Abstract
This study aimed to determine the impact of horticultural land use on the taxonomic composition and the composition of functional feeding groups of freshwater macroinvertebrate assemblages in lowland streams. Three streams running through intensively cultivated plots were compared to three less impacted streams, two of which run through a UNESCO Biosphere Reserve. Nutrient and pesticide measured concentrations, as well as the macroinvertebrate assemblages associated with aquatic vegetation, were analyzed in each stream. These concentrations were higher at the horticultural streams, negatively correlated to the richness, density, and diversity of the macroinvertebrate assemblages, leading to a dominance of tolerant families. The trophic structure of the macroinvertebrates assemblages was dominated by collector-gatherers in the horticultural streams, while all functional feeding groups were more equally distributed in less impacted streams. We concluded that horticulture reduces biodiversity and changes the trophic structure of the macroinvertebrate assemblage, eventually affecting stream functions. Mitigation measures are required to preserve the structure and function of these ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aquatic ecosystems are threatened by human activities (Dudgeon, 2019). About 40% of the world’s surface is used for crop production (Foley et al., 2005), and this agricultural practice is considered one of the leading causes of global biodiversity loss (Dolédec & Statzner, 2010). Increased crop production in recent decades has intensified freshwater deterioration (Tilman, 1999; Davis et al., 2015). Rivers and streams with agricultural land use in their basins show altered flow dynamics (Langhammer, 2003; Keesstra et al., 2005), poor water quality (Jordan et al., 1997; Narangarvuu et al., 2014; Thomas et al., 2018), changes in the composition of biological communities (Roy et al., 2003; Nessimian et al., 2008; Miserendino et al., 2011), and in their metabolism (Young & Huryn, 1999; Gücker et al., 2009). These environmental consequences might have a stronger impact in developing countries because agriculture is essential to their economic development (Barbier, 2004).
Horticulture, the production of fresh vegetables for human consumption, is the second-largest farming activity worldwide in terms of total production, following extensive cereal production (FAO, 2013). However, its productivity (i.e., production per area) is about five times larger than the productivity of cereals (FAOSTAT, 2020). In Argentina, the horticultural output is roughly 10 million tons a year, produced in 600,000 ha, and it is carried out in “green belts” surrounding large urban centers (Castagnino et al., 2020). The Horticultural Belt around the city of La Plata (HBLP) is located in the center-east of Argentina, covering roughly 8600 ha (Baldini et al., 2021). Crops are grown in open fields and under-cover (i.e., in greenhouses); therefore, several harvests are carried out per year. The cultivation is an input-dependent system, involving intensive agrochemical applications.
The use of agrochemicals in Argentina has increased in the last decades. For example, fertilizer application has increased tenfold since 1990, reaching 3,000,000 tons in 2013 (CIAFA, 2013). Similarly, pesticide application rose from 150,000 to 330,000 tons between 2001 and 2011, with chlorpyrifos being the most widely applied insecticide in the country (CASAFE, 2013). In the HBLP, under-cover crops increased from ~ 25% of the total horticultural surface in 2005 to ~ 50% in 2015 (Baldini et al., 2021). This has led to a higher number of crop cycles per year, thus intensifying the application of agrochemicals. More than 160 different pesticides are used in HBLP, mainly insecticides and fungicides (DP, 2015).
Agrochemicals applied to crops can reach streams by runoff, which is considered one of the main sources of stream contamination (Schulz, 2001). Fertilizers might increase nutrient concentrations leading to the eutrophication of freshwater ecosystems. Pesticides are retained in bottom sediments (Friberg et al., 2003) and may affect epibenthic fauna (Liess et al., 2008; Egler et al., 2012; Schäfer et al., 2012). The most frequent pesticides detected in streams sediments of the study area are chlorpyrifos, endosulfan, and its degradation product, endosulfan-sulfate (Hunt et al., 2016; Solis et al., 2016; Silva-Barni et al., 2018). Other persistent organic compounds like dichlorodiphenyltrichloroethane (DDTs) are also detected in Pampean streams (González et al., 2013; Silva-Barni et al., 2016). Because of their high persistence, bioaccumulation, and biomagnification ability, they are considered a threat to the non-target biodiversity (Wania & MacKay, 1996).
Macroinvertebrates are an essential component of freshwater biota. They play a key role in stream functioning by recycling nutrients, promoting organic matter breakdown, feeding on primary producers and zooplankton, supplying food to higher trophic levels, and providing biodiversity to stream ecosystems (Covich et al., 1999; Graça, 2001; Macadam & Stockan, 2015). Macroinvertebrates are one of the most widely used indicators of water quality in streams because of their abundance, diversity, easy collection and identification (Barbour et al., 1999). Pesticides have been shown to affect the structure of macroinvertebrate communities by reducing the abundance of sensitive species and increasing the abundance of tolerant ones (Liess & von der Ohe, 2005; Schäfer et al., 2012; Beketov et al., 2013). For example, Genito et al. (2002) reported lower richness and abundance of sensitive taxa in the Susquehanna River basin (USA) with increased agricultural area in the basin. Egler et al. (2012) also reported reduced richness and abundance of most taxa when comparing agricultural and forested sites of three streams in Brazil. These effects were related to the expansion of agricultural land as well as to pesticide pollution. Hence, macroinvertebrate metrics (i.e., richness, density, equitability, diversity) and composition (i.e., different taxa) are implemented to address the anthropogenic impact on stream communities (Kenney et al., 2009). Previous research in the study area indicated that the macroinvertebrate community related to aquatic vegetation turned out to be a sensitive indicator of agricultural land use (Cortelezzi et al., 2013; Solis et al., 2016, 2017, 2018; Altieri et al., 2021).
Macroinvertebrates can also be classified into Functional Feeding Groups (FFGs) based on food acquisition strategies (Cummins et al., 2005; Cummins, 2018), which provide information about ecosystem functioning (Wallace & Webster, 1996; Heino, 2008). Both taxonomic and FFGs information represent complementary tools to assess land use impact on aquatic systems (Rawer-Jost et al., 2000; Cummins, 2016). Several studies reported differences in the composition of FFGs in agricultural and undisturbed streams (Helson & Williams, 2013; Fu et al., 2016; Solis et al., 2019). Typically, impacted sites showed higher abundances of collector-gatherers and reduced numbers of shredders and predators (Helson & Williams, 2013; Fu et al., 2016). Moreover, pesticides might affect sensitive shredders, which in turn could impair the leaf litter decomposition process (Schäfer et al., 2007). Therefore, changes in macroinvertebrate composition might also compromise ecosystem functions.
Agricultural intensification driven by the global food demand leads to increased degradation of natural environments. The impact of agriculture on freshwater ecosystems has been deeply studied (Allan, 2004; Davis et al., 2015). However, the impact of horticulture has received only little attention on a global scale, especially in South America. A few studies have described the impact of horticulture on water quality, finding increased nutrient concentrations on streams in Kenya (Muriithi & Yu, 2015) and China (Wang et al., 2019). Also, the presence of pesticides in streams draining horticultural basins was reported in Honduras (Kammerbauer & Moncada, 1998), Sweden (Kreuger et al., 2010) Australia (Wightwick et al., 2012; Allinson et al., 2014), and Argentina (Mac Loughlin et al., 2017). Still, the impact of horticulture on biological communities has not been studied in depth. In the previous research, Arias et al. (2020a) determined the differential composition of macroinvertebrates in horticultural streams compared with less disturbed basins. To our knowledge, except for this preliminary contribution, the impact of horticultural land use on streams remains underreported.
