Abstract
The number of breast cancer (BC) survivors has been increasing lately, due to the improvement in early detection strategies and oncological treatments. However, BC survivors are 3 times as likely to develop heart failure (HF) within 5 years of cancer diagnosis, and 7/100 of them will develop HF in a median follow-up of 8.5 years. Furthermore, HF in BC survivors has a worse prognosis compared to other causes of HF. Anthracyclines and trastuzumab have been proven to improve survival. However, they are also considered as the main causative factors of HF in BC survivors. Old patients, those with a pre-existing cardiovascular (CV) risk factors/disease, prior exposure to chemotherapy and radiotherapy are at increased risk. Serial evaluation of troponins and cardiac imaging parameters using echocardiography and cardiovascular magnetic resonance can significantly contribute to the early diagnosis of cardiac involvement before overt HF will develop. Assessment and immediate treatment of traditional CV risk factors is the first step for cardiotoxicity prevention. In BC survivors with known heart disease, the clinical stabilization is strongly recommended for cardiotoxicity prevention. Finally, in high-risk CV patients, primary prevention including cardioprotectants and/or CV drugs should be applied. According to recent studies, the early start of ACE inhibitors and β-blockers and the modification of anti-cancer treatment can prevent the decline in left ventricular ejection fraction. However, further multicenter studies are needed to establish both prevention and treatment protocols to successfully overcome HF development in BC survivors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In Greek mythology, Sisyphus, the mythical king of the city of Corinth, was the craftiest and most cunning of all mortals and ruled his city with an iron fist. Of his numerous awe-inspiring deeds, he most notably even managed to cheat the coming of death. However, his self-aggrandizing drew the ire of the gods of Olympus and especially their king, Zeus. For his hubris in believing himself clever than Zeus, Sisyphus was condemned to ceaselessly roll a boulder up the slope of a steep hill. The boulder, however, was enchanted by Zeus to slip Sisyphus’s grasp just before reaching the top, thus consigning him to an eternity of fruitless effort; for no punishment more dreadful for a living person could be conceived, than futile and “absurd” labor without any hope of reward or escape.
Doctors nowadays find themselves in a very similar position to the mythical figure of Sisyphus. They fathom themselves similarly endowed with intelligence and cunning (and an entire arsenal of diagnostic tests and interventions), oftentimes coming head to head with death, which they sometimes manage to cheat. They share a fate equally as absurd however. In spite of their ingenuity, they, like Sisyphus, inevitably remain pawns to the whims of the gods, like leaves swept away by the blowing wind, powerless yet rebellious against the inevitable end, with clear knowledge of their cruel destiny. Regardless of the strenuousness of their effort, the wit of their ideas, and the dedication to their convictions, the boulder will eventually slip their grasp, yet again reaffirming the futility that defines the purpose of their profession.
The field of oncology is no exception to this unfortunate realization. The number of breast cancer (BC) survivors has steadily been following an increasing trend in recent years, as a consequence of improved early detection strategies and innovations in pharmacologic treatments. However, this comes at the cost of a higher incidence of cardiovascular disease (CVD) and heart failure (HF) as well as increased associated mortality [1]. Cardio-oncology has emerged as a co-operative endeavor between oncologists and cardiologists aiming to improve the early identification, treatment, and follow-up of cancer patients with cardiotoxicity and is in a position to provide the basis for dealing with this manifestation of the “myth of Sisyphus.” Increasing understanding behind the pathophysiology of cardiotoxicity due to cancer therapeutics, also referred to as cancer therapeutics–related cardiac dysfunction (CTRCD), has permitted its categorization into two distinct groups; type I CTRCD which is characterized by irreversible myocardial damage and type II CTRCD which is characterized by reversible myocardial dysfunction.
Anthracyclines are the prototypical agents causing type I CTRCD [2]. The irreversibility of myocardial damage is supported by the typical histopathological evidence of structural abnormalities in the myocardium, such as myofibril loss, distention of the sarcoplasmic reticulum, and vacuolization of the cytoplasm [2]. It is also dose-dependent and the incidence of HF after its use is as high as 5% at a cumulative dose of 400 mg/m2, rising to 16% at a dose of 500 mg/m2, 26% at a dose of 550 mg/m2, and 48% at a dose of 700 mg/m2. It confers a poor prognosis, compared with other forms of cardiomyopathy, such as ischemic or dilated cardiomyopathy, with a 5-year survival rate reported to be < 50% [2]. It may be either acute, occurring during or within the first days of administration of anthracyclines, or chronic, becoming evident usually within the first 30 days after the last dose, but not uncommonly also a number of years after the end of treatment [3]. CTRCD-associated cardiomyopathies have also been implemented in the MOGE(S) classification of cardiomyopathies [4].
Trastuzumab is a monoclonal antibody that binds to the extracellular domain of the human epidermal growth factor receptor II (HER2) and is a prime example of a causative agent of Type II CTRCD [4]. Type II CTRCD is not accompanied by structural myocardial changes as it occurs in isolation. However, significant concern arises from the additive cardiotoxic effect of concomitant or sequential use of Type II CTRCD causative agents in patients with Type I CTRCD [2, 3]. Discontinuation of trastuzumab due to CTRCD has been reported to range from 2.1 to 18.9% [3].
The exact cause of cardiotoxicity after radiotherapy for breast cancer is not clear [5]. Experimental evidence suggests indirect harmful effects of microvascular and macrovascular damage on the myocytes. Radiotherapy leads to an acute inflammation within the heart blood capillaries and to continuous inflammatory processes, resulting in endothelial cell proliferation and formation of fibrin thrombi with obstruction of the myocardial capillary lumen leading to ischemia, myocardial cell death, and fibrosis. Furthermore, radiation induces an inflammatory process in the coronary arteries leading to accelerated atherosclerosis [5].
The cardio-oncological approach in breast cancer survivors
A number of position papers on the appropriate approach to CTRCD have been published [6,7,8,9,10,11]. According to these, the management of CTRCD should focus on the following key points:
-
1)
risk stratification,
-
2)
prevention (primary or secondary),
-
3)
early diagnosis,
-
4)
early start of cardiac treatment,
-
5)
balance between oncologic treatment and cardiovascular (CV) safety, and
-
6)
identification of those patients who will benefit from closer surveillance and/or early treatment.
Baseline risk stratification
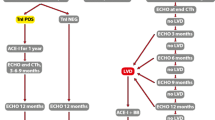
BC survivors are perhaps the most relevant patient group for cardio-oncology as their condition is highly prevalent and they usually have excellent survival. Patient evaluation starts with a baseline estimation of a priori CTRCD risk. Current use of anthracyclines and trastuzumab is associated with the highest rates of CTRCD. Older patients, those with pre-existing CV risk factors/disease and those with prior exposure to chemotherapy (CT) and radiotherapy (RT), are also at increased risk for CTRCD [8]. Assessment and immediate treatment of traditional CV risk factors is the first step of cardiotoxicity prevention [12, 13].
