Abstract
Agriculture and livestock may contribute to water quality degradation in adjacent waterbodies and produce changes in the resident invertebrate composition. The objective of the present study was to assess land use effects on the stream invertebrate assemblages in rural areas of the Argentine Pampa. The four sampling events were performed at six sites in four streams of the Pampa plain; two streams were sampled inside a biosphere reserve, and another one was surrounded by extensive livestock fields. The fourth stream was sampled at three sites; the upstream site was adjacent to agricultural plots, the following site was adjacent to an intensive livestock plot and the downstream site was adjacent to extensive breeding cattle plots. Higher pesticide concentrations were found at the site adjacent to agricultural plots and higher nutrient concentrations at the sites adjacent to agricultural and intensive breeding cattle plots. The invertebrate fauna were also different at these sites. Multivariate analysis showed a relationship between nutrient concentrations and taxonomic composition. Amphipoda (Hyalella curvispina) was the dominant group in the reserve and extensive breeding cattle sites, but was not present in the agricultural site. Also, Chironomidae were absent from the agricultural site while present at other sites. Gasteropoda (Biomphalaria peregrina), Zygoptera, and Hirudinea were dominant at the most impacted agricultural and intensive breeding cattle sites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Agriculture (croplands and pasture) represents the world’s largest terrestrial biome (Stehle and Schulz 2015). Non-point source contamination with agrochemicals applied in agricultural production is increasingly recognized as the main cause of water quality degradation in inland waters (Schulz 2004). Deterioration of aquatic environments represents at present the largest challenge for the sustainability of ongoing agricultural intensification (Stehle & Schulz 2015). Runoff is an important route of entry pesticides and exposure scenario for the non-target fauna (Schulz 2001). As a consequence, the highest pesticide concentrations occur as short-term exposure pulses during precipitation events, and the effect of pesticides is expected to be higher in small streams due to a close proximity to the crop (Schulz 2004). The overall effect of agrochemicals on the resident communities is difficult to assess because of the ephemeral nature of the exposure and the natural variability of the assemblages (Beketov and Liess 2008; Beketov et al. 2009). Variation in the sensitivity of different species suggests that exposure should result in changes in species composition of invertebrate assemblages, and therefore the relative proportion of different taxonomic groups may be used in evaluation of environmental impact (Azrina et al. 2006). Several studies have shown an effect of insecticide exposure on species composition in streams from Europe, Asia, and Australia (Schafer et al. 2012). Increased nutrient loads to aquatic ecosystems cause euthrophication (Chalar et al. 2011) and might also affect the invertebrate assemblages (Nordin 1985). Biomass increase (Bourassa and Cattaneo 1998), changes in the invertebrate community structure (Allan 2004; Chambers et al. 2006), and loss of diversity (Nijboer and Verdonschot 2004) have been reported.
Agricultural practices have been intensified in recent decades in Argentina. Traditionally, farmers employed a mixed system of livestock and wheat/corn production. The genetically modified glyphosate-resistant soybean was introduced into the market in 1996 and its cultivation steadily increased to attain at present roughly half the total agricultural production in Argentina (MAGyP 2015). Argentina is the world’s third largest producer and exporter of soy, after the USA and Brazil. Genetically modified soy represents 95 % of the total soy production in Argentina (Bindraban et al. 2009). Wheat and soy varieties with a short growing period allow two harvests per year, wheat followed by soy. Livestock has been moved to marginal areas or concentrated in intensive production systems. Pesticide consumption increased from 6 million kilograms in 1992 (Pengue 2000) to 32 million kilograms in 2012 (CASAFE 2013).
Repeated agrochemical applications represent a risk to adjacent waterbodies. The overall effect of the present agriculture intensification on the resident aquatic fauna in Argentine remains unreported. The objective of the present contribution was to assess the effect of land use on the water quality and the invertebrate assemblages of adjacent streams in the Argentine Pampa plain.
