Abstract
Cleaning associations are one of the most dynamic and complex mutualistic interactions of reef environments and are often influenced by local conditions. In the Western Atlantic (WE) most studies concentrate in tropical areas, with little attention to subtropical areas. We examined an assemblage of cleaner fish and their clients on the rocky reefs of the coast of Santa Catarina state, South Brazil, the southern limit of tropical reef fishes in the WE. We recorded 150 cleaning interactions, in which four fish species and one shrimp species acted as facultative cleaners. The grunt Anisotremus virginicus and the angelfish Pomacanthus paru serviced most clients. Fifteen fish species acted as clients, among which the most frequent was the planktivorous grunt Haemulon aurolineatum (31%). Cleaning interactions occurred mostly (87%) with non-carnivorous clients and the number of interactions was not related to the abundance of the species involved. The absence of dedicated cleaner fishes at the study sites and the replacement of their roles by facultative cleaners may be related to local conditions, including cold currents and reduction of rock cover. Under these circumstances, clients take advantage of the services offered by facultative cleaners, a characteristic of temperate areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reef environments are highly productive systems that harbour a great diversity of organisms, which present a great complexity in their relations (Luckhurst and Luckhurst 1978). These relations can be negative – predation and competition, or positive – commensalism and mutualism (Stachowicz 2001). Among the best-known mutualistic relationships is the cleaning activity (e.g., Losey 1972; Poulin and Grutter 1996; Vaughan et al. 2017), which occurs when a cleaner animal, such as fish or shrimp, removes and ingests ectoparasites, mucus, dead tissue or unwanted particles from a client animal, generally a fish (Losey 1972; Côté 2000; Vaughan et al. 2017). These interactions contribute to increasing reef fish diversity, affecting the small-scale distribution of the involved species, and helping to maintain healthy communities (Côté 2000; Grutter et al. 2003; Waldie et al. 2011). About 200 fish species and 50 shrimp species are recognized as cleaners in the marine environment (Vaughan et al. 2017). Cleaner fishes may clean throughout their lifetime (obligate or dedicated cleaners) or play the cleaning role at the juvenile stage only and feed on alternative sources (facultative cleaners; Côté 2000; Vaughan et al. 2017).
In tropical regions such as the Indo-Pacific and the Caribbean, the fish species richness is high and there are dedicated cleaners in both regions: cleaning wrasses of the genus Labroides in the Indo-Pacific and cleaner gobies of the genus Elacatinus in the Caribbean, the latter reaching the southwestern Atlantic (Potts 1973; Wicksten 1995; Côté 2000; Sazima et al. 2008; Côté and Soares 2011; Vaughan et al. 2017). Species of the genera Labroides and Elacatinus are the most important cleaners where they occur, as they service the species richest and most diverse fish client assemblages (Potts 1973; Kuwamura 1976; Johnson and Ruben 1988; Wicksten 1995; Grutter and Poulin 1998; Sazima et al. 2000, 2008, 2010b). On the other hand, in subtropical and temperate regions dedicated cleaners are missing, and cleaning services are provided by facultative cleaners of several families, which generally service small and not much diverse fish client assemblages (e.g., Ayling and Grace 1971; Moosleitner 1980; Côté 2000; Zander and Sötje 2002; Shepherd et al. 2005; Krajewski 2007; Bertoncini et al. 2009; Narvaez et al. 2015; Vaughan et al. 2017; Walsh et al. 2017). The same appears to be the case of very isolated islands such as Ascension, Rocas Atoll, Cape Verde and São Tomé Island in the Atlantic (Quimbayo et al. 2012; Morais et al. 2017; Quimbayo et al. 2017b), and Malpelo Island in Eastern Pacific (Quimbayo et al. 2017a).
Species richness and abundance in a community play a fundamental role in the structure of cleaning interactions, since abundant species are more likely to meet cleaner organisms (Floeter et al. 2007b). Moreover, parasite load and cleaner preferences for some clients are factors that also influence the structure of cleaning interactions, since these two factors determine how much time cleaners and clients can spend in these interactions (Grutter 2002). However, these factors may vary according to regional and local conditions, and thus the factors that may modulate the strength and diversity of these relationships vary among localities (Guimarães et al. 2007).
