Abstract
Somatic cybridization is a technique based on inducted or spontaneous fusion between nuclear genome of one parent and either the cytoplasmic genome of the other parent or a combination of both donors. This approach is considered a powerful biotechnological tool in plant improvement since it enables to obtain novel intergenomic recombinations that are impossible to achieve by conventional crosses. Twenty-five plants were regenerated following protoplast fusion of Cleopatra mandarin (Citrus reshni Hort. ex Tan.) and sour orange (Citrus aurantium L.) and a new 2n cybrid has been identified (4 % of plants obtained). A complete phytochemical investigation of the new genotype and both parents was carried out analyzing the polyphenol content of juices by liquid-chromatography–ultraviolet–diode array detector–mass spectrometry and the peel essential oils using a combination of gas chromatography equipped with a flame ionization detector and a mass spectrometry. On the whole 10 flavonoids (7 flavanone and 3 flavones) and 4 hydroxycinnamic acids were detected and quantified, while 67 components were fully characterized and grouped into four classes (monoterpene hydrocarbons, oxygenated monoterpenes, sesquiterpenes, and others) for an easier comparison of all oils. Comparing the new cybrid to both parents an increase of the monoterpene hydrocarbons and a corresponding decrease of oxygenated components were shown. The aim of the present work was to evaluate whether the genome rearrangements in the new cybrid, obtained through not conventional strategies, can produce superior traits and improved performance such as enhanced presence and quality of bioactive components.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Numerous epidemiological studies conducted in many countries indicate that a diet rich in fruits and vegetables, including fruit and vegetable products, delays the ageing process and reduces the risk of various lifestyle diseases (Szajdek and Borowska 2008). In this context, several studies have associated the consumption of polyphenols with lower risks of different types of cancer (Nichenametla et al. 2006) and cardiovascular diseases (Kris-Etherton et al. 2002), and have shown that polyphenols possess antioxidant, anti-inflammatory and anti-ageing activity (Benavente-García and Castillo 2008). Citrus fruits are among the richest dietary sources of flavonoids (Tripoli et al. 2007) and other bioactive compounds such as limonoids, carotenoids, amines, organic acids, sterols and furocoumarins (Kris-Etherton et al. 2002). Over the past decades, a large number of studies have been carried out on the biomedical properties of many Citrus species, among them sour orange (Citrus aurantium L.) and more recently Cleopatra mandarin (Citrus reshni Hort. ex Tan.) (Barreca et al. 2011; Ram et al. 2011; Hamdan et al. 2013).
The breeding programs success is mainly dependent on the rate of the genetic variation available. Although the nuclear genome has a predominant role on the inheritance of most plant traits, cytoplasmic factors and cytoplasm/nucleus interactions are also important. Chloroplast and mitochondrial genes, interacting with nuclear genes, are implicated in the control of several morphological, physiological and agronomic traits. However, the influence of the cytoplasm on the phenotypic traits is largely unexplored. The role for nucleus × cytoplasm interactions has been reported in some cases (Shonnard and Gepts 1994; Matsui et al. 2002; Zhang et al. 2003; Atienza et al. 2008; Wang et al. 2010), nevertheless more aspects have to be still clarified.
Somatic hybridization by protoplast fusion has progressed steadily over the past 30 years. Considerable successful instances of applying this technique for breeding in plant of economic interest, including rice, potato, wheat, rapeseed and citrus were reported (Johnson and Veilleux 2001; Grosser and Gmitter 2011; Wang et al. 2013). In Citrus, somatic hybridization has been employed to circumvent the problems encountered using conventional breeding approaches, such as sexual incompatibility, nucellar polyembryony, male or female sterility and others (Grosser et al. 2000).
Traditional symmetric fusion, combining entire genomes of both parents, often contains many unwanted nuclear encoded agronomic traits. Citrus somatic hybrids are often sterile, show morphologically abnormal traits and may produce uncontrolled genomic instabilities, thus hampering their use for breeding of varieties. Therefore, asymmetric somatic cybridization, a technique based on the induction of unilateral chromosome elimination, has been developed as a means to create morphologically normal hybrids, called cybrids, that contain the whole genome of one species and either the cytoplasmic genome of the other (non-nuclear) parent or that of a combination of both parental species. The potential of cybrids in citrus improvement could be relevant since it might be possible to breed specific traits associated to the cytoplasmic genomes while maintaining cultivar integrity (Saito et al. 1993; Guo et al. 2004). In Citrus interspecific, intraspecific and intergeneric symmetric protoplast fusion can spontaneously produce cybrids. To date, more than 40 citrus cybrids plants have been unexpectedly regenerated following symmetric somatic fusion experiments (Guo et al. 2013). Most of these cybrids have the nuclear genome of the leaf parent, the mitochondrial genome of the embryogenic callus parent and the chloroplast genome randomly inherited (Guo et al. 2013; Moreira et al. 2000), however, several investigations revealed the co-existence or recombination of parental mitochondrial genomes in cybrid plants (Iovene et al. 2007; Zubko et al. 2003). Positive variation for important agronomic traits such as maturity data and number of seeds (Grosser et al. 2000), tolerance to diseases (Tusa et al. 2000), metabolic, organoleptic and aromatic features (Fanciullino et al. 2005; Bassene et al. 2008, 2011; Wang et al. 2010) have been observed in citrus cybrids
Pursuing our activity (Ruberto et al. 1999; Tusa et al. 2007; Abbate et al. 2012; Fatta Del Bosco et al. 2013) on the characterization of metabolic profiles of new Citrus hybrids, aimed to the selection new Citrus fruits with an improved content of nutraceutical components and/or with new aromatic profiles, as well as further tool to establish quality and typicalness of these new fruits. Here the results of a combined chemical and genetic study of a new diploid cybrid obtained from the protoplast fusion of Cleopatra mandarin and sour orange and its corresponding parents (Fig. 1) are reported, in an effort to evaluate the effect on juices polyphenols and peel essential oil characteristics associated to the new nucleo-cytoplasmic arrangement of the cybrid.