Materials and methods
Study sites
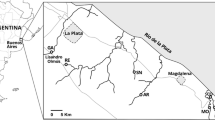
Four streams were studied in the Rio de la Plata coastal fringe, between La Plata and Magdalena, in Buenos Aires province, Argentina (Fig. 1). The streams run parallel west-east direction towards the Rio de la Plata. The climate is mild and humid; mean monthly temperatures range from 9.9 °C in July to 22.4 °C in January. Mean annual rainfall is 1010 mm featuring small seasonal variations (Hurtado et al. 2006).
The streams were sampled four times throughout the soybean growing period, in December 2011, January, March, and April 2012. Three sites were sampled along the Pescado stream, which has a length of 36 km and a basin of 211 km2, (Hurtado et al. 2006). An upstream site was sampled at Remes (35° 1′ 31.87″ S; 57° 59′ 39.6″ W), where the stream is surrounded by intensively cultivated plots. Soy is the main crop, although wheat, corn, and vegetables (mainly tomatoes) are also grown. Soil is cultivated up to a few meters from the stream. At Poblet, 3.8 km downstream, (35° 2′ 2.45″ S; 57° 56′ 34.3″W), the stream runs through a breeding cattle plot, which contained high cow densities throughout the studied period. The stream was the only water source for cattle. The cattle moved freely within the plot and gathered along the stream banks for drinking. This site will be called hereafter the “intensive livestock” site. At Ignacio Correa (35° 1′23.97″S; 57°51′27.42″W), 8 km downstream, the stream is deeper and wider (Table 1), and the adjacent land use is breeding cattle at low densities, over natural grasslands without fertilization. A fence separates the livestock plots from the stream leaving a 5–20 m strip of ungrazed grassland. The Arregui stream (35° 7′ 22.1″ S; 57° 41′ 11.6″W) has a basin which is similar to the landscape described for the Pescado lower stretch at Ignacio Correa, and adjacent land use is breeding cattle at low densities on natural grasslands. The stream was separated from the cattle plots by a fence leaving 5–20 m strip of ungrazed grassland. Ignacio Correa and Arregui will be referred to hereafter as “extensive livestock” sites. Juan Blanco (35° 8′30.23″S; 57°26′23.98″W) and Destino (35° 7′ 53.10″S; 57° 24′ 1.47″W) streams run through the biosphere reserve “Parque Costero Sur”. The landscape is grassland with small patches of forest. All studied streams have small slope and borders covered by riparian vegetation, including emergent (mainly Typha and Scirpus), submerged (mainly Potamogeton, Ceratophyllum, and Egeria), and also floating vegetation (mainly Eichornia, Lemna, and Azolla).
Environmental variables
Dissolved oxygen and temperature (YSI 51B), conductivity (Hanna instruments 8733), and pH (Orion 250 A) in stream water were measured in situ. Water samples were immediately filtered through Whatman GF/C filters, and carried in coolers to the laboratory. Suspended matter (SM) was determined as the weight difference after filtration. Dissolved nutrients were determined in the filtrate. Soluble reactive phosphorus (SRP, molybdate-ascorbic), nitrate (NO3 −, hydrazine reduction followed by diazotation); ammonium (NH4 +, indophenol blue), and bicarbonate (HCO3 −, Gran titration) were determined following APHA (2005).
Pesticide analysis
Cypermethrin, chlorpyrifos, and endosulfan concentration in bottom sediments from the studied streams were reported by Hunt et al. (2016). Sediment samples were collected with a stainless steel scoop from the top 2 cm, and placed in pesticide-free amber glass jars with Teflon lids, which were kept in coolers on ice until arrival at the laboratory where they were kept refrigerated until extraction (maximum of 5 d). Aliquots were taken from each sample for total organic carbon and grain size analysis. Sediments were extracted with a mixture of acetone and methylene chloride following You et al. (2004b). Clean-up procedures were carried out using Envi-Carb II/primary—secondary amine solid-phase extraction (SPE) cartridges. Insecticides analysis was performed on an Agilent 6890 series GC equipped with an Agilent 7683 autosampler and a micro-ECD (Agilent Technologies, Palo Alto, CA, USA). A detailed methodological description is provided by Hunt et al. (2016).