Follow-up and the role of troponins
Cardiac troponins T/I (cTnT/cTnI) are the biomarkers of choice for the detection of damage to cardiomyocytes, and available evidence suggests that they may potentially detect CTRCD at a pre-clinical stage [14,15,16,17]. A study investigating the role of troponins in the early detection of CTRCD included 211 BC patients with poor-prognosis disease, scheduled for high-dose CT [18]. In this population, plasma cTnI was measured immediately before and 3 days after each CT dose. In patients with elevated cTnI, left ventricular ejection fraction (LVEF) was significantly reduced after the first month of treatment and worsened even further during the follow-up period, which was not the case in patients with non-elevated cTnI. In another study, TnI was measured 3 days after CT (early evaluation) and after 1 month (late evaluation) [19], and the greater magnitude of LVEF reduction and cardiac event rate was observed in BC survivors with persistent troponin positivity at both time points. Finally, in a cohort of 251 BC survivors treated with trastuzumab, 14% had elevated cTnI levels, most frequently immediately after the first administration (45%). Notably, those with elevated cTnI developed LV dysfunction more frequently compared to those with non-elevated cTnI (62% vs. 5%; p < 0.001) and were less likely to recover from cardiac dysfunction, despite optimal HF treatment [20]. More recent high-sensitivity troponin assays have a significantly lower limit of minimum detection compared to older assays, which allows them to detect even very small elevations in cTnT/cTnI levels [21].
The role of cardiac imaging in the evaluation of CTRCD
Current practice guidelines recommend repeated echocardiographic assessment of LVEF for the detection of CTRCD in BC patients. However, these and other recommendations remain incompletely defined. Notably, the frequency of the echocardiographic evaluation is not specified. Additionally, even if more clearly defined recommendations are instituted, the use of symptomatic decreases of LVEF as indicators of CTRCD, despite their definite clinical importance, remains suboptimal, as their appearance usually indicates more advanced disease states and heralds worse prognosis [22, 23]. Characteristically, a recent prospective study of a large (n = 2625), unselected cohort of patients treated with anthracyclines (51% BC patients) showed that close monitoring of LVEF after the end of CT enabled almost all CTRCD cases to be identified during the first 12 months of follow-up [24]. The study also showed that initiation of treatment with ACE inhibitors (enalapril) and beta-blockers (carvedilol or bisoprolol) led to relative normalization of heart function in most cases (82%). Nevertheless, only 13% of patients who showed normalized LVEF had attained the same or higher LVEF value as before CT. These data suggest that diagnostic approaches employing LVEF in isolation might not be sensitive enough to identify CTRCD at an early enough stage, which in turn limits the possibility and/or effectiveness of medical intervention. This is due to the fact that LVEF changes can be observed only after cardiac function deteriorates beyond a critical point after which initial compensatory mechanisms are overwhelmed. Furthermore, subsequent normalization of LVEF cannot preclude the possibility of later cardiac dysfunction [23].
CTRCD is a potentially permanent pathologic process starting with myocardial injury and progressively leading to LVEF decline and finally to symptomatic HF. Taking the aforementioned limitations of LVEF into account, the diagnostic approach to CTRCD should be broadened to include serial measurements of biochemical indicators of myocardial injury (hs-cTnT/hs-cTnI) and serial evaluation with cardiac imaging modalities evaluating parameters beyond systolic function [7]. The main cardiovascular imaging modalities currently employed are the following.
Two-dimensional transthoracic echocardiography
Two-dimensional transthoracic echocardiography (2D TTE) remains the cornerstone of cardiac imaging, due to its wide availability, portability, low cost, lack of exposure to ionizing radiation and high levels of familiarity, and expertise among cardiologists. Additionally, newer and more robust echocardiographic techniques have become part of clinical routine and have been incorporated in cardio-oncologic recommendations [25]. However, there is a widespread preoccupation with the determination of the exact significance of LVEF changes and how these should guide modifications in therapeutic regimens [25]. Recently, a decrease in LVEF of > 10% to a value < 53%, confirmed by repeated echocardiographic evaluation, has been proposed for the diagnosis of chemotherapy-induced CTRCD [25].
Three-dimensional transthoracic echocardiography
The superiority of three-dimensional transthoracic echocardiography (3D TTE) over 2D TTE in the calculation of LVEF has been demonstrated in various studies [26]. Compared with 2D TTE, 3D TTE has significantly more agreement with cardiovascular magnetic resonance (CMR), which is the modality of choice for accurate systolic function and ventricular volumetric assessment [26]. This is due to the lack of assumptions about LV geometry, which may be inaccurate in asymmetric/remodeled ventricles [27]. Additionally, apical foreshortening artifacts are also eliminated via the use of complex multiarray transducers, which also contributes to the effectiveness of 3D TTE. Furthermore, a recent study demonstrated that LVEF, as assessed by 3D TTE, has significantly better reproducibility than that assessed by 2D TTE [27] and should therefore be determined using 3D TTE wherever possible. However, despite the better correlation with CMR measurements, 3DTTE still underestimates LV volumes and has lower reproducibility than CMR, possibly due to lower spatial resolution [28]. A meta-analysis comparing LV volume and LVEF measurements between 3D TTE and CMR similarly concluded that 3D TTE is superior to 2DE TTE in volumetric assessment of the LV, with a lower inter- and intra-observer variability in LVEF calculations. However, it confirmed that 3D TTE still underestimates LV volumes compared to CMR, especially in patients with suboptimal acoustic window and LV remodeling [28]. These limitations combined with the need for high-quality apical grayscale images not available in BC patients post oncological surgery or breast reconstruction [29] and the fact that LVEF calculation is more time-consuming with 3D TTE than with 2D TTE discourage its routine implementation in the evaluation of BC patients.
The role of tissue Doppler imaging and myocardial tissue deformation indices
Myocardial tissue deformation indices such as strain, strain rate, and left ventricular torsion have been evaluated as early markers of myocardial dysfunction in several clinical scenarios. Tissue Doppler imaging (TDI) measures regional myocardial velocities and can estimate regional myocardial deformation and deformation rate through the measurement of strain rate and strain [30]. Several studies have reported early alterations in TDI-derived strain and strain rate measurements in chemotherapy-treated patients [30]. However, TDI carries several limitations such as angle-dependency and only allows for evaluation of longitudinal deformation, whereas the radial and circumferential function measurements are feasible only in specific myocardial segments [31]. The limitations of TDI led to the use of two-dimensional speckle tracking echocardiography (2D STE) for the evaluation of myocardial deformation, since this new technique is angle-independent and allows for quantification of strain and strain rate in any direction within the imaging plane [32].
Two-dimensional speckle tracking echocardiography
2D STE is based on tracking the motion of small bright spots in the myocardium (speckles) on the grayscale image as they move during the cardiac cycle. Myocardial deformation quantification is based on the calculation of the distance between two speckles over the cardiac cycle. It is angle independent and allows measurement of strain/strain rate values in all myocardial segments in the longitudinal, radial, and circumferential direction, using two-dimensional grayscale apical and parasternal short axis images. Moreover, compared to TDI, it enables easier data acquisition and offline analysis [31, 32]. Therefore, it has recently been introduced in clinical practice for the quantification of myocardial deformation (Figs. 1 and 2).