Materials and methods
Plant material
The Citrus plants investigated in this study were obtained by somatic protoplast fusion between Cleopatra mandarin (C. reshni—CLEO) and sour orange (C. aurantium—SO). The CLEO protoplasts were isolated from suspension culture initiated from nucellus-derived embryogenic callus while the SO’s protoplasts were derived from leaves of young nucellar seedlings germinated in vitro. CLEO and SO protoplasts were purified, mixed and fused using polyethylene glycol (PEG) method. Fusion cultures, embryogenic colonies, embryoids and large somatic embryos developing from recovered hybrid calli were grown. Developing somatic embryos were first transferred and cultured on 1500 medium for enlargement and after on B+ or DBA3 medium to induce shoot growth. Shoots were rooted in Magenta boxes (Magenta Corp., Chicago, IL, USA) and, finally the rooted plants were transferred to a commercial potting mixture and maintained at high humidity for acclimation according to the procedure described in Siragusa et al. (2007). Twenty-five regenerated plants were developed.
Plants obtained from the protoplast fusion event and relative parents were cultivated in the experimental station of the Istituto di Bioscienze e BioRisorse (IBBR-CNR), Collesano, Palermo. CLEO, SO and prescreened putative cybrid plants morphologically resembling SO (Grosser et al. 1996) were selected and the fruits were collected between February 2012 and March 2013.
Flow cytometry analysis
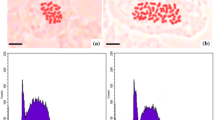
The samples’ ploidy was evaluated by flow cytometry analysis, using diploid SO as reference standard. The analysis was carried out with the Partec PAS flow cytometer (Partec, http://sysmex-europe.com/partec), equipped with a mercury lamp. Fully expanded leaves (0.1 g) were chopped in a glass Petri dish with 400 μL nuclei extraction buffer (Partec solution CyStain® UV Precise P, 250 tests). The solution was filtered through a 30 μm Cell-Trics disposable filter Partec and 1.6 mL of staining solution containing 4,6-diamidino-2-phenylindole was added. Routinely, 4000–5000 nuclei for each sample were measured (Galbraith et al. 1998) and histograms of DNA content were generated using the Partec software package (FloMax). Three replicates for each sample were carried out.
DNA extraction
DNA was isolated from young, fresh leaves collected in the field and, after lyophilization, stored at −80 °C until required. The extraction was carried out by the Doyle and Doyle CTAB method (1990). The extract was treated with DNase-free RNase (Roche Diagnostics, Germany) and quantified by using a NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA).
Genetic analysis
A total of 22 simple sequence repeats (SSRs), 8 nuclear (nSSR), 7 chloroplast (cpSSR) and 7 mitochondrial (mtSSR) loci respectively, were chosen to investigate the genetic profile of CLEO, SO and putative cybrids obtained by protoplast fusion. The used markers (Table 1) were selected based on their dispersal map location and polymorphism rate (Cheng et al. 2003, 2005; Guo et al. 2006; Jannati et al. 2009; Froelicher et al. 2011). Multiplex PCRs were performed, amplifying several loci simultaneously, using the Qiagen multiplex PCR kit (Qiagen GmbH, Hilden, Germany). One of each pair was fluorescently labelled with FAM, JOE, TAMRA or ROX. PCR reactions were carried out in reaction volumes of 25 µL containing 20 ng of DNA and 0.2 µM each of forward and reverse primers, in a 2× Qiagen multiplex Master Mix. Reactions were performed under the following conditions: 15 min at 95 °C; 35 cycles of 30 s at 94 °C, 1 min 30 s at 57 °C, 1 min at 72 °C, followed by 30 min at 60 °C. Amplification products were checked and quantified by electrophoresis on 2 % agarose gel using Low DNA mass Ladder (Invitrogen). The fragments were separated by capillary electrophoresis and genotyped with an ABI PRISM 3500 genetic analyzer.