Macroinvertebrate sampling
Macroinvertebrates were sampled from emergent vegetation by means of a D-net of 500 μm pore size, 30 cm diameter, and a net opening area of approximately 600 cm2. At each site, three sweeps were collected, covering an area of approximately 1 m2 per sample. Samples were preserved with 80 % alcohol and taken to the laboratory for sorting. All invertebrates were later identified under a stereoscopic microscope. Taxa were identified to family or genus following Domínguez & Fernández (2009) and Merritt et al. (2008).
Statistical analysis
Environmental measurements at the different sites were compared by means of the analysis of variance (ANOVA) followed by the Tukey test at a significance level of p ≤ 0.05. Macroinvertebrate assemblage data were analyzed using the multivariate statistical package PRIMER version 5 (Clarke & Gorley, 2001). Total abundance for each taxon was estimated and reported in terms of number of individuals per surface unit (ind/m2). Data were transformed by a log (x + 1) function to reduce the contribution of the dominant groups. Data were analyzed using the similarity index of Bray & Curtis (1957). With the object of assessing habitat delimitation as a function of land use, a cluster analysis was performed. Differences among sites were assessed by means of the analysis of similarity (ANOSIM). The percentage of similarity (SIMPER) was calculated to assess the taxa that contributed most to the differences in the assemblages among sites (Clarke & Warwick, 2001). Relationships between environmental variables and taxonomic composition were studied by multivariate analysis using the CANOCO program, version 4.53 (ter Braak & Smilauer 1998). The taxa with total relative abundance lower than 0.1 % were deleted from the analysis to reduce the influence of rare species. All data used were transformed by a log (x + 1) function. A detrended correspondence analysis (DCA) was performed, followed by a redundancy analysis RDA (Ter Braak & Smilauer 1998; Leps & Smilauer 2003). The global significance of the model was tested for all the canonical axes and for the first axis alone. Finally, RDA was performed with the environmental variables to assess their contributions to the taxonomic variations and the differences among sites.
Results
Environmental variables
No significant differences were detected in the conductivity, pH, and suspended matter among the different sites (Table 1). Mean water pH ranged from neutral (7.1, Remes) to moderately alkaline (7.9, Juan Blanco). Conductivity ranged from 195 μS/cm (Ignacio Correa) to 813 μS/cm (Arregui).
Nutrient concentrations showed significant differences among sites. SRP concentrations were significantly higher at the three sites in Pescado stream than at the other streams: Arregui, Blanco, and Destino (ANOVA, Tukey p ≤ 0,005). Nitrite concentrations were significantly higher in Poblet than in Arregui and Ignacio Correa (ANOVA, Tukey p ≤ 0,005). Apparently higher nitrate and ammonium concentrations in the Pescado stream were not statistically significant.
Pesticides
Endosulfan and cypermethrin were not detected in the streams located within the reserve or draining basins with extensive livestock (Table 2). Chlorpyrifos was sometimes detected at low concentrations in these streams. By contrast, in the Pescado stream, all three analyzed insecticides were detected; concentrations being highest at Remes, adjacent to agricultural plots, and decreasing downstream. Endosulfan sulfate concentrations were higher than those of endosulfan. Both endosulfan and endosulfan sulfate concentrations were higher than those of cypermethrin and chlorpyrifos. Endosulfan sulfate in Remes was an order of magnitude higher than those in Poblet, 3.8 km downstream, and fell below detectable limits in Ignacio Correa, 8 km downstream.
Macroinvertebrate assemblages
Fifty-four taxa were identified throughout the studied period (Table 3). Trichoptera was detected only in the reserve. The family Hyalellidae was the dominant taxon in the reserve and extensive livestock sites. By contrast, Hyalellidae was absent from Remes, and present at low densities in Poblet. Heteroptera was poorly represented at the former site and abundant in the latter. Chironomidae was also absent from Remes while present at all other sites. In the Pescado stream Platyhelminthes (Dugesiidae), Annelida (Hirudinea), and Gastropoda (Planorbidae, B. peregrina) were the groups better represented. At Remes, Mollusca was the dominant group. Dugesiidae was most abundant at Ignacio Correa and Coleoptera in Arregui.
