved that apigenin, a natural flavonoid present in fruits and vegetables, may prevent LPS-induced IL-1β production by blocking caspase-1 activation through the disruption of the NLRP3 inflammasome assembly [39].
NLRP3 and Sepsis: Effects on the Mitochondrial Energetic Response
Mitochondrial energy dysfunction plays an important role in the pathogenesis of sepsis, especially by the ability to increase mitochondrial ROS production [40]. Evidence suggests that mitochondrial uncoupling proteins (UCPs), a superfamily of mitochondrial anion carrier proteins, are related to NLRP3 activation. Mitochondrial uncoupling protein-2 (UCP2) has been involved in numerous physiopathological conditions including metabolic disorders, inflammation, ischemic shock, cancer, and aging, even being pointed as a possible central modulator in metabolic processes[41]. In macrophages, UCP2 deficiency suppressed NLRP3 expression, the data suggest that UCP2 regulates NLRP3-mediated caspase-1 activation through fatty acid synthase (FASN)-mediated lipid synthesis. The study further demonstrated that regulation of downstream AKT and p38 MAPK activation by FASN is critical for NLRP3 expression and caspase-1 activation [42].
Mitochondrial damage was associated with inflammation through of NLRP3 activation. Excessive or sustained calcium (Ca2+) uptake can lead to mitochondrial damage characterized by increased production of ROS, mitochondrial permeability transition, and eventually rupture of the mitochondria. In addition, mitochondria have a principal role in regulating Ca2+ levels by taking Ca2+ that is released from the endoplasmic reticulum, causing elevation of their levels within mitochondria [43]. With the production of ROS, lysosomes can rupture, structures that function as Ca2+ deposits, and this Ca2+ mobilization in turn leads to the activation of NLRP3 [44].
In other hand, autophagy is a degradation system of cytoplasmic constituents that involves sequestration of cellular components, fusion to lysosomes, degradation of the cellular materials by hydrolytic enzymes, and reutilization of degradation products [45]. Evidence suggests that autophagy negatively regulates NLRP3 activation [45, 46]. A hypothesis suggests that inflammasomes or pro-IL-1β molecules are degraded by autophagosomes [43, 47], and the second hypothesis proposes that mitochondria-selective autophagy, termed mitophagy, eliminates dysfunctional/damaged mitochondria, avoiding the cytoplasm release of inflammasome activation DAMPs like ROS and thus preventing activation of NLRP3 inflammasome [48]. The ROS formation are critical events associated with NLRP3 inflammasome activation. However, the mitochondrial dysfunction and inflammasome activation and their contribution to the pathogenesis of sepsis thus remain incompletely understood.
NLRP3 and Sepsis: Effects on the Cardiovascular System
The in vitro stimulation of cardiac fibroblasts with LPS elevates the expression of NLRP3 and caspase-1, and this alteration is reverted by the administration of glyburide, a NLRP3 inhibitor [49]. NLRP3 knockout animals showed reduced myocardial damage when treated with melatonin [50]. Cortistatin, a neuropeptide and an immunomodulatory factor, demonstrated an interesting ability to inhibit NF-kB and NLRP3 activity, prevent the production and release of pro IL-1β, and attenuate sepsis-associated myocardial injury [51]. A recent study demonstrated that NLRP3 is activated in platelets of septic rodents, and it is associated with inflammation, endothelial permeability, and multiple organ injury [52].
NLRP3 and Sepsis: Effects on the Gastrointestinal System
Recent studies have shown the inflammatory effects of NLRP3 inflammasome on the gastrointestinal system. The hepatic levels of NLRP3 as well as proteins related to pyrocytosis seem to increase as sepsis evolves, thus inhibiting these proteins and blocking NLRP3 activation can alleviate acute liver injury induced by sepsis and may increase survival [53]. In fact, silencing NLRP3 gene 48 h prior to sepsis induction improved hyperbileacidemia and reduced cytokine levels, neutrophil infiltration, and macrophages pyrocytosis in hepatic tissue [54]. Of note, bile acids are recognized as a class of DAMPS that synergize with ATP in activating the NLRP3 inflammasome in a Ca2+ influx-dependent manner; however, bile acids are also ligands of the farnesoid X receptor (FXR), and, once stimulated, FXR seems to physically interact with NLRP3 inflammasome components to repress NLRP3 activation [55].