In the southwestern Atlantic, the state of Santa Catarina in Brazil harbours low species richness and is the southern limit of distribution of many reef fishes with tropical affinities (Floeter et al. 2004; Ferreira et al. 2004; Anderson et al. 2015). This area is influenced by the warm tropical water from Brazil Current in summer and the cold water from the South Atlantic Central Water in winter, which causes a decrease in water temperature and, consequently, severe disturbances on fish fauna (Anderson et al. 2015). Besides, Santa Catarina is also the southern limit of rocky reefs, which causes a reduction of the benthic cover and space used by reef fishes. The temperature variations and the reduction of cover make this region an ideal area for studies on interactions among reef organisms, including those of cleaners and their clients.
The Santa Catarina coastal area is practically a blank space from the cleaning interaction perspective, as there is only one study of this relationship for this region (Krajewski 2007). In view of the lack of information about cleaning activity and the importance of this region as a transition area between warm-water and cold-water fish fauna (Floeter et al. 2004; Ferreira et al. 2004), our study aims for a description of cleaning interactions at this southern distribution limit of reef fish fauna. More specifically, we intend to answer (1) how many species act as cleaners and how many act as clients? (2) how does the cleaning frequency vary among cleaners? (3) which trophic groups of clients are attended by the cleaners and what are the most serviced? (4) are cleaning interactions influenced by the abundance of cleaners and clients? The answers to these questions provide new insights on cleaning interactions in subtropical areas of the southwestern Atlantic.
Materials and methods
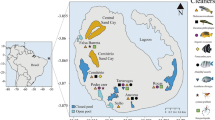
Cleaning interactions were recorded at 10 sites along the Santa Catarina state (25°59’S, 29°19’S; Fig. 1) coastal area, which is the southernmost distribution limit of reef organisms with tropical affinities, such as fish (Floeter et al. 2008) and corals (Capel et al. 2012). This limit is influenced both by warm and cold currents, which affect the distribution of tropical marine organisms (Anderson et al. 2015). All study sites are rocky reefs with small cover of coral colonies and large cover of algae and sponges (Floeter et al. 2001).
The study sites at the southern limit of reef fish distribution in the southwestern Atlantic. (A) Coast of Brazil limits. The square stands for the state of Santa Catarina. (B) Coast of Santa Catarina limits. Circles are our study sites: SE = Praia da Sepultura, GA = Ilha da Galé, DE = Deserta, SA = Saco do Capim, RN = Rancho Norte, BF = Baía do Farol, BL = Barra da Lagoa, XA = Ilha do Xavier, PS = Pântano do Sul
We analysed cleaning interactions from 300 min of direct observations during scuba diving, 203 min video recordings, and 128 photographs along three years (2014–2016). We considered that a cleaning interaction began when cleaner and client had physical contact and ended when some of these two parts withdrew (Losey 1972). We classified each client species within the trophic groups defined by Ferreira et al. (2004). Finally, we estimated species richness and abundance of clients through underwater visual censuses (UVCs; N = 450) along 40 m2 (20 × 2 m) transects (Floeter et al. 2007a).
Data analyses
The frequency of cleaning interactions was evaluated through one bipartite interaction network between cleaners and clients, considering the abundance of each client species and the number of interactions between both parts. We carried out this analysis using the function “plotwed” within the package bipartite (Dormann et al. 2008). To evaluate the relationship between the proportion of cleaning interactions and the abundance of clients, we used a linear regression with 95% confidential intervals over model predictions. Species falling above or below this interval were considered, respectively, as interacting disproportionally more or less than expected given their abundance. All the analyses were performed in R software version 3.2.4 (R Core Team 2016).
Results
We recorded 150 cleaning interactions during our study. A small assemblage of five facultative cleaner species was recorded at our study sites in Santa Catarina. Four fishes, the porkfish Anisotremus virginicus and the silver porgy Diplodus argenteus (Haemulidae), the french angelfish Pomacanthus paru (Pomacanthidae), the sergeant major Abudefduf saxatilis (Pomacentridae) and one shrimp species, Lysmata ankeri (Hyppolitidae) serviced an assemblage of 15 client species (Table 1; Fig. 2). The French angelfish and the porkfish tended cleaning stations near the bottom and serviced their clients at the stations or in midwater not far from these. The silver porgy and the sergeant major serviced the clients at variable locations from near the bottom to midwater. The shrimp tended cleaning stations in crevices (Fig. 3).