Comparison of assemblages at different sites
The cluster analysis registered differences among the invertebrate assemblages in sites with different land use (Fig. 2). A group was formed by all samples from Remes and one sample at Poblet. Another group was formed with the samples from livestock sites. The samples from the reserve sites join together in two smaller groups.
The ANOSIM analysis showed significant differences among the different sites (Global R = 0,408; p = 0,001). Significant differences were detected among pairs formed by sites of contrasting land use: Remes was significantly different from the reserve and extensive breeding sites (Table 4), Poblet was significantly different from the extensive breeding sites and Arregui was significantly different from Juan Blanco.
The SIMPER analysis identified 20 taxa as the main contributors to the dissimilarity among sites, which results were significantly different among the sampling sites in relation to adjacent land use (Table 5).
Relations of the assemblage to environmental variables
The multivariate analysis (RDA) showed that bicarbonate, nutrients, pH, stream width, stream depth, and suspended matter correlated significantly with the canonical axes explaining 56 % of the overall variability (p = 0.006). However, because several variables contribute similarly to the overall variability, it is difficult to discern, at this level of resolution, any clear temporal or spatial pattern in the invertebrate assemblages.
Considering that the three contrasting land uses (agriculture, intensive livestock, and extensive livestock) were present at different sites of the same stream, the multivariate analysis was repeated for the three sites of Pescado stream (Remes, Poblet, and Ignacio Correa). SRP, stream depth, suspended matter, nitrite, and ammonium were significantly correlated with the canonic axes (p ≤ 0.006). These variables explain 72 % of the total variance of the first two axes. The first axis (eigenvalue 0.37) explains the variability given by the taxonomic abundance; PRS (−0.74) and depth (0.71) were correlated with this axis. Nitrite (0.45), ammonium (0.40), and suspended matter (0.63) were correlated with the second axis.
Figure 3 shows that at the Remes agricultural site, high PRS, and low inorganic nitrogen concentrations coincide with the dominance of Scirtidae, B. peregrina, and P. canaliculata. The intensive livestock site Poblet has high inorganic nitrogen and suspended matter, and intermediate PRS concentrations, with dominance of Zygoptera, Notonecta, Baetidae, Hirudinea, Belostoma, Libelullidae, and Chironomidae, while the extensive livestock site Ignacio Correa shows lower nutrient concentration, higher depths and dominance of Curculionidae, Hyalellidae, Anacaenini, Stratiomydae, Ephydridae, and Platyhelminthe.
Discussion
SRP concentrations were significantly higher in the stream with agriculture in its upper basin. Mugni et al. (2013) measured higher PRS concentrations in a first order stream when wheat/soy was grown and fertilized with 120 kg/ha of diamonic phosphate in the surrounding plot than in the following year, when unfertilized soy was grown. Hart et al. (2004) measured SRP concentrations five times higher in a first order stream when the surrounding pasture was fertilized with 4 monthly doses of 50 kgP/ha, and peak SRP concentrations (3.3 mg/L) coincided with rains that occurred within a few days after applications. Mugni (2009) showed that the higher SRP concentrations in the Pescado stream at Remes were contributed by the runoff events occurring after fertilization of adjacent crops. Consistently, Castela et al. (2008) showed that the benthic macroinvertebrate community correlated negatively with increasing SRP concentration and changes in the riparian vegetation.
Downstream of Remes, at Poblet, nitrite concentrations were significantly higher than in the other sites and high ammonium concentrations were measured in coincidence with a drought in January 2012. The higher ammonium concentration measured at Poblet was likely not transported from upstream, as the concentration measured at Remes was lower in January. The stream was the only source of water for the cattle, which gathers in the stream to drink. Livestock trampling deteriorated the riparian vegetation, and cattle manure accumulated in the riparian buffer zone. Mugni et al. (2013) reported higher inorganic nitrogen concentrations in a first order stream surrounded by cattle than in an upstream site adjacent to unfertilized soy, in Arrecifes (Argentina). Concentrations were also higher than the next year at the same site, when unfertilized soy was grown.