Global longitudinal strain (GLS) seems to be the most sensitive marker of subclinical myocardial damage. Its use in the clinical management of BC patients has been extensively evaluated with promising results in early diagnosis of subclinical myocardial dysfunction. Several studies report early alterations in myocardial deformation [33, 34] in patients treated with anthracyclines and/or trastuzumab. More importantly, early alterations in GLS have been reported to predict future deterioration of global LV systolic function [34, 35]. A reduction of > 15% of GLS has been proposed as an echocardiographic index for defining early CTRCD [36]. Impaired GLS has also been reported to be a predictor of major adverse cardiac events (MACE) in a cohort of 158 patients under treatment with anthracycline, with borderline to low normal 2D LVEF (50–59%) [37]. Rotational deformation impairment has been also proposed as an early marker of LV dysfunction in BC survivors [38].
Three-dimensional speckle tracking echocardiography
Although 2D STE seems to have an established role in the clinical management of BC patients, the evaluation of myocardial deformation with three-dimensional speckle tracking echocardiography (3D STE) has emerged as another promising technique in this field. Despite its lower temporal resolution in comparison with 2D STE, initial clinical results indicate that 3D STE may have important advantages over 2D STE, allowing a faster and potentially more accurate analysis of myocardial function, by overcoming the limitations of 2D STE and by allowing the simultaneous analysis of a significantly greater number of myocardial segments in the longitudinal, radial, and circumferential direction since the entire LV can be analyzed from a single volume of data, obtained from the apical position [32, 39]. However, despite the initial promising results of 3D STE, further investigation is required to assess its clinical impact in the management of BC survivors.
Cardiovascular magnetic resonance
Cardiovascular magnetic resonance (CMR) is the ideal imaging modality for the serial evaluation of biventricular volumes and EF, while also providing the possibility to characterize myocardial tissues with regard to edema and fibrosis [40]. In addition, CMR may identify the etiology behind newly discovered abnormal myocardial mass, pericardial disease process, and/or valvular abnormality [40]. BC survivors may particularly benefit from CMR evaluation, because of its high spatial resolution, reproducibility, and ability to accurately detect even small changes in LVEF that may occur as a result of CT [41,42,43,44,45]. CMR also permits the evaluation of myocardial deformation. Recently, Romano et al. [46] demonstrated that CMR-derived GLS was a strong, independent predictor of all-cause mortality in those with ischemic and nonischemic cardiomyopathy after accounting for both LVEF and the presence of fibrosis in the form of late gadolinium enhancement (LGE). Furthermore, Jolly et al. [47] showed that GLS worsened in CT patients from baseline to 3-month follow-up (− 18.8 ± 2.9% vs. − 17.6 ± 3.1%, p = 0.001) [47]. In a prospective study of 53 BC patients, CMR-derived GLS deteriorated along with subclinical declines in LVEF within 1–6 months following the initiation of treatment with low to moderate doses of an anthracycline [42]. However, at present, CMR strain measures are not widely used in clinical practice.
Tissue characterization indices convey important diagnostic information. T2 mapping is the earliest marker of anthracycline-induced cardiotoxicity, as it identifies intracardiomyocyte edema in the absence of pathologic T1 mapping, extracellular volume fraction (ECV), or LV function (Fig. 3). The identification of diffuse myocardial edema in the absence of myocardial fibrosis, represented by pathologic T2 mapping values in isolation, represents acute and reversible myocardial injury [2], which in turn demonstrates the potential of this CMR marker for guiding tailored CT [2]. Furthermore, the combination of native T1 and T2 mapping can be valuable in detecting and monitoring CTRCD, by identifying either an inflammatory component (elevated native T1 and T2) and/or interstitial fibrosis/remodeling (raised native T1 but not T2), respectively [48].
Another tissue characterization index with potential diagnostic implications in this context is the ECV, which is derived from T1 mapping measurements before (native) and after the administration of paramagnetic contrast agent (post-contrast) and adjusted for the hematocrit value. In a cohort of 42 adult cancer survivors previously treated with anthracyclines (17% treated for BC) and undergoing CMR for clinical reasons (HF or atrial fibrillation), over a median of 89 ± 40 months after anthracycline-based CT, ECV was significantly higher compared with healthy controls (36 ± 3% vs. 28 ± 2%, p < 0.0001) [49]. More recently, a cross-sectional analysis of patients 3 years after anthracycline-based treatment, newly diagnosed or untreated cancer patients, and healthy controls, demonstrated that native T1 mapping and ECV were significantly higher in previously treated cancer survivors after accounting for demographics, cardiovascular risk factors, and other markers of myocardial remodeling (p < 0.01 for all). This study suggests that an elevated ECV is associated with prior anthracycline chemotherapy [50].
Additionally, the observed pattern of myocardial fibrosis (focal or diffuse) provides important diagnostic information about the underlying disease, when quantified using mapping techniques or LGE [51]. CT has been shown to injure the vascular endothelium in a dose-dependent manner [52]. This damaged endothelium influences LV perfusion, which might be further exacerbated by vascular damage caused by thorax RT (especially left-sided). CMR can perform an accurate assessment of myocardial perfusion by means of adenosine-based stress perfusion studies [53]. However, CMR perfusion studies have not been systematically studied in BC survivors.
CMR imaging is of particular value relative in the evaluation of right ventricular (RV) dysfunction in BC patients. RV dysfunction in BC patients may be due to primary or metastatic neoplasms or secondary to chemotherapy. A CMR study of 41 adult BC patients, treated with trastuzumab, showed transient changes in RV function. Compared with baseline, RVEF was reduced at 6 and 12 months after trastuzumab with a near resolution at the 18-month CMR evaluation [54]. In this study, the changes in RVEF occurred independently of LVEF changes [54]. However, its prognostic value still remains unknown. It is also recognized that prior anthracycline exposure is associated with future declines in LV mass. CMR can quantify LV mass in an accurate, reproducible manner [54]. In a study of 61 patients undergoing anthracycline CT, a decline in LV mass and not in LVEF 6 months after chemotherapy was independently associated with worsening HF [55].
CMR is also of great significance for the evaluation of pericardial disease among patients receiving CT. It can detect and assess pericardial inflammation with or without effusion and it can exclude constrictive pericarditis after thoracic RT [56]. Differentiating pericardial inflammation from fibrosis in the presence of pericarditis is difficult, even using CMR [57,58,59,60]. It has been shown, however, that pericardial LGE is most frequently associated with pericardial inflammation [61], whereas end-stage or chronic fibrotic constrictive pericarditis does not enhance after administration of paramagnetic contrast agent but may show morphologic findings of constriction [62].
Can we avoid the fate of Sisyphus in cardio-oncology?
The current situation of oncology is very reminiscent of the myth of Sisyphus, in the sense that one problem is solved (neoplasia) only to be faced with another setback (CTRCD). But how can the physician of tomorrow avoid the fate that befell Sisyphus? The following approaches can form the basis of effectively facing these challenging issues in the field of cardio-oncology:
-
1)
Primary prevention: reduction of the direct cardiotoxic effect
-
a)
Anthracycline cumulative dose limitation
-
a)
The risk of doxorubicin-induced HF increases proportionally to the amount of the cumulative dose of administered anthracycline and is further increased if trastuzumab is co-administered [1,2,3,4]. However, some patients develop cardiotoxicity even at standard doses, while others can tolerate a total dose twofold greater than the conventional dose, suggesting that genetic variation or other hitherto unknown factors might modify the risk of CTRCD [1,2,3,4]. Minimizing anthracycline exposure or avoiding anthracycline-based regimens in HER-2+ BC should be considered in patients with high a priori CTRCD risk (previous anti-neoplastic treatments, older age, high CVD risk) [1,2,3].
