Abstract
Industrialization and urbanization are the main sources of pollutions worldwide and particularly in developing countries. This study aims the determination of anthropogenic inputs with trace metals in aquatic ecosystems at the Plain of Annaba (NE Algeria), which is known as one of the largest industrial areas in Africa. Samples of surface waters and upper sediments were conducted in six stations: four in Meboudja wadi and two in Seybouse wadi. Contents of iron, copper, chromium, nickel, zinc, and manganese were measured by atomic absorption spectrophotometry, whereas Cd and Pb were determined using electrothermal atomic absorption spectrometry. Measurements of Hg were carried out using atomic fluorescence. Spatiotemporal variations of metal concentrations were tested using generalized linear models (GLM), whereas the influence of water pollution on sediment contamination was tested with generalized additive models (GAMs). Metal contents measured in surface water and sediments varied differently from upstream to downstream of the study wadis and between seasons. The results showed that the surface water was polluted with high levels of iron, nickel, chromium, lead, and cadmium. Values of the contamination index revealed that the surface sediments were contaminated by iron, chromium, lead, and cadmium. The GAMs indicated that water-phase metal concentrations had no significant effects on trace metal concentrations in the sediment. This suggests that seasonal metal concentrations in water phase, which are measured during the study period (2012) and are time-dependent, contribute increasingly and gradually over time—not immediately—to the accumulation of metals into the sediments. Therefore, the long-term accumulation of metals in the sediments resulted from the continuous discharges of metals in the water phase. The anthropogenic impacts are marked by high contaminations of Meboudja wadi particularly in downstream areas of the steel factory and the nearby industrial areas. The direct industrial discharges into the water and atmosphere (iron, lead, cadmium) as well as urban disposals and agricultural activities are at the origin of these contaminations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Heavy metals are distributed in all abiotic compartments (water, sediment) and biotic (flora and fauna) and are found in soluble, colloidal, and particulate components; mainly in the form of metal cations (Du Laing et al. 2009). Sediments accumulate high concentrations in estuarine ecosystems and wetlands due to their ecological function of pollutant sink; thus, the sediment occupies a key place in studies of the hydrosphere (Sprovieri et al. 2007). Industrialization, urban development, and intensification of agricultural activities are the main causes behind the increase in pressure threatening these environments (Gao and Chen 2012; Varol and Şen 2012). Industries, especially metallurgical mass producing and large metal consuming, salt out, deliberately, in the environment of large amounts of toxic metals such as Pb, Cd, Cu, Zn, Ni, Fe, and Cr. The entry of trace metal elements into aquatic habitats can also be the result of either direct discharges into marine and freshwater ecosystems due to some point source discharges such as factories or other human activities or indirect path such as the case wet or dry landfills (Praveena et al. 2010).
The presence of metals in waters and sediments can induce toxic effects impacting various organs in aquatic organisms resulting in (i) the appearance of abnormal forms such as necrosis, ulceration, and atrophy.; (ii) alteration of cell membranes with signs of oxidative stress; and (iii) perturbations of metabolism, photosynthesis, and DNA repair (Förstner and Wittmann 1981). During the accumulation of trace metals (TM) in human, several metabolic, endocrine, and urinary dysfunctions are manifested. Among these disorders, lead poisoning is one of the most common (Jaishankar et al. 2014). Heavy metals differently affect living beings. Thus, in a polluted habitat, plants show reduced growth, altered metabolism (inhibition of photosynthesis), lower biomass production, and metal accumulation (Nagajyoti et al. 2010). In aquatic invertebrates, the toxic effects of TM are manifested by delayed growth and reproductive disturbance resulting from tissue damage and metabolic alterations (Mance 1987; Rainbow 2002). Fish are affected by behavioral changes and disturbances in spermatogenesis with carcinogenic or mutagenic effects (Mance 1987).
The areas of El-Hadjar and Annaba are among the largest industrial zones in Africa (CNERU 1980). Discharges of wastewaters contaminate the wadis and the littoral environment, and the pollutants can lead to the eutrophication of water resources (Argillier et al. 2013). At very low levels, heavy metals are essential to the development of the life, but at high concentrations, they are toxic (Varol and Şen 2012; Diop et al. 2015) because they cannot be eliminated through biological processes and accumulate in various components of the ecosystem (water, sediment, flora, and fauna). Because of the accumulative effects of TM in plants and in the food chain, they generate serious ecological risks even for human health and well-being (Romić and Romić 2003; Grisey et al. 2010, 2012; Ahmed et al. 2012; Lenoble et al. 2013; Diop 2014).
Anthropogenic activities generating TM emissions are various and mainly related to urbanization, agricultural intensification, and industrial activities, which lead to increased levels of organic pollutants and heavy metals in the soil, air, and water resources (Gueddah 2003; Romić and Romić 2003; Ahmed et al. 2012; Grisey et al. 2012). Moreover, urban wastewaters drain out huge quantities of TM and pollutant materials (Rodier et al. 1996). The hydrological network and the sediments reflect the various physicochemical processes taking place at the scale of the watershed as well as in aquatic environments. Therefore, the sediments represent excellent indicators of water quality due to their capacity to accumulate heavy metals (Calmano et al. 1994).
In addition, industrial activities cause degradations of increasing importance to the quality of ecosystems. This degradation is mainly caused by massive and uncontrolled pollutions, related to natural organic matter and/or industrial synthetic products (Djabri et al. 2009; Ahmed et al. 2012). When industrial pollution is associated with contaminations from agricultural and urban activities, all combined, they affect all compartments of hydrosystems (water, sediments, flora, and fauna) (Débièche 2002; Gueddah 2003).
The purpose of this investigation is to evaluate the levels of contamination by iron, copper, zinc, manganese, chromium, nickel, lead, cadmium, and mercury in the surface waters and surface sediments of Meboudja and Seybouse wadis, which drain off waters into the Mediterranean Sea through the industrial and urban areas of El-Hadjar–Annaba (Algeria). Since we are interested here in assessing trace metal pollution at stations located along the water stream of wadis, TM concentrations are expected higher near potentially polluting sources (plants or towns experiencing intensive industrial and/or urbanization activities). Regarding the seasonal trends of water/sediment pollutions, TM concentrations are expected to decrease in autumn and winter, but to increase in spring and summer, because of the dilution of TM with the upsurge of water flow following heavy rainfall occurring in winter and autumn. In addition, TM concentrations are expected to increase in spring and summer for the reason that TM are released in the water after the degradation of organic matters of the colloidal and dissolved phases when water temperatures, salinity, and pH increase (Du Laing et al. 2009; Lesven et al. 2009; Diop 2014). In addition, the effect of short-term water pollution on the contamination of the sediment is tested in this survey. Accordingly, this study assesses the state and quality of the watershed of the plain of Annaba (Seybouse watershed), in which natural habitats are experiencing different forms of degradation and pollution resulted from the expansion and intensification of urbanization, industrialization, agriculture, and tourism (Louhi et al. 1998; Belabed et al. 2013a, b).
Material and methods
Study area
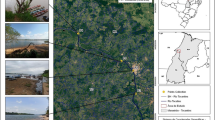
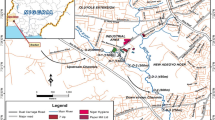
The industrial area of El-Hadjar is located at 10 km south of the city center of Annaba (North-east Algeria). It is bypassed in the East by Seybouse wadi, in the North-West by Meboudja wadi, and in the South by Rassoul wadi. This industrial zone is located in the low plain of Seybouse which is limited by the Mediterranean Sea in the North; in the West, the massifs of Belelieta (287 m), Bouhamra (152 m), and Edough (1008 m); in the South, the oriental Numidia massif (1411 m); and in the East, the aquifer system of Annaba-Bouteldja. The climate is Mediterranean humid with more than 600 mm of precipitation per year and an average temperature of 17.3 °C (Table S1). With more than 1.5 M inhabitants, agricultural and industrial activities mostly characterized by the area of Annaba. The study area is located in the final parts of Meboudja and Seybouse wadis between the city of El-Hadjar and the Mediterranean Sea (Fig. 1).