Pesticides were detected in bottom sediments; higher concentrations were measured at Remes, decreasing downstream. Higher concentrations of endosulfan than chlorpyrifos and cypermethrin are likely a consequence of its longer persistence and higher application rates (Leonard et al. 2000). Higher concentrations of endosulfan sulfate than endosulfan concentrations were measured. Similarly, Leonard et al. (1999) determined higher endosulfan sulfate concentrations in a river several weeks after endosulfan application in the basin. Both endosulfan and endosulfan sulfate concentrations measured in Remes on December 2011 were higher than the LC50 of Chironomus tentans in sediments (You et al. 2004a); consistent with the absence of Chironomids in Remes while they are present in all other studied sites, where insecticide concentrations were lower or undetectable.
H. curvispina has repeatedly been used as a sentinel organism for environmental impact assessment in Pampasic streams because of its extensive geographic distribution, its commonly high abundance and its high sensitivity to pesticide exposure (Jergentz et al. 2004b). Mugni et al. (2011) reported the occurrence of toxicity pulses to H. curvispina in stream and runoff water in coincidence with the first rains after pesticide application in the adjacent crop at Remes, the same site sampled in the present work. Jergentz et al. (2004a) detected toxicity pulses associated to rain events in other Pampasic streams during the period of insecticide application. Mugni et al. (2012) and Paracampo et al. (2012) studied the toxicity persistence in runoff water from experimental soy plots following endosulfan, chlorpyrifos, and cypermethrin crop application, and reported that runoff water was toxic to H. curvispina up to roughly 1 month after application. Higher endosulfan sulfate than endosulfan concentrations suggest that the insecticide was applied several weeks before being transported to the stream by runoff (Leonard et al. 1999). Soy is sown in October–November, and pesticides are applied at time of sowing. The last rain before the sampling amounted to 38 mm and occurred on 20/11/2011. Therefore, the pesticides detected in the stream bottom in December were likely introduced into the stream at least 2 weeks earlier. Runoff contribution undergoes several processes in the stream such as downstream transport, sedimentation, and decay. Therefore, exposure at the runoff event was higher than that measured on the sampling date. Available information is consistent with the absence of H. curvispina in Remes, while it was the dominant taxon in the reserve and extensive livestock sites, and suggests insecticide toxicity as the cause.
Leonard et al. (1999) reported that the density of Ephemeroptera and Trichoptera in the Namoi River were negatively correlated with the presence of endosulfan from cotton crops within its basin. Castillo et al. (2006) studied the invertebrate fauna of a stream adjacent to a banana plantation in Costa Rica. Pesticides were detected in stream water at concentrations lower than reported lethal concentrations for crustaceans in the literature. However, they reported changes in the invertebrate assemblages after field applications and differences in comparison with nearby streams taken as reference sites. They reported among other changes, an increase in gastropods proportion, amounting to 40 % of the total abundance in the impacted sites. Similarly, in the present study, gasteropoda comprised 57 % of the total invertebrate abundance in Remes, compared to 1–8 % at the less impacted sites.
The composition of invertebrate assemblages is commonly used to assess environmental impact because species composition shifts in response to perturbations acting as continuous monitoring of the resident site and allowing an overall evaluation of impact (Hering et al., 2004).
Liess & Von der Ohe (2005) studied pesticide concentrations and invertebrate assemblages in streams from Braunschweig, Germany, and classified the invertebrate fauna according to sensitivity to pesticides. They showed that measured insecticide concentrations as low as 1/100 LC50 for D. magna caused significant changes in the invertebrate composition. Schäfer et al. (2007) analyzed the invertebrate community of streams in France and Finland concluding that pesticide exposition caused a decrease in the most sensitive taxa. Schäfer et al. (2012) compiled data from field studies in Europe, Siberia, and Australia concluding that the abundances of sensitive macroinvertebrates in the communities were reduced by 27 to 61 % at concentrations less than 1/100 of the LC50 for D. magna. It is assumed that the changes in the community composition occur because of differential sensitivity to agrochemical exposure at low concentrations.
The families that were well represented in the invertebrate assemblages of Remes in the present study (B. peregrina (Planorbidae), Scirtidae, Hirudinea, P. canaliculata (Ampullariidae), Aeshnidae, Dugesiidae, Libelullidae) were considered as tolerant by Liess & Von der Ohe (2005). On the contrary, families considered as sensitive by Liess & Ohe (2005) as Caenagrionidae, Corixidae, Stratiomydae, and Caenidae were absent or poorly represented at the Remes site.