Bipartite network of cleaning interactions at the southern limit of reef fishes in the southwestern Atlantic, showing the number of interactions and the abundance of cleaners and clients. The cleaners are arranged in order of the total number of interactions, from highest to lowest. Clients are organized in order of abundance. The lines represent the number of interactions and the circles represent an estimate of total abundance of the species at the study sites in Santa Catarina. The size of the circles and the thickness of the lines represent the logarithm of the values of abundance and number of interactions
Interactions between cleaners and clients in Santa Catarina. a Anisotremus virginicus juveniles clean a posing Haemulon aurolineatum at Xavier Island. Photo: Floeter SR (b) Adult A. virginicus services a group of Priacanthus arenatus, which darken requesting the cleaner’s services. Photo: Floeter SR (c) Pomacanthus paru about to clean a H. aurolineatum individual that poses with an open mouth. Photo: Morais RA (d) Cantherhines macrocerus poses requesting cleaning by P. paru (in the lower right corner). Photo: Nunes LT (e) Diplodus argenteus cleans Kyphosus sectatrix while the client darkens (compare with the individual at the right, which waits for cleaning services with stretched fins and a habitual colour. Photo: Krajewski JP (f) Lysmata ankeri cleaning the right mouth corner of Gymnothorax moringa. Photo: Krajewski JP
The cleaner that displayed the highest number of interactions was A. virginicus, which interacted 75 times (50% of the interactions), followed by P. paru, which interacted 47 times (31% of the interactions) and D. argenteus, which interacted 21 times (14% of interactions). The other two species, which had a lower number of interactions, were L. ankeri, which interacted five times (3.5% of the interactions) and A. saxatilis, which interacted only twice (1.5% of the interactions; Fig. 2). Anisotremus virginicus was the only cleaner that cleaned both in the juvenile and adult stages (Fig. 3a, b).
All recorded interactions were initiated by the clients, which displayed characteristic swimming and/or posing, besides displaying colour changes (Fig. 3). We recorded 15 client species in 12 families and five trophic groups (Table 1) serviced by the cleaner assemblage. Most of the cleaning interactions (55%) were with mobile invertebrate feeders (MINV), 26% were with omnivores (OMNI) and 14% were with herbivore-detritivores (HERB). The other two trophic groups attended by the cleaners were sessile invertebrate feeders (SINV), with 4% of interactions and carnivores (MCAR), with 1% of interactions (Table 1).
We did not observe a clear relationship between the number of cleaning interactions and the abundance of clients (R2 = 0.19, p = 0.05, Fig. 4). However, some species such as Priacanthus arenatus and Kyphosus sectatrix interacted more than expected from their abundance. In contrast, species such as Sparisoma frondosum and Chaetodon striatus interacted less than expected from their abundance (Fig. 4).
Linear regression between the number of cleaning interactions and the abundance of client species recorded in Santa Catarina. Each point stands for a client species. Line represents the linear model and shaded area represents the confidence intervals of 95% generated based on standard errors of model’s predictions. Species that are outside of the confidence intervals are highlighted with the name and figure of the species. KYP SEC = Kyphosus sectatrix, PRI ARE = Priacanthus arenatus, FIS TAB = Fistularia tabacaria, SPA FRO = Sparisoma frondosum, CHA STR = Chaetodon striatus
Discussion
Our study reveals that only facultative cleaners (sensu Côté 2000; Vaughan et al. 2017) perform cleaning services at the southern limit of tropical reef fishes in the southwestern Atlantic. From this viewpoint, our results are similar to those recorded at temperate sites in the Northern Atlantic and the Mediterranean Sea (e.g. Moosleitner 1980; Zander and Sötje 2002; Bertoncini et al. 2009; Narvaez et al. 2015), and probably indicate a trend. Further studies will likely strengthen the suggestion that in subtropical areas or at very isolated islands, facultative cleaners replace the role played by dedicated cleaners (Morais et al. 2017; Quimbayo et al. 2017a). Moreover, these studies may shed light on the role the facultative cleaners play and what factors can modulate their behaviour in different regions (see Walsh et al. 2017). Although the dedicated cleaner goby Elacatinus figaro occurs in Santa Catarina (based on one specimen, Sazima et al. 1997), we found none at our study area. It would be of interest to search for this goby on Santa Catarina coast and assess its importance as a cleaner in the reef community, given that it may be very rare or the only individual found might be a stray (Anderson et al. 2015).