The aim of this study was, therefore, to assess the impact of horticultural land use on the taxonomic composition and the composition of functional feeding groups of macroinvertebrate assemblages in Pampean lowland streams. We compared the macroinvertebrate assemblages associated with aquatic vegetation, nutrients and pesticide concentrations between horticultural and less disturbed basins. The previous work in the study area reported pesticide presence and changes in macroinvertebrates composition in agricultural streams (Hunt et al., 2017; Solis et al., 2018). Changes in FFGs, such as increasing collector-gatherers and declining shredder abundances in agriculture compared to livestock and reserve streams, have also been reported for these streams (Solis et al., 2019). Accordingly, we assume that the effects of pesticides on macroinvertebrate composition in the horticultural streams would be similar to the ones observed in agricultural streams. We, therefore, hypothesized that horticulture changes the taxonomic and trophic composition of macroinvertebrate assemblages. We expected that horticultural streams would exhibit lower richness, density, and diversity than less disturbed streams.
Materials and methods
Study area
Six streams were studied within the Pampas ecoregion, a large grassland plain that covers the central area of Argentina. The climate is humid temperate, the mean annual air temperature is 16 °C, and rainfall ranges from 700 to 1200 mm (Morello et al., 2012). The Pampas are covered by loessic eolic sediments from the Holocene (Hurtado et al., 2006), and soils show a high organic matter content (Imbellone et al., 2010). Streams running through the Pampa plain are denominated “Pampean streams” because of several features that differentiate them from others (Feijoó & Lombardo, 2007). This region has a remarkably low slope (< 1%), and therefore, stream current flow is slow. The stream substrate is rather homogeneous and lacking pebbles or stones. Bottom sediments are composed of fine materials, mainly silt and clay (Feijoó & Lombardo, 2007). The studied streams are narrow (3–10 m) and shallow (0.1–0.4 m depth) and lack forested borders. Abundant and diverse aquatic macrophytes are developed in these streams, providing the habitat heterogeneity that structures biological communities rather than substrate type (Giorgi et al., 2005; Ferreiro et al., 2011; Cortelezzi et al., 2013). The most common macrophytes are the rooted emergent Ludwigia peploides (Kunth) P.H. Raven, Gymnocoronis spilantoides (D. Don ex Hook. & Arn.) DC., and Myriophyllum aquaticum (Vell.) Verdc, which develop dense patches on the margins and sometimes cover the entire stream surface (Arias, 2019).
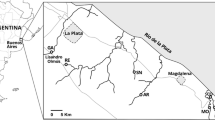
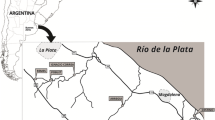
Three streams, Martin (34°55′20.80ʹʹS; 58°4′58.42ʹʹW), Gato (34°59′7.12ʹʹS; 58°3′16.17ʹʹW), and Sauce (35°1′42.52ʹʹS; 58°0′25.99ʹʹW), which run through the HBLP were sampled at sites adjacent to intensively cultivated plots (Fig. 1). These streams are referred to as “horticultural streams” hereinafter. The other three streams, Sin Nombre (35°2′23.04ʹʹS; 57°42′40.51ʹʹW), Destino (35°8′20.97ʹʹS; 57°23′28.08ʹʹW), and Morales (35°8′20.11ʹʹS; 57°24′0.23ʹʹW), are located in less impacted basins and are referred to as “less disturbed streams” hereinafter (Fig. 1). No horticultural activity is carried out in any of the latter basins (Arias, 2019); instead, extensive livestock raising on natural grasslands is the main land use. The sampling sites in Morales and Destino streams were located within the “Parque Costero del Sur” UNESCO Biosphere Reserve (Athor, 2009), while the Sin Nombre site was surrounded by cattle-raising fields.
Sampling design
Samplings were performed four times in each of the six studied streams (i.e., November 2016, December 2016, January 2017 and March 2017). A 50 m reach was sampled per stream. Samplings were carried out during the period of largest horticultural production, hence the period of largest agrochemical applications. In December 2016, the macrophyte cover in the Martin stream was scarce, and in March 2017, the Morales stream was almost dry; consequently, macroinvertebrates were not sampled. Therefore, a total of 22 samples, 11 for each land use, were available for the present study.
Environmental variables
Water temperature and dissolved oxygen were measured in situ with a Dissolved Oxygen Meter (YSI Model 51B), conductivity with a HI8733 Conductivity Meter (Hanna Instruments), and pH with a HI98103 Checker (Hanna Instruments). Vegetation cover was estimated visually, and depth was determined with a graduated rod in the same 1 m2 where macroinvertebrates were then sampled. Water samples were taken in triplicate in each sampling for suspended matter, chlorophyll, and nutrient analysis and were transported to the laboratory under refrigeration. Water was filtered through previously weighted Whatman GF/C fiberglass filters (1.2 µm) for soluble reactive phosphorus (SRP), ammonia, nitrite, and nitrate analytical determination, following APHA (2012). The suspended matter was determined by weight difference after filtration. Chlorophyll was extracted with 90% acetone and measured by the spectrophotometric method (Lorenzen, 1967).
Sediment samples were taken from the first 5 cm of the stream bottom with a stainless-steel scoop and transported to the laboratory under refrigeration. In the laboratory, samples were homogenized by hand, and sub-samples of 5 g were taken for organic matter analysis. Organic matter in sediment was determined by weight difference after being dried at 105 °C and muffled at 550 °C for 2 h (Heiri et al., 2000). Sediment sub-samples were frozen (− 20 °C) until pesticide analysis.
Pesticide analysis
Sediment samples (20 g) were extracted following You et al. (2004), with a mixture of dichloromethane and methanol. Cleanup procedures were performed under solid-phase extraction with Fluorisil®. Extracts were dried under a nitrogen flux and suspended in n-hexane (1 ml). Purification was carried out through silica gel chromatography.
Samples were analyzed for chlorpyrifos, endosulfans (α-, β- and endosulfan-sulfate), dichlorodiphenyltrichloroethanes (p,pʹ-DDT, p,pʹ-DDD and p,pʹ-DDE), hexachlorcyclohexanes (α- and γ-HCH), heptachlors (heptachlor and heptachlor epoxide), chlordanes (α- and γ- isomers), and dieldrin. Before injection, samples were treated with activated Cu for sulfur extraction, following Metcalfe & Metcalfe (1997) with modifications (Miglioranza et al., 2003). A Shimadzu–17A gas chromatograph with an electron capture detector (ECD) equipped with a Supelco DB-5 column (30 m × 0.25 mm i.d. × 0.25 μm film thickness; carrier gas: He; velocity: 1.5 ml min−1) was used in splitless mode. The temperature program was an initial 100 °C (1 min), an increase of 5 °C min−1 reaching 150 °C (1 min), and finally an increase of 1.5 °C min−1 until 240 °C. Quantification was performed using the analytical standards Ultra Scientific® and PCB #103 (Accustandard Absolute Standards®). Procedural and instrumental blanks were analyzed, and compound levels were below the detection limits. Recoveries of the internal standard (PCB #103) were estimated at values greater than 90%. Instrumental detection limits were calculated according to Keith et al. (1983). Concentrations and detection limits were reported in ng g−1 dry weight of the sediment samples (ng g−1 dw). Detection limits were 0.007–0.029 ng g−1 DW for all compounds. Two samples from the Gato stream and one from the Sin Nombre stream were not analyzed due to the highly organic matrix extracted. Therefore, 19 samples were available for pesticide analyses.
Macroinvertebrate assemblages
In each sampling, three macroinvertebrate samples were taken with a 500 μm D-net in a 1 m2 quadrant by sweeping the surface of the aquatic vegetation. Vegetation patches of similar size and composition within each stream were selected. Organisms were fixed in 96% ethanol and transported to the laboratory. Identification was performed under a Leica® EZ4 stereo microscope at the family level, except for Oligochaeta, Acari, Collembola and Copepoda, following Merrit et al. (2008) and Domínguez & Fernández (2009). Each sample was kept and analyzed separately and then averaged for calculations. The mean density of each taxon was determined for each stream per date. Identified macroinvertebrates were then classified following available information for South American taxa (Cummins et al., 2005; Tomanova et al., 2006; Ramírez & Gutiérrez-Fonseca, 2014), into five functional feeding groups: collector-filterers, collector-gatherers, scrapers, shredders, and predators.