With regard to treatment options, mice subjected to LPS-induced acute liver injury and treated with bone marrow mesenchymal stem cells showed reduced levels of NLRP3, ASC, and caspase-1 as a result of prostaglandin E2 (PGE2) secretion and enhanced IL-10 levels [56]. The oral administration of Cinnamomum osmophloeum essential oil to mice before LPS injection reduced plasma levels of IL-1β and IL-18 and diminished the expression of ASC, caspase-1, and NLRP3 in the intestinal mucosa [57].
NLRP3 and Sepsis: Effects on the Renal System
A few studies have demonstrated the effects of NLRP3 modulation on kidney disease caused by sepsis. The viability of mitochondria is mainly sustained by sirtuin 3 (SIRT3), a member of NAD+-dependent deacetylases family, and its overexpression in renal tissue improves mitochondrial dynamics by decreasing reactive species. In this sense, Zhao and colleagues (2016) [58] demonstrated that sepsis induced a higher upregulation of NLRP3 inflammasome in SIRT3 knockout rats than in wild type animals, and this was reversed by an antioxidant treatment.
Wang and colleagues (2015) [59] tested the effects of carbon monoxide (CO) on rats with sepsis-induced acute kidney failure. Their results indicated a decrease in serum creatinine and urea levels and reduced apoptotic and histological scores, followed by diminished NLRP3 expression and higher survival rate.
Hyperin, a flavonoid substance found in Ericaceae, Guttifera, and Celastraceae plants, was tested in mice subjected to LPS-induce acute kidney injury. Hyperin treatment significantly inhibited TNF-α, IL-6, and IL-1β production through modulation of TLR4 expression and NF-κB activation. In addition, hyperin treatment blocked LPS-induced NLRP3 signaling pathway in a dose-dependent manner [60].
NLRP3 and Sepsis: Effects on the Respiratory System
Septic patients can have their respiratory function severely compromised due to increased neutrophil infiltration, edema formation, and elevated levels of IL-1β and IL-18 in the lung tissue, and the heme oxygenase-1 (HO-1) enzyme seems to be protective due to its anti-inflammatory features. The administration of hemin, a HO-1 inducer, downregulated the expression for NLRP3, ASC, and caspase-1 and the activity of NF-κB and NLRP3 inflammasome [61].
The study of different medicinal plants and their products to treat or prevent respiratory damage is increasing. In a recent research, cinnamaldehyde, a phenolic compound of Cinnamon species, was able to reduce the expression of NLRP3 and IL-1β in vitro and in the lungs of LPS-injected mice [62]. Another study demonstrated protective effects of dihydromyricetin, an isolated substance from the tender stem and leaves of the Ampelopsis grossedentata species, against CLP-induced acute lung injury by inhibiting NLRP3 inflammasome activation and subsequent pyroptosis [63].
NLRP3 and Sepsis: Effects on the Central Nervous System
Resveratrol, a phenolic compound found in grapes, significantly inhibited NLRP3 expression and IL-1β cleavage in the hippocampus of mice subjected to a sepsis-associated encephalopathy model, while treating BV2 cell lines with resveratrol prevents ATP induced NLRP3 activation and IL-1β cleavage [64]. Recently, Fu and colleagues (2019) [65] demonstrated that inhibiting the formation of NLRP3 inflammasome may positively contribute to the reduction of neurological and cognitive impairment of septic animals. The authors suggested that the NLRP3/caspase-1 pathway-induced pyroptosis mediates cognitive deficits, at least in mice. However, further studies are crucial to elucidate the potential effects of inflammasome inhibitors on cognitive damage after sepsis.