Meboudja wadi, with 14-km length, represents a discharge system of the Lake Fetzara. It crosses the industrial areas of Sidi Amar, El-Hadjar, and El-Bouni and also receives two small wadis: Taiba and Rassoul. With 240-km length, the watershed of Seybouse wadi stretches over 6471 km2. The industrial area of El-Hadjar–Annaba comprises five groups of industrial activities of which three are located in the area drained by Meboudja wadi and two in the area of Seybouse wadi. The watershed of Meboudja wadi includes El-Hadjar complex, a mega-factory of iron and steel, which produces nearly 2-M tons of steel per year. It also includes the industrial area of Chaiba comprising various factories (mechanics, cement, glass, paper, wood, recycling of ferrous products) and the industrial area of Pont Bouchet integrating various factories (pharmaceutical, plastic, mechanical repairs, recycling of car batteries). Moreover, Meboudja wadi receives domestic wastewaters and disposals from the towns of El-Hadjar, Sidi Amar, and Chaiba. In the drainage area of Seybouse wadi, there are two industrial areas. The first one in the South consists of two tomato paste pants and the second one in the North includes two plants of milk products and metallic equipment. In this part of Seybouse wadi, flow also domestic wastewaters and disposals from El Bouni town (Fig. 1).
Sampling stations
Six stations were sampled through the study area (Fig. 1). Four stations are selected along Meboudja wadi according to their location towards industrial and urban areas: (i) station M1 (07° 39′ 20.5″ E; 36° 46′ 9.84″ N) is located upstream of the iron and steel El-Hadjar complex; (ii) station M2 (07° 44′ 0.03″ E; 36° 48′ 51.5″ N) downstream the later complex; (iii) station M3 (07° 53′ 6.02″ E; 36° 52′ 7.27″ N) at the exit of the industrial area of Chaïba; and (iv) station M4 (07° 45′ 0.71″ E; 36° 54′ 41.4″ N) is situated near the industrial area of Pont Bouchet, whereas two stations belong to Seybouse wadi: (v) station S5 (07° 47′ 0.01″ E; 36° 54′ 17.7″ N) is located upstream the adjunction point with Meboudja wadi and (vi) station S6 (07° 46′ 0.66″ E; 36° 51′ 6.47″ N) downstream at approximately 1 km from its embouchure in the Mediterranean Sea.
Sampling and analysis of surface waters
From autumn 2011 to summer 2012 and for each season, surface waters and upper sediments of each station were seasonally sampled using standard sampling procedures. Samples were collected at the mid of October, February, April, and July for the seasons autumn (2011), winter spring, and summer (2012), respectively. Water samples were conducted at a depth of 50 cm in deep water current or at semi-depth in shallow waters (Rodier et al. 1996). Samples were collected in decontaminated polyethylene bottles. When measurements were not carried out immediately, samples were preserved at 4 °C.
Water samples were acidified using HNO3 (D = 1.4 g/cm3) to reach a pH lower than 2. Using a vacuum pump, 0.5% (v/v) was filtered with a SARTORIUS membrane of 0.45 μm porosity and then preserved at 4 °C prior to TM analysis. Contaminations by Fe, Cu, Cr, Ni, Zn, and Mn were measured using atomic absorption spectrophotometry, whereas Cd and Pb with electrothermal atomic absorption spectrometry (PerkinElmer A Analyst 100). Measurements of Hg were carried out following “International Atomic Energy Agency (IAEA) and United Nations Environment Program (UNEP)” (IAEA–UNEP) method (IAEA 1997, 2001) using cold vapor atomic fluorescence spectrometry in presence of stannous chloride. Results were compared to the reference values of the European Community (EC) (Débièche 2002).
Collection and analysis of upper sediments
Samples (∼ 500 g) of fine-grained materials were scraped from the surface sediments with a plastic shovel. Sediment samples were collected concurrently with water samples at a frequency of one sample per season and per station. The topmost centimeters of sediment that we sampled represent the most recent deposit that provides accurate assessment on the spatial and temporal changes in TM contamination. Samples were collected in polyethylene containers and transported in a cooler box to the lab where they were stocked at 4 °C until analysis. After drying, crushing, and sieving, sediment samples were mineralized through a wet process by acid attack in order to eliminate organic materials (AFNOR 1986).
A sample of 0.5 g of sediment, with fractions less than 63 μm, was added to a mixture of pure acids HNO3/HF/HCl (1 v/1 v/2 v). The mixture was heated at 150 °C for 2 h and then evaporated to almost dryness. Mineralized residues were submitted to successive rinsing in bi-distilled water, filtered without ash using a Whatman filter (0.45 mm), and then supplemented to a final volume of 100 mL. Contents of Pb, Cd, Cr, and Cu were measured using atomic absorption spectrophotometry with graphite furnace, whereas contents of Zn, Ni, and Fe were determined with flame atomic absorption spectrophotometry. Validity of the analytical methods was checked using a reference sediment sample from a Canadian lake (Lake Sediment No. LKSD-1) by (i) CANMET that certified TM contents that were given after extraction and (ii) external control using several inter calibrations (IAEA 1997, 2001). The minimum detection limits for heavy metals in the sediment were 0.06, 0.02, 0.01, 0.04, 0.03, 0.05, 0.04, 0.02, and 0.06 mg/kg for Pb, Cd, Hg, Fe, Cu, Zn, Mn, Cr, and Ni, respectively.
Evaluation of index of contamination (IC)
Reference (or guide) values used in this study for surface sediments are those of the “Agence du Bassin Rhône Méditerranée Corse” (ABRMC 1991). The index of contamination (IC) represents the ratio between the reference value of a given heavy metal and its content in the studied sediment. If IC is lower than 3, the sediment is considered of class 1 (not contaminated). When the IC range is between 3 and 10, the sediment is of class 2 (polluted). If IC is higher than 10, the sediment is of class 3 (high contamination risks). Reference values of the IC are indicated (ABRMC 1991).
Effects of weather conditions on water and sediment contaminations
In order to understand the effect of weather parameters on seasonal variations of TM in both waters and the sediment, we obtained data from the TuTiempo.net database (http://en.tutiempo.net/climate/ws-603600.html). Three climatic parameters, namely average temperature, precipitation, and wind speed (measured at 10 m above ground level), were considered out of the rest of climate variables because these ones are the main factors that significantly influence changes of water flow/evaporation and thus determine metal mobilization in the water and sediment. Daily meteorological data of 3 months preceding water/sediment sampling were used to compute seasonal values of average temperature (°C), precipitation (mm), and wind speed (km/h). Daily temperatures and wind speeds were averaged, whereas precipitations were cumulated to calculate the seasonal values that were linked to seasonal TM concentrations of the water phase and the sediment.