Data analysis
Environmental variables were compared between land uses through Student’s t-tests; whenever variables did not meet the assumptions of normality or homogeneity, the Mann–Whitney U tests were performed instead. Seasonal differences were previously assessed; except for dissolved oxygen, no significant differences were detected for environmental variables between spring and summer therefore, we did not consider seasonal differences in environmental variables between the studied streams.
Pesticides were compared between land uses by Mann–Whitney U tests. Whenever the pesticide concentration fell below the detection limits, the lower value was used for statistical analysis (i.e., 0.007 µg L−1).
Macroinvertebrate metrics family richness (S), density (individuals m−2), evenness (Pielou index, Jʹ), and Shannon diversity index (Hʹ) were calculated using PRIMER (Plymouth Routines Multivariate Ecological Research, version 6) (Clarke & Gorley, 2001). Also, the richness and relative abundance of each functional feeding group (FFG) were calculated for each sample. Macroinvertebrate metrics and the relative abundance of each FFG were compared between land uses through the Student’s t-test or Mann–Whitney U test when variables did not meet the assumptions of normality or homogeneity. The relationships of metrics and FFGs to nutrients and pesticide concentrations were assessed through Pearson correlation analysis and plotted using corrplot (Wei et al., 2017) in R statistical software, version 4.1.2 (R Development Core Team, 2022).
For multivariate analyses, taxa present in only one sample were dismissed (Clarke & Warwick, 2001). Taxa density was transformed by log (x + 1) and FFGs relative abundances were transformed by the arc. sin function to reduce the contributions of very abundant taxa. The Bray–Curtis similarity index was then applied. The differences in assemblage composition and FFGs between land uses were analyzed with ANOSIM (factor: land use). Comparison between land uses was carried out without considering season as a factor since previous analysis with ANOSIM did not detect statistical differences between spring and summer. Percentage similarity (SIMPER) analysis was then applied to determine the contribution of each family or FFG to the dissimilarity between land uses. Finally, non-metric multidimensional scaling (NMDS) was performed to analyze the ordination of samples based on taxonomic or functional data. Multivariate analyses were performed using vegan (Oksanen et al., 2013) in R statistical software, version 4.1.2 (R Development Core Team, 2022).
Results
Environmental variables and pesticides
Table 1 shows the environmental variables measured in the horticultural and less disturbed streams. Nitrate, ammonium, and SRP were significantly higher in the horticultural than in the less disturbed streams (P = 0.012, P = 0.022, and P = 0.002, respectively), while suspended matter was higher in the less disturbed streams (P = 0.005).
Measured pesticide concentrations were roughly an order of magnitude higher in the horticultural than in the less disturbed streams (Table 2). Chlorpyrifos was determined in all analyzed samples and was the pesticide measured at the highest concentrations, the highest one in Martin horticultural stream (9127 ng g−1 dw) and the lowest one (6 ng g−1 dw) in the less disturbed Sin Nombre stream. Endosulfan-sulfate, total endosulfan, and p,p’-DDE concentrations were significantly higher in the horticultural streams (P = 0.008, P = 0.003, and P = 0.026, respectively).
Macroinvertebrate assemblages
A total of 13,706 specimens were counted, belonging to 18 orders and 51 families. Mean family richness was significantly lower in the horticultural (19 families) than in the less disturbed streams (28 families, P < 0.001; Fig. 2). Mean macroinvertebrate density was also significantly lower in the horticultural (273 ind m−2) than in the less disturbed streams (972 ind m−2, P < 0.001, Fig. 2). The lowest richness (11 families) and density (63 ind m−2) were observed in the horticultural Martin stream, while the largest richness (33 families) was attained in the less disturbed Sin Nombre stream. The highest density (1500 ind m−2) was determined in the less disturbed Destino stream, flowing within the Reserve. There were no statistical differences in diversity (Hʹ) and evenness (Jʹ) between land uses (Fig. 2).
Daphniidae (Cladocera) followed by Planorbidae (Gastropoda) and Hyallelidae (Amphipoda) were the most abundant taxa in the less disturbed streams. These families were present at low densities in the horticultural streams, where Entomobryoidea (Collembola) was dominant (Table 3).
Functional feeding groups
The relative abundance of collector-gatherers was significantly higher in the horticultural streams, while collector-filterers (P = 0.005) and scrapers (P = 0.026) were significantly lower than in the less disturbed streams. Collector-gatherers were dominant for both land uses; however, they represented 64% of the relative abundance in the horticultural streams and 26% in the less disturbed ones (Fig. 3).
The most abundant collector-gatherers in horticultural streams were Entomobryoidea and Cyprididae (Ostracoda), while in the less disturbed streams they were Oligochaeta, Hydrophilidae (Coleoptera), and Chironomidae (Diptera). Predators were the most diverse group (21 families), with Coenagrionidae (Odonata) and Dytiscidae (Coleoptera) being dominant in the less disturbed streams, and Dugesiidae (Platyhelminthes) being dominant in the horticultural streams. For both land uses, collector-filterers, scrapers, and shredders characteristically showed Daphniidae, Planorbidae, and Hyallelidae, respectively, attaining higher densities in the less disturbed streams (Table 3).
Correlation analysis
Macroinvertebrates metrics showed negative correlations with nutrients and pesticides. Richness (S) was negatively correlated to nitrate, SRP and chlorpyrifos, while density (N) was negatively correlated to endosulfan-sulfate, nitrate and SRP. Regarding functional groups, collector-filterers (CF) were negatively correlated to nitrite, SRP, endosulfans, and DDTs, and scrapers (SC) were negatively correlated to α-endosulfan. Collector-gatherers (CG) were positively related to SRP, nitrite, endosulfans, and DDTs, and negatively correlated to macroinvertebrates metrics. In addition, endosulfan-sulfate and DDE showed positive correlations with nitrate and SRP (Fig. 4).
Correlation plot of nutrients, pesticides, macroinvertebrate metrics and relative densities of FFGs. N-NO2−: nitrite; N-NO3−: nitrate; N-NH4+: ammonia; SRP: soluble reactive phosphorus; endosulfan-S: endosulfan sulfate; DDE: p.p’-DDE; DDT: p.p’-DDT; S: family richness; N: density (individuals m−2); J’: evenness; H’: Shannon diversity; CF: collector-filterers; CG: collector-gatherers; PR: predators; SC: scrapers; SH: shredders. (*) indicates significant correlations at P < 0.05
Multivariate analysis
The taxonomic composition of the assemblages was significantly different between land uses (ANOSIM: Global R = 0.70, P = 0.001). SIMPER analysis showed an average dissimilarity of 67% (Table 4). Planorbidae, Daphniidae, Entomobrioidea, and Hyalellidae were the families that contributed the most to the dissimilarity between land uses.
ANOSIM based on FFGs also showed significant differences between land uses (Global R = 0.49; P = 0.001) attaining a dissimilarity of 30%. The NMDS analysis showed the different assemblage compositions, with the horticultural sites showing more dispersion than the less disturbed sites in both approaches. Taxonomic data separate land uses, while functional data showed less dispersion in less disturbed sites and overcrossing samples between land uses (Fig. 5).
Discussion
The present results support our hypothesis that horticulture affects the stream macroinvertebrate community and might compromise the functioning of these streams.
The studied streams are located within the same geomorphological region, and therefore, most of the measured environmental variables were not different among land uses. However, the suspended matter was higher in the less impacted streams. The reserve was surrounded by livestock areas, and cattle had access to the stream channel. Cattle might cause the removal of sediment from the bottom, resuspending fine particles into water, which could have triggered the higher suspended matter found in these streams. Similar interpretations were made by Molina et al. (2017), who reported higher suspended matter content in streams surrounded by cattle-raising areas than those in cropped basins.