CONCLUSION
The evidences presented here demonstrated that NLRP3 inflammasome activation may induce important alterations in different systems affected by sepsis. So far, the mechanistic evaluations indicate that inhibiting the assembly and activation of this inflammasome could prevent the inflammatory response normally visualized in sepsis. Still, further studies are necessary to demonstrate the detailed influence exerted by the NLPR3 inflammasome on sepsis pathophysiology and to design new treatment approaches.
References
Ziesmann, Markus T., and John C. Marshall. 2018. Multiple organ dysfunction: the defining syndrome of sepsis. Surgical Infections 19: sur.2017.298. doi:https://doi.org/10.1089/sur.2017.298.
Sartelli, Massimo, Yoram Kluger, Luca Ansaloni, Timothy C. Hardcastle, Jordi Rello, Richard R. Watkins, Matteo Bassetti, et al. 2018. Raising concerns about the sepsis-3 definitions. World Journal of Emergency Surgery 13: 1–9. https://doi.org/10.1186/s13017-018-0165-6.
Hotchkiss, Richard S., and I.E. Karl. 2003. The pathophysiology and treatment of sepsis. The New England Journal of Medicine 348: 138–150. https://doi.org/10.1056/NEJMra021333.
Censoplano, Nina, Conrad L. Epting, and Bria M. Coates. 2014. The role of the innate immune system in sepsis. Clinical Pediatric Emergency Medicine 15. Elsevier Inc.: 169–176. https://doi.org/10.1016/j.cpem.2014.04.007.
Kuzmich, Nikolay, Konstantin Sivak, Vladimir Chubarev, Yuri Porozov, Tatiana Savateeva-Lyubimova, and Francesco Peri. 2017. TLR4 signaling pathway modulators as potential therapeutics in inflammation and sepsis. Vaccines 5: 34. https://doi.org/10.3390/vaccines5040034.
Beutler, B., X. Du, and A. Poltorak. 2001. Identification of Toll-like receptor 4 (TLR4) as the sole conduit for LPS signal transduction: genetic and evolutionary studies. Journal of Endotoxin Research 7: 277–280.
Beutler, Bruce. 2002. Toll-like receptors: how they work and what they do. Current Opinion in Hematology 9: 2–10.
Kumar, Vijay. 2018. Inflammasomes: Pandora’s box for sepsis. Journal of Inflammation Research 11: 477–502. https://doi.org/10.2147/JIR.S178084.
Place, David E., and Thirumala Devi Kanneganti. 2018. Recent advances in inflammasome biology. Current Opinion in Immunology 50. Elsevier Ltd:: 32–38. https://doi.org/10.1016/j.coi.2017.10.011.
Mangan, Matthew S.J., Edward J. Olhava, William R. Roush, H. Martin Seidel, Gary D. Glick, and Eicke Latz. 2018. Targeting the NLRP3 inflammasome in inflammatory diseases. Nature Reviews Drug Discovery 17: 588–606. https://doi.org/10.1038/nrd.2018.97.
Song, Limin, Lei Pei, Shanglong Yao, Yan Wu, and You Shang. 2017. NLRP3 inflammasome in neurological diseases, from functions to therapies. Frontiers in Cellular Neuroscience 11: 1–17. https://doi.org/10.3389/fncel.2017.00063.
Bauer, Christian, Peter Duewell, Hans-Anton Lehr, Stefan Endres, and Max Schnurr. 2012. Protective and aggravating fffects of NLRP3 inflammasome activation in IBD models: influence of genetic and environmental afctors. Digestive Diseases 30: 82–90. https://doi.org/10.1159/000341681.
Duewell, Peter, Hajime Kono, Katey J. Rayner, Cherilyn M. Sirois, Gregory Vladimer, Franz G. Bauernfeind, George S. Abela, et al. 2010. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464: 1357–1361. https://doi.org/10.1038/nature08938.