-
b)
Use of less cardiotoxic anthracycline analogues
Epirubicin-induced CTRCD occurs after relatively higher doses when compared to doxorubicin, but higher doses must be administered to achieve the same clinical response (90 mg/mg epirubicin = 60 mg/mg doxorubicin) [1, 9]. Administration of liposomes containing CT agents is a promising alternative manner of administration, which has the innate advantage that non-fenestrated capillaries such as those in cardiac tissue have very low permeability for liposomes. Thus, the tendency to accumulate in cardiac cells is reduced, lowering the risk of CTRCD. Pegylated liposomal doxorubicin showed lower cardiotoxicity compared to standard doxorubicin and should be considered in BC subjects at increased risk or if higher anthracycline doses are needed [3]. In a meta-analysis, liposomal doxorubicin showed a lower risk of both asymptomatic and symptomatic LV dysfunction, compared to standard doxorubicin [11]. Although the non-inferiority of liposomal doxorubicin to non-liposomal doxorubicin has been established, the improved toxicology profile may come at a higher cost of treatment [11]
-
c)
Alternatives to trastuzumab for HER-2+ breast cancer
In BC patients with completely or partially recovered LV dysfunction due to trastuzumab, that requires continuation of HER-2 blockade, a less toxic pharmacologic alternative should be used. The MARIANNE trial investigated taxanes in combination with trastuzumab (TH) vs. trastuzumab-emtansine (T-DM1) alone or T-DM1 in combination with pertuzumab in patients with advanced HER-2+ BC [63]. Both T-DM1regimens were equal to TH regarding survival and were associated with a lower rate of LV dysfunction [24]. This suggests that T-DM1 might be lead to less CTRCD in BC patients that require long-term treatment with trastuzumab and have high a priori risks for CTRCD.
-
d)
Pharmacologic prevention
Agents with potential cardioprotective actions that can reduce the cardiotoxic effect of anti-neoplastic agents are of great value in the field of cardio-oncology, as an alternative to reducing or temporarily interrupting anti-neoplastic treatments. Dexrazoxane markedly reduces anthracycline-related cardiotoxicity in adults with various solid tumors and in children with acute lymphoblastic leukemia and Ewing sarcoma [64,65,66]. A large amount of evidence shows that patients who received dexrazoxane have a reduced incidence of HF compared to those that did not. Nevertheless, dexrazoxane has not seen wide adoption by the medical community, and it is only recommended by the American Society of Clinical Oncology in patients with metastatic BCa cumulative dose of > 300 mg/m2 doxorubicin [66], due to its not-well established potential to reduce the effectiveness of anthracyclines [66].
Beta-blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor antagonists, statins, and aldosterone antagonists have been reported as potentially cardioprotective in BC patients treated with anthracyclines or trastuzumab [67,68,69,70,71,72,73,74,75,76]. In a cohort of 40 BC patients, carvedilol prevented strain abnormalities after anthracycline use [77]. However, in a similar population, the prophylactic use of the drug failed to prevent an LVEF reduction > 10%, but blunted troponin increases and preserved diastolic function [78]. Nebivolol started 7 days before anthracycline treatment and administered for 6 months that prevented reductions in LVEF, while in untreated patients, LVEF was significantly reduced [68]. In a retrospective study that included 318 BC patients, the continuation of beta-blocker during CT with anthracyclines, trastuzumab, or both was associated with a lower rate of HF at 5-year follow-up [69].
The angiotensin II receptor blocker telmisartan, started 1 week before treatment with epirubicin in 25 patients with various solid tumors (mostly BC), was able to prevent the deterioration of TDI-derived myocardial deformation indices [72]. Recently, the PRADA study has reported that candesartan administrated with adjuvant CT, with or without trastuzumab, protects against early LVEF reductions according to CMR data [73], but it has not been confirmed by a follow-up randomized study involving a similar population [79]. The Canadian study MANTICORE-101 compared perindopril vs. bisoprolol in the prevention of LV dysfunction in patients with HER2+ BC receiving trastuzumab [70]. Neither drug has been shown to prevent LV remodeling. However, at multivariable analysis, the use of both drugs was associated with a preserved LVEF. Spironolactone cardioprotective action has been reported. In a recent randomized trial including 43 BC patients, those receiving spironolactone 1 week before anthracycline-based CT did not show significant LVEF reduction after 3 weeks of CT completion [74]. Their diastolic function was also unaffected and no increases in cTnI were identified.
The cardioprotective effect of statins depends on their pleiotropic effects, particularly their antioxidant properties. In a retrospective analysis, the continuation of statin was associated with a reduction in HF incidence and cardiac mortality during follow-up [75]. In another prospective observational study, a lower reduction in LVEF was observed only in those who were under treatment with statins [76].
-
2)
Secondary prevention
Secondary prevention should be applied in selected high-risk BC survivors with signs of cardiotoxicity, presented with a biomarker increase and/ or strain decrease. The sole example in this field is a randomized trial including 473 patients with various tumors (BC 30%), treated with high-dose CT in which enalapril was evaluated [71]. In patients treated with enalapril, nobody presented a LVEF reduction of 10 absolute points below 50% and the incidence of MACE was significantly low. In the enalapril-treated group, after a follow-up period of 12 months, LVEF was equal to baseline value in 88% of cases (both in patients with transient and persistent troponin rise), supporting that enalapril can achieve preservation of systolic function.
-
3)
Primary vs. secondary prevention
It has been shown that enalapril, started early after evidence of troponin elevation and continued for 12 months, can prevent the development of LV dysfunction and related MACE [71]. However, to identify the troponin elevation, repeated samples are needed, as this marker may increase at different times during treatment. On the other side, primary prevention, extended to all patients who need to be treated with potentially cardiotoxic anti-cancer therapies, does not have this limitation. The ICOSONE (International CardioOncology Society-one) randomized study was prospectively conducted to compare the effectiveness of two different strategies: to verify whether enalapril, initiated in all patients before CT (prevention group) was capable of preventing troponin elevation and subsequent LV dysfunction and whether this approach was more effective than enalapril treatment, initiated only after troponin elevation during CT (Troponin-triggered Group). The main finding was that the 2 strategies were equally effective in preventing LV dysfunction and MACE.
On the other side, a secondary prevention, guided by a rise in troponin, although it has the disadvantage of repeated blood sampling, has a very high negative, predictive value [71]. This strategy is cost-effective and allows the exclusion of low-risk patients, the reduction of overmedicalization, and the unnecessary anxiety. A primary prevention, although it does not require serial troponin evaluation, needs continuous monitoring for drug titration and exposes less susceptible to cardiotoxicity BC survivors to potential side-effects [80].