Statistical analyses
Spatial and seasonal values of trace metals measured both in surface waters and upper sediments were visualized as balloon plots using the package {ggplt2} in R (Chang 2013). The variation of these values among study stations and seasons was tested using generalized linear models (GLM) with an identity link function and assuming a Gaussian error distribution (Zuur et al. 2009). Being located upstream of pollution sources (whether of industrial and/or urban origin), compared to other stations, the station “M1” as well as the season of study start “autumn” was included in the intercept of each GLM. These two terms were precisely selected as the initial conditions in order to estimate the variation of metal concentrations along the rest of stations and seasons. Finally, the effects of the factors “stations” and “seasons” on the variation of TM in water and IC values were investigated using type III F tests. The Tukey post hoc test was carried out to classify categories of each factor with a significant effect (P < 0.05). The effect of water contamination of each metal on its contamination in the sediment was tested using a non-parametric model. In generalized additive models (GAMs), metal contents of the sediment were fitted as a smooth function of water contents (Crawley 2013) using a Gaussian distribution with “identity” as link function. GAMs were carried out using the package {mgcv}. The effects of seasonal climate parameters (temperature, precipitation, and wind speed) of seasonal TM concentrations were tested using Pearson correlations. All statistical tests were performed using the software R (R Core Team 2016).
Results
Heavy metals in surface waters
The analysis of surface waters from Meboudja and Seybouse wadis showed that TM concentrations fluctuated following seasons and study stations (Fig. 2).
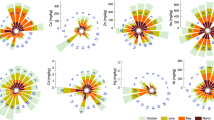
Spatial and seasonal variations of trace metal concentrations (in ppm) in surface waters of Meboudja and Seybouse wadis at the mouth of the Seybouse Watershed in NE Algeria. Italic letters between square brackets indicate results of the Tukey HSD tests. The same letters are not significantly different at P > 0.05. Uppercase and small letters indicate differences between study sites and seasons, respectively. TM reference values (in ppm) were determined following ABRMC (1991): Fe = 0.2, Cu = 1, Zn = 5, Mn = Ni = Cr = Pb = 0.05, Cd = 0.05, and Hg = 0.001
Iron
In Meboudja wadi, the maximum and minimal values of iron were recorded respectively in summer and spring. Values of iron in autumn, winter, and springs do not exceed 2 ppm. During summer, iron values increased to factor 2 in stations M1 and M3 and to factor 5 in M4. In stations of Seybouse wadi, contents of iron were not different. However, in summer, an increase of more than 50% occurred in S6 compared to S5 (Fig. 2). The contents of iron in surface waters of Seybouse were, however, lower than those observed in Meboudja, except for the spring period where iron contents were close to the European Community (EC) reference value (0.2 ppm). It is obvious that the surface waters of the two wadis were highly contaminated during all the year. The GLM revealed a significant increase of iron contents during summer (t = 3.54, P = 0.003) with significant seasonal variations (Table 1), whereas differences between stations were not significant (F test: F = 1.18, P = 0.366).
Copper
In all study stations, contents of copper in surface waters reached maximum values in summer period (GLM: t = 2.63, P = 0.019). However, values of copper were significantly higher in M1 and M2 compared to the rest of stations (F test: F = 5.14, P = 0.006). The Tukey test indicated that these values were low in M1, increased in M2 (from factor 4 to 10 according to seasons), and then dropped gradually in M3 and M4. In Seybouse wadi, copper values in S6 were slightly higher than in S5 particularly in autumn (Fig. 2). However, contents of copper of the two wadis do not exceed 0.2 ppm indicating lower values than the EC reference value (1 ppm). The GLM revealed a significant decrease of copper contents during spring compared to autumn (Table 1). Copper values significantly varied between seasons (F = 17.07, P < 0.001).
Zinc
Zinc contents in surface waters of both wadis have not exceeded 1 ppm and were lower than the EC reference value (5 ppm). Values recorded during spring were significantly the lowest (GLM: t = −2.24, P = 0.041), but no significant difference was observed between seasons (F test: F = 1.69, P = 0.212). Regarding stations, zinc concentrations were the highest in M4 (t = 3.06, P = 0.008), where they increased during autumn and summer. The Tukey test indicated that values in M2, M3, and S5 were the lowest with a significant variation between stations (F test: F = 3.76, P = 0.021) (Fig. 2, Table 1).
Manganese
Manganese contents in surface waters of the two wadis were higher than the EC reference value (0.05 ppm), except during spring in Seybouse wadi. Mn values increased along stations of Meboudja wadi and then decreased when waters of the later merged with Seybouse wadi at S5. However, the GLM revealed no significant variation between stations (F test: F = 1.78, P = 0.177), whereas the season factor had a significant effect (F test: F = 3.97, P = 0.029). The Tukey test showed that the lowest values of manganese were recorded during spring, while summer enclosed the highest values (Fig. 2, Table 1).
Nickel
Nickel contents in surface waters of Meboudja and Seybouse wadis clearly exceed the EC reference value (0.05 ppm). GLM revealed that nickel concentrations decreased downstream in both wadis; however, the variation of nickel between study stations was not significant (F test: F = 1.63, P = 0.212). Regarding seasonal changes in nickel values, minimal and maximum contents of nickel were detected during winter and summer, respectively (Fig. 2). The GLM indicated that nickel values increased significantly in summer (t = 2.95, P = 0.01), whereas the increase observed in spring was not significant (Table 1). According to the Tukey test, nickel values of these two seasons were significantly higher than autumn and winter. Indeed, F test showed that nickel contents differed significantly from one season to another (F = 5.18, P = 0.012).
Chromium
In Meboudja surface waters, the highest values of chromium were recorded in winter and summer in M4 and during autumn in M3 (Fig. 2). The GLM indicated a significant increase in M4 (P = 0.038) and with a significant decrease in spring (P = 0.011) (Table 1). Indeed, chromium contents during spring were lower than the EC reference value (0.05 ppm). However, during autumn, chromium concentrations exceed the EC standard in all sampling stations. In winter and summer, chromium contents rise in S6 exceeding the EC standard, particularly during the cold season in S5 and S6. F test, revealed no significant variation between stations (P = 0.143), whereas the effect of season was significant (F = 4.01, P = 0.028). For the latter factor, the Tukey test indicated that chromium values of the spring were the lowest (group a), while autumnal values were the highest (group b).
Lead
Lead concentrations in surface waters of Meboudja vary between 0.09 ppm (M2 in autumn) and 1.5 ppm (M3 in winter). In M3 and M4, values were higher than those recorded in M1 and M2 (Fig. 2, Table 1). Overall, lead contents in all study stations were higher than the EC reference value (0.05 ppm). Peaks were recorded during autumn, winter in M3 and M4, and during summer in M1 and M3. In S5 and S6, lead concentrations were close to the European standard except in spring where values are 20 to 30 times higher than those recorded during the rest of study seasons. The GLM showed that there is no significant difference in lead contents between study sites (F test: F = 1.08, P = 0.412) and seasons (F = 0.20, P = 0.896).
Cadmium
Cadmium values in surface waters of Meboudja wadi exceeded EC standard (0.005 ppm) except during spring where Cd contents were lower than this reference value. The lowest contents are recorded in M1, but progressive increase of cadmium occurs in winter and summer in M2 and M3 (Fig. 2). The GLM, revealed that Cd increased significantly downstream in M2 (P = 0.034) and M3 (P = 0.010) compared to M1 (Table 1), with a significant change between stations (F test: F = 2.98, P = 0.046). In Seybouse wadi, cadmium contents were detected only in S5 during autumn and in S6 during summer, where both values (0.005 ppm) exceeded the EC standard. GLM revealed that seasonal fluctuations of cadmium were significant (F test: F = 6.82, P = 0.004), where Cd concentration decreased significantly during the spring season (t = −2.43, P = 0.028).