Danielski, Lucineia Gainski, Amanda Della Giustina, Marwa Badawy, Tatiana Barichello, João Quevedo, Felipe Dal-Pizzol, and Fabrícia Petronilho. 2017. Brain barrier breakdown as a cause and consequence of neuroinflammation in sepsis. Molecular Neurobiology. Molecular Neurobiology 55: 1–9. https://doi.org/10.1007/s12035-016-0356-7.
Karasawa, Tadayoshi, and Masafumi Takahashi. 2017. Role of NLRP3 inflammasomes in atherosclerosis. Journal of Atherosclerosis and Thrombosis 24: 443–451. https://doi.org/10.5551/jat.RV17001.
Patel, Seema. 2017. Inflammasomes, the cardinal pathology mediators are activated by pathogens, allergens and mutagens: A critical review with focus on NLRP3. Biomedicine and Pharmacotherapy 92. Elsevier Masson SAS:: 819–825. https://doi.org/10.1016/j.biopha.2017.05.126.
Sutterwala, Fayyaz, Stefanie Haasken, and Suzanne Cassel. 2014. Mechanism of NLRP3 inflammasome activation. 1319: 82–95. https://doi.org/10.1111/nyas.12458.Mechanism.
He, Yuan, Hideki Hara, and Gabriel Núñez. 2016. Mechanism and regulation of NLRP3 inflammasome activation. Trends in Biochemical Sciences 41: 1012–1021. https://doi.org/10.1016/j.tibs.2016.09.002.
Hafner-Bratkovič, Iva, and Pablo Pelegrín. 2018. Ion homeostasis and ion channels in NLRP3 inflammasome activation and regulation. Current Opinion in Immunology 52: 8–17. https://doi.org/10.1016/j.coi.2018.03.010.
Shao, Bo Zong, Zhe Qi Xu, Bin Ze Han, Su Ding Feng, and Chong Liu. 2015. NLRP3 inflammasome and its inhibitors: A review. Frontiers in Pharmacology 6: 1–9. https://doi.org/10.3389/fphar.2015.00262.
Liu, Qiuyun, Danyan Zhang, Diyu Hu, Xiangmei Zhou, and Zhou Yang. 2018. The role of mitochondria in NLRP3 inflammasome activation. Molecular Immunology 103: 115–124. https://doi.org/10.1016/j.molimm.2018.09.010.
Pellegrini, Carolina, Luca Antonioli, Gloria Lopez-Castejon, Corrado Blandizzi, and Matteo Fornai. 2017. Canonical and non-canonical activation of NLRP3 inflammasome at the crossroad between immune tolerance and intestinal inflammation. Frontiers in Immunology 8. https://doi.org/10.3389/fimmu.2017.00036.
Gong, Tao, Yanqing Yang, Tengchuan Jin, Wei Jiang, and Rongbin Zhou. 2018. Orchestration of NLRP3 inflammasome activation by ion fluxes. Trends in Immunology 39: 393–406. https://doi.org/10.1016/j.it.2018.01.009.
Katsnelson, Michael A., Kristen M. Lozada-Soto, Hana M. Russo, Barbara A. Miller, and George R. Dubyak. 2016. NLRP3 inflammasome signaling is activated by low-level lysosome disruption but inhibited by extensive lysosome disruption: roles for K+ efflux and Ca2+ influx. American Journal of Physiology. Cellular Physiology 311: C83–C100. https://doi.org/10.1152/ajpcell.00298.2015.
Yi, Young-Su. 2017. Caspase-11 non-canonical inflammasome: a critical sensor of intracellular lipopolysaccharide in macrophage-mediated inflammatory responses. Immunology 152: 207–217. https://doi.org/10.1111/imm.12787.