Fruit collection, juice preparation and standards
Fruits of SO, CLEO and their cybrids, were collected at the proper ripening stage between February 2012 and March 2013, from three trees for each genotype. Only the true cybrids, verified using cytometry analysis and molecular markers, were processed. The juice from fruits (4 or 5 for each tree) was obtained by a domestic squeezer and used freshly or stored at −18 °C.
HPLC grade diosmin, vitexin, caffeic acid, eriocitrin, narirutin, naringenin 7-O-glucoside, naringin, hesperidin, and didymin were purchased from Extrasynthese—Z.I. (Lyon Nord, France), sinapic acid and coumaric acid were obtained from Fluka Chemie (Buchs SG, Switzerland), ascorbic acid, ethylene diamine tetracetate disodium salt, neohesperidin, ferulic acid and isoferulic acid were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Sample preparation for analyses of hydroxycinnamic acids
To a 1 mL of filtered (0.45 μm) juice, 1 mL of a 2 N NaOH solution (containing 10 mM EDTA and 57 mM ascorbic acid) and 10 μL of 1 mM isoferulic acid in MeOH as internal standard were added. The solution was maintained at 40 °C for 30 min under 300 rpm stirring. Each sample was then adjusted to pH 7 with 2 N HCl and freeze-dried. After suspending the residue in methanol (1 mL) and filtering on 0.45 μm filter, the solution was directly injected to HPLC system.
Sample preparation for analyses of flavanones and flavones
The juice was filtered on 0.45 μm filter and directly injected to the HPLC system. Flavonoid quantification was achieved from the recorded absorbance in the chromatogram compared with its external standard curve. Diosmetin 6,8-di-C-glucosyde and vicenin-2, whose reference samples were not available, were quantified against vitexin assuming a response factor equal to 1.
HPLC–UV and HPLC–MS–ESI analyses
HPLC analyses were performed on a thermostated (40 °C) Phenomenex Luna® C18 250 × 4.6 mm (5 μm) column at 1 mL/min flow rate using water-formic acid 9:1 (v/v) as eluant A and acetonitrile-formic acid, 9:1 (v/v) as eluant B with the following gradient of composition: t0min B (5 %), t5min B (5 %), t20min B (15 %), t28min B (15 %), t38min B (30 %), t50min B (100 %), t55min B (100 %), injection volume: 20 μL.
Qualitative analyses with simultaneous ESI–MS and UV–DAD detection were carried out on a waters 1525 pump (Waters Associates) equipped with a waters 996 photodiode array detector and waters micromass ZQ2000 mass spectrometer detector. ESI–MS detection was performed in negative mode setting a capillary voltage of 3.5 kV, a cone voltage of 40 V, a vaporizer temperature of 250 °C, a carrier gas flow (nitrogen) of 500 L/h and mass acquisition between 100 and 1500 Da. DAD analyses were carried out in the range between 200 and 700 nm, setting the detector at 280 nm for flavanones, 340 nm for flavones and 320 nm for hydroxycinnamic acids respectively. The identification of juice constituents was carried out by comparison of the spectral properties (UV and ESI–MS) of analytes with those of reference samples.
Quantitative determination of the citrus juice composition was carried out on a Dionex HPLC system equipped with a P680 pump, a UV-170U detector and ASI 100 auto-sampler. All determinations were carried out in triplicate, using internal standard method for hydroxycinnamic acids and external standard method for flavanones and flavones.
Isolation of essential oils
Fresh rind tissue (flavedo, 100 g) of each sample was subjected to hydrodistillation until there was no significant increase in the volume of oil collected (3 h). The oils were dried over anhydrous sodium sulfate and stored under N2 in a sealed vial at −20 °C until required.
Gas chromatography (GC) of essential oils
Essential oils were analyzed in the fast mode on a Shimadzu gas chromatograph, Model 17-A equipped with a flame ionization detector (FID), operating software class VP chromatography date system version 4.3 (Shimadzu Co., Kyoto, Japan). Analytical conditions: SPB-5 capillary column (15 m × 0.10 mm × 0.10 μm), helium as carrier gas. Injection in split mode (1:200), injected volume 1 μL (25 μL of oil in 400 μL of CH2Cl2), injector and detector temperature 250 and 280 °C, respectively. Linear velocity in column 51 cm/s. The oven temperature was held at 60 °C for 1 min, then programmed from 60 to 280 °C at 10 °C/min. Percentages of compounds were determined from their peak areas in the GC–FID profiles.
Gas-chromatography–mass spectrometry (GC–MS) of essential oils
GC–MS was carried out in the fast mode on a Shimadzu GC–MS mod. GCMS-QP5050A, operating software GCMS solution version 1.02 (Shimadzu). Ionization voltage in electronic impact mode 70 eV, electron multiplier 1000 V, transfer line temperature 280 °C, injection in split mode (1:96), constant linear velocity in column 50 cm/s. Analytical conditions were the same as GC.
Identification of essential oil components
The identity of components was based on their retention indexes relative to C9–C22 n-alkanes (Alltech Italy) on the SPB-5, computer matching of spectral MS data those from NIST MS 107 and NIST 21 libraries (NIST 1998), the comparison of the fragmentation patterns with those reported in literature (Adams 2007) and, whenever possible, co-injections with authentic standards, which were purchased from Aldrich Chemical Co., Extrasynthese, France, and Fluka Chemie AG, Switzerland.