Mercury
In all stations of Meboudja wadi, mercury contents increased downstream to peak in waters of M4 (GLM: t = 4.23, P = 0.001). In the later, peaks occur during the four seasons with a maximum value of 0.26 ppm in autumn (Fig. 2, Table 1). The Tukey test indicated that mercury concentrations in the latter station were significantly the highest, followed by concentrations of M3 (group b), whereas means of Hg in the rest of study stations was statistically different (F test: F = 6.63, P = 0.002). In spring, Hg contents decreased significantly (GLM: t = −2.89, P = 0.011) and were lower than the EC reference value. In Seybouse wadi, mercury contents in S5 and S6 were quite similar except in summer where they increased with more than 50% in S6. Except for spring, mercury contents in surface waters of Seybouse wadi exceeded the EC reference value. Variations of Hg concentrations were significant between seasons (F = 5.61, P = 0.009), where hot seasons (summer and autumn) were significantly the highest (group b according to the Tukey test).
Contamination by trace metals in the upper sediment
Iron
In upper sediment of Meboudja wadi, iron concentrations varied from 29,080 to 54,760 mg/kg dry weight (dw) with the highest values recorded in M2 and M3. In Seybouse wadi, values recorded in the sediment S6 are two times higher than those in S5 station during summer and spring, but in autumn and winter, they are close to each other. Iron IC of the surface sediments (Table 2) varied from 14.54 to 27.60 in Meboudja wadi and from 9.93 to 16.46 in Seybouse wadi. Since all IC values were higher than 10, it is obvious that Meboudja and Seybouse wadis involve high risks. The GLM revealed high significant differences between study stations (F = 110.07, P < 0.001) and seasons (F = 7.92, P = 0.002), where IC values significantly increased in Meboudja stations (M2 and M3) and decreased in S5 and S6 (P < 0.001).
Copper
In Meboudja wadi, copper contents in the sediment have similar seasonal trend from one station to another. The GLM revealed no difference between seasons (F test: F = 2.53, P = 0.096). Levels of copper were weak in M1, increase gradually in M2, reach a peak in M3, and then decrease in M4. According to the GLM, all variations of Cu concentrations downstream along stations were significant (Table 2), with a significant change of the factor station (F test: F = 37.53, P < 0.001). The highest concentrations were recorded in winter and summer, with a significant increase during winter (t = 2.64, P = 0.018). In Seybouse wadi, copper contents were lower than Meboudja stations. Copper IC of surface sediments of Meboudja and Seybouse wadis was relatively low (Fig. 3). Therefore, sediments were not contaminated by copper, except for M3 station during winter where copper IC reached 3.22.
Spatial and seasonal variations of contamination index for trace metals measured in upper sediments of Meboudja and Seybouse wadis at the mouth of the Seybouse Watershed in NE Algeria. Italic letters between square brackets indicate results of the Tukey HSD tests. Values with the same letter are not significantly different at P > 0.05. Uppercase letters indicate differences between study sites, whereas small letters between seasons. TM reference values (in mg/kg dw) were determined following Boust (1981): Fe = 2000, Cu = 26, Zn = 88, Mn = 400, Ni = 45, Cr = 45, Pb = 22, Cd = 0.6, and Hg = 0.2
Zinc
In the surface sediments of study wadis, zinc IC varied between 0.45 and 2.89. IC levels displayed particular spatial and seasonal fluctuations. A progressive increase in zinc level was observed in M2 and M3 followed by a slight diminution in M4. According to GLM, all increases that occurred in these stations were significant (Fig. 3, Table 2). The highest values were recorded in autumn and winter, with maximum values in M3. The Tukey test showed that these seasons were significantly higher than spring and summer. In Seybouse wadi, S6 station presents double zinc values of those found in S5 during autumn and winter. Zinc contents measured in spring and summer decreased significantly (P < 0.001); they were less than 100 mg/kg dw. The GLM revealed very high significant differences in Zn between study stations (F test: F = 30, P < 0.001) and seasons (F test: F = 22.9, P < 0.001).
Manganese
In each season, manganese concentrations showed similar trends for study stations. Manganese values were low in M1, then significantly increased in M2, with a slight reduction fall in M3 and M4 (Fig. 3). The GLM revealed that increases of Mn in M2, M3, and M4 compared to initial conditions of M1 were significant (Table 2). Mn variations were significant between stations (F test: F = 13.53, P < 0.001), but no differences were found between seasons (F = 0.67, P < 0.586). In surface sediments of Seybouse wadi, manganese content in S6 is close to that in M4 but represents less of the one third of the content in M2 in spring. Manganese IC of the surface sediments of the two wadis varies from 0.31 to 1.72. As all IC values <3, the upper sediments of the two wadis were considered as not contaminated with manganese (class 1).
Nickel
In Meboudja wadi, nickel levels in the surface sediment vary from 40 to 96 mg/kg dw. They increased significantly in M2 (GLM: t = 4.13, P = 0.001), reach a peak in M3 (GLM: t = 5.35, P < 0.001), and then slightly fall in M4. In Seybouse wadi, levels of nickel in the surface sediment were lower than the guide value. Values of nickel IC varied significantly between study stations (F test: F = 10.53, P < 0.001). The Tukey test indicated that M1 and S5 held the lowest Ni IC values, whereas M3 had the highest IC values (Fig. 3). During winter and summer, nickel significantly increases in sediments (Table 2), with a significant effect of seasons (F = 8.49, P = 0.002). Following the Tukey test, IC values of both seasons were significantly higher than spring. Values recorded in the latter do not exceed the standard guide value. Moreover, as nickel IC of the surface sediments of both wadis varied between 0.85 and 2.01, they were not contaminated by this metal (Class 1).
Chromium
In the sediments of Meboudja wadi, chromium contents present similar seasonal trend between the stations (Fig. 3). Chromium concentrations increased significantly in M2 (GLM: t = 3.70, P = 0.002), reach a peak in M3 (GLM: t = 8.92, P < 0.001), and then decreased in M4 but with values slightly greater than those of M1 (Table 2). Chromium IC varied significantly between study stations (F test: F = 25.96, P < 0.001), where IC values of M1, M4, S5, and S6 were significantly the lowest following the Tukey test. The GLM revealed that Cr values in the sediments decreased significantly during spring (t = −3.25, P = 0.005) and increased in winter and summer. Accordingly, the effect of season on the variations of Cr IC values was significant (F test: F = 8.49, P = 0.002). Like the rest of trace metals, chromium contents in Seybouse wadi were low compared to Meboudja wadi. Chromium IC of the surface sediments of both wadis varied from 1.30 to 4.48. IC was higher than 3 in M3 during all seasons except spring, characterizing the station as contaminated by chromium.
Lead
Lead concentrations displayed similar seasonal fluctuations from study stations. In M1, they were low, then increased to a peak in M3, and then decrease in M4 (Fig. 3). All increases observed in Meboudja stations were significant (GLM: P < 0.05) (Table 2). In stations S5, lead contents increased compared to M1, but in S6, the increase was significant (t = 2.70, P = 0.017). Lead concentrations varied significantly between stations (F test: F = 105, P < 0.001). According to the Tukey test, lead contents in M3 and M4 were the highest, where they exceeded the EC reference value. Lead IC of the surface sediments in Meboudja and Seybouse wadis showed various classes: class 1 for M1 and S5; class 2 for M4 and S6; and class 3 for M2 and M3 (zones at risk). Changes in lead IC were not significant between seasons (F test: F = 0.23, P = 0.871).