Our observations on the shrimp Lysmata ankeri (recently recorded in Santa Catarina - Giraldes and Freire 2015) are the first records of cleaning activity of this crustacean in the southwestern Atlantic. However, the number of clients and cleaning events of this species can be underestimated since other species of cleaners shrimp of this genus exhibit nocturnal activity, and thus, interact little with diurnal clients. The four fish cleaner species recorded in our study have been recorded in cleaning interactions elsewhere in the southwestern Atlantic (e.g. Sazima et al. 1999; Krajewski 2007; Sampaio et al. 2017). The cleaner assemblage at our Santa Catarina sites may be considered large (2% cf. Anderson et al. 2015) when compared to the total number of reef fish species recorded at the study area (Anderson et al. 2015). For instance, at the oceanic Trindade Island and Rocas Atoll the cleaner assemblage are smaller (about 1% cf. Gasparini and Floeter 2001; Quimbayo et al. 2017b). Assessing the percentage of cleaner species versus total reef fish fauna for most if not all sites in the southwestern Atlantic will much probably reveal cleaner assemblages smaller than that we found in Santa Catarina.
The porkfish Anisotremus virginicus adults engaged in cleaning activities is an unexpected result in our study, since this species is reported as cleaner only during juvenile stages (Côté 2000; Feitoza et al. 2002; Krajewski 2007; Luiz et al. 2008; Sazima et al. 2010b; Vaughan et al. 2017). Cleaning by facultative cleaners both as juveniles and adults is known for several other species, a few examples being the king angelfish Holacanthus passer at Malpelo Island in the Central Pacific (Quimbayo et al. 2017a), and the Noronha wrasse Thalassoma noronhanum at Rocas Atoll in the West Atlantic (Quimbayo et al. 2017b). The high number of cleaning interactions recorded here for A. virginicus is likely related to the behaviour of juveniles, which form schools of up to about 10 individuals near large rocks (cleaning stations) and, thus, call attention of clients that search for a ready and fast service.
The high number of cleaning interactions we recorded for the french angelfish P. paru supports findings along the coast as well as at oceanic islands in the southwestern Atlantic, at which this angelfish is an important cleaner (Sazima et al. 1999; Luiz et al. 2008; Sazima et al. 2010b; Morais et al. 2017; Quimbayo et al. 2017b; Sampaio et al. 2017). At the Abrolhos Archipelago, P. paru acts as a specialized cleaner and services an assemblage of 31 fish client species (Sazima et al. 1999, 2010b). This number is comparable to the species richness of client assemblages serviced by the cleaner gobies of the genus Elacatinus, which are the only dedicated cleaners in the southwestern Atlantic (Sazima et al. 2000; Gasparini and Floeter 2001; Sazima et al. 2008; Campos and Sá-Oliveira 2011). The similarity of client assemblage richness of the french angelfish and the gobies is likely associated with some traits shared by both these cleaners, such as yellow stripes on black body colour, which increase the signal for cleaning services and contrast the cleaner against the substrate (Cheney et al. 2009). Although at our study sites in Santa Catarina P. paru serviced the richest client assemblage, this species richness barely compares to those found in northward coastal areas (Sazima et al. 1999, 2010b). Nevertheless, our results indicate that the french angelfish, despite being an important cleaner in the subtropical Santa Catarina, seems to not devote much time to cleaning interaction (see Morais et al. 2017 at the isolated Ascension Island) and feed on alternative sources instead.
The low number of cleaning interactions displayed by the sergeant major Abudefduf saxatilis would be expected, as a few previous records of this cleaner were mostly with sea turtles (Sazima 1988; Sazima et al. 2010a; Quimbayo et al. 2017b). However, at Fernando de Noronha Archipelago, the sergeant major regularly services a few fish species including the flat needlefish Ablennes hians (Sazima et al. 2013). On the other hand, the low number of interactions recorded here for the shrimp Lysmata ankeri may be due to our diurnal observations and the cryptic and/or nocturnal habits of cleaner shrimps (Williams 1984; Bonaldo et al. 2015; Giraldes and Freire 2015). This cryptic and possibly nocturnal shrimp was the only cleaner we recorded interacting with potentially dangerous clients such as the spotted moray Gymnothorax moringa. However, cleaner shrimps interacting with morays are a common sight worldwide (e.g., Côté 2000; Morais et al. 2017) and much probably morays do not pose a real danger to shrimps or other small cleaners such as juvenile spotfin hogfish Bodianus pulchellus or the noronha goby Elacatinus phthirophagus (Luiz et al. 2008; Sazima et al. 2013).