Conclusions
Land use in the basin affects the invertebrate assemblage composition in Pampasic streams. Higher pesticide concentrations were determined in sites adjacent to agriculture. Higher insecticide concentration resulted from adjacent crop applications. Higher SRP concentrations in the agricultural site were related with the soy fertilization. Being a symbiotic atmospheric nitrogen fixer, soy is not fertilized with N. Higher inorganic N at the site with adjacent intensive livestock was related with the cattle contribution through manure. The invertebrate assemblage composition in these sites was significantly different from those located in a reserve and in basins with extensive breeding cattle. Extensive livestock over natural pastures separated from the stream by a fence leaving a pasture ungrazed strip did not produce discernible effects in the invertebrate assemblages. Present evidence suggest that exposure to agrochemicals is the main cause of the observed differences in the invertebrate assemblage composition. Available information suggest that the ongoing agricultural intensification induces a change towards a fauna with increased abundance of Planorbidae, Ampullariidae, Hirudinea, and Aeshnidae, and the absence of Hyalellidae, Chironomidae, Caenagrionidae, Caenidae, Polycentropodidae, and Stratiomydae. It is suggested that these taxa represent good candidates for being utilized as regional indicators of agrochemical contamination.
References
Allan, D. (2004). Landscapes and riverscapes: the influence of land use on stream ecosystems. Annual Review of Ecology, Evolution, and Systematics, 35, 57–84. doi:10.1146/annurev.ecolsys.35.120202.110122.
APHA. American Public Health Association (2005). Standard methods for the examination of water and wastewater (20 ed.). Washington, DC: Am. Public Health Assoc.
Azrina, M. Z., Yap, C. K., Rahim Ismail, A., Ismail, A., & Tan, S. G. (2006). Anthropogenic impacts on the distribution and biodiversity of benthic macroinvertebrates and water quality of the Langat River, Peninsular Malaysia. Ecotoxicology and Environmental Safety, 64, 337–347.
Beketov, M. A., & Liess, M. (2008). An indicator for effects of organic toxicants on lotic invertebrate communities: independence of confounding environmental factors over an extensive river continuum. Environmental Pollution, 156, 980–987.
Beketov, M. A., Foit, K., Schafer, R. B., Schriever, C. A., Sacchi, A., Capri, E., Biggs, J., Wells, C., & Liess, M. (2009). SPEAR indicates pesticide effects in streams—comparative use of species- and family-level biomonitoring data. Environmental Pollution, 157, 1841–1848.
Bindraban, P. S., Franke, A. C., Ferraro, D. O., Ghersa, C. M., Lotz, L. A. P., Nepomuceno, A., Smulders, M. J. M., van de Wiel, C. C. M. (2009). G-M related sustainability: agro ecological impacts, risks and opportunities of soy production in Argentina and Brazil. Plant Research International B.V., Wageningen. 56 pp.
Bourassa, N., & Cattaneo, A. (1998). Control of periphyton biomass in Laurentian streams. Journal of the North American Benthological Society, 17, 420–429.
Bray, J. R., & Curtis, J. T. (1957). An ordination of the upland forest communities in southern Wisconsin. Ecological Monographs, 27, 325–349.
CASAFE, 2013. Cámara de Sanidad Agropecuaria y Fertilizantes, Buenos Aires, Argentina. http://www.casafe.org/sobrelaindustria.htm. Accessed on November 2013.
Castela, J., Ferreira, V., & Graça, M. (2008). Evaluation of stream ecological integrity using litter decomposition and benthic invertebrates. Environmental Pollution, 153, 440–449.
Castillo, L. E., Martinez, E., Ruepert, C., Savage, C., Gilek, M., Pinnock, M., & Solis, E. (2006). Water quality and macroinvertebrate community response following pesticide applications in a banana plantation, Limon. Costa Rica. Sci. Total Environ., 367, 418–432.