Cadmium
The lowest cadmium values were recorded in M1 and S5. A progressive increase is recorded in M2 and M3 followed by a slight fall in M4 (Fig. 3). The GLM showed a positive link of Cd contents in M2, M3, M4, and S6 (Table 2), with a significant variation between stations (F test: F = 24.12, P = 0.001). The maximum values were recorded in station M3 followed by M4. It is noteworthy that cadmium contents in M2 exceeded the standard reference value (0.6 mg/kg dw), whereas they are below the reference value in Seybouse wadi except in S6 during winter. Overall, cadmium IC varied from 0.32 to 6.80. Stations M1, S5, and S6 thus are not contaminated by cadmium. M2 is class 2 in spring and summer whereas M3 and M4 are of class 2 during the whole year and thus are contaminated by cadmium. Variations of cadmium between seasons were not found significant (F = 1.23, P = 0.333).
Mercury
Mercury concentrations in the surface sediments showed a range difference of 0.34 mg/kg dw between min and max values. According to the GLM, Hg increased significantly downstream along all stations of Meboudja wadi and peaked in M4 (Fig. 3), whereas Hg contents were not linked to Seybouse stations (Table 2). Mercury contents varied significantly between stations (F test: F = 18.60, P < 0.001), but this variation followed the same trend between seasons (F = 3.26, P = 0.051) though a significant increase was detected during summer (t = 2.87, P = 0.012). The Tukey test indicated that sediments of M4 then M3 possessed higher lead contents than M1, S5, and S6 that were the lowest. Overall, the surface sediments of study stations (excluding M4 in summer, IC >3) were not contaminated by mercury.
Effects of water pollution on sediment contamination
Overall, seasonal variations of trace metal concentrations revealed that the contamination of surface waters in the study wadis had no significant effect on the contamination of upper sediments. All GAMs showed no significant effect (P > 0.05) (Fig. 4). This may be explained by the influence of sediment sampling procedures of this study in which samples were scraped and collected from the sediment surface that is expected to be often disturbed or re-suspended, causing the metal concentrations in these samples potentially less relevant to the long-term pollution and accumulation. Also, the complexation and different forms of metals as well as the presences of salts in the water could also be influential. All that said to indicate that the metal concentrations in the water led to the metal pollutions in the sediments, although GAMs suggested that contamination of the sediment is caused by a long-term and cumulative process of water pollution. Despite these non-significant effects, it is noteworthy that lead concentrations in waters influenced sediment pollution more rapidly than other trace metals considering that the GAM revealed 40.6% of deviance explained.
Relationships between concentrations of trace metals in surface waters and upper sediments in wadis of the Seybouse watershed in North Africa. The solid lines represent loess (locally weighted polynomial) curves smoothed with a GAM fit (generalized additive model) and 95% confidence region in light gray. F-statistics (F) and P value (P) are results of GAMs
Effects of seasonal climatic conditions on TM concentrations
The relationships between climatic parameters and TM concentrations differed between surface water and upper sediment. For surface waters, the average temperature was negatively correlated with TM concentrations with significant correlations (P < 0.005) found with Fe, Cu, Mn, Cr, Cd, and Hg. The precipitation and wind speed positively influenced TM concentration, with significant correlations between rainfall and Cd, Hg, Fe, Cu, Ni, and Cr and between wind speed and Fe, Cu, Ni, and Cd. Regarding upper sediments, TM concentrations were not deemed influenced by climatic parameters, except for zinc in relation to rainfall (r = 0.446, P = 0.029) (Table 3).
Discussion
The current study shows that surface waters of Meboudja and Seybouse wadis are highly contaminated by iron during the whole year because the recorded values exceeded the European reference value (0.2 ppm). These high concentrations originated from iron dust emission from El-Hadjar complex. Similarly, Louhi et al. (1998) confirm the presence of high iron levels in waters of Meboudja near the iron and steel complex. Along Meboudja wadi, the lowest iron concentrations in surface sediments are located in M1 upstream of the industrialized zones, whereas the highest concentrations occurred in M2, M3, and M4 just downstream of El-Hadjar complex. Furthermore, discharges of industrial effluents and wastewaters without any treatment by this metallurgical industry, major producers of heavy metals, represent the main source of iron in water and sediments (Djabri et al. 2009). The high levels of iron may be also explained by the presence of iron dust in the air. Heavy rains, flows of water, and turbulent currents create high levels of oxygenation that increase the precipitation of Fe2+ into iron oxides facilitating the formation and aggregation of mineral particles, thus its sedimentation (Salvarredy-Arangren 2008; Djabri et al. 2009; Belabed et al. 2011, 2013a; Diop 2014). Taking all the previously mentioned together, the iron and steel complex of El-Hadjar is clearly pinpointed as the point source of iron pollutions in the area. In Seybouse wadi, the most significant iron variations are in spring and in summer, but contents are lower than those in Meboudja wadi. In S6 station, nearby the embouchure with the Mediterranean Sea, iron concentration in the sediments is the highest due to the dynamic supplies through the hydrologic network. The IC by iron of the surface sediments of the two wadis shows that these sediments are of class 3, thus polluted by iron with higher values compared to other heavy metals. High concentrations of iron can be related to hydrodynamic and water physicochemical conditions, especially to the precipitation of iron oxides (Asfers et al. 2016).
Zinc and copper concentrations in surface waters and upper sediments of surveyed wadis are homogeneous and vary slightly. They do not exceed the reference values, but the highest values are recorded in Meboudja wadi during summer. The presence of these two metals in stations located downstream industrial areas confirms their anthropogenic origin. Similarly, Louhi et al. (1998) report very high levels of copper contents above the European reference value in various sampling stations with a maximum record nearby El-Hadjar iron and steel complex. Water contamination is likely related to the technological processes of this complex which originate heavy pollution by copper and zinc. This pollution has a heavy impact on groundwater quality of the region (Bougherira et al. 2016).
There are various data on zinc content from North African and European hydrosystems (Appendix Table 4): in Tunisia, Azzaoui (1993) indicates 100 mg/kg dw in lake Ghar El Melh and Ben M’Barek (1995) 180 mg/kg dw in Lake Ichkeul. In Morocco, Benbouih et al. (2005) record 210 mg/kg dw in Lake Fouarat whereas in Egypt, Saad and Fahmy (1985) indicate 106 mg/kg dw in Lake Nozha and 94 mg/kg dw in Lake Mariout. No data for Libya were reviewed. In the area of Constantine (NE Algeria), Afri-Mehennaoui et al. (2006) report that industrial and urban effluents substantially influence waters of Rhumel and Boumerzoug wadis, where copper concentrations ranged between 44 and 778 μg/L.
For copper, the annual average contents in Meboudja (21.61 mg/kg dw) and Seybouse (25.38 mg/kg dw) are very close to those of lake Bouseham in Morocco (26 mg/kg dw) (Benbouih et al. 2005). For Meboudja and Seybouse wadis, the IC by copper and zinc show that the values do not exceed 3, characterizing the sediments of the two wadis as class 1, thus not contaminated by these two heavy metals. Angelidis and Catsiki (2002) indicate that copper and zinc distribution depends on the nature of the sediment whose proportions of clay and organic matter play a determining role. They also report high values of copper and zinc in sandy-clay sediments located near disposals. Also, copper and zinc are associated to sandy facieses directly exposed to waste discharges (Lesven et al. 2009; Diop 2014). Several studies (Salvarredy-Arangren 2008; Djabri et al. 2009; Belabed et al. 2011, 2013a; Diop 2014) indicate that deterioration of sulfates (especially chalcopyrite and galenite) can increase sulfates and acidity justifying the presence of copper, zinc, and lead, which increase is mainly related to the mining activity inducing trace metal mobilization.