At our study sites in Santa Catarina, the cleaner fishes apparently preferred to stay safe by cleaning client species that pose no (or slight) predation risk, although piscivorous and carnivorous clients habitually interact with cleaners in the southwestern Atlantic and other regions (Grutter 2004; Cheney et al. 2008; Francini-Filho and Sazima 2008), even with facultative cleaners (Sazima et al. 1999; Quimbayo et al. 2010; Morais et al. 2017). The interaction with potentially dangerous clients may be related to the degree of specialization of the cleaner (Darcy et al. 1974; Sazima et al. 1999), since clients may prey on cleaners (Francini-Filho et al. 2000).
Despite the absence of a clear relationship between the number of cleaning interactions and the abundance of clients, our results indicate some noteworthy interactions between cleaners and clients. For instance, the silver porgy Diplodus argenteus had a high number of interactions with the Bermuda chub Kyphosus sectatrix, which indicates that there may be a preference for the chub client (see Sazima et al. 1999 for preferences not related to abundance). The bigeye Priacanthus arenatus was the only client species that interacted with adult cleaning porkfish Anisotremus virginicus and was one of the species that interacted more than expected from its abundance, although abundance is an important factor for frequent interactions at the large scale (Floeter et al. 2007a, b).
In conclusion, our study shows the importance of facultative cleaners in subtropical areas (where dedicated cleaners are missing), and the variable behaviour of a cleaner species previously considered as cleaner only during juvenile stages. Moreover, our results indicate that low species richness in subtropical areas may have an influence on cleaning networks, since few cleaner species interact with few client species. This result indicates the possibility that in subtropical areas cleaning networks are less nested than in tropical areas (e.g. Guimarães et al. 2007).
References
Anderson AB, Carvalho-Filho A, Morais RA, Nunes LT, Quimbayo JP, Floeter SR (2015) Brazilian tropical fishes in their southern limit of distribution: checklist of Santa Catarina’s rocky reef ichthyofauna, remarks and new records. Check List 11:1–25. https://doi.org/10.15560/11.4.1688
Ayling AM, Grace RV (1971) Cleaning symbiosis among New Zealand fishes. New Zeal J Mar Freshw Res 5:205–218. https://doi.org/10.1080/00288330.1971.9515377
Bertoncini AA, Machado LF, Barreiros JP, Hostim-Silva M, Verani JR (2009) Cleaning activity among Labridae in the Azores: the rainbow wrasse Coris julis and the Azorean blue wrasse Centrolabrus caeruleus. J Mar Biol Assoc UK 89:859–861. https://doi.org/10.1017/S002531540900040X
Bonaldo RM, Grutter AS, Sazima I, Krajewski JP (2015) 24/7 service: nocturnal cleaning in a tropical Indo-Pacific reef. Mar Biodivers 45:611–612. https://doi.org/10.1007/s12526-014-0289-1
Campos CEC, Sá-Oliveira JC (2011) Atividade de limpeza e clientes de Elacatinus figaro (Pisces: Gobiidae) nos recifes de coral dos parrachos de Muriú, Nordeste do Brasil. Biota Neotrop 11:47–51. https://doi.org/10.1590/S1676-06032011000100004
Capel KCC, Segal B, Bertuol P, Lindner A (2012) Corallith beds at the edge of the tropical South Atlantic. Coral Reefs 31:75. https://doi.org/10.1007/s00338-011-0818-3
Cheney KL, Bshary R, Grutter AS (2008) Cleaner fish cause predators to reduce aggression toward bystanders at cleaning stations. Behav Ecol 19:1063–1067. https://doi.org/10.1093/beheco/arn067
Cheney KL, Grutter AS, Blomberg SP, Marshall NJ (2009) Blue and yellow signal cleaning behavior in coral reef fishes. Curr Biol 19:1283–1287. https://doi.org/10.1016/j.cub.2009.06.028
Côté IM (2000) Evolution and ecology of cleaning symbiosis in the sea. Oceanogr Mar Biol 38:311–355