Statistical analysis
SPSS software, version 14.1, was used to carry out statistical analysis of the data. ANOVA and HSD Tukey test were applied to the data to determine significant differences between the analyzed components; the model was statistically significant with a value of p ≤ 0.01.
Results
Ploidy analysis
The ploidy of the 25 regenerated plants was estimated by flow cytometry using diploid SO as internal standard. The results displayed that 21 genotypes (84 %) obtained by somatic protoplast fusion between CLEO and SO were diploid (2n = 2x = 18) since showed the same profile of the control (2n = 2x = 18). The other 4 plants (16 %), showing a 4n ploidy level, were classified as hybrids and kept out from the present study.
Molecular analysis
Nuclear, chloroplastic and mitochondrial genomes were analyzed by using 22 SSR markers (Table 1) to characterize the genomic constitution of putative 2n cybrids selected and their parental (CLEO and SO). Three out of eight nuclear markers (TAA27, CAC15 and CAT01) enabled to distinguish the parents of the somatic protoplast fusion and thus the hybrid status of the regenerated plants could be checked. The results showed that the nuclear profile of seven (24 %) regenerated diploid plant was identical to the leaf parent (SO) confirming its origin as donor of nuclear genome to the regenerated plants. The other 14 prescreened samples presented the same allelic profile of CLEO therefore were excluded from the analysis.
To study in depth the nature of seven putative cybrids, the constitution of their cytoplasmic genome was also investigated. Two out of seven chloroplast microsatellite loci selected (SPCC9 and SPCC11) showed polymorphism between CLEO and SO, thus the identification of the parental genomes was possible. Six out of seven regenerated plants had the same plastidial genome profile of SO, therefore these plants were not true cybrids. Instead, in the remaining sample, the plastidial genome was inherited from both parents, since the putative cybrid, named CLEO–SO, displayed a heterozygous profile for SPCC9 and SPCC11 markers with alleles belonging to both parents (Table 2). In the case of the mitochondrial genome, the profiles of the Citrus specie used to isolate protoplasts from embryogenic calli (CLEO) were observed in the CLEO–SO plant (Table 2) for three polymorphic markers (rrn18-1/rrn5, nad 2/3-4 and nad71-2). Therefore, molecular results confirmed that the Citrus plant named CLEO–SO, obtained by somatic protoplast fusion between CLEO and SO, was a cybrid genotype, with nuclear genome from SO, chloroplast genome from both parents while the mitochondria were inherited from CLEO.
Chemicals profile
Polyphenol profile and essential oil composition of the new cybrid selected were then investigated comparing its chemical profile with both parents, to evaluate the effect associated to the new nucleo-cytoplasmic organization.
Polyphenol profiles of juices
Table 3 lists the polyphenols, ten flavonoids (TF) and four hydroxycinnamic acids (HCA) reported in Fig. 2, analyzed in this study with their spectral features utilized for characterization and quantitative determination, whereas Table 4 reports the results on the content of the aforesaid components of the two parents: CLEO and SO, and their diploid cybrid (CLEO–SO).
The profile of SO is characterized by the predominance of two flavanones, naringin and neohesperidin the two bitter components which characterize this Citrus species, whereas vicenin-2 and diosmetin 6,8-di-C-glucoside are the main flavones. Amongst HCA, the ferulic is the main one, coumaric, caffeic and sinapic, in that order, follow at a considerable gap. CLEO shows a consistent lower amount of flavonoids with respect to SO. In this case hesperidin and narirutin are the main flavanones, and vicenin-2 is largely the main flavone. Concerning HCA, also in this the ferulic is largely the main acid, sinapic, caffeic and coumaric follows at large distance, whereas caffeic and sinapic show a comparable and low amount.
The first and significant compositional aspect of the cybrid is the higher content of flavonoids with respect to both parents. In fact, flavanones and flavones reach the amount of 300.97 and 79.70 mg/L, respectively. From a qualitative point of view the main flavanones of the cybrid are naringin and neohesperidin, as for SO. Also flavones are present in higher amount with respect to both parents, also in this case vicenin-2 and diosmetin 6,8-di-C-glucoside, are the main components as for sour orange. Concerning the HCA, ferulic acid is still the main component followed by coumaric, caffeic and sinapic as previously observed for SO parent.
A comprehensive evaluation of these data allows to establish that the new cybrid concerning its polyphenol profile maintain substantially that of SO leaf parent.