In waters of study wadis, the lowest manganese values are recorded in M1 and S5 with a maximum content in M2. Except for the spring period, concentrations are higher than European standard (0.05 ppm). Louhi et al. (1998) indicate that in stations of Meboudja wadi near El-Hadjar iron and steel complex, waters are of black color and manganese contents are far higher than the previous standard. In waters of Rhumel and Boumerzoug wadis, manganese values ranged between 10 and 471 μg/L (Afri-Mehennaoui et al. 2006). In the surface sediment of Meboudja and Seybouse wadis, manganese contents present variations according to both sampling station and season. The lowest values are in Seybouse wadi whose highest values correspond to the values in M1 upstream the industrial areas. In M2, manganese doubles or triples in values according to seasons with the maximum concentrations occurring in autumn and spring. Values of contamination index by manganese do not exceed 3 characterizing the sediments of the two wadis as non-contaminated. Djabri et al. (2009) confirm that iron and steel activity is the principal anthropogenic source of pollution by manganese as it is the case in Val de Milluni in Bolivia (Salvarredy-Arangren 2008).
Because of the contiguity to El-Hadjar complex, nickel and chromium in surface waters in M2 and M3 show higher values than the respective European reference value. Indeed, nickel concentrations increase in M2 and M3 at levels close to 100 mg/kg dw during summer and winter. Concentrations higher than the standards are also reported by Louhi et al. (1998) in surface waters of Meboudja wadi nearby El-Hadjar complex (Appendix Table 4). However, sediment contamination indexes by nickel do not exceed 3 characterizing the sediments of Meboudja and Seybouse as not contaminated. On the other hand, chromium shows high-level contents in stations located nearby the industrial area. The indexes of contamination of the sediments of the two wadis show pollution by chromium in M3 in autumn, winter, and summer.
Callender (2003) indicated that even if natural provision of nickel is related to soil erosion, its contents can also come via wastewaters from steel, foundry, and paint factories. Thus, the presence of nickel in study wadis can only be explained by the impact of industrial wastes. Furthermore, high levels of chromium can also come from the sedimentation of dead organisms as well as from the geological origin of rocks (Callender 2003; Belabed et al. 2011, 2013a). This investigation incriminates the local industrial activities as being the source of this pollution source.
Lead contents exceed the European reference value (0.05 ppm) in surface waters of all Meboudja sampling stations and in Seybouse wadi except for the spring period. This is quite similar to findings of Louhi et al. (1998) in waters of Meboudja where lead concentrations were lower than the accepted standards of industrial wastes. In surface waters of Rhumel and Boumerzoug wadis (Afri-Mehennaoui et al. 2006), lead concentrations vary from 0.6 to 1.1 μg/L (Appendix Table 4).
In the surface sediments of Seybouse wadi, lead contents represent about a third of those recorded in Meboudja where in M3 during all seasons. The seasonal variations of lead contents in the sediments are similar in the two wadis. The IC by lead characterizes the surface sediments of Meboudja and Seybouse wadis as being sediments of class 1 (M1 and S5), of class 2 (M4 and S6), and of class 3 (M2 and M3 being zones at risks). The results of this study are slightly higher than those recorded in previous studies (Appendix Table 4) such as the port of Skikda (NE Algeria) where lead contents vary from 118 to 145 mg/kg (Gueddah 2003).
Data of the World Health Organization (WHO 2001) indicate varying lead contents from 0.175 to 0.520 μg/g dw in sediments of the Moroccan coasts; from 23 to 3300 μg/g dw in Spain, from 18 to 1250 μg/g dw in Marseille (France), from 18 to 470 μg/g dw in Gulf of Trieste (Italy), and from 25 to 130 μg/g dw in Gulf of Thermaikos (Greece). Roussiez et al. (2005) pointed out that lead contents in the sediment of Lion Golf (France) reach 39.2 mg/kg dw. Our findings revealed high levels of lead in the sediments of the two wadis, which indicate the presence of emission sources of lead. The pollution with this heavy metal originates essentially from various industrial activities. Moreover, the use of lead as anti-explosive in fuels for cars leads to lead concentration increases in the air, which represents the atmospheric route of water and sediment contamination (Li et al. 2003; Belabed et al. 2011, 2013b; Varol and Şen 2012).
Cadmium contents in surface waters of Meboudja wadi exceed the European reference value (i.e., 5 ppm, Débièche 2002), but in spring, values are lower than this standard (Appendix Table 4). In surface waters of Rhumel and Boumerzoug wadis, cadmium concentrations are lower than 0.5 μg/L (Afri-Mehennaoui et al. 2006). Results of this investigation are close to those indicated in Tunisia by Azzaoui (1993) in lake Ghar El Melh (Cd = 1.5 mg/kg dw) and by Ben M’Barek (1995) in Lake Ichkeul (1 mg/kg dw). In Morocco in lake Fouarat, Benbouih et al. (2005) indicate cadmium concentration of 0.4 mg/kg dw. In Egypt, in Lake Mariout and lake Nozha, cadmium contents are, respectively, 0.2 and 0.15 mg/kg dw (Saad and Fahmy 1985). The presence of cadmium in the sediments can be linked to the leaching of agriculture lands enriched with phosphate fertilizers. Moreover, the iron and steel industry is also the main source of emission of cadmium, which becomes easily water-soluble by acid drainages (Callender 2003; Salvarredy-Arangren 2008; Djabri et al. 2009; Wahsha et al. 2012). It is significant to stress that cadmium is highly nephrotoxic even at low doses (Gunawardana et al. 2006). The principal sources of mercury in the environment are natural degassing of earth’s crust and the industrial and agricultural activities (Cossa and Ficht 1999).
The major anthropogenic sources of contamination by trace metals in continental waters are the mining activity, metallurgy, and the iron and steel industry, which are also the principal sources of heavy metals inducing atmospheric pollution (Callender 2003). Thus, as it has been confirmed in many previous studies in North Africa (e.g., Djabri et al. 2009; Belabed et al. 2011, 2013b) or in Europe (Velasco et al. 2005), this investigation confirms that the iron and steel complex of El-Hadjar is at the origin of the presence of Fe, Zn, Cu, Cd, Mn, and Ni in the surface waters and upper sediments of the regional hydrological network.
Conclusion
The results of this investigation show the impact of the industrial and urban complexes on the levels of contamination by TM of surface waters and sediments of Meboudja and Seybouse wadis (Seybouse watershed, Algeria). The use of multiple sampling stations along the stream of wadis is a relevant technique for assessing pollutions and therefore detects the location of industrial effluents and/or urban wastewater discharges responsible for the contamination of the waters and/or the sediments. Accordingly, because TM concentrations increase downstream the study wadis, the industrial discharges of some factories are pinpointed as the polluting sources in the plain of Annaba. Iron, manganese, nickel, chromium, lead, cadmium, and mercury contents are higher than the reference values, indicating polluted waters with specific metallic pollutants. The most polluted stations were those located downstream of Meboudja wadi (M2, M3, M4) nearby El-Hadjar complex and close to the industrial park of Chaiba and Pont Bouchet. The sediment is classified at high contamination risk with regard to iron and lead, but at medium risk for chromium and cadmium. These findings characterize of the regional hydrological network as being polluted by the discharge effluents from the urban and industrial areas, particularly from El-Hadjar iron and steel complex. Seasonal monitoring of TM contaminations in waters and sediments of lotic hydrosystems deepens our understanding about the processes, such as mobilization, complexation, re-suspension, and accumulation, that control metal concentrations and therefore may be exploited for the remediation of heavy metal pollutions.