Yang, Jieling, Yue Zhao, and Feng Shao. 2015. Non-canonical activation of inflammatory caspases by cytosolic LPS in innate immunity. Current Opinion in Immunology 32: 78–83. https://doi.org/10.1016/j.coi.2015.01.007.
Conway-Morris, Andrew, Julie Wilson, and Manu Shankar-Hari. 2018. Immune activation in sepsis. Critical Care Clinics 34: 29–42. https://doi.org/10.1016/j.ccc.2017.08.002.
Babelova, Andrea, Kristin Moreth, Wasiliki Tsalastra-greul, Jinyang Zeng-brouwers, Oliver Eickelberg, Marian F. Young, Peter Bruckner, Josef Pfeilschifter, Roland M. Schaefer, and Hermann-josef Gro. 2009. Biglycan , a danger signal that activates the NLRP3 inflammasome via Toll-like and P2X receptors. 284: 24035–24048. https://doi.org/10.1074/jbc.M109.014266.
Chaly, Yury, Fu Yu, Anthony Marinov, Bruce Hostager, Wei Yan, Brian Campfield, and John A. Kellum, et al. 2014. Follistatin-like protein 1 enhances NLRP3 inflammasome-mediated IL-1β secretion from monocytes and macrophages. European Journal of Immunology 44: 1467–1479. https://doi.org/10.1002/eji.201344063.
Kim, Min-Ji, Joo-Heon Yoon, and Ji-Hwan Ryu. 2016. Mitophagy: a balance regulator of NLRP3 inflammasome activation. BMB Reports 49: 529–535. https://doi.org/10.5483/BMBRep.2016.49.10.115.
Kim, Min-Ji, Soo Han Bae, Jae-Chan Ryu, Younghee Kwon, Oh. Ji-Hwan, Jeongho Kwon, Jong-Seok Moon, et al. 2016. SESN2/sestrin2 suppresses sepsis by inducing mitophagy and inhibiting NLRP3 activation in macrophages. Autophagy 12: 1272–1291. https://doi.org/10.1080/15548627.2016.1183081.
Jin, Liliang, Sanjay Batra, and Samithamby Jeyaseelan. 2017. Deletion of NLRP3 augments survival during polymicrobial sepsis by decreasing autophagy and enhancing phagocytosis. The Journal of Immunology 198: 1253–1262. https://doi.org/10.4049/jimmunol.1601745.
Lee, Seonmin, Kiichi Nakahira, Jesmond Dalli, Ilias I. Siempos, Paul C. Norris, Romain A. Colas, Jong Seok Moon, et al. 2017. NLRP3 inflammasome deficiency protects against microbial sepsis via increased lipoxin B4 synthesis. American Journal of Respiratory and Critical Care Medicine 196: 713–726. https://doi.org/10.1164/rccm.201604-0892OC.
Gentile, Lori F., Angela L. Cuenca, Alex G. Cuenca, C. Dina, Ricardo Ungaro, Philip A. Efron, Lyle L. Moldawer, and Shawn D. Larson. 2015. Improved emergency myelopoiesis and survival in neonatal sepsis by caspase-1 / 11 ablation: 300–311. https://doi.org/10.1111/imm.12450.
Mao, Kairui, Shuzhen Chen, Mingkuan Chen, Yonglei Ma, Yan Wang, Bo Huang, Zhengyu He, et al. 2013. Nitric oxide suppresses NLRP3 inflammasome activation and protects against LPS-induced septic shock: 201–212. https://doi.org/10.1038/cr.2013.6.
Ahn, Huijeong, Jeeyoung Kim, Eui-bae Jeung, and Geun-shik Lee. 2014. Immunobiology Dimethyl sulfoxide inhibits NLRP3 inflammasome activation. Immunobiology 219. Elsevier GmbH.:: 315–322. https://doi.org/10.1016/j.imbio.2013.11.003.