Essential oil composition
Table 5 lists the composition of the essential oils of all samples. In total, 67 components were fully identified and grouped into four classes: monoterpene hydrocarbons with 11 components, oxygenated monoterpenes, the most numerous class, with 25 compounds, sesquiterpenes and others, with 10 and 21 compounds, respectively. Monoterpenes hydrocarbons were in all cases the most highly represented class. Limonene, as usual for most of Citrus peel essential oil, is the main component, ranging between 90 and 93 %. Myrcene is the second monoterpene hydrocarbons in all samples, SO shows β-pinene and γ-terpinene slightly over the 1 %, whereas in the other two samples, CLEO and new cybrid CLEO–SO, all components are below 1 %. Oxygenated monoterpenes, which normally give a quality mark to the Citrus oils, are represented at very low extent: CLEO shows the highest amount (ca. 4 %), being linalool the main component, other slight significant components are terpineol and terpinen-4-ol (Lota et al. 2001; Hamdan et al. 2013). SO shows a lower content of these components, linalool is still the main one, followed by geraniol and terpineol. In CLEO–SO the lowest amount of these oxygenated compounds has been evidenced, in fact all components are below 1 % being linalool and terpineol the main ones (Table 5). The other two classes of components, namely sesquiterpenes and others (not terpenoid compounds), even though totally represented by 31 components are in almost all samples below 1 %.
Discussion
The aim of this work was to evaluate whether superior traits and improved performance can be obtained through the nucleo-cytoplasmic arrangement that characterize the cybrid. Genetic and chemical analysis of diploid cybrid and its corresponding parents was conducted to evaluate the effect on juices polyphenols and peel essential oil production associated to the new nucleo-cytoplasmic organization in the cybrid analyzed.
Primarily, the genetic arrangement of the genotype obtained by somatic protoplast fusion to verify the true cybrid nature of the genotype was evaluated. As reported in previous studies, cybrids harbor one parental nuclear genome and either the plastidial genome of the other parent or a combination of both parents (Guo et al. 2004; Fanciullino et al. 2005; Bassene et al. 2011; Grosser et al. 1996; Cheng et al. 2005). Molecular analysis confirmed the cybrid nature of the genotype obtained from protoplast fusion of CLEO and SO. Indeed, CLEO–SO showed nuclear genome of the leaf parent SO, the mitochondria genome was inherited from the callus parent CLEO while, regarding the plastidial genome, SSR marker analysis revealed the coexistence of both parents. According to available data, the chloroplast genome in the cybrid are derived mainly uniparentally from either of the fusion parents, although coexistence of the chloroplast genomes from both parents in the hybrids is also reported (Guo et al. 2007). Therefore, these results indicated that the CLEO–SO is a true cybrid.
Bio-molecular studies on the secondary metabolic profile and on volatile components, of Citrus somatic hybrids are rather limited (Fanciullino et al. 2005; Gancel et al. 2003, 2005), therefore it is difficult to find common aspects in the accumulation mechanism of these components at the moment. In fact, previous studies on the volatile compounds from leaves and peels of interspecific somatic hybrids showed a strongly inhibition of some components (i.e. sesquiterpene hydrocarbons), as well as an overproduction of the other ones (i.e. citronellal); these contrasting results prompted some authors to claim that Citrus somatic hybrids do not retain their parental traits (Gancel et al. 2005). In the present study, a slight overproduction of total juice flavonoids (Table 4), an increase of the monoterpene hydrocarbons and a corresponding decrease of oxygenated components with respect to both parents in the essential oils (Table 5) were observed in CLEO–SO. Our results show, analogously to many similar ones, just how difficult it is to establish an inheritance mechanism related to the biosynthetic accumulation of secondary metabolites throughout various breeding methodologies of Citrus (Rapisarda et al. 2003; Fabroni et al. 2012). This is probably due to the complexity and genetic changeability of this genus (Guo et al. 2006; Barkley et al. 2006). In any case these comparisons clearly provide an insight into the spectrum of changes associated with the genetic manipulation by protoplast fusion.
Besides, the molecular constitution of cybrids provides a novel and promising approach for citrus breeding. In fact, although the nuclear genome has a predominant role on the inheritance of most plant traits, it is recognized that plant cell function depends on the coordinate action and communication between nuclear and cytoplasmic genomes. There is increasing awareness of the intricate networks and signal transduction pathways that link plant organelles, called “retrograde regulation” (Strand 2004; Liu and Butow 2006; Rhoads and Subbaiah 2007).
Recent investigations underline how the chloroplast and their redox state are involved in the regulation of important biological processes, including nuclear/plastid gene expression, hormone signalling and stress responses (Suzuki et al. 2002). Similarly, mitochondrial respiration is important for neutralizing excess of photosynthetic reducing power, preventing oxidative damage (Dinakar et al. 2010), highlighting the key role of plant mitochondria during biotic stress responses. Indeed, there are evidences that signals initiated by plant–pathogen interactions may directly or indirectly transfer to mitochondrial components consequently increasing generation of mitochondrial reactive oxygen species (mtROS). The mtROS or mitochondrial dysfunction may act as cellular signals that initiate nuclear gene expression involved in defense and hypersensitive responses (Amirsadeghi et al. 2007). Redox regulation and ROS metabolism are interlinked and involved in optimizing the function of mitochondria, chloroplast and other organelles. This network needs implied regulation and high level of coordination especially under stress conditions.