Chalar, G., Arocena, R., Pacheco, J. P., & Fabián, D. (2011). Trophic assessment of streams in Uruguay: a trophic State Index for Benthic Invertebrates (TSI-BI). Ecol. Indic., 11, 362–369.
Chambers, P. A., Meissner, R., Wrona, F. J., Rupp, H., Guhr, H., Seeger, J., Culp, J. M., & Brua, R. B. (2006). Changes in nutrient loading in an agricultural watershed and its effects on water quality and stream biota. Hydrobiologia, 556, 399–415.
Clarke, K. R., Gorley, R. N. (2001). PRIMER v5: User Manual/Tutorial PRIMER-E: Plymouth Routines in Multivariate Ecological Research, 91 pp.
Clarke, K. R., Warwick, R. M. (2001). Change in marine communities, an approach to statistical analysis and interpretacion, 2nd Edition. PRIMER-E: Plymouth Routines in Multivariate Ecological Research.
Dominguez, E., Fernandez, H. R. (2009). Macroinvertebrados bentónicos. Sistemática y biología. 1ª ed. Tucumán. Fundación Miguel Lillo, 656 p.
Hart, M. R., Quin, B. F., & Nguyen, M. L. (2004). Phosphorus runoff from agricultural land and direct fertilizer effects: a review. Journal of Environmental Quality, 33, 1954–1972.
Hering, D., Moog, O., Sandin, L., & Verdonschot, P. (2004). Overview and application of the AQEM assessment system. Hydrobiología, 516, 1–20.
Hunt, L., Bonetto, C., Resh, V. H., Forsin Buss, D., Fanelli, S., Marrochi, N., & Lydy, M. J. (2016). Insecticide concentrations in stream sediments of soy production regions of South America. The Science of the Total Environment, 547, 114–124.
Hurtado, M. A., Giménez, J. E., Cabral, M. G. (2006). Análisis ambiental del partido de La Plata: Aportes al ordenamiento territorial. 1ª ed. Buenos Aires: Consejo Federal de Inversiones, 125p.
Jergentz, S., Mugni, H., Bonetto, C., & Schulz, R. (2004a). Runoff-related endosulfan contamination and aquatic macroinvertebrate response in rural basins near Buenos Aires. Argentina. Arch. Environ. Contam. Toxicol., 46(3), 345–353.
Jergentz, S., Pessacq, P., Mugni, H., Bonetto, C., & Schulz, R. (2004b). Linking in situ bioassays and dynamics of macroinvertebrates to assess agricultural contamination in streams of the Argentine pampa. Ecotoxicology and Environmental Safety, 59, 133–141.
Leonard, A. W., Hyne, R. V., Lim, R. P., Pablo, F., & Van der Brink, P. J. (2000). Riverine endosulfan concentration in the Namoi River, Australia: link to cottton field runoff and macroinvertebrate population densities. Envrion. Toxicol. Chem., 19, 1540–1551.
Leonard, A., Hyne, V., Lim, R., & Chapman, J. (1999). Effect of endosulfan runoff from cotton fields on macroinvertebrates in the Namoi River. Ecotoxicol. Envrion. Saf., 42, 125–134.
Leps, J., & Smilauer, P. (2003). Multivariate analysis of ecological data using CANOCO. Cambridge: Cambridge University Press.
Liess, M., & Von der Ohe, P. (2005). Analyzing effects of pesticides on invertebrate communities in streams. Environmental Toxicology and Chemistry, 24, 954–965.
MAGyP (2015). Ministerio de Agricultura, Ganadería y Pesca Series y Estadísticas http://old.siia.gov.ar/index.php/series-por-tema/agricultura Accessed on November 2015
Merritt, R. W., Cummins, K. W., & Berg, M. B. (2008). An introduction to the aquatic insects of North America (p. 1158). Dubuque, Iowa: Kendall-Hunt.
Mugni, H. (2009). Concentración de nutrientes y toxicidad de pesticidas en aguas superficiales de cuencas rurales. Tesis doctoral, Universidad de La Plata. 140 pp.