At the moment, there are no evidence-based studies presenting how to overcome the “myth of Sisyphus” in BC patients. Current recommendations are focused mainly on the continuation, withdraw, or modification of CT according to the LVEF [80]. ACE inhibitors and beta-blockers have been recently evaluated in prospective studies [81] showing that in patients who developed anthracycline cardiomyopathy, the treatment with these medications is necessary and should be started immediately. According to this study, 64% of patients treated within 2 months after the end of CT showed a recovery of LVEF; however, in patients treated later, this percentage decreased progressively and complete recovery was not observed in patients treated after 6 months [81]. Finally, the combined role of biochemical indices and new CMR parametric indices should be prospectively evaluated. In this context, native T1 and T2 mapping can be valuable in detecting and monitoring cardiac involvement with cancer-related treatment, providing distinct information of early inflammatory involvement (raised native T1 and T2) and/or interstitial fibrosis and remodeling (raised native T1 but not T2), respectively [48].
It seems that the management of cancer is no more limited solely to the treatment of malignancy. With increasing numbers of cancer survivors living longer, cardiologists are faced with the management of early and late cardiotoxicity that finally leads to rapidly progressive HF and increased morbidity/mortality. These data emphasize the early diagnosis/treatment as a prerequisite to overcome the “Sisyphous myth” in cardio-oncology. To achieve this target, we currently have many diagnostic modalities, which although cannot stop the inevitable end, they are not absurd. Through them, we can improve the quality of life, extend life expectancy, and clarify the complex pathophysiology of cardiac involvement post CT, which may be treated more effectively. Even if some of them look rather expensive such as CMR, the potential to early identify patients prone to cardiotoxicity-induced HF can counterbalance the high cost.
Conclusions
As survival after BC continues to increase, it has become clear that BC survivors are 3 times as likely to develop HF within 5 years of cancer diagnosis compared to the general population. Anthracyclines, trastuzumab, and radiotherapy are the main causative factors of HF secondary to CTRCD in BC survivors. Existing literature suggests that early identification of CTRCD and initiation of treatment with ACE inhibitors and β-blockers can potentially prevent further deterioration in cardiac function. However, additional multicenter studies are needed to establish both prevention and treatment protocols in order for the physicians of the future to avoid the same cruel fate as the mythical figure of Sisyphus.
References
Mehta LS, Watson KE, Barac A, Beckie TM, Bittner V, Cruz-Flores S, Dent S, Kondapalli L, Ky B, Okwuosa T, Piña IL, Volgman AS, American Heart Association Cardiovascular Disease in Women and Special Populations Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Quality of Care and Outcomes Research (2018) Cardiovascular disease and breast cancer: where these entities intersect: a scientific statement from the American Heart Association. Circulation 137(8):e30–e66
Galán-Arriola C, Lobo M, Vílchez-Tschischke JP, López GJ, de Molina-Iracheta A, Pérez-Martínez C, Agüero J, Fernández-Jiménez R, Martín-García A, Oliver E, Villena-Gutierrez R, Pizarro G, Sánchez PL, Fuster V, Sánchez-González J, Ibanez B (2019) Serial magnetic resonance imaging to identify early stages of anthracycline-induced cardiotoxicity. J Am Coll Cardiol 73(7):779–791
Chatterjee K, Zhang J, Honbo N, Karliner JS (2010) Doxorubicin cardiomyopathy. Cardiology. 115(2):155–162
Westphal JG, Rigopoulos AG, Bakogiannis C, Ludwig SE, Mavrogeni S, Bigalke B, Doenst T, Pauschinger M, Tschöpe C, Schulze PC, Noutsias M (2017) The MOGE(S) classification for cardiomyopathies: current status and future outlook. Heart Fail Rev 22(6):743–752
Sardaro A, Petruzzelli MF, D'Errico MP, Grimaldi L, Pili G, Portaluri M (2012) Radiation-induced cardiac damage in early left breast cancer patients: risk factors, biological mechanisms, radiobiology, and dosimetric constraints. Radiother Oncol 103:133–142
Lenihan D (2017) Cardio-oncology: what is the best practice we can all strivre for? Int J Cardiol 241:393–394
Zamorano JL, Lancellotti P, Rodriguez Muñoz D, Aboyans V, Asteggiano R, Galderisi M, Habib G, Lenihan DJ, Lip GYH, Lyon AR, Lopez Fernandez T, Mohty D, Piepoli MF, Tamargo J, Torbicki A, Suter TM, Zamorano JL, Aboyans V, Achenbach S, Agewall S, Badimon L, Barón-Esquivias G, Baumgartner H, Bax JJ, Bueno H, Carerj S, Dean V, Erol Ç, Fitzsimons D, Gaemperli O, Kirchhof P, Kolh P, Lancellotti P, Lip GYH, Nihoyannopoulos P, Piepoli MF, Ponikowski P, Roffi M, Torbicki A, Vaz Carneiro A, Windecker S, Authors/Task Force Members, ESC Committee for Practice Guidelines (CPG), Document Reviewers (2017) 2016 ESC position paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: the task force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur J Heart Fail 19:9–42
Fradley MG, Brown AC, Shields B et al (2017) Developing a comprehensive cardio-oncology program at a cancer institute: the Moffit cancer center experience. Oncol Rev 11:340
Barros-Gomez S, Herrmann J, Mulvagh SL et al (2016) Rationale for setting up a cardio-oncology unit: our experience at Mayo Clinic. Cardio-Oncology 2:5
Barac A, Murthag G, Carver JR et al (2015) Cardiovascular health of patients with cancer and cancer survivors. J Am Coll Cardiol 65:2739–2746
Andreopoulou E, Gaiotti D, Kim E, Volm M, Oratz R, Freedberg R, Downey A, Vogel CL, Chia S, Muggia F (2007) Feasibility and cardiac safety of pegylated liposomal doxorubicin plus trastuzumab in heavily pretreated patients with recurrent HER2-overexpressing metastatic breast cancer. Clin Breast Cancer 7(9):690–696
Hamo CE, Bloom MW, Cardinale D et al (2016) Cancer therapy-related cardiac dysfunction and heart failure: part 2: prevention, treatment, guidelines, and future directions. Circ Heart Fail 9:e002843
Guenancia C, Lefevbvre A, Cardinale D et al (2016) Obesity as a risk factor for anthracyclines and trastuzumab cardiotoxicity in breast cancer: a systematic review and meta-analysis. J Clin Oncol 34:3157–3165