The horticultural streams showed higher nutrients and some pesticide concentrations compared to the less disturbed ones. Pesticides are often applied together with fertilizers and transported into streams by runoff (Mugni et al., 2013), which was also reported for horticultural basins (Mangiafico et al., 2009; Palma et al., 2010). Moreover, nutrients and pesticides showed inverse correlations with most of the community attributes; the lowest richness and density were recorded in the stream where the highest pesticide concentrations were detected. Nutrients interact with pesticides showing antagonistic and synergistic effects on stream communities in mesocosm studies (Alexander et al., 2013; Barmentlo et al., 2018). However, macroinvertebrates assemblage showed no differences in abundance, dominance, or diversity before and after experimental fertilization in a Pampean stream (Cortelezzi et al., 2015). Moreover, higher nutrient concentrations in the horticultural streams did not cause an increase in chlorophyll concentrations. We suggest that the inverse correlations between nutrients and pesticides with richness or density indicate the incorporation of fertilizers and pesticides into streams, thus reflecting the impact of horticulture.
Chlorpyrifos was ubiquitous and was the pesticide detected at higher concentrations, which is consistent with the fact of being the most applied insecticide in Argentina (CASAFE, 2013). Its half-life in sediments was reported between 24 and 223 days (Bondarenko & Gan, 2004). Endosulfan (α- and β- and endosulfan-sulfate) and DDTs were also frequently detected. Endosulfan isomers and their derivate products (half-lives 6 months; Silva & Beauvais, 2010) were forbidden in Argentina since 2013 (SENASA, 2011), which enhanced the utilization of other products, like chlorpyrifos. DDT is still measured in the bottom sediments of the Pampean streams because of the extremely long persistence (half-life in soil is 2–15 years; EPA, 2022).
At environmentally relevant concentrations pesticides might affect feeding (Nyman et al., 2013), growth (Singh et al., 2021), or reproduction of macroinvertebrates. In laboratory studies, chlorpyrifos (5 µg L−1) reduced hatching and prolonged the time of hatching of snails (Rivadeneira et al., 2013), while endosulfan (0.2–0.4 ug L−1) reduced by 30–40% the number of gravid decapods females (Wirth et al., 2002). Effects on reproduction might change population dynamics and, consequently, alter the community composition (Schäfer et al., 2011).
We suggest that horticulture changes the composition of macroinvertebrate assemblages, by reducing the richness or abundance of several taxa considered sensitive and increasing the abundance of those considered tolerant. Reduced richness is a common pattern in macroinvertebrate assemblages exposed to pesticides in field studies on agricultural basins (Jergentz et al., 2004; Egler et al., 2012; Beketov et al., 2013; Rizo-Patrón et al., 2013). Hunt et al. (2017) described that the SPEAR index (SPEcies At Risk; Liess & von der Ohe, 2005) decreased with increasing pesticide concentrations in sediments in intensively cropped areas in Argentina. Similar findings were described by Münze et al. (2015) in streams in Germany, where the reduced abundances of sensitive species were related to pesticide toxicity. In addition, Macchi et al. (2018) determined high concentrations of chlorpyrifos in irrigation channels of fruit orchards in Río Negro province, Argentina, together with reduced taxon richness compared with upstream, non-cropped areas.
The macroinvertebrate assemblages were different between land uses: While Planorbidae, Hyallelidae, and Daphniidae were abundant in the less disturbed streams, they were strongly reduced in the horticultural sites. Hyalellidae species are sensitive to pesticides (Mugni et al., 2011; Peluso et al., 2013), and are considered indicators of water quality (Graça et al., 2002; Di Marzio et al., 2005). Daphniidae species are also highly sensitive (Zhou et al., 2008; Rubach et al., 2010; Rico & van den Brink, 2015; Arias et al., 2020b). The reduced density of Hyallelidae and Daphniidae in the horticultural streams was presumably caused by agrochemical exposure. Although usually considered tolerant to pollution, freshwater snails like Planorbidae might be vulnerable to stressors because of their low dispersion ability, according to Rico and van der Brink (2015). Entomobryoidea, Cyprididae, and Dugessidae were the most abundant taxa in the horticultural streams. Dugesiidae is considered tolerant to pesticides by Liess & von der Ohe (2005). Similarly, Egler et al. (2012) determined higher densities of Collembola in agricultural streams than in forestry and pasture streams in Brazil. The higher density of Entomobryoidea might also reflect faster recolonization after toxic pulses and/or higher availability of resources because of the disappearance of sensitive taxa, as reported in soil by Endlweber et al. (2006).
All functional feeding groups were present in both land uses. However, their relative abundances were different, being more equally distributed in the less disturbed streams and dominated by collector-gatherers in horticultural streams. The changes in the relative abundances of FFG suggest changes in ecosystem functions (Cummins, 2018). Collector-filterers, mainly represented by cladocerans, were abundant in the less disturbed streams and reduced in the horticultural streams, and they were negatively correlated to nutrients. This finding could be attributed to the low availability of food related to the lower suspended matter content in horticultural streams, in addition to the high sensitivity of cladocerans. Scrapers were also reduced in horticultural streams. Collector-gatherers were positively correlated to pesticides and nutrients, suggesting an increase in fine organic matter processing in horticultural streams. Together with the reduced abundance of scrapers, the higher relative abundance of collector-gatherers suggests a shift to heterotrophy (Cummins et al., 2005). In this sense, the large decrease in amphipod density in the horticultural streams suggests a reduced coarse organic matter breakdown rate (Piscart et al., 2009; Swan et al., 2021). Predators changed their composition from a dominance of Coenagrionidae, considered sensitive, in the less impacted streams, to Dugesiidae, considered tolerant, in the horticultural streams. Together with sensitivity, the lower macroinvertebrate density diminished prey availability, likely contributing to the decreased predators’ relative abundance in the horticultural streams. Further studies are needed for a better understanding of the impacts of horticultural practices on the functionality of these streams, particularly the effect of pesticides in processes like primary production and decomposition. This would contribute to the development of management measures for freshwater ecosystems.
The use of family–level taxonomic resolution allowed us to detect differences in assemblage composition when comparing land use. However, different species within a family might show different feeding strategies (Ramírez & Gutierrez-Fonseca, 2014). Identification to lower taxonomic levels together with broader information about the trophic ecology of neotropical taxa would allow the determination of FFGs more accurately and would improve the knowledge of functional attributes of these streams.
Increasing temperatures and rainfalls due to climate change are predicted for the Pampean region (Barros et al., 2013). Consequently, an increase in surface runoff events is also expected (Rodrigues Capitulo et al., 2010; Andrade et al., 2021). The HBLP has expanded in recent years and production has intensified (Baldini et al. 2021); thus, agrochemicals use is expected to increase as well. These projections threaten the integrity of the streams in the region, both for water quality and for the structure and function of these ecosystems (Rodrigues Capitulo et al., 2010).
The present results highlight the need for the implementation of mitigation measures in surface waters. Riparian buffer zones are one of the most simple and cost-effective mitigation strategies (Reichenberger et al., 2007). They have been proven to reduce stream contamination by retaining sediments and nutrients (Vought et al., 1995) and have been implemented as an effective measure to protect streams from runoff in Brazil (Hunt et al., 2017) and Finland (Kuglerová et al., 2020). In the Pampean region, riparian zones can retain up to 74% of glyphosate in experimental systems (Giaccio et al., 2016). Riparian wetlands also reduced pesticide pollution and increased the abundance of macroinvertebrates in a Pampean stream (Solis et al., 2021). Restoration of deteriorated riparian zones and preservation of wetlands, together with restrictions on pesticide use, are necessary for the protection of the structure and function of streams in food production regions.
Conclusions
Horticultural production in the Pampean region increases nutrient and pesticide concentrations, affecting taxonomic and trophic macroinvertebrate composition by reducing richness and density. Overall, horticulture affects community structure and functioning, producing a shift towards detritivory. Macroinvertebrates provide information about the structure and function of regional ecosystems. Taxonomic and functional assessments evidenced the effect of land use on the macroinvertebrate community. Macroinvertebrates resulted in a useful tool to address the impact of land uses and particularly, the effect of pesticides. Our results strongly emphasize the need to improve mitigation measures and restrictions on pesticide use along with the development of sustainable production systems, to preserve freshwater environments in food production regions. Future studies should focus on the effect of land uses and pesticides on the functioning of Pampean streams.