References
ABRMC. (1991). Pollution du Rhône. Synthèse des connaissances, Ministère de l’Environnement Français. Lyon: Agence du Bassin du Rhône Méditerranée Corse ‘ABRMC’.
AFNOR (1986). Essai drs eaux. Dosage de dix éléments métalliques (Cr, Mn, Fe, Co, Ni, Cu, Zn, Ag, Cd, Pb) par spectrométrie d’absorption atomique dans la flamme NF-T. La Plaine St Denis: AFNOREditions, pp. 90–112.
Afri-Mehennaoui, F. Z., Sahli, L., & Mehennaoui, S. (2006). Assessments of sediment trace metal level and biological quality of Rhumel River by using multivariate analysis. Environmetrics, 15, 435–446.
Ahmed, G., Miah, M. A., Anawar, H. M., Chowdhury, D. A., & Ahmad, J. U. (2012). Influence of multi-industrial activities on trace metal contamination: an approach towards surface water body in the vicinity of Dhaka Export Processing Zone (DEPZ). Environmental Monitoring and Assessment, 184, 4181–4190.
Alkan, N., & Örnek, V. (2013). Investigation of metal accumulation in surface sediment in Sürmene Bay of the eastern Black Sea. Report Comm. Int. Mediterranean Sea.
Angelidis, M. O., & Catsiki, A. (2002). Metal bioavailability and bioaccumulation in the marine environment: methodological questions (pp. 33–41). Ancona: Workshop Monograph n°19 of the mediterranean science comission ‘CIESM’, metal and radionuclides bioaccumulation in marine organisms.
Argillier, C., Caussé, S., Gevrey, M., Pédron, S., De Bortoli, J., Brucet, S., et al. (2013). Development of a fish-based index to assess the eutrophication status of European lakes. Hydrobiologia, 704, 193–211.
Asfers, Y., Taouil, H., Ibn, A. S., & Chakit, M. (2016). Evaluation metallic pollution caused by iron, copper, lead and cadmium of Oum-Rabia river water. Journal of Bio Innovation, 5, 59–67.
Azzaoui, S. (1993). Géochimie de certains métaux lourds dans les sédiments de la lagune de Ghar El Melh (N-E Tunisie). Dissertation of DEUA, University of Tunis 2, Tunisia.
Belabed, B. E., Bendjema, A., Boudjelida, H., Djabri, & Bensouilah, M. (2011). Evaluation of the metal contaminations in the surface sediments of the Oubeira lagoon, National park of El Kala, Algeria. Archives of Applied Science Research, 3, 51–62.
Belabed, B. E., Frossard, V., Dhib, A., Turki, S., & Aleya, L. (2013a). What factors determine trace metal contamination in Lake Tonga (Algeria)? Environmental Monitoring and Assessment, 185, 9905–9915.
Belabed, B. E., Laffray, X., Dhib, A., Fertouna-Belakhal, M., Turki, S., & Aleya, L. (2013b). Factors contributing to heavy metal accumulation in sediments and in the intertidal mussel Perna perna in the Gulf of Annaba (Algeria). Marine Pollution Bulletin, 74, 477–489.
Ben M’barek, N. (1995). Impact des ouvrages sur l’équilibre de l’écosystème: Lac Ichkeul. Dissertation of DEA. University of Tunis 2.
Benbouih, H., Nassali, H., Leblans, M., & Shiri, A. (2005). Contamination en métaux traces des sédiments du lac Fouaret-Maroc. Afrique Science, 1, 109–125.
Bougherira, N., Hani, A., Nechem, D., Chaffai, H. (2016). Modelling of ground water contamination, plants case of El Hadjar, N East of Algeria. In S. Sauvage, J. M. Sánchez-Pérez, A. E Rizzoli. (Eds.), Proceedings of the 8th International congress on environmental modelling and software (pp. 1287–1294). July 10–14, Toulouse: France.
Boust, D. (1981). Métaux traces dans l'estuaire de la Seine et ses abords. Ph.D. thesis, University of Caen.
Callender, E. (2003). Heavy metals in the environment. Historical trends. In B. S. Lallor (Ed.), Environmental geochemistry. Treatise on geochemistry. Oxford: Elsevier-Pergamon 67 pp.
Calmano, W., Forstner, U., & Hong, J. (1994). Mobilization and scavenging of heavy metals following resuspension of anoxic sediments from the Elbe River, ACS Symp. Ser (Vol. 550, pp. 298–321). Washington D.C.: American Chemical Society.
Casado-Martinez, M. C., Buceta, J. L., Belzence, M. J., & Del Valls, T. A. (2006). Using sediment quality guidelines for dredged material management in commercial ports from Spain. Environmental International, 32, 388–396.
Chang, W. (2013). R graphics cookbook. Sebastopol: Media O’Reilly Edition.
CNERU (1980). Etudes et réalisations en urbanisme. Recueil sur la zone industrielle d’El-Hadjar: 42 p.
Core Team, R. (2016). R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing Available at www.r-project.org.
Cossa, D., & Ficht, A. (1999). La dynamique du mercure. Programme Scientifique Seine-Aval, Editions Ifremer, Plouzané, France.
Crawley, M. J. (2013). The R book (Second ed.pp. 666–680). Chichester: John Wiley & Sons Ltd.
Débièche, H. (2002). Evolution de la qualité des eaux (salinité, azote et métaux lourds) sous l’effet de la pollution saline, agricole et industrielle. Doctoral thesis, University of Franche-Comté, France.
Diop, C. (2014). Etude de la contamination, de la spéciation et de la biodisponibilité des éléments traces métalliques dans les eaux et sédiments côtiers et estuariens au Sénégal: Evaluation de la toxicité potentielle. Doctoral thesis, University of Lille 1, France.
Diop, C., Dewaelé, D., Cazier, F., Diouf, A., & Ouddane, B. (2015). Assessment of trace metals contamination level, bioavailability and toxicity in sediments from Dakar coast and Saint Louis estuary in Senegal, West Africa. Chemosphere, 138, 980–987.
Djabri, L., Ghorraib, L., Hani, A., Lamouroux, G., & Sharour, I. (2009). Contamination des eaux souterraines par les métaux lourds d’une mine de fer abandonnée: Cas de la région de Bekkaria (Tébessa). Revue Francophone d’Ecologie Industrielle, 50, 31–35.
Du Laing, G., Rinklebe, J., Vandecasteele, B., Meers, E., & Tack, F. M. G. (2009). Trace metal behaviour in estuarine and riverine floodplain soils and sediments: a review. Science of the Total Environment, 407, 3972–3985.
Förstner, U., & Wittmann, G. T. (1981). Metal pollution in the aquatic environment. Berlin: Springer.
Gao, X., & Chen, C. T. A. (2012). Heavy metal pollution status in surface sediments of the coastal Bohai Bay. Water Research, 46, 1901–1911.
Gharsallah, Z. (2005). Evaluation de la pollution du littoral d’Annaba: qualité microbiologique de l’eau et teneur en métaux lourds du sédiment superficiel. Magister Thesis, University of Annaba, Algeria.
Grisey, E., Belle, E., Dat, J., Mudry, J., & Aleya, L. (2010). Survival of pathogenic and indicator organisms in groundwater and landfill leachate through coupling bacterial enumeration with tracer tests. Desalination, 261, 162–168.
Grisey, E., Laffray, X., Contoz, O., Cavalli, E., Mudry, J., & Aleya, L. (2012). The bioaccumulation performance of reeds and cattails in a constructed treatment wetland for removal of heavy metals in landfill leachate treatment (Etueffont, France). Water, Air, and Soil Pollution, 223, 1723–1741.