Christenson ES, James T, Agrawal V, Park BH (2015) Use of biomarkers for the assessment of chemotherapy-induced cardiac toxicity. Clin Biochem 48:223–235
Cardinale D, Biasillo G, Salvatici M, Sandri MT, Cipolla CM (2017) Using biomarkers to predict and to prevent cardiotoxicity of cancer therapy. Expert Rev Mol Diagn 17:245–256
Daubert MA, Jeremias A (2010) The utility of troponin measurement to detect myocardial infarction: review of the current findings. Vasc Health Risk Manag 6:691–699
Lipshultz SE, Rifai N, Sallan SE, Lipsitz SR, Dalton V, Sacks DB, Ottlinger ME (1997) Predictive value of cardiac troponin T in pediatric patients at risk for myocardial injury. Circulation 96:2641–2648
Cardinale D, Sandri MT, Martinoni A, Borghini E, Civelli M, Lamantia G, Cinieri S, Martinelli G, Fiorentini C, Cipolla CM (2002) Myocardial injury revealed by plasma troponin I in breast cancer treatment with high dose chemotherapy. Ann Oncol 13:710–715
Cardinale D, Sandri MT, Colombo A, Colombo N, Boeri M, Lamantia G, Civelli M, Peccatori F, Martinelli G, Fiorentini C, Cipolla CM (2004) Prognostic value of Troponin I in cardiac risk stratification of cancer patients undergoing high-dose chemotherapy. Circulation 109:2749–2754
Cardinale D, Colombo A, Torrisi R, Sandri MT, Civelli M, Salvatici M, Lamantia G, Colombo N, Cortinovis S, Dessanai MA, Nolè F, Veglia F, Cipolla CM (2010) Trastuzumab-induced cardiotoxicity: clinical and prognostic implications of Troponin I evaluation. J Clin Oncol 28:3910–3916
Kitayama H, Kondo T, Sugiyama J, Kurimoto K, Nishino Y, Kawada M, Hirayama M, Tsuji Y (2017) High-sensitive troponin T assay can predict anthracycline- and trastuzumab-induced cardiotoxicity in breast cancer patients. Breast Cancer 24(6):774–782
Armenian SH, Lacchetti C, Barac A, Carver J, Constine LS, Denduluri N, Dent S, Douglas PS, Durand JB, Ewer M, Fabian C, Hudson M, Jessup M, Jones LW, Ky B, Mayer EL, Moslehi J, Oeffinger K, Ray K, Ruddy K, Lenihan D (2017) Prevention and monitoring of cardiac dysfunction in survivors of adult cancers: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol 35:893–911
Cardinale D, Cipolla CM (2016) Chemotherapy-induced cardiotoxicity: importance of early detection. Expert Rev Cardiovasc Ther 14:1297–1299
Cardinale D, Colombo A, Bacchiani G, Tedeschi I, Meroni CA, Veglia F, Civelli M, Lamantia G, Colombo N, Curigliano G, Fiorentini C, Cipolla CM (2015) Early detection of anthracycline cardiotoxicity and improvement with heart failure therapy. Circulation 131:1981–1988
Gavila J, Seguí MÁ, Calvo L, López T, Alonso JJ, Farto M, Sánchez-de la Rosa R (2017) Evaluation and management of chemotherapy-induced cardiotoxicity in breast cancer: a Delphi study. Clin Transl Oncol 19(1):91–104
Jenkins C, Moir S, Chan J, Rakhit D, Haluska B, Marwick TH (2009) Left ventricular volume measurement with echocardiography: a comparison of left ventricular opacification, three-dimensional echocardiography, or both with magnetic resonance imaging. Eur Heart J 30(1):98–106
Aggeli C, Felekos I, Kastellanos S, Panagopoulou V, Oikonomou E, Tsiamis E, Tousoulis D (2015) Real-time three-dimensional echocardiography: never before clinical efficacy looked so picturesque. Int J Cardiol 198:15–21
Dorosz JL, Lezotte DC, Weitzenkamp DA, Allen LA, Salcedo EE (2012) Performance of 3-dimensional echocardiography in measuring left ventricular volumes and ejection fraction: a systematic review and meta-analysis. J Am Coll Cardiol 59(20):1799–1808
Pignatti M, Mantovani F, Bertelli L, Barbieri A, Pacchioni L, Loschi P, De Santis G (2013) Effects of silicone expanders and implants on echocardiographic image quality after breast reconstruction. Plast Reconstr Surg 132(2):271–278
Jurcut R, Wildiers H, Ganame J, D'hooge J, De Backer J, Denys H, Paridaens R, Rademakers F, Voigt JU (2008) Strain rate imaging detects early cardiac effects of pegylated liposomal Doxorubicin as adjuvant therapy in elderly patients with breast cancer. J Am Soc Echocardiogr 21(12):1283–1289
Pavlopoulos H, Nihoyannopoulos P (2008) Strain and strain rate deformation parameters: from tissue Doppler to 2D speckle tracking. Int J Cardiovasc Imaging. 24(5):479–491
Mor-Avi V, Lang RM, Badano LP, Belohlavek M, Cardim NM, Derumeaux G, Galderisi M, Marwick T, Nagueh SF, Sengupta PP, Sicari R, Smiseth OA, Smulevitz B, Takeuchi M, Thomas JD, Vannan M, Voigt JU, Zamorano JL (2011) Current and evolving echocardiographic techniques for the quantitative evaluation of cardiac mechanics: ASE/EAE consensus statement on methodology and indications endorsed by the Japanese Society of Echocardiography. Eur J Echocardiogr 12(3):167–205
Laufer-Perl M, Derakhshesh M, Milwidsky A, Mor L, Ravid D, Amrami N, Sherez J, Keren G, Topilsky Y, Arbel Y (2018) Usefulness of global longitudinal strain for early identification of subclinical left ventricular dysfunction in patients with active cancer. Am J Cardiol 122(10):1784–1789
Fallah-Rad N, Walker JR, Wassef A, Lytwyn M, Bohonis S, Fang T, Tian G, Kirkpatrick ID, Singal PK, Krahn M, Grenier D, Jassal DS (2011) The utility of cardiac biomarkers, tissue velocity and strain imaging, and cardiac magnetic resonance imaging in predicting early left ventricular dysfunction in patients with human epidermal growth factor receptor II-positive breast cancer treated with adjuvant trastuzumab therapy. J Am Coll Cardiol 57(22):2263–2270
Galanti G, Pedrizzetti G, Pedri S, Stefani L (2017) 2D longitudinal LV speckle tracking strain pattern in breast cancer survivors: sports activity vs exercise as prescription model. Intern Emerg Med 12(8):1149–1157
Thavendiranathan P, Poulin F, Lim KD, Plana JC, Woo A, Marwick TH (2014) Use of myocardial strain imaging by echocardiography for the early detection of cardiotoxicity in patients during and after cancer chemotherapy: a systematic review. J Am Coll Cardiol 63(25 Pt A):2751–2768
Mousavi N, Tan TC, Ali M, Halpern EF, Wang L, Scherrer-Crosbie M (2015) Echocardiographic parameters of left ventricular size and function as predictors of symptomatic heart failure in patients with a left ventricular ejection fraction of 50-59% treated with anthracyclines. Eur Heart J Cardiovasc Imaging 16(9):977–984