Data availability
We declare that results are supported by data. The datasets analyzed during the current study are not published but are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Alexander, A. C., A. T. Luis, J. M. Culp, J. D. Baird & A. J. Cessna, 2013. Can nutrients mask community responses to insecticide mixtures? Ecotoxicology 22: 1085–1100.
Allan, J. D., 2004. Landscapes and riverscapes: The Influence of Land Use on Stream Ecosystems. Annual Review of Ecology, Evolution, and Systematics 35: 257–284.
Allinson, G., A. Bui, P. Zhang, G. Rose, A. M. Wightwick, M. Allinson & V. Pettigrove, 2014. Investigation of 10 herbicides in surface waters of a horticultural production catchment in southeastern Australia. Archives of Environmental Contamination and Toxicology 67: 358–373.
Altieri, P., L. E. Paz, R. F. Jensen, J. Donadelli & A. Rodrigues Capitulo, 2021. Transplanting macrophytes as a rehabilitation technique for lowland streams and their influence on macroinvertebrate assemblages. Anais Da Academia Brasileira De Ciências. https://doi.org/10.1590/0001-3765202120191029.
Andrade, V. S., M. F. Gutierrez, L. Regaldo, A. R. Paira, M. R. Repetti & A. M. Gagneten, 2021. Influence of rainfall and seasonal crop practices on nutrient and pesticide runoff from soybean dominated agricultural areas in Pampean streams. Argentina. Science of the Total Environment 788: 147676.
APHA, 2012. American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 22nd ed., Washington DC.
Arias, M., 2019. Efecto del uso de fertilizantes y pesticidas sobre organismos acuáticos en arroyos de la zona hortícola de La Plata. Doctoral Thesis, Universidad Nacional de La Plata, Argentina. 147 pp.
Arias, M., A. Scalise, M. Solis, A. Paracampo, M. Indaco, S. Fanelli, H. Mugni & C. Bonetto, 2020a. Horticulture affects macroinvertebrate assemblages in adjacent streams (Buenos Aires, Argentina). Knowledge & Management of Aquatic Ecosystems 421: 5.
Arias, M., C. Bonetto & H. Mugni, 2020b. Sublethal effects on Simocephalus vetulus (Cladocera: Daphnidae) of pulse exposures of cypermethrin. Ecotoxicology and Environmental Safety 196: 110546.
Athor, J., 2009. Parque Costero del Sur—Naturaleza, conservación y patrimonio cultural, Fundación de Historia Natural “Félix de Azara,” Buenos Aires:
Baldini, C., E. M. Marasas & A. A. Drozd, 2021. Three decades of landscape change across the largest peri-urban horticultural region of Argentina: urban growth, productive intensification and the need for resilient landscape management. Journal of Environmental Planning and Management 1: 40.
Barbier, E. B., 2004. Explaining Agricultural Land Expansion and Deforestation in Developing Countries. American Journal of Agricultural Economics 86: 1347–1353.
Barbour, M. T., J. Gerritsen, B. D. Snyder & J. B. Stribling, 1999. Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish, 2nd ed. U.S. Environmental Protection Agency, Washington:
Barmentlo, S. H., M. Schrama, E. R. Hunting, R. Heutink, P. M. Van Bodegom, G. R. de Snoo & M. G. Vijver, 2018. Assessing combined impacts of agrochemicals: Aquatic macroinvertebrate population responses in outdoor mesocosms. Science of the Total Environment 631: 341–347.
Barros, V. R., C. R. Garavaglia & M. E. Doyle, 2013. Twenty-first century projections of extreme precipitations in the Plata Basin. International Journal of River Basin Management 11: 373–387.
Beketov, M. A., B. J. Kefford, R. B. Schäfer & M. Liess, 2013. Pesticides reduce regional biodiversity of stream invertebrates. Proceedings of the National Academy of Science of the United States 110: 11039–11043.
Bondarenko, S. & J. Gan, 2004. Degradation and sorption of selected organophosphate and carbamate insecticides in urban stream sediments. Environmental Toxicology and Chemistry: an International Journal 23: 1809–1814.
CASAFE 2013. Cámara de Sanidad Agropecuaria y Fertilizantes, Buenos Aires, Argentina. Retrieved July 16, 2020, from http://www.casafe.org/publicaciones/estadisticas/
Castagnino, A. M., K. Díaz, J. Fernandez Lozano, A. P. Guisolis, O. Liverotti, M. B. Rosini & S. Sasale, 2020. Panorama of the argentine horticultural sector: 1. Characterization and priorities of national horticulture. Horticultura Argentina 39: 76–102.
CIAFA, 2013. Cámara de la Industria Argentina de Fertilizantes y Agroquímicos. Retrieved from http://www.ciafa.org.ar
Clarke, K. R. & R. N. Gorley, 2001. PRIMER v5: User Manual/Tutorial, PRIMER-E, Plymouth:
Clarke, K. R. & R. M. Warwick, 2001. Change in marine communities: an approach to statistical analysis and interpretation, 2nd ed. PRIMER-E, Plymouth:
Cortelezzi, A., M. V. Sierra, N. Gómez, C. Marinelli & A. Rodrigues Capítulo, 2013. Macrophytes, epipelic biofilm, and invertebrates as biotic indicators of physical habitat degradation of lowland streams (Argentina). Environmental Monitoring and Assessment 185: 5801–5815.
Cortelezzi, A., C. Ocón, M. V. Oosterom, R. Cepeda & A. R. Capítulo, 2015. Nutrient enrichment effect on macroinvertebrates in a lowland stream of Argentina. Iheringia. Série Zoologia 105: 228–234.
Covich, A. P., M. A. Palmer & T. A. Crowl, 1999. The role of benthic invertebrate species in freshwater ecosystems. BioScience 49: 119–127.
Cummins, K. W., 2016. Combining taxonomy and function in the study of stream macroinvertebrates. Journal of Limnology 75: 235–241.
Cummins, K. W., 2018. Functional Analysis of Stream Macroinvertebrates. In Gokce, D. (ed), Limnology-Some New Aspects of Inland Water Ecology. Intech Open, London.
Cummins, K. W., R. W. Merritt & P. C. Andrade, 2005. The use of invertebrate functional groups to characterize ecosystem attributes in selected streams and rivers in south Brazil. Studies on Neotropical Fauna and Environment 40: 69–89.
Davis, J., A. P. O’Grady, A. Dale, A. H. Arthington, P. A. Gell, P. D. Driver, N. Bond, M. Casanova, M. Finlayson, R. J. Watts, S. J. Capon, I. Nagelkerken, R. Tingley, B. Fry, T. J. Page & A. Specht, 2015. When trends intersect: The challenge of protecting freshwater ecosystems under multiple land use and hydrological intensification scenarios. Science of the Total Environment 534: 65–78.
Di Marzio, W. D., M. Sáenz, J. Alberdi, M. Tortorelli & G. Silvana, 2005. Risk assessment of domestic and industrial effluents unloaded into a freshwater environment. Ecotoxicology and Environmental Safety 61: 380–391.
Dolédec, S. & B. Statzner, 2010. Responses of freshwater biota to human disturbances: contribution of J-NABS to developments in ecological integrity assessments. Journal of the North American Benthological Society 29: 286–311.
Domínguez, E. & H. R. Fernández (eds), 2009. Macroinvertebrados bentónicos. Sistemática y Biología, 1ª edición. Fundación Miguel Lillo, Tucumán.