Chloroplast and mitochondrial genes, or their interactions with nuclear genes, are clearly implicated in the control of several morphological, physiological and agronomic traits (Woodson and Chory 2008). Cytoplasmic male sterility (CMS) is, by far, the most known cytoplasmically inherited trait. The drought tolerance in Brassica (Uprety and Tomar 1993), heat tolerance in common bean (Shonnard and Gepts 1994), productivity and tolerance to iron deficiency in wheat (Zhang et al. 2003; Jones et al. 1998) and disease resistance in barley (Matsui et al. 2002), were also associated to cytoplasmically inherited characters. In wheat significant differences for seed lutein content between euplasmic and their respective alloplasmic lines were also observed (Atienza et al. 2008), underscoring the important role of the cytoplasm to determine the seed carotenoid accumulation. These evidences prove the useful use of cybridization as sources of cytoplasmic variability for broadening the genetic diversity in plant, underlining the important role of the nuclear–cytoplasm interaction.
As reported in Citrus cybrid combinations, cytoplasm substitution shows positive effects on important agronomic traits, as CMS (Yamamoto et al. 1997), aroma and essential oil characteristics (Fanciullino et al. 2005; Abbate et al. 2012), fruit taste and nutritional quality (Bassene et al. 2008), therefore cybridization can be used in citrus cultivar improvement as a strategy to transfer specific traits associated with mitochondrial and chloroplast genomes.
Conclusions
The results obtained in this work point out the role of cytoplasm and/or the nucleus–cytoplasmic interactions in determining new skins with respect to polyphenol distribution and essential oil composition in citrus cybrid fruit. Since the somatic cybridization represents a suitable alternative strategy against the major biotic/abiotic constraints of the Mediterranean area, developing in meantime genotypes with interesting chemical traits, the cytoplasm constitution of the new CLEO–SO cybrid could have positive potential effect on different features.
In conclusion, the regeneration of somatic cybrid CLEO–O is an important beginning step that can allow enlarging the citrus germplasm patrimony available for further studies at the biochemical, agronomic and genetic levels.
References
Abbate L, Tusa N, Fatta Del Bosco S, Strano T, Renda A, Ruberto G (2012) Genetic improvement of Citrus fruits: new somatic hybrids from Citrus sinensis (L.) Osb. and Citrus limon (L.) Burm. f. Food Res Int 48:284–290
Adams RP (2007) Identification of essential oil components by gas chromatography/mass spectrometry, 4th edn. Allured Publishing Corporation, Carol Stream
Amirsadeghi S, Robson CA, Vanlerberghe GC (2007) The role of the mitochondrion in plant responses to biotic stress. Physiol Plant 129:253–266
Atienza SG, Martín A, Pecchioni N, Platani C, Cattivelli L (2008) The nuclear–cytoplasmic interaction controls carotenoid content in wheat. Euphytica 159:325–331
Barkley NA, Roose ML, Krueger RR, Federici CT (2006) Assessing genetic diversity and population structure in a citrus germplasm collection utilizing simple sequence repeat markers (SSRs). Theor Appl Genet 112:1519–1531
Barreca D, Bellocco E, Caristi C, Leuzzi U, Gattuso G (2011) Distribution of C- and O-glycosyl flavonoids, (3-hydroxy-3-methylglutaryl) glycosyl flavanones and furocoumarins in Citrus aurantium L. juice. Food Chem 124:576–582
Bassene J-B, Berti L, Carcouet E, Dhuique-Mayer C, Fanciullino A-L, Bouffin J, Ollitrault P, Froelicher Y (2008) Influence of mitochondria origin on fruit quality in a citrus cybrid. J Agric Food Chem 56:8635–8640
Bassene J-B, Froelicher Y, Navarro L, Ollitrault P, Ancillo G (2011) Influence of mitochondria on gene expression in a citrus cybrid. Plant Cell Rep 30:1077–1085
Benavente-García O, Castillo J (2008) Update on uses and properties of Citrus flavonoids: new findings in anticancer, cardiovascular, and anti-inflammatory activity. J Agric Food Chem 56:6185–6205
Cheng Y, Guo W, Deng X (2003) cpSSR: a new tool to analyse chloroplast genome of Citrus somatic hybrids. Acta Bot Sin 45:906–909
Cheng Y, De Vicente MC, Meng H, Guo W, Tao N, Deng X (2005) A set of primers for analyzing chloroplast DNA diversity in Citrus and related genera. Tree Physiol 25:661–672
Dinakar C, Abhaypratap V, Yearla SR, Raghavendra AS, Padmasree K (2010) Importance of ROS and antioxidant system during the beneficial interactions of mitochondrial metabolism with photosynthetic carbon assimilation. Planta 231:461–474