Han, Ji-won, Do-Wan Shim, Eun-jeong Shim, Myong-ki Kim, Yong-kook Shin, Su-Bin Kwak, Sushruta Koppula, Tack-Joong Kim, Tae-Bong Kang, and Kwang-Ho Lee. 2015. Syneilesis palmata (Thunb.) Maxim. extract attenuates inflammatory responses via the regulation of TRIF-dependent signaling and inflammasome activation. Journal of Ethnopharmacology 166: 1–4. https://doi.org/10.1016/j.jep.2015.02.056.
Kumar, Naresh, Geetika Gupta, Kotha Anilkumar, Naireen Fatima, Roy Karnati, Gorla Venkateswara Reddy, Priyanka Voori Giri, and Pallu Reddanna. 2016. 15-Lipoxygenase metabolites of α-linolenic acid, [13-(S)-HPOTrE and 13-(S)-HOTrE], mediate anti-inflammatory effects by inactivating NLRP3 inflammasome. Scientific Reports 6: 31649. https://doi.org/10.1038/srep31649.
Zhang, Xiaoxuan, Guangji Wang, Emily C. Gurley, and Huiping Zhou. 2014. Flavonoid apigenin inhibits lipopolysaccharide-induced inflammatory response through multiple mechanisms in macrophages. 9: 1–18. https://doi.org/10.1371/journal.pone.0107072.
Costa, P., and S. Ana Paula. 2008. A Santiago, Rodrigo T Amâncio, Antonio Galina, Marcus F Oliveira, and Fernando A Bozza. Sepsis induces brain mitochondrial dysfunction 36: 1925–1932. https://doi.org/10.1097/CCM.0b013e3181760c4b.
Vozza, Angelo, Giovanni Parisi, Francesco De Leonardis, Francesco M. Lasorsa, and Alessandra Castegna. 2014. UCP2 transports C4 metabolites out of mitochondria, regulating glucose and glutamine oxidation. PNAS 111: 960–965. https://doi.org/10.1073/pnas.1317400111.
Moon, Jong-seok, Seonmin Lee, Mi-ae Park, Ilias I. Siempos, Maria Haslip, Patty J. Lee, Mijin Yun, et al. 2015. UCP2-induced fatty acid synthase promotes NLRP3 inflammasome activation during sepsis. 125: 665–680. https://doi.org/10.1172/JCI78253.interact.
Harris, James, Tali Lang, Jacinta P W Thomas, Maria B Sukkar, Neel R Nabar, and John H Kehrl. 2017. Autophagy and inflammasomes. Molecular Immunology 86. Elsevier Ltd: 10–15. doi:https://doi.org/10.1016/j.molimm.2017.02.013.
Murakami, Tomohiko, Johan Ockinger, Jiujiu Yu, Vanessa Byles, Aisleen Mccoll, and Aldebaran M. Hofer. 2012. Critical role for calcium mobilization in activation of the NLRP3 inflammasome. 109: 11282–11287. https://doi.org/10.1073/pnas.1117765109.
Lavera, Isabel De, Ana Delgado Pavón, Marina Villanueva Paz, Manuel Oropesa-ávila, Mario De, Elizabet Alcocer-gómez, Juan Garrido-maraver, David Cotán, Mónica Álvarez-córdoba, and José A Sánchez-alcázar. 2017. The connections among autophagy, inflammasome and mitochondria: 1030–1038. doi:https://doi.org/10.2174/1389450117666160527143143.
Cosin-roger, Jesus, Simona Simmen, Hassan Melhem, Kirstin Atrott, Isabelle Frey-wagner, Martin Hausmann, Cheryl De Vallière, et al. autophagy activation. Nature Communications, 1–13. US: Springer. https://doi.org/10.1038/s41467-017-00213-3.
Shi, Chong-shan, Kevin Shenderov, Ning-na Huang, Juraj Kabat, Mones Abu-asab, Katherine A. Fitzgerald, Alan Sher, and John H. Kehrl. 2014. Activation of autophagy by inflammatory signals limits IL-1β production by targeting ubiquitinated inflammasomes for destruction. Nature Immunology 13: 255–263. https://doi.org/10.1038/ni.2215.Activation.