DP, 2015. Defensoría del Pueblo de la Provincia de Buenos Aires. Relevamiento de la utilización de agroquímicos en la Provincia de Buenos Aires. Mapa de situación e incidencia sobre la salud. Retrieved April 20, 2020, from https://www.agro.unlp.edu.ar/sites/default/files/paginas/informe_agroquimicos_comprimido.pdf
Dudgeon, D., 2019. Multiple threats imperil freshwater biodiversity in the Anthropocene. Current Biology 29(19): R960–R967.
Egler, M. A., D. F. Buss, J. C. Moreira & D. F. Baptista, 2012. Influence of agricultural land use and pesticides on benthic macroinvertebrate assemblages in an agricultural river basin in southeast Brazil. Brazilian Journal of Biology 72: 437–443.
Endlweber, K., M. Schädler & S. Scheu, 2006. Effects of foliar and soil insecticide applications on the collembolan community of an early set-aside arable field. Applied Soil Ecology 31: 136–146.
EPA, 2022. Environmental Protection Agency, United States. Ingredients used in pesticide products. Retrieved from https://www.epa.gov/ingredients-used-pesticide-products
FAO, 2013. Food and Agriculture Organization. Statistical Year Book, World Food and Agriculture, Food and Agriculture Organization of the United Nations, Rome:, 307.
FAOSTAT, 2020. Food and Agriculture Data. Food and Agriculture Organization of the United Nations. Retrieved April 20, 2020, from http://www.fao.org/faostat/en/#data
Feijoó, C. S. & R. J. Lombardo, 2007. Baseline water quality and macrophyte assemblages in Pampean streams: a regional approach. Water Research 41: 1399–1410.
Ferreiro, N., C. Feijoó, A. Giorgi & L. Leggieri, 2011. Effects of macrophyte heterogeneity and food availability on structural parameters of the macroinvertebrate community in a Pampean stream. Hydrobiologia 664: 199–211.
Foley, J. A., R. DeFries, G. P. Asner, C. Barford & G. P. K. BonanSnyder, 2005. Global consequences of land use. Science 309: 570–574.
Friberg, N., M. Lindstrøm, B. Kronvang & S. E. Larsen, 2003. Macroinvertebrate/sediment relationships along a pesticide gradient in Danish streams. In Kronvang, B. (ed), The Interactions between Sediments and Water Developments in Hydrobiology, Springer, Dordrecht: 169. https://doi.org/10.1007/978-94-017-3366-3_15.
Fu, L., Y. Jiang, J. Ding, Q. Liu, Q. Z. Peng & M. Y. Kang, 2016. Impacts of land use and environmental factors on macroinvertebrate functional feeding groups in the Dongjiang River basin, southeast China. Journal of Freshwater Ecology 31: 21–35.
Genito, D., W. J. Gburek & A. N. Sharpley, 2002. Response of Stream Macroinvertebrates to Agricultural Land Cover in a Small Watershed. Journal of Freshwater Ecology 17: 109–119.
Giaccio, G. C. M., P. Laterra, V. C. Aparicio & J. L. Costa, 2016. Glyphosate retention in grassland riparian areas is reduced by the invasion of exotic trees. Phyton - International Journal of Experimental Botany 85: 108–116.
Giorgi, A., C. Feijoo & G. Tell, 2005. Primary producers in a Pampean stream: temporal variation and structuring role. Biodiversity and Conservation 14: 1699–1718.
González, M., K. S. B. Miglioranza, S. I. Grondona, M. F. Silva-Barni, D. E. Martinez & A. Peña, 2013. Organic pollutant levels in an agricultural watershed: the importance of analyzing multiple matrices for assessing streamwater pollution. Environmental Science: Processes & Impacts 15: 739.
Graça, M. A. S., 2001. The role of invertebrates on leaf litter decomposition in streams – a review. International Review of Hydrobiology 86(4–5): 383–393.
Graça, M. A. S., A. Rodrı́gues-Capı́tulo, C. Ocón & N. Gómez, 2002. In situ tests for water quality assessment: a case study in Pampean rivers. Water Research 36: 4033–4040.
Gücker, B., I. G. Boëchat & A. Giani, 2009. Impacts of agricultural land use on ecosystem structure and whole-stream metabolism of tropical Cerrado streams. Freshwater Biology 54: 2069–2085.
Heino, J., 2008. Patterns of functional biodiversity and function-environment relationships in lake littoral macroinvertebrates. Limnology and Oceanography 53: 1446–1455.
Heiri, O., A. F. Lotter & G. Lemcke, 2000. Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. Journal of Paleolimnology 25: 101–110.
Helson, J. E. & D. D. Williams, 2013. Development of a macroinvertebrate multimetric index for the assessment of low-land streams in the neotropics. Ecological Indicators 29: 167–178.
Hunt, L., C. Bonetto, V. H. Resh, D. F. Buss, S. Fanelli, N. Marrochi & M. J. Lydy, 2016. Insecticide concentrations in stream sediments of soy production regions of South America. Science of the Total Environment 547: 114–124.
Hunt, L., C. Bonetto, N. Marrochi, A. Scalise, S. Fanelli, M. Liess, M. Lydy, C. Chiu & V. H. Resh, 2017. Species at Risk (SPEAR) index indicates effects of insecticides on stream invertebrate communities in soy production regions of the Argentine Pampas. Science of the Total Environment 580: 699–709.
Hurtado, M. A., J. E. Gimenez & M. G. Cabral, 2006. Análisis ambiental del partido de La Plata: Aportes al ordenamiento territorial, 1ra edición, Consejo Federal de Inversiones, Buenos Aires:
Imbellone, P. A., J. E. Giménez & J. L. Panigatti, 2010. Suelos de la Región Pampeana: Procesos de Formación, ED INTA, Buenos Aires:, 320.
Jergentz, S., H. Mugni, C. Bonetto & R. Schulz, 2004. Runoff-related endosulfan contamination and aquatic macroinvertebrate response in rural basins near Buenos Aires, Argentina. Archives of Environmental Contamination and Toxicology 46: 345–352.
Jordan, T. E., D. L. Correll & D. E. Weller, 1997. Relating nutrient discharges from watersheds to land use and streamflow variability. Water Resources Research 33: 2579–2590.
Kammerbauer, J. & J. Moncada, 1998. Pesticide residue assessment in three selected agricultural production systems in the Choluteca River Basin of Honduras. Environmental Pollution 103: 171–181.
Keesstra, S. D., J. van Huissteden, J. Vandenberghe, O. Van Dam, J. de Gier & I. D. Pleizier, 2005. Evolution of the morphology of the river Dragonja (SW Slovenia) due to land-use changes. Geomorphology 69: 191–207.
Keith, L. H., W. Crummett, J. Deegan, R. A. Libby, J. K. Taylor & G. Wentler, 1983. Principles of environmental analysis. Analytical Chemistry 55: 2210–2218.
Kenney, M. A., A. E. Sutton-Grier, R. F. Smith & S. E. Gresens, 2009. Benthic macroinvertebrates as indicators of water quality: The intersection of science and policy. Terrestrial Arthropod Reviews 2: 99.
Kreuger, J., S. Graaf, J. Patring & S. Adielsson, 2010. Pesticides in surface water in areas with open ground and greenhouse horticultural crops in Sweden. Ekohydrologi 117: 49.
Kuglerová, L., J. Jyväsjärvi, C. Ruffing, T. Muotka, A. Jonsson, E. Andersson & J. S. Richardson, 2020. Cutting edge: A comparison of contemporary practices of riparian buffer retention around small streams in Canada, Finland, and Sweden. Water Resources Research 56: e2019WR026381.
Langhammer, J., 2003. Antropogenic transformation of River Network in the Otava River Basin. Acta Universitatis Carolinae Geographica 2: 139–156.
Liess, M. & P. O. von der Ohe, 2005. Analyzing effects of pesticides on invertebrate communities in streams. Environmental Toxicology and Chemistry 24: 954–965.
Liess, M., R. B. Schäfer & C. A. Schriever, 2008. The footprint of pesticide stress in communities—species traits reveal community effects of toxicants. Science of the Total Environment 406: 484–490.