Gueddah, D. (2003). Evaluation de la pollution industrielle et urbaine dans la région de Skikda: impact sur l’écosystème marin côtier. MSc dissertation, University of Annaba, Algeria.
Gunawardana, C. G., Martinez, R. E., Xiao, W., & Templeton, D. M. (2006). Cadmium inhibits both intrinsic and extrinsic apoptotic pathways in renal mesangial cells. American Journal of Physiology-Renal Physiology, 290, 1074–1082.
IAEA. (1997). Standard operating procedure for trace metals determination. Wien: Marine Environmental Laboratory, International Atomic Energy Agency IAEA/UNEP.
IAEA. (2001). The analytical performance study for the MEDPOL region: determination of trace elements and methyl mercury in estuarine sediment sample. Wien: International Atomic Energy Agency IAEA/UNEP.
Jaishankar, M., Tseten, T., Anbalagan, N., Mathew, B. B., & Beeregowda, K. N. (2014). Toxicity, mechanism and health effects of some heavy metals. Interdisciplinary Toxicology, 7, 60–72.
Larrose, A., Coynel, A., Schafer, J., Blanc, L., Massé, L., & Maneux, E. (2010). Assessing the current state of the Gironde Estuary by mapping priority contaminant distribution and risk potential in surface sediment. Applied Geochemistry, 25, 1912–1922.
Lenoble, V., Omanović, D., Garnier, C., Mounier, S., Đonlagić, N., Le Poupon, C., & Pižeta, I. (2013). Distribution and chemical speciation of arsenic and heavy metals in highly contaminated waters used for health care purposes (Srebrenica, Bosnia and Herzegovina). Science of the Total Environment, 443, 420–428.
Lesven, L., Lourino-Cabana, B., Billon, G., Proix, N., Recourt, P., Ouddane, B., et al. (2009). Water-quality diagnosis and metal distribution in a strongly polluted zone of Deûle river (northern France). Water, Air, and Soil Pollution, 198, 31–44.
Li, X. D., Wang, D. C., Wai, O. M. H., Li, Y. S., Liu, W. X., & Shen, Z. G. (2003). Multivariate statistical study of heavy metal enrichment in sediments of the earl river estuary. Environmental Pollution, 121, 377–388.
Louhi, A., Mokhnache, S. A., Djabri, L., Gheid, A., & Messadi, D. (1998). Pollutions des Oueds Meboudja et Seybouse dans la zone industrielle de Annaba (Nord-Est Algérien). Partie 1: Utilisation de la spectroscopie d'absorption atomique avec flamme. Journal de la Societe Algerienne de Chimie, 8, 45–55.
Mance, G. (1987). Pollution threat of heavy metals in aquatic environments. Berlin: Springer.
Nagajyoti, P. C., Lee, K. D., & Sreekanth, T. V. M. (2010). Heavy metals, occurrence and toxicity for plants: a review. Environmental Chemistry Letters, 8, 199–216.
Onyari, J. M., & Wandiga, S. O. (1989). Distribution of Cr, Pb, Cd, Zn, Fe and Mn in Lake Victoria sediments, East Africa. Bulletin of Environmental Contamination and Toxicology, 42, 807–813.
Praveena, S. M., Aris, A. Z., & Radojevic, M. (2010). Heavy metals dynamics and source in intertidal mangrove sediment of Sabah, Borneo Island. Environ Asia, 3, 79–83. doi:10.14456/ea.2010.44.
Rainbow, P. S. (2002). Trace metal concentrations in aquatic invertebrates: why and so what? Environmental Pollution, 120, 497–507.
Rodier, J., Bazin, C., Broutin, J. P., Chambou, P., Champsaur, H., & Rodier, L. (1996). L’analyse de l’eau: eaux naturelles, eaux résiduaires et eaux de mer (8th ed.). Paris: Collection Dunod.
Romić, M., & Romić, D. (2003). Heavy metals distribution in agricultural topsoils in urban area. Environmental Geology, 43, 795–805.
Roussiez, V., Ludwig, W., Probst, J. L., & Monaco, A. (2005). Background levels of heavy metals in surficial sediments of the Gulf of Lions (NW Mediterranean): an approach based on 133 Cs normalization and lead isotope measurements. Environmental Pollution, 138, 167–177.
Saad, M. A. H., & Fahmy, M. A. (1985). Occurrence of some heavy metals in surficial sediments of the Nile delta lakes, Egypt. Journal of Water, Air, and Soil Pollution, 24, 27–33.
Saad, M. A. H., McComas, S. R., & Eisenreich, S. J. (1985). Metals and chlorinated hydrocarbons in surficial sediments of three Nile delta lakes, Egypt. Water Air and Soil Pollution, 24, 27–39.
Salvarredy-Arangren, M.M. (2008). Contamination en métaux lourds des eaux de surface et des sédiments du Val de Milluni (Andes Boliviennes) par des déchets miniers. Approches géochimique, minéralogique et hydrochimique. PhD thesis, University of Toulouse 3, France.
Sprovieri, M., Feo, M. L., Prevedello, L., Manta, D. S., Sammartino, S., Tamburrino, S., & Marsella, E. (2007). Heavy metals, polycyclic aromatic hydrocarbons and polychlorinated biphenyls in surface sediments of the Naples harbour (southern Italy). Chemosphere, 67, 998–1009.
Varol, M., & Şen, B. (2012). Assessment of nutrient and heavy metal contamination in surface water and sediments of the upper Tigris River, Turkey. Catena, 92, 1–10.
Velasco, F., Alvaro, A., Suarez, S., Herrero, J. M., & Yusta, I. (2005). Mapping Fe-bearing hydrated sulphate minerals with short wave infrared (SWIR) spectral analysis at San Miguel mine environment, Iberian Pyrite Belt (SW Spain). Journal of Geochemical Exploration, 87, 45–72.
Wahsha, M., Bini, C., Fontana, S., Wahsha, A., & Zilioli, D. (2012). Toxicity assessment of contaminated soils from a mining area in Northeast Italy by using lipid peroxidation assay. Journal of Geochemical Exploration, 113, 112–117.
WHO. (2001). Depleted uranium: sources, exposure and health effects. Geneva: World health organization ‘WHO’.
Zonta, R., Botter, M., Cassin, D., Pini, R., Scattolin, M., & Zaggia, L. (2007). Sediment chemical contamination of a shallow water area close to the industrial zone of Porto Marghera (Venice Lagoon, Italy). Marine Pollution Bulletin, 55, 529–542.
Zuur, A. F., Ieno, E. N., Walker, N. J., Saveliev, A. A., & Smith, G. M. (2009). Mixed effects models and extensions in ecology with R. New York: Springer.
Acknowledgments
We thank Mr. Abdelkrim Arar (University of Batna 2, Algeria) for his help in the preparation of the map used in Fig. 1.
Author information
Authors and Affiliations
Contributions
BEB and BS designed the study. BEB collected water and sediment samples and conducted laboratory analyses. HC analyzed data. BEB, HC, and AM drafted the manuscript. HC and BEB revised and prepared the final version. All authors read and approved the manuscript.
Corresponding author
Electronic supplementary material
Table S1
Climatic information of Annaba (Longitude: 7.81°, Latitude: 36.867°, Altitude: 0 m), northeastern Algeria (DOCX 131 kb)
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Belabed, BE., Meddour, A., Samraoui, B. et al. Modeling seasonal and spatial contamination of surface waters and upper sediments with trace metal elements across industrialized urban areas of the Seybouse watershed in North Africa. Environ Monit Assess 189, 265 (2017). https://doi.org/10.1007/s10661-017-5968-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-5968-5