Motoki H, Koyama J, Nakazawa H, Aizawa K, Kasai H, Izawa A, Tomita T, Miyashita Y, Kumazaki S, Takahashi M, Ikeda U (2012) Torsion analysis in the early detection of anthracycline-mediated cardiomyopathy. Eur Heart J Cardiovasc Imaging 13(1):95–103
Xu Y, Shi J, Zhao R, Zhang C, He Y, Lin J, Zhang Q, Shu X, Cheng L (2019) Anthracycline induced inconsistent left ventricular segmental systolic function variation in patients with lymphoma detected by three-dimensional speckle tracking imaging. Int J Cardiovasc Imaging 35:771–779. https://doi.org/10.1007/s10554-018-1510-2
Mavrogeni SI, Markousis-Mavrogenis G, Kolovou G (2018) “Save the last dance” for cardiovascular magnetic resonance. Eur Cardiol 13(2):95–97
Wassmuth R, Lentzsch S, Erdbruegger U, Schulz-Menger J, Doerken B, Dietz R, Friedrich MG (2001) Subclinical cardiotoxic effects of anthracyclines as assessed by magnetic resonance imaging—a pilot study. Am Heart J 141:1007–1013
Drafts BC, Twomley KM, D’Agostino R Jr et al (2013) Low to moderate dose anthracycline-based chemotherapy is associated with early noninvasive imaging evidence of subclinical cardiovascular disease. J Am Coll Cardiol Img 6:877–885
Bellenger N, Burgess M, Ray S, Lahiri A, Coats AJ, Cleland JG, Pennell DJ (2000) Comparison of left ventricular ejection fraction and volumes in heart failure by echocardiography, radionuclide ventriculography and cardiovascular magnetic resonance. Are they interchangeable? Eur Heart J 21:1387–1396
Wang K, Eblan MJ, Deal AM, Lipner M, Zagar TM, Wang Y, Mavroidis P, Lee CB, Jensen BC, Rosenman JG, Socinski MA, Stinchcombe TE, Marks LB (2017) Cardiac toxicity after radiotherapy for stage III non-small-cell lung cancer: pooled analysis of dose escalation trials delivering 70 to 90 Gy. J Clin Oncol 35:1387–1394
Adams MJ, Hardenbergh PH, Constine LS, Lipshultz SE (2003) Radiation-associated cardiovascular disease. Crit Rev Oncol Hematol 45:55–75
Romano S, Judd RM, Kim RJ, Kim HW, Klem I, Heitner J, Shah DJ, Jue J, White BE, Shenoy C, Farzaneh-Far A (2017) Association of feature-tracking cardiac magnetic resonance imaging left ventricular global longitudinal strain with all-cause mortality in patients with reduced left ventricular ejection fraction. Circulation 135:2313–2315
Jolly M-P, Jordan JH, Meléndez GC, McNeal GR, D’Agostino RB, Hundley WG (2017) Auto-mated assessments of circumferential strain from cine CMR correlate with LVEF declines in cancer patients early after receipt of cardio-toxic chemotherapy. J Cardiovasc Magn Reson 19:59
Haslbauer JD, Lindner S, Valbuena-Lopez S, Zainal H, Zhou H, D'Angelo T, Pathan F, Arendt CA, Bug G, Serve H, Vogl TJ, Zeiher AM, Carr-White G, Nagel E, Puntmann VO (2019) CMR imaging biosignature of cardiac involvement due to cancer-related treatment by T1 and T2 mapping. Int J Cardiol 275:179–186
Neilan TG, Coelho OR, Shah RV et al (2013) Myocardial extracellular volume by cardiac magnetic resonance imaging in patients treated with anthracycline-based chemotherapy. Am J Cardiol 111:717–722
Jordan JH, D’Agostino RB, Hamilton CA et al (2014) Longitudinal assessment of concurrent changes in left ventricular ejection fraction and left ventricular myocardial tissue characteristics after administration of cardiotoxic chemotherapies using t1-weighted and t2-weighted cardiovascular magnetic resonance. Circ Cardiovasc Imaging 7:872–879
Mavrogeni S, Apostolou D, Argyriou P, Velitsista S, Papa L, Efentakis S, Vernardos E, Kanoupaki M, Kanoupakis G, Manginas A (2017) T1 and T2 mapping in cardiology: “mapping the obscure object of desire”. Cardiology. 138(4):207–217
Vasu S, Hundley WG (2013) Understanding cardio-vascular injury after treatment for cancer: an overview of current uses and future directions of cardiovascular magnetic resonance. J Cardiovasc Magn Reson 15:66–83
Mavrogeni S, Bratis K, Koutsogeorgopoulou L, Karabela G, Savropoulos E, Katsifis G, Raftakis J, Markousis-Mavrogenis G, Kolovou G (2017) Myocardial perfusion in peripheral Raynaud’s phenomenon. Evaluation using stress cardiovascular magnetic resonance. Int J Cardiol 228:444–448
Barthur A, Brezden-Masley C, Connelly KA, Dhir V, Chan KKW, Haq R, Kirpalani A, Barfett JJ, Jimenez-Juan L, Karur GR, Deva DP, Yan AT (2017) Longitudinal assessment of right ventricular structure and function by cardiovascular magnetic resonance in breast cancer patients treated with trastuzumab: a prospective observational study. J Cardiovasc Magn Reson 19:44
Jordan JH, Castellino SM, Meléndez GC et al (2018) Left ventricular mass change after anthracycline chemotherapy. Circ Heart Fail 11:e004560
White CS (1996) MR evaluation of the pericardium and cardiac malignancies. Magn Reson Imaging Clin N Am 4:237–251
Imazio M, Demichelis B, Parrini I, Favro E, Beqaraj F, Cecchi E, Pomari F, Demarie D, Ghisio A, Belli R, Bobbio M, Trinchero R (2005) Relation of acute pericardial disease to malignancy. Am J Cardiol 95:1393–1394
Maisch B, Ristic A, Pankuweit S (2010) Evaluation and management of pericardial effusion in patients with neoplastic disease. Prog Cardiovasc Dis 53:157–163
Kojima S, Yamada N, Goto Y (1999) Diagnosis of constrictive pericarditis by tagged cine magnetic resonance imaging. N Engl J Med 341:373–374
Wang ZJ, Reddy GP, Gotway MB, Yeh BM, Hetts SW, Higgins CB (2003) CT and MR imaging of pericardial disease. Radiographics 23 Spec No:S167–S180
Zurick AO, Bolen MA, Kwon DH, Tan CD, Popovic ZB, Rajeswaran J, Rodriguez ER, Flamm SD, Klein AL (2011) Pericardial delayed hyperenhancement with CMR imaging in patients with constrictive pericarditis undergoing surgical pericardiectomy: a case series with histopathological correlation. J Am Coll Cardiol Img 4:1180–1191
Bogaert J, Francone M (2009) Cardiovascular magnetic resonance in pericardial diseases. J Cardiovasc Magn Reson 11:14
Perez EA, Barrios C, Eiermann W, Toi M, Im YH, Conte P, Martin M, Pienkowski T, Pivot X, Burris HA, Petersen JA, Stanzel S, Strasak A, Patre M, Ellis P (2017) Trastuzumab emtansine with or without pertuzumab versus trastuzumab plus taxane for human epidermal growth factor receptor 2-positive, advanced breast cancer: primary results from the phase III MARIANNE Study. J Clin Oncol 35:141–148
Lipshultz SE, Rifai N, Dalton VM, Levy DE, Silverman LB, Lipsitz SR, Colan SD, Asselin BL, Barr RD, Clavell LA, Hurwitz CA, Moghrabi A, Samson Y, Schorin MA, Gelber RD, Sallan SE (2004) The effect of dexrazoxane on myocardial injury in doxorubicin-treated children with acute lymphoblastic leukemia. N Engl J Med 351:145–153