Côté IM, Soares MC (2011) Gobies as cleaners. In: Patzner RA, Van Tassel JL, Kovacic M, Kapoor BG (eds) The biology of gobies. Science Publishers, Jersey
Darcy GH, Maisel E, Ogden JC (1974) Cleaning preferences of the gobies Gobiosoma evelynae and G. prochilos and the juvenile wrasse Thalassoma bifasciatum. Copeia 1974:375–379
Dormann CF, Fruend J, Bluethgen N, Gruber B (2008) Introducing the bipartite package: Analysing ecological networks. R news 8(2):8–11
Feitoza BM, Dias TLP, Rocha LA, Gasparini JL (2002) First record of cleaning activity in the slippery dick, Halichoeres bivittatus (Perciformes : Labridae), off northeastern Brazil. Aquat J Ichthyol Biol 5:73–76
Ferreira CEL, Floeter SR, Gasparini JL, Ferreira BP, Joyeux JC (2004) Trophic structure patterns of Brazilian reef fishes: a latitudinal comparison. J Biogeogr 31:1093–1106. https://doi.org/10.1111/j.1365-2699.2004.01044.x
Floeter SR, Guimarães RZP, Rocha LA et al (2001) Geographic variation in reef-fish assemblages along the Brazilian coast. Glob Ecol Biogeogr 10:423–431. https://doi.org/10.1046/j.1466-822X.2001.00245.x
Floeter SR, Ferreira CEL, Dominici-Arosemena A, Zalmon IR (2004) Latitudinal gradients in Atlantic reef fish communities: trophic structure and spatial use patterns. J Fish Biol 64:1680–1699. https://doi.org/10.1111/j.1095-8649.2004.00428.x
Floeter SR, Krohling W, Gasparini JL, Ferreira CEL, Zalmon IR (2007a) Reef fish community structure on coastal islands of the southeastern Brazil: the influence of exposure and benthic cover. Environ Biol Fish 78:147–160. https://doi.org/10.1007/s10641-006-9084-6
Floeter SR, Vázquez DP, Grutter AS (2007b) The macroecology of marine cleaning mutualisms. J Anim Ecol 76:105–111. https://doi.org/10.1111/j.1365-2656.2006.01178
Floeter SR, Rocha LA, Robertson DR, Joyeux JC, Smith-Vaniz WF, Wirtz P, Edwards AJ, Barreiros JP, Ferreira CEL, Gasparini JL, Brito A, Falcón JM, Bowen BW, Bernardi G (2008) Atlantic reef fish biogeography and evolution. J Biogeogr 35:22–47. https://doi.org/10.1111/j.1365-2699.2007.01790.x
Francini-Filho RB, Moura RL, Sazima I (2000) Cleaning by the wrasse Thalassoma noronhanum, with two records of predation by its grouper client Cephalopholis fulva. J Fish Biol 56:802–809. https://doi.org/10.1006/jfbi.1999.1197
Francini-Filho RB, Sazima I (2008) A comparative study of cleaning activity of two reef fishes at Fernando de Noronha Archipelago, tropical West Atlantic. Environ Biol Fish 83(2):213–220
Gasparini JL, Floeter SR (2001) The shore fishes of Trindade Island, western South Atlantic. J Nat Hist 35:1639–1656
Giraldes BW, Freire AS (2015) Extending the southern range of four shrimps (Crustacea: Decapoda: Stenopodidae, Hippolytidae and Alpheidae) in southwestern Atlantic (27° S) and confirming the presence of Mediterranean Stenopus spinosus Risso, 1827 in Brazil. Zootaxa 3972:419–431. https://doi.org/10.11646/zootaxa.3972.3.7
Grutter AS (2002) Cleaning symbioses from the parasites’ perspective. Parasitology 124:s61–s81. https://doi.org/10.1017/S0031182002001488
Grutter AS (2004) Cleaner fish use tactile dancing behavior as a preconflict management strategy. Curr Biol 14:1080–1083. https://doi.org/10.1016/j.cub.2004.05.048
Grutter AS, Poulin R (1998) Cleaning of coral reef fishes by the wrasse Labroides dimidiatus: influence of client body size and phylogeny. Copeia 1998:120–127
Grutter AS, Murphy JM, Choat JH (2003) Cleaner fish drives local fish diversity on coral reefs. Curr Biol 13:64–67. https://doi.org/10.1016/S0960-9822(02)01393-3
Guimarães PR, Sazima C, dos Reis SF, Sazima I (2007) The nested structure of marine cleaning symbiosis: is it like flowers and bees? Biol Lett 3:51–54. https://doi.org/10.1098/rsbl.2006.0562