Lorenzen, C. J., 1967. Determination of chlorophyll and phaeopigments: spectrophotometric equations. Limnology and Oceanography 12: 343–346.
Mac Loughlin, T. M., L. Peluso & D. Marino, 2017. Pesticide impact study in the peri-urban horticultural area of Gran La Plata, Argentina. Science of the Total Environment 598: 572–580.
Macadam, C. R. & J. A. Stockan, 2015. More than just fish food: ecosystem services provided by freshwater insects. Ecological Entomology 40: 113–123.
Macchi, P., R. M. Loewy, B. Lares, L. Latini, L. Monza, N. Guiñazú & C. M. Montagna, 2018. The impact of pesticides on the macroinvertebrate community in the water channels of the Río Negro and Neuquén Valley, North Patagonia (Argentina). Environmental Science and Pollution Research 25: 10668–10678.
Mangiafico, S. S., J. Newman, D. J. Merhaut, J. Gan, B. Faber & L. Wu, 2009. Nutrients and pesticides in stormwater runoff and soil water in production nurseries and citrus and avocado groves in California. HortTechnology 19: 360–367.
Merrit, R. W., K. W. Cummins & M. B. Berg, 2008. An Introduction to the Aquatic Insects of North America, Kendall-Hunt, Dubuque:
Metcalfe, T. L. & C. D. Metcalfe, 1997. The trophodynamics of PCBs including mono and non-ortho congeners in the food web of north-Central Lake Ontario. Science of the Total Environment 201: 245–272.
Miglioranza, K. S. B., J. E. Aizpún de Moreno & V. J. Moreno, 2003. Dynamics of organochlorine pesticides in soils from a southeastern region of Argentina. Environmental Toxicology and Chemistry 22: 712–717.
Miserendino, M. L., R. Casaux, M. Archangelsky, C. Di Prinzio, C. Brand & A. M. Kutschker, 2011. Assessing land-use effects on water quality, in-stream habitat, riparian ecosystems and biodiversity in Patagonian northwest streams. Science of the Total Environment 409: 612–624.
Molina, M. C., C. A. Roa-Fuentes, J. O. Zeni & L. Casatti, 2017. The effects of land use at different spatial scales on instream features in agricultural streams. Limnologica 65: 14–21.
Morello, J., S. D. Matteucci, A. F. Rodriguez, M. E. Silva, P. Mesopotámica & P. Llana, 2012. Ecorregiones y complejos Ecosistémicos de Argentina, Orientación Gráfica Editora, Buenos Aires:
Mugni, H., A. Ronco & C. Bonetto, 2011. Insecticide toxicity to Hyalella curvispina in runoff and stream water within a soybean farm (Buenos Aires, Argentina). Ecotoxicology and Environmental Safety 74: 350–354.
Mugni, H., A. Paracampo & C. Bonetto, 2013. Nutrient concentrations in a pampasic first order stream with different land uses in the surrounding plots (Buenos Aires, Argentina). Bulletin of Environmental Contamination and Toxicology 91: 391–395.
Münze, R., P. Orlinskiy, R. Gunold, A. Paschke, O. Kaske, M. A. Beketov, M. Hundt, C. Bauer, G. Schüürmann, M. Möder & M. Liess, 2015. Pesticide impact on aquatic invertebrates identified with Chemcatcher® passive samplers and the SPEARpesticides index. Science of the Total Environment 537: 69–80.
Muriithi, F. K. & D. Yu, 2015. Understanding the impact of intensive horticulture land-use practices on surface water quality in Central Kenya. Environments 2: 521–545.
Narangarvuu, D. C., B. Hsu, S. H. Shieh, F. C. Wu & P. S. Yang, 2014. Macroinvertebrate assemblage patterns as indicators of water quality in the Xindian watershed, Taiwan. Journal of Asia-Pacific Entomology 17: 505–513.
Nessimian, J. L., E. M. Venticinque, J. Zuanon, P. De Marco, M. Gordo, L. Fidelis & L. Juen, 2008. Land use, habitat integrity, and aquatic insect assemblages in Central Amazonian streams. Hydrobiologia 614: 117.
Nyman, A. M., A. Hintermeister, K. Schirmer & R. Ashauer, 2013. The insecticide imidacloprid causes mortality of the freshwater amphipod Gammarus pulex by interfering with feeding behavior. PLoS ONE 8: e62472.
Oksanen, J., F. G. Blanchet, R. Kindt, P. Legendre, P. R. Minchin, R. B. O’hara, G. L. Simpson, P. Solymos, Henry, M., H. Stevens & H. Wagner, 2013. Vegan: Community Ecology Package. R package version 2.0–7. http://CRAN.R-project.org/package=vegan
Palma, P., P. Alvarenga, V. Palma, C. Matos, R. M. Fernandes, A. Soares & I. R. Barbosa, 2010. Evaluation of surface water quality using an ecotoxicological approach: a case study of the Alqueva Reservoir (Portugal). Environmental Science and Pollution Research 17: 703–716.
Peluso, L., G. B. Rossini, A. Salibián & A. Ronco, 2013. Physicochemical and ecotoxicological based assessment of bottom sediments from the Luján River basin, Buenos Aires, Argentina. Environmental Monitoring and Assessment 185: 5993–6002.
Piscart, C., R. Genoel, S. Dolédec, E. Chauvet & P. Marmonier, 2009. Effects of intense agricultural practices on heterotrophic processes in streams. Environmental Pollution 157: 1011–1018.
R Development Core Team. (2022). R: a Language and Environment for Statistical Computing. R Foundation for Statistical Computing. https://www.R-project.org/
Ramírez, A. & P. E. Gutiérrez-Fonseca, 2014. Functional feeding groups of aquatic insect families in Latin America: a critical analysis and review of existing literature. Revista De Biología Tropical 62: 155–167.
Rawer-Jost, C., J. Böhmer, J. Blank & H. Rahmann, 2000. Macroinvertebrate functional feeding group methods in ecological assessment. Hydrobiologia 422: 225–232.
Reichenberger, S., M. Bach, A. Skitschak & H. G. Frede, 2007. Mitigation strategies to reduce pesticide inputs into ground-and surface water and their effectiveness; a review. Science of the Total Environment 384(1–3): 1–35.
Rico, A. & P. J. van den Brink, 2015. Evaluating aquatic invertebrate vulnerability to insecticides based on intrinsic sensitivity, biological traits, and toxic mode of action. Environmental Toxicology and Chemistry 34: 1907–1917.
Rivadeneira, P. R., M. Agrelo, S. Otero & G. Kristoff, 2013. Different effects of subchronic exposure to low concentrations of the organophosphate insecticide chlorpyrifos in a freshwater gastropod. Ecotoxicology and Environmental Safety 90: 82–88.
Rizo-Patrón, F. V., A. Kumar, M. B. M. Colton, M. Springer & F. A. Trama, 2013. Macroinvertebrate communities as bioindicators of water quality in conventional and organic irrigated rice fields in Guanacaste, Costa Rica. Ecological Indicators 29: 68–78.
Rodrigues Capitulo, A., N. Gómez, A. Giorgi & C. Feijoo, 2010. Global changes in pampean lowland streams (Argentina): implications for biodiversity and functioning. Hydrobiologia 657: 53–70.
Roy, A. H., A. D. Rosemond, M. J. Paul, D. S. Leigh & J. B. Wallace, 2003. Stream macroinvertebrate response to catchment urbanisation (Georgia, U.S.A.). Freshwater Biology 48: 329–346.
Rubach, M. N., D. J. Baird & P. J. van den Brink, 2010. A new method for ranking mode-specific sensitivity of freshwater arthropods to insecticides and its relationship to biological traits. Environmental Toxicology and Chemistry 29: 476–487.
Schäfer, R. B., T. Caquet, K. Siimes, R. Mueller, L. Lagadic & M. Liess, 2007. Effects of pesticides on community structure and ecosystem functions in agricultural streams of three biogeographical regions in Europe. Science of the Total Environment 382: 272–285.