Mugni, H., Demetrio, P., Paracampo, A., Pardi, M., Bulus, G., & Bonetto, C. (2012). Toxicity persistence in runoff water and soil in experimental soybean plots following chlorpyrifos application. Bulletin of Environmental Contamination and Toxicology, 89, 208–212. doi:10.1007/s00128-012-0643-6.
Mugni, H., Paracampo, A., & Bonetto, C. (2013). Nutrient concentrations in a Pampasic first order stream with different land uses in the surrounding plots (Buenos Aires, Argentina) bull. Environ. Contam. Toxicol., 91(4), 391–395. doi:10.1007/s00128-013-1079-3.
Mugni, H., Ronco, A., & Bonetto, C. (2011). Insecticide toxicity to Hyalella curvispina in runoff and stream water within a soybean farm (Buenos Aires, Argentina). Ecotoxicol. Environ. Saf., 74, 350–354. doi:10.1016/j.ecoenv.2010.07.030.
Nijboer, R. C., & Verdonschot, P. F. M. (2004). Variable selection for modelling effects of eutrophication on stream and river ecosystems. Ecological Modelling, 177, 17–39.
Nordin, R. N. (1985). Water Quality Criteria for Nutrients and Algae (Technical Appendix) http://www.elp.gov.bc.ca/wat/wq/BCguidelines/nutrients.html.
Paracampo, A., Mugni, H., Demetrio, P., Pardi, M., Bulus, G., Asborno, M., & Bonetto, C. (2012). Toxicity persistence in runoff from experimental soybean plots following pesticide applications. J. Environ. Sci. Health, Part B, 47, 761–768. doi:10.1080/03601234.2012.676358.
Pengue, W. (2000). Cultivos transgénicos ¿Hacia dónde vamos? (p. 190). Buenos Aires: Lugar Editorial S. A.
Schafer, R. B., Caquet, T., Siimes, K., Mueller, R., Lagadic, L., & Liess, M. (2007). Effects of pesticides on community structure and ecosystem functions in agricultural streams of three biogeographical regions in Europe. The Science of the Total Environment, 382, 272–285.
Schafer, R. B., von der Ohe, C. P., Rasmussen, J., Kefford, B. J., Beketov, M. A., Schulz, R., & Liess, M. (2012). Thresholds for the effects of pesticides on invertebrate communities and leaf breakdown in stream ecosystems. Environmental Science & Technology, 46, 5134–5142.
Schulz, R. (2001). Rainfall-induced sediment and pesticide input from orchards into the Lourens River, Western Cape, South Africa: importance of a single event. Water Research, 35, 1869–1876.
Schulz, R. (2004). Field studies on exposure, effects, and risk mitigation of aquatic nonpoint-source insecticide pollution: a review. Journal of Environmental Quality, 33, 419–448.
Stehle, S., Schulz, R. (2015). Agricultural insecticides threaten surface waters at the global scale. Edited by Jules M. Blais, University of Ottawa, Ottawa, Canada, March 2015. www.pnas.org/cgi/doi/10.1073/pnas.1500232112.
ter Braak, C. J. F., Smilauer, P. (1998). CANOCO Reference Manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination (ver. 4). Ithaca, NY: Microcomputer Power.
You, J., Schuler, L. J., & Lydy, M. J. (2004a). Acute toxicity of sediment-sorbed endrin, methoxychlor, and endosulfan to Hyalella azteca and Chironomus tentans. Bulletin of Environmental Contamination and Toxicology, 73(3), 457–464.
You, J., Weston, D. P., & Lydy, M. J. (2004b). A sonication extraction method for the analysis of pyrethroid, organophosphate, and organochlorine pesticides from sediment by gas chromatography with electron-capture detection. Archives of Environmental Contamination and Toxicology, 47(2), 141–147.
Acknowledgments
This study was supported by grants from the Agencia Nacional de Promoción Científica y Tecnológica (Argentina — PICT 2010-0446) and the Concejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina- PIP 2011 N° 0180).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Solis, M., Mugni, H., Hunt, L. et al. Land use effect on invertebrate assemblages in Pampasic streams (Buenos Aires, Argentina). Environ Monit Assess 188, 539 (2016). https://doi.org/10.1007/s10661-016-5545-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-016-5545-3