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Fabroni S, Ruberto G, Rapisarda P (2012) Essential oil profiles of new citrus hybrids, a tool for genetic citrus improvement. J Essent Oil Res 24:159–169
Fanciullino A-L, Gancel A, Froelicher Y, Luro F, Ollitrault P, Brillouet JM (2005) Effects of nucleo-cytoplasmic interactions on leaf volatile compounds from citrus somatic diploid hybrids. J Agric Food Chem 53:4517–4523
Fatta Del Bosco S, Abbate L, Tusa N, Strano T, Renda A, Ruberto G (2013) Genetic improvement of Citrus fruits: the essential oil profiles in a Citrus limon backcross progeny derived from somatic hybridization. Food Res Int 50:344–350
Froelicher Y, Mouhaya W, Bassene J, Costantino G, Kamiri M, Luro F, Morillon R, Ollitrault P (2011) New universal mitochondrial PCR markers reveal new information on maternal citrus phylogeny. Tree Genet Genomes 7:49–61
Galbraith DW, Lambert GM, Macas J, Doležel J (1998) Analysis of nuclear DNA content and ploidy in higher plants. In: Robinson JP, Darzynkiewicz Z, Dean PN, Dressler LG, Orfao A, Rabinovitch PS, Stewart CC, Tanke HJ, Wheeless LL (eds) Current protocols in cytometry. Wiley, New York, pp 7.6.1–7.6.22
Gancel AL, Ollitrault P, Froelicher Y, Tomi F, Jacquemond C, Luro F, Brillouet J-M (2003) Leaf volatile compounds of seven citrus somatic tetraploid hybrids sharing willow leaf mandarin (Citrus deliciosa Ten.) as their common parent. J Agric Food Chem 51:6006–6013
Gancel A-L, Ollitrault P, Froelicher Y, Tomi F, Jacquemond C, Luro F, Brillouet J-M (2005) Citrus somatic allotetraploid hybrids exhibit a differential reduction of leaf sesquiterpenoid biosynthesis compared with their parents. Flavour Fragr J 20:626–632
Grosser JW, Gmitter FG Jr (2011) Protoplast fusion for production of tetraploids and triploids: applications for scion and rootstock breeding in citrus. Plant Cell Tissuebiotechnology of plant organelles Org 104:343–357
Grosser JW, Gmitter FG Jr, Tusa N, Reforgiato Recupero G, Cucinotta P (1996) Further evidence of a cybridization requirement for plant regeneration from citrus leaf protoplasts. Plant Cell Rep 15:672–676
Grosser JW, Ollitrault P, Olivares-Fuster O (2000) Somatic hybridization in citrus: an effective tool to facilitate variety improvement. In Vitro Cell Dev Plant 36:434–449
Guo WW, Cai XD, Grosser JW (2004) Somatic cell cybrids and hybrids in plant improvement. In: Daniel H, Chase CD (eds) Molecular biology and biotechnology of plant organelles. Kluwer, Dordrecht, pp 635–659
Guo WW, Cheng Y, Chen C, Deng X (2006) Molecular analysis revealed autotetraploid, diploid and tetraploid cybrid plants regenerated from an interspecific somatic fusion in Citrus. Sci Hortic 108:162–166
Guo WW, Wu RC, Cheng YJ, Deng XX (2007) Production and molecular characterization of Citrus intergeneric somatic hybrids between red tangerine and citrange. Plant Breed 126:72–76
Guo WW, Xiao SX, Deng XX (2013) Somatic cybrid production via protoplast fusion for citrus improvement. Sci Hortic 163:20–26
Hamdan DI, Abdulla RH, Mohamed ME, El-Shazly AM (2013) Chemical composition and biological activity of essential oils of Cleopatra mandarin (Citrus reshni) cultivated in Egypt. J Pharmacogn Phytother 5(5):83–90
Iovene M, Savarese S, Cardi T, Frusciante L, Scotti N, Simon PW, Carputo D (2007) Nuclear and cytoplasmic genome composition of Solanum bulbocastanum (+) S. tuberosum somatic hybrids. Genome 50:443–450
Jannati M, Fotouhi R, Abad AP, Salehi Z (2009) Genetic diversity analysis of Iranian citrus varieties using micro-satellite (SSR) based markers. J Hortic For 1:120–125
Johnson AAT, Veilleux RE (2001) Somatic hybridization and applications in plant breeding. Plant Breed Rev 20:167–225
Jones P, Keane EM, Osborne BA (1998) Effects of alien cytoplasmic variation on carbon assimilation and productivity in wheat. J Exp Bot 49:1519–1528
Kris-Etherton PM, Hecker KD, Bonanome A, Coval SM, Binkoski AE, Hilpert KF, Griel AE, Etherton TD (2002) Bioactive compounds in foods: their role in the prevention of cardiovascular disease and cancer. Am J Med 113 (Suppl 9B):71S–88S
Liu Z, Butow RA (2006) Mitochondrial retrograde signaling. Ann Rev Genet 40:159–185
Lota M-L, de Rocca SD, Tomi F, Casanova J (2001) Chemical variability of peel and leaf essential oils of 15 species of mandarins. Biochem Syst Ecol 29:77–104
Matsui K, Yoshida M, Ban T, Komatsuda T, Kawada N (2002) Role of male-sterile cytoplasm in resistance to barley yellow mosaic virus and Fusarium head blight in barley. Plant Breed 121:237–240