Nakahira, Kiichi, Jeffrey Adam Haspel, Vijay A.K. Rathinam, Seon-jin Lee, Hilaire C. Lam, Joshua A. Englert, Marlene Rabinovitch, et al. 2011. Autophagy proteins regulate innate immune response by inhibiting NALP3 inflammasome-mediated mitochondrial DNA release. Nature Immunology 12: 222–230. https://doi.org/10.1038/ni.1980.Autophagy.
Zhang, Wenbo, Xuemei Xu, Raymond Kao, Tina Mele, Peter Kvietys, and Claudio M. Martin. 2014. Cardiac fibroblasts contribute to myocardial dysfunction in mice with sepsis : the role of NLRP3 inflammasome activation. Vol. 9. https://doi.org/10.1371/journal.pone.0107639.
Rahim, Ibtissem, Bahia Djerdjouri, Ramy K. Sayed, Marisol Fernández-Ortiz, Beatriz Fernández-Gil, Agustín Hidalgo-Gutiérrez, Luis C. López, Germaine Escames, Russel J. Reiter, and Darío Acuña-Castroviejo. 2017. Melatonin administration to wild-type mice and nontreated NLRP3 mutant mice share similar inhibition of the inflammatory response during sepsis. Journal of Pineal Research 63: e12410. https://doi.org/10.1111/jpi.12410.
Zhang, B.O., Y.U.E. Liu, Yu-bin Sui, Huai-qiu Cai, Wen-xiu Liu, Minling Zhu, and Xin-hua Yin. 2015. Cortistatin inhibits NLRP3 inflammasome activation of cardiac fibroblasts during sepsis. Journal of Cardiac Failure 21. Elsevier Inc:: 426–433. https://doi.org/10.1016/j.cardfail.2015.01.002.
Cornelius, Denise C., Cedar H. Baik, Olivia K. Travis, Dakota L. White, Cassandra M. Young, W. Austin Pierce, Corbin A. Shields, Bibek Poudel, and Jan M. Williams. 2019. NLRP3 inflammasome activation in platelets in response to sepsis. 7: 1–9. https://doi.org/10.14814/phy2.14073.
Chen, Yuan-li, Guo Xu, Xiao Liang, Juan Wei, Jing Luo, Guan-nan Chen, and Xiao-di Yan. 2016. Inhibition of hepatic cells pyroptosis attenuates CLP-induced acute liver injury. Vol. 8, 5685–5695.
Wu, Y., J. Ren, B. Zhou, C. Ding, J. Chen, G. Wang, G. Gu, X. Wu, S. Liu, D. Hu, and J. Li. 2015. Gene silencing of non-obese diabetic receptor family (NLRP3) protects against the sepsis-induced hyper-bile acidaemia in a rat model. Clinical and Experimental Immunology 179: 277–293. https://doi.org/10.1111/cei.12457.
Hao, Haiping, Lijuan Cao, Changtao Jiang, Yuan Che, Songyang Zhang, Shogo Takahashi, Guangji Wang, and Frank J. Gonzalez. 2017. Farnesoid X Receptor regulation of the NLRP3 inflammasome underlies cholestasis-associated sepsis. Cell Metabolism 25: 856–867.e5. https://doi.org/10.1016/j.cmet.2017.03.007.
Miao, Chun-mu, Xiao-wei Jiang, Kun He, Pei-zhi Li, Zuo-jin Liu, Ding Cao, Ou Zhi-bing, Jian-ping Gong, Chang-an Liu, and Yao Cheng. 2016. Bone marrow stromal cells attenuate LPS-induced mouse acute liver injury via the prostaglandin E 2-dependent repression of the NLRP3 inflammasome in Kupffer cells. Immunology Letters 179: 102–113. https://doi.org/10.1016/j.imlet.2016.09.009.