Schäfer, R. B., P. J. van den Brink & M. Liess, 2011. Impacts of pesticides on freshwater ecosystems. In Sánchez-Bayo, F., P. J. van der Brink & R. M. Mann (eds), Ecological impacts of toxic chemicals Bentham Science Publishers Ltd., USA: 111–137.
Schäfer, R. B., C. P. von der Ohe, J. J. Rasmussen, B. J. Kefford, M. A. Beketov, R. Schulz & M. Liess, 2012. Thresholds for the effects of pesticides on invertebrate communities and leaf breakdown in stream ecosystems. Environmental Science & Technology 46: 5134–5142.
Schulz, R., 2001. Rainfall-induced sediment and pesticide input from orchards into the Lourens River, Western Cape, South Africa: importance of a single event. Water Research 35: 1869–1876.
SENASA (2011). Servicio Nacional de Sanidad y Calidad Agropecuaria. Resolution 511/2011. Retrieved from http://www.senasa.gov.ar/
Silva, M. & S. Beauvais, 2010. Risk assessment for acute, subchronic, and chronic exposure to pesticides: Endosulfan. In Krieger, R. (ed), Hayes’ Handbook of Pesticide Toxicology Academic Press, London: 499–522.
Silva-Barni, M. F., P. M. Ondarza, M. Gonzalez, R. Da Cuña, F. Meijide, F. Grosman & K. S. Miglioranza, 2016. Persistent organic pollutants (POPs) in fish with different feeding habits inhabiting a shallow lake ecosystem. Science of the Total Environment 550: 900–909.
Silva-Barni, M. F., M. Gonzalez, F. Wania, Y. D. Lei & K. S. Miglioranza, 2018. Spatial and temporal distribution of pesticides and PCBs in the atmosphere using XAD-resin based passive samplers: a case study in the Quequén Grande River watershed, Argentina. Atmospheric Pollution Research 9: 238–245.
Singh, D. S., M. Alkins-Koo, L. V. Rostant & A. Mohammed, 2021. Sublethal effects of malathion insecticide on growth of the freshwater crab Poppiana dentata (Randall, 1840) (Decapoda: Trichodactylidae). Nauplius. https://doi.org/10.1590/2358-2936e2021030.
Solis, M., H. Mugni, L. Hunt, N. Marrochi, S. Fanelli & C. Bonetto, 2016. Land use effect on invertebrate assemblages in Pampasic streams (Buenos Aires, Argentina). Environmental Monitoring and Assessment 188: 539.
Solis, M., H. Mugni, S. Fanelli & C. Bonetto, 2017. Effect of agrochemicals on macroinvertebrate assemblages in Pampasic streams, Buenos Aires, Argentina. Environmental Earth Sciences 76: 1–13.
Solis, M., C. Bonetto, N. Marrochi, A. Paracampo & H. Mugni, 2018. Aquatic macroinvertebrate assemblages are affected by insecticide applications on the Argentine Pampas. Ecotoxicology and Environmental Safety 148: 11–16.
Solis, M., M. Arias, S. Fanelli, C. Bonetto & H. Mugni, 2019. Agrochemicals’ effects on functional feeding groups of macroinvertebrates in Pampas streams. Ecological Indicators 101: 373–379.
Solis, M., N. Cappelletti, C. Bonetto, M. Franco, S. Fanelli, J. Amalvy & H. Mugni, 2021. Attenuation of insecticide impact by a small wetland in a stream draining a horticultural basin in Argentina. Science of the Total Environment 785: 147317.
Swan, C. M., L. Boyero & C. Canhoto, 2021. The ecology of plant litter decomposition in stream ecosystems, Springer, Switzerland:, 523.
Thomas, K. E., R. Lazor, P. A. Chambers & A. G. Yates, 2018. Land-use practices influence nutrient concentrations of southwestern Ontario streams. Canadian Water Resources Journal 3: 2–17.
Tilman, D., 1999. Global environmental impacts of agricultural expansion: the need for sustainable and efficient practices. Proceedings of the National Academy of Sciences 96: 5995–6000.
Tomanova, S., E. Goitia & J. Helešic, 2006. Trophic levels and functional feeding groups of macroinvertebrates in neotropical streams. Hydrobiologia 556: 251–264.
Vought, L. B. M., G. Pinay, A. Fuglsang & C. Ruffinoni, 1995. Structure and function of buffer strips from a water quality perspective in agricultural landscapes. Lands U Plan 31: 323–331.
Wallace, J. B. & J. R. Webster, 1996. The role of macroinvertebrates in stream ecosystem function. Annual Review of Entomology 41: 115–139.
Wang, R., J. Min, H. J. Kronzucker, Y. Li & W. Shi, 2019. N and P runoff losses in China’s vegetable production systems: loss characteristics, impact, and management practices. Science of the Total Environment 663: 971–979.
Wania, F. & D. MacKay, 1996. Tracking the distribution of persistent organic pollutants. Environmental Science and Technology 30: 390A-396A.
Wei, T., V. Simko, M. Levy, Y. Xie, Y. Jin & J. - Zemla, 2017. Package ‘corrplot.’ Statistician 56(316): e24.
Wightwick, A. M., A. D. Bui, P. Zhang, G. Rose, M. Allinson, J. H. Myers & G. Allinson, 2012. Environmental fate of fungicides in surface waters of a horticultural-production catchment in southeastern Australia. Archives of Environmental Contamination and Toxicology 62: 380–390.
Wirth, E. F., S. A. Lund, M. H. Fulton & G. I. Scott, 2002. Reproductive alterations in adult grass shrimp, Palaemonetes pugio, following sublethal, chronic endosulfan exposure. Aquatic Toxicology 59: 93–99.
You, J., D. P. Weston & M. J. Lydy, 2004. A sonication extraction method for the analysis of pyrethroid, organophosphate, and organochlorine pesticides from sediment by gas chromatography with electron-capture detection. Archives of Environmental Contamination and Toxicology 47: 141–147.
Young, R. G. & A. D. Huryn, 1999. Effects of land use on stream metabolism and organic matter turnover. Ecological Applications 9: 1359–1376.
Zhou, Q., J. Zhang, J. Fu, J. Shi & G. Jiang, 2008. Biomonitoring: an appealing tool for assessment of metal pollution in the aquatic ecosystem. Analytica Chimica Acta 606: 135–150.
Acknowledgements
The authors thank the reviewers and the editor for their valuable comments and suggestions. We thank Paula Altieri, Carolina Ocón, and Joaquín Cochero for their suggestions in data analysis. We also thank Juan Martin Paredes del Puerto for improving the artwork. We thank our colleagues Hannah Chmiel for proofreading and Collins Ogbeide for the language edition as a native speaker.
Funding
This work was supported by the National Scientific and Technical Research Council (CONICET) (PIP 2011-0180), the Argentine National Agency for the Promotion of Science and Technology (ANPCyT) (PICT 2010-0446), and the National University of La Plata (UNLP) (N772). Marina Arias was granted a scholarship from the National Scientific and Technical Research Council (CONICET).
Author information
Authors and Affiliations
Contributions
MA contributed to conceptualization, methodology, formal analysis, investigation, writing; CB contributed to conceptualization, writing, funding acquisition, supervision; SLF contributed to methodology; LS contributed to methodology; KSBM contributed to methodology. HM contributed to conceptualization, investigation, funding acquisition, supervision.
Corresponding author
Ethics declarations
Conflict of interest
This research does not present conflict of interest, and publication elsewhere has not been considered.
Ethical approval
Not applicable.
Additional information
Handling editor :Verónica Ferreira
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arias, M., Bonetto, C., Fanelli, S.L. et al. Macroinvertebrate assemblages in lowland streams under horticultural impact (Buenos Aires, Argentina). Hydrobiologia 850, 399–416 (2023). https://doi.org/10.1007/s10750-022-05081-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-05081-7