Moreira CD, Chase CD, Gmitter FG Jr, Grosser JW (2000) Inheritance of organelle genomes in citrus somatic cybrids. Mol Breed 6:401–405
Nichenametla SN, Taruscio TG, Barney DL, Exon JH (2006) A review of the effects and mechanism of polyphenolics in cancer. Crit Rev Food Sci Nutr 46:161–183
NIST (1998) National Institute of Standard and Technology. Mass Spectral Library (Version 1998)
Partec (2015). Symsex Europe GmbH, Germany. http://www.sysmex-europe.com/partec
Ram MU, Guddadarangavvanahally KJ, Bhimanagouda SP (2011) Rapid separation method of polymethoxyflavones from citrus using flash chromatography. Sep Purif Technol 81:151–158
Rapisarda P, Pannuzzo P, Romano G, Russo G (2003) Juice components of a new pigmented Citrus hybrid Citrus sinensis (L.) Osbeck × Citrus clementina Hort. ex Tan. J Agric Food Chem 51:1611–1616
Rhoads DM, Subbaiah CC (2007) Mitochondrial retrograde regulation in plants. Mitochondrion 7:177–194
Ruberto G, Starrantino A, Rapisarda P (1999) Citrus improvement—chemical and genetic aspects. In: Pandalai SG (ed) Recent research developments in agricultural and food chemistry. Research Signpost, Trivandrum, pp 445–470
Saito W, Ohgawara T, Shimizu J, Ishii S, Kobayashi S (1993) Citrus cybrid regeneration following cell fusion between nucellar cells and mesophyll cells. Plant Sci 88:195–201
Shonnard GC, Gepts P (1994) Genetics of heat tolerance during reproductive development in common bean. Crop Sci 34:1168–1175
Siragusa M, Carra A, Salvia L, Puglia AM, De Pasquale F, Carimi F (2007) Genetic instability in calamondin (Citrus madurensis Lour.) plants derived from somatic embryogenesis induced by diphenylurea derivative. Plant Cell Rep 26:1289–1296
Strand A (2004) Plastid-to-nucleus signaling. Curr Opin Plant Biol 7:621–625
Suzuki N, Koussevitzky S, Mitter R, Miller G (2002) ROS and redox signaling in the response of plants to abiotic stress. Plant Cell Environ 35:259–270
Szajdek A, Borowska EJ (2008) Bioactive compounds and health-promoting properties of berry fruits: a review. Plant Food Hum Nutr 63:147–156
Tripoli E, La Guardia M, Giammanco S, Di Majo D, Giammanco M (2007) Citrus flavonoids: molecular structure, biological activity and nutritional properties: a review. Food Chem 104:466–479
Tusa N, Fatta Del Bosco S, Nigro F, Ippolito A (2000) Response of cybrids and a somatic hybrid of lemon to Phoma tracheiphila infections. HortScience 35:125–127
Tusa N, Abbate L, Renda A, Ruberto G (2007) Polyphenol distribution in juices from Citrus allotetraploid somatic hybrids and their sexual hybrids. J Agric Food Chem 55:9089–9094
Uprety DC, Tomar VK (1993) Photosynthesis and drought resistance of Brassica carinata and its parent species. Photosynthetica 29:321–327
Wang L, Pan ZY, Guo WW (2010) Proteomic analysis of leaves from a diploid cybrid produced by protoplast fusion between Satsuma mandarin and pummelo. Plant Cell Tissue Org 103:165–174
Wang J, Jiang J, Wang Y (2013) Protoplast fusion for crop improvement and breeding in China. Plant Cell Tissue Org 112:131–142
Woodson JD, Chory J (2008) Coordination of gene expression between organellar and nuclear genomes. Nat Rev Genet 9:383–395
Yamamoto M, Matsumoto R, Okudai N, Yamada Y (1997) Aborted anthers of Citrus result from gene–cytoplasmic male sterility. Sci Hortic 70:9–14
Zhang A, Yu F, Zhang F (2003) Alien cytoplasm effects on phytosiderophore release in two spring wheats (Triticum aestivum L.). Gen Res Crop Evol 50:767–772
Zubko MK, Zubko EI, Adler K, Grimm B, Gleba YY (2003) New CMS phenotypes in cybrids Nicotiana tabacum L. (Hyoscyamus niger L.). Ann Bot Lond 92:281–288
Acknowledgments
The authors wish to thank Mrs. Tonia Strano, Mr. Agatino Renda (ICB-CNR, Catania) and Dr. Antonio Motisi (IBBR-CNR, Palermo) for their skilful technical assistance. This work was financially supported by Consiglio Nazionale delle Ricerche (CNR, Rome, Italy).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Sergio Fatta Del Bosco and Edoardo Napoli have equally contributed to this work.
Rights and permissions
About this article
Cite this article
Fatta Del Bosco, S., Napoli, E., Mercati, F. et al. Somatic cybridization for Citrus: polyphenols distribution in juices and peel essential oil composition of a diploid cybrid from Cleopatra mandarin (Citrus reshni Hort. ex Tan.) and sour orange (Citrus aurantium L.). Genet Resour Crop Evol 64, 261–275 (2017). https://doi.org/10.1007/s10722-015-0348-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-015-0348-x