Johnson WS, Ruben P (1988) Cleaning behavior of Bodianus rufus, Thalassoma bifasciatum, Gobiosoma evelynae, and Periclimenes pedersoni along a depth gradient at Salt River submarine canyon, St. Croix. Environ Biol Fish 23:225–232
Krajewski JP (2007) Cleaning by the occasional cleaner Diplodus argenteus (Perciformes: Sparidae) in South Brazil: why so few client species? J Mar Biol Assoc UK 87:1013–1016. https://doi.org/10.1017/S0025315407055981
Kuwamura T (1976) Different responses of inshore fishes the the cleaning wrasse, Labroides dimidiatus, as observed in Shirahama. Publ Seto Mar Biol Lab 23:119–144
Losey GS (1972) The ecological importance of cleaning symbiosis. Copeia (4):960–970
Luckhurst E, Luckhurst K (1978) Analysis of the influence of substrate variables on coral reef fish communities. Mar Biol 323:317–323. https://doi.org/10.1007/BF00455026
Luiz OJ, Carvalho-Filho A, Ferreira CEL et al (2008) The reef fish assemblage of the Laje de Santos marine State Park, southwestern Atlantic: annotated checklist with comments on abundance, distribution, trophic structure, symbiotic associations, and conservation. Zootaxa 1807:1–25
Moosleitner VH (1980) Putzerfische und –garnelen im Mittelmeer. Zool Anzeiger 205:219–240
Morais RA, Brown J, Ferreira CEL et al (2017) Mob rulers and part-time cleaners: two reef fish associations at the isolated Ascension Island. J Mar Biol Assoc UK 94(4):799–811. https://doi.org/10.1017/S0025315416001041
Narvaez P, Furtado M, Neto A, Moniz I, Azevedo JMN, Soares MC (2015) Temperate facultative cleaner wrasses selectively remove ectoparasites from their client-fish in the Azores. Mar Ecol Prog Ser 540:217–226. https://doi.org/10.3354/meps11522
Potts GW (1973) The ethology of Labroides dimidiatus (Cuv. & Val.) (Labridae) on Aldabra. Anim Behav 21:250–291
Poulin R, Grutter AS (1996) Cleaning symbioses: proximate and adaptive explanations. BioScience 46(7):512–517
Quimbayo JP, Zapata FA, Floeter SR, Bessudo S, Sazima I (2010) First record of cleaning by a triplefin blenny in the tropical Pacific. Coral Reefs 29:909–909. https://doi.org/10.1007/s00338-010-0656-8
Quimbayo JP, Floeter SR, Noguchi R, Rangel CA, Gasparini JL, Sampaio CLS, Ferreira CEL, Rocha LA (2012) Cleaning mutualism in Santa Luzia (Cape Verde archipelago) and São Tomé Islands, tropical eastern Atlantic. Mar Biodivers Rec e118:5. https://doi.org/10.1017/S175526721200108X
Quimbayo JP, Dias MS, Schlickmann OC, Mendes TC (2017a) Fish cleaning interactions on a remote island from the tropical eastern Pacific. Mar Biodivers 43:603–608. https://doi.org/10.1007/s12526-016-0493-2
Quimbayo JP, Nunes LT, Ozekoski R, et al. (2017b) Cleaning interactions at the only atoll in the South Atlantic. Environ Biol Fishes. https://doi.org/10.1007/s10641-017-0612-3
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Sampaio CLS, Loiola M, Colman LP, Medeiros DV, Quimbayo JP, Miranda RJ, Reis-Filho JA, Nunes JACC (2017) Cryptobenthic fish as clients of french angelfish Pomacanthus paru (Pomacanthidae) during cleaning behaviour. Mar Biodivers Rec 10:8. https://doi.org/10.1186/s41200-017-0109-y
Sazima I (1988) Similarities in feeding behaviour between some marine and freshwater fishes in two tropical communities. J Fish Biol 29:53–65
Sazima I, Moura RL, Rosa RS (1997) Elacatinus figaro sp. n. (Perciformes: Gobiidae), a new cleaner goby from the coast of Brazil. aqua J. Ichthyol. Aquat Biol 2:33–38
Sazima I, Moura RL, Sazima C (1999) Cleaning activity of juvenile angelfish, Pomacanthus paru, on the reefs of the Abrolhos archipelago, western South Atlantic. Environ Biol Fish 56:399–407. https://doi.org/10.1023/A:1007531925845
Sazima I, Sazima C, Francini-Filho RB, Moura RL (2000) Daily cleaning activity and diversity of clients of the barber goby, Elacatinus figaro, on rocky reefs in southeastern Brazil. Environ Biol Fish 59:69–77. https://doi.org/10.1023/A:1007655819374.