Sieswerda E, van Dalen EC, Postma A et al (2011) Medical interventions for treating anthracycline-induced symptomatic and asymptomatic cardiotoxicity during and after treatment for childhood cancer [serial online]. Cochrane Database Syst Rev 9
Hensley ML, Hagerty KL, Kewalramani T, Green DM, Meropol NJ, Wasserman TH, Cohen GI, Emami B, Gradishar WJ, Mitchell RB, Thigpen JT, Trotti A III, von Hoff D, Schuchter LM (2009) American Society of Clinical Oncology 2008 clinical practice guideline update: use of chemotherapy and radiation therapy protectants. J Clin Oncol 27:127–145
Kalay N, Basar E, Ozdogru I, Er O, Cetinkaya Y, Dogan A, Oguzhan A, Eryol NK, Topsakal R, Ergin A, Inanc T (2006) Protective effects of carvedilol against anthracycline-induced cardiomyopathy. J Am Coll Cardiol 48:2258–2262
Kaya MG, Ozkan M, Gunebakmaz O, Akkaya H, Kaya EG, Akpek M, Kalay N, Dikilitas M, Yarlioglues M, Karaca H, Berk V, Ardic I, Ergin A, Lam YY (2013) Protective effects of nebivolol against anthracycline-induced cardiomyopathy: a randomized control study. Int J Cardiol 167:2306–2310
Seicean S, Seicean A, Alan N, Plana JC, Budd GT, Marwick TH (2013) Cardioprotective effect of β-adrenoceptor blockade in patients with breast cancer undergoing chemotherapy: follow-up study of heart failure. Circ Heart Fail 6:420–426
Pituskin E, Mackey JR, Koshman S, Jassal D, Pitz M, Haykowsky MJ, Pagano JJ, Chow K, Thompson RB, Vos LJ, Ghosh S, Oudit GY, Ezekowitz JA, Paterson DI (2017) Multidisciplinary approach to novel therapies in cardio-oncology research (MANTICORE 101-breast): a randomized trial for the prevention of Trastuzumab-associated cardiotoxicity. J Clin Oncol 35:870–877
Cardinale D, Colombo A, Sandri MT, Lamantia G, Colombo N, Civelli M, Martinelli G, Veglia F, Fiorentini C, Cipolla CM (2006) Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation 114:2474–2481
Cadeddu C, Piras A, Mantovani G et al (2010) Protective effects of the angiotensin II receptor blocker telmisartan on epirubicin-induced inflammation, oxidative stress, and early ventricular impairment. Am Heart J 160:487.e1-7
Akpek M, Ozdogru I, Sahin O, Inanc M, Dogan A, Yazici C, Berk V, Karaca H, Kalay N, Oguzhan A, Ergin A (2015) Protective effects of spironolactone against anthracycline-induced cardiomyopathy. Eur J Heart Fail 17:81–89
Seicean S, Seicean A, Plana JC, Budd GT, Marwick TH (2012) Effect of statin therapy on the risk for incident heart failure in patients with breast cancer receiving anthracycline chemotherapy: an observational clinical cohort study. J Am Coll Cardiol 60:2384–2390
Chotenimitkhun R, D’Agostino R Jr, Lawrence JA et al (2015) Chronic statin administration may attenuate early anthracycline-associated declines in left ventricular ejection function. Can J Cardiol 31:302–307
Elitok A, Oz F, Cizgici AY, Kilic L, Ciftci R, Sen F, Bugra Z, Mercanoglu F, Oncul A, Oflaz H (2014) Effect of carvedilol on silent anthracycline-induced cardiotoxicity assessed by strain imaging: a prospective randomized controlled study with six-month follow-up. Cardiol J 21:509–515
Avila MS, Ayub-Ferreira SM, de Barros Wanderley MR Jr et al (2018) Carvedilol for prevention of chemotherapy-related cardiotoxicity: the CECCY Trial. J Am Coll Cardiol 71:2281–2290
Boekhout AH, Gietema JA, Milojkovic Kerklaan B, van Werkhoven ED, Altena R, Honkoop A, Los M, Smit WM, Nieboer P, Smorenburg CH, Mandigers CMPW, van der Wouw AJ, Kessels L, van der Velden AWG, Ottevanger PB, Smilde T, de Boer J, van Veldhuisen DJ, Kema IP, de Vries EGE, Schellens JHM (2016) Angiotensin II-receptor inhibition with candesartan to prevent trastuzumab-related cardiotoxic effects in patients with early breast cancer: a randomized clinical trial. JAMA Oncol 2:1030–1037
Cardinale D, Ciceri F, Latini R, Franzosi MG, Sandri MT, Civelli M, Cucchi GF, Menatti E, Mangiavacchi M, Cavina R, Barbieri E, Gori S, Colombo A, Curigliano G, Salvatici M, Rizzo A, Ghisoni F, Bianchi A, Falci C, Aquilina M, Rocca A, Monopoli A, Milandri C, Rossetti G, Bregni M, Sicuro M, Malossi A, Nassiacos D, Verusio C, Giordano M, Staszewsky L, Barlera S, Nicolis EB, Magnoli M, Masson S, Cipolla CM, Cipolla CM, Cardinale D, Ciceri F, Latini R, Sandri MT, Maggioni AP, Labianca R, Tettamanti M, Senni M, Finzi A, Grosso F, Vago T, Civelli M, Gramenzi S, Masson S, Balconi G, Bernasconi R, Salvatici M, Nicolis E, Barlera S, Magnoli M, Buratti MG, Ojeda Fernandez ML, Franzosi MG, Staszewsky L, Vasamì A, Malossi A, Sicuro M, Thiebat B, Barè C, Corzani A, Coccolo F, Colecchia S, Pellegrini C, Bregni M, Appio L, Caico I, G.Rossetti, Mesenzani O, Campana C, Giordano M, Gilardoni M, Scognamiglio G, Corrado G, Battagin D, de Rosa F, Carpino C, Palazzo S, Monopoli A, Milandri C, Giannessi PG, Zipoli G, Ghisoni F, Rizzo A, Pastori P, Callegari S, Sesenna C, Colombo A, G.Curigliano, Fodor C, Mangiavacchi M, Cavina R, Guiducci D, Mazza R, Turazza FM, Vallerio P, Marbello L, Sala E, Fragasso G, Trinca S, Aquilina M, Rocca A, Farolfi A, Andreis D, Gori S, Barbieri E, Lanzoni L, Marchetti F, Falci C, Bianchi A, Mioranza E, Banzato A, Re F, Gaibazzi N, Gullo M, Turina MC, Gervasi E, Giaroli F, Nassiacos D, Verusio C, Barco B, Bertolini A, Cucchi G, Menatti E, Sinagra G, Aleksova A, Guglielmi A, Pinotti G, Gueli R, Mongiardi C, Vallini I (2018) ICOS-ONE study investigators anthracycline-induced cardiotoxicity: a multicenter randomised trial comparing two strategies for guiding prevention with enalapril: the International CardioOncology Society-one trial. Eur J Cancer 94:126–137
Curigliano G, Cardinale D, Suter T et al (2012) Cardiovascular toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidelines. Ann Oncol 23:vii155–vii166
Cardinale D, Colombo A, Lamantia G, Colombo N, Civelli M, de Giacomi G, Rubino M, Veglia F, Fiorentini C, Cipolla CM (2010) Anthracycline-induced cardiomyopathy. Clinical relevance and response to pharmacologic therapy. J Am Coll Cardiol 55:213–220
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mavrogeni, S.I., Sfendouraki, E., Markousis-Mavrogenis, G. et al. Cardio-oncology, the myth of Sisyphus, and cardiovascular disease in breast cancer survivors. Heart Fail Rev 24, 977–987 (2019). https://doi.org/10.1007/s10741-019-09805-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-019-09805-1