Lee, Shih-chieh, Jie-sheng Hsu, Chien-chun Li, Ke-ming Chen, and Cheng-tzu Liu. 2015. Protective effect of leaf essential oil from Cinnamomum osmophloeum Kanehira on endotoxin-induced intestinal injury in mice associated with suppressed local expression of molecules in the signaling pathways of. 3: 1–17. https://doi.org/10.1371/journal.pone.0120700.
Zhao, Wen-yu, Lei Zhang, Ming-xing Sui, You-hua Zhu, and Li Zeng. 2016. Protective effects of sirtuin 3 in a murine model of sepsis-induced acute kidney injury. Scientific Reports 6: 33201. https://doi.org/10.1038/srep33201.
Wang, Peng, Jian Huang, Yi Li, Ruiming Chang, Haidong Wu, and Jiali Lin. 2015. Exogenous carbon monoxide decreases sepsis-induced acute kidney injury and inhibits NLRP3 inflammasome activation in rats. Vol. 3, 20595–20608. https://doi.org/10.3390/ijms160920595.
Chunzhi, Gong, Li Zunfeng, Qin Chengwei, Bu Xiangmei, and Jingui Yu. 2016. Hyperin protects against LPS-induced acute kidney injury by inhibiting TLR4 and NLRP3 signaling pathways. 7: 82602–82608.
Luo, Yun-peng, Lei Jiang, Kai Kang, Dong-sheng Fei, Xiang-lin Meng, Chuan-chuan Nan, Shang-ha Pan, Ming-ran Zhao, and Ming-yan Zhao. 2014. International Immunopharmacology Hemin inhibits NLRP3 inflammasome activation in sepsis-induced acute lung injury , involving heme oxygenase-1. International Immunopharmacology 20. Elsevier B.V.: 24–32. https://doi.org/10.1016/j.intimp.2014.02.017.
Xu, Feng, Feng Wang, Taoqun Wen, Wentao Sang, Dejian Wang, and Nan Zeng. 2017. Inhibition of NLRP3 inflammasome: a new protective mechanism of cinnamaldehyde in endotoxin poisoning of mice. Immunopharmacology and Immunotoxicology 39: 296–304. https://doi.org/10.1080/08923973.2017.1355377.
Wang, Yu-chang, Qin-xin Liu, Qiang Zheng, Tao Liu, Xi-e Xu, Xing-hua Liu, Wei Gao, Xiang-jun Bai, and Zhan-fei Li. 2019. Dihydromyricetin alleviates sepsis-induced acute lung injury through inhibiting NLRP3 inflammasome-dependent pyroptosis in mice model. Inflammation 42: 1301–1310. https://doi.org/10.1007/s10753-019-00990-7.
Sui, Da-ming, Qun Xie, Wen-jing Yi, Sahil Gupta, Xi-ya Yu, Jin-bao Li, Jun Wang, Jia-feng Wang, and Xiao-ming Deng. 2016. Resveratrol protects against sepsis-associated encephalopathy and inhibits the NLRP3/IL-1 β axis in microglia. Mediators of Inflammation 2016: 1–10. https://doi.org/10.1155/2016/1045657.
Fu, Qun, Jing Wu, Xiao-Yan Zhou, Mu-Huo Ji, Qing-Hong Mao, Qing Li, Man-Man Zong, Zhi-Qiang Zhou, and Jian-Jun Yang. 2019. NLRP3/Caspase-1 pathway-induced pyroptosis mediated cognitive deficits in a mouse model of sepsis-associated encephalopathy. Inflammation 42: 306–318. https://doi.org/10.1007/s10753-018-0894-4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest in this publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Danielski, L.G., Giustina, A.D., Bonfante, S. et al. The NLRP3 Inflammasome and Its Role in Sepsis Development. Inflammation 43, 24–31 (2020). https://doi.org/10.1007/s10753-019-01124-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-019-01124-9