Sazima I, Carvalho-Filho A, Sazima C (2008) A new cleaner species of Elacatinus (Actinopterygii: Gobiidae) from the southwestern Atlantic. Zootaxa 1932:27–32
Sazima C, Grossman A, Sazima I (2010a) Turtle cleaners: reef fishes foraging on epibionts of sea turtles in the tropical southwestern Atlantic, with a summary of this association type. Neotrop Ichthyol 8:187–192. https://doi.org/10.1590/S1679-62252010005000003
Sazima C, Guimarães PRJ, Reis SF, Sazima I (2010b) What makes a species central in a cleaning mutualism network? Oikos 119:1319–1325. https://doi.org/10.1111/j.1600-0706.2009.18222.x
Sazima I, Krajewski JP, Bonaldo RM, Sazima C (2013) A vida dos peixes em Fernando de Noronha. Terra da Gente, Campinas, Brazil
Shepherd SA, Teale J, Muirhead D (2005) Cleaning symbiosis among inshore fishas at Althorpe Island, South Australia and elsewhere. Trans Royal Soc S Aust 129:193–201
Stachowicz JJ (2001) Mutualism, facilitation, and the structure of ecological communities. Bioscience 51:235–246. https://doi.org/10.1641/0006-3568(2001)051[0235:MFATSO]2.0.CO;2
Vaughan DB, Grutter AS, Costello MJ, Hutson KS (2017) Cleaner fishes and shrimp diversity and a re-evaluation of cleaning symbioses. Fish Fish 18(4):698–716. https://doi.org/10.1111/faf.12198
Waldie PA, Blomberg SP, Cheney KL, Goldizen AW, Grutter AS (2011) Long-term effects of the cleaner fish Labroides dimidiatus on coral reef fish communities. PLoS One 6(6):e21201. https://doi.org/10.1371/journal.pone.0021201
Walsh CAJ, Pinheiro HT, Rocha LA, Goodbody-Gringley G (2017) Cleaning service gaps in Bermuda, North Atlantic. Ecology 98:1973–1974. https://doi.org/10.1002/ecy.1841
Wicksten MK (1995) Associations of fishes and their cleaners on coral reefs of Bonaire, Netherlands Antilles. Copeia 1995:477–481
Williams AB (1984) Shrimps, lobsters, and crbs of the Altantic coast of the eastern United States, Maine to Florida. Smithson Inst Press. https://doi.org/10.2307/1352125, 8, 77
Zander CD, Sötje I (2002) Seasonal and geographical differences in cleaner fish activity in the Mediterranean Sea. Helgol Mar Reser 55:232–241. https://doi.org/10.1007/s101520100084
Acknowledgements
This study was carried out under “Projeto Ilhas do Sul” (PI: Sergio R. Floeter, Conselho Nacional de Desenvolvimento Científico e Tecnológico 475367/2006-8), “Projeto MAArE – Monitoramento Ambiental do Arvoredo e Entorno” (PI: Barbara Segal, a condition set by the ICMBio in the context of IBAMA’s environmental licensing process), “Projeto Biodiversidade Marinha do Estado de Santa Catarina” (PI: Alberto Lindner, FAPESC 4302/2010-8), and SISBIOTA-Mar (PI: Sergio R. Floeter, CNPq 563276/2010-0; Fundação de Amparo à Pesquisa do Estado de Santa Catarina 6308/2011-8). We also acknowledge LBMM staff for field assistance and comments during the development of this study. R.A. Morais., L.T. Nunes, and J.P. Krajewski for pictures used in Fig. 3. JPQ and ORCS thanks Coordenação de Aperfeiçoamento de Pessoal de Nível Superior and Conselho Nacional de Desenvolvimento Científico e Tecnológico for financial support. IS thanks Conselho Nacional de Desenvolvimento Científico e Tecnológico and Fundação de Amparo à Pesquisa do Estado de São Paulo for previous financial support to study cleaning symbiosis in the southwestern Atlantic.
Author information
Authors and Affiliations
Corresponding author
Additional information
JPQ and ORCS should be considered joint first authors.
Rights and permissions
About this article
Cite this article
Quimbayo, J.P., Schlickmann, O.R.C., Floeter, S.R. et al. Cleaning interactions at the southern limit of tropical reef fishes in the Western Atlantic. Environ Biol Fish 101, 1195–1204 (2018). https://doi.org/10.1007/s10641-018-0768-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-018-0768-5