Abstract
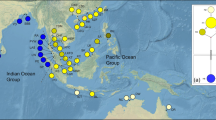
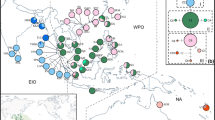
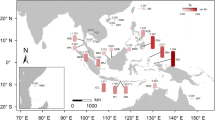
The genetic structure of mangrove species is greatly affected by their geographic history. Nine natural populations of Ceriops tagal were collected from Borneo, the Malay Peninsula, and India for this phylogeographic study. Completely different haplotype compositions on the east versus west coasts of the Malay Peninsula were revealed using the atpB-rbcL and trnL-trnF spacers of chloroplast DNA. The average haplotype diversity (Hd) of the total population was 0.549, nucleotide diversity (θ) was 0.030, and nucleotide difference (π) was 0.0074. The cladogram constructed by the index of population differentiation (G ST) clearly separated the South China Sea populations from the Indian Ocean populations. In the analysis of the minimum spanning network, the Indian Ocean haplotypes were all derived from South China Sea haplotypes, suggesting a dispersal route of C. tagal from Southeast Asia to South Asia. The Sunda Land river system and surface currents might be accountable for the gene flow directions in the South China Sea and Bay of Bengal, respectively. The historical geography not only affected the present genotype distribution but also the evolution of C. tagal. These processes result in the genetic differentiation and the differentiated populations that should be considered as Management Units (MUs) for conservation measurements instead of random forestation, which might lead to gene mixing and reduction of genetic variability of mangrove species. According to this phylogeographic study, populations in Borneo, and east and west Malay Peninsula that have unique genotypes should be considered as distinct MUs, and any activities resulting in gene mixing with each other ought to be prevented.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alfaro ME, Karn DR, Voris HK, Abernathy E, Sellins SL (2004) Phylogeny of Cerberus (Serpentes: Homalopsinae) and phylogeography of Cerberus rynchops: diversification of a coastal marine snake in Southeast Asia. J Biogeogr 31:1277–1292
Chiang TY, Chiang YC, Chen YJ, Chou CH, Havanond S, Hong TN, Huang S (2001) Phylogeography of Kandelia candel in East Asiatic mangroves based on nucleotide variation of chloroplast and mitochondrial DNAs. Mol Ecol 10:2697–2710
Clarke PJ, Kerrigan RA, Westphal CJ (2001) Dispersal potential and early growth in 14 tropical mangroves: do early life history traits correlate with patterns of adult distribution? J␣Ecol 89:648–659
Couper A (1983) The times atlas of the ocean. Van Norstrand Reinhold Co., New York
Dodd RS, Rafii ZA (2002) Evolutionary of genetics of mangroves: continental drift to recent climate change. Trends Ecol Evol 16:80–86
Duke NC (1992) Mangrove floristics and biogeography. In: Robertson A, Alongi D (eds) Tropical mangrove ecosystem. American Geographical Union, Washington DC, pp 63–100
Duke NC (1995) Genetic diversity, distributional barriers and rafting continents – more thoughts on the evolution of mangroves. Hydrobiologia 295:167–181
Duke NC, Benzie JAH, Goodall JA, Ballment ER (1998) Genetic structure and evolution of species in the mangrove genus Avicennia (Avicenniaceae) in the Indo-West Pacific. Evolution 52(6):1612–1626
Duke NC, Lo EYY, Sun M (2002) Global distribution and genetic discontinuities of mangroves-emerging pattern in the evolution of Rhizophora. Trees 16:65–79
Excoffier L, Smouse PE (1994) Using allele frequencies and geographic subdivision to reconstruct gene trees within a species: molecular variance parsimony. Genetics 136:343–359
Felsenstein J (1978) Cases in which parsimony or compatibility methods will be positively misleading. Syst Zool 27:401–410
Fraser DJ, Bernatchez L (2001) Adaptive evolutionary conservation: towards a unified concept for defining conservation units. Mol Ecol 10:2741–2752
Ge X-J, Sun M (2001) Population genetic structure of Ceriops tagal (Rhizophoraceae) in Thailand and China. Wetlands Ecol Manage 9:203–209
Huang S, Chen YJ, Havanond S (1999) Patterns of genetic variation of Ceriops tagal in Ranong and Eastern Malay Peninsula. Paper presented at Ecotone VIII. May 23–29, 1999, Ranong, Thailand
Kado T, Fujimoto A, Giang LH, Tuan M, Hong PN, Harada K, Tachida H (2004) Genetic structures of natural populations of three mangrove species, Avicennia marina, Kandelia candel and Lumnitzera racemosa, in Vietnam revealed by maturase sequences of plastid DNA. Plant Species Biol 19:91–99
Kumar S, Tamura K, Nei M (2004) MEGA3: Integrated Software for Molecular Evolutionary Genetics Analysis and Sequence Alignment. Briefings Bioinformatics 5:150–163
Lourie SA, Green DM, Vincent ACJ (2005) Dispersal, habitat differences, and comparative phylogeography of Southeast Asian seahorses (Syngnathidae: Hippocampus). Mol Ecol 14:1073–1094
Maguire TL, Saenger P, Baverstock P, Henry R (2000) Microsatellite analysis of genetic structure in the mangrove species Avicennia marina (Forsk.) Vierh. (Avicenniaceae). Mol Ecol 9:1853–1862
McGuinness KA (1997) Dispersal, establishment and survival of Ceriops tagal propagules in a north Australian mangrove forest. Oecologia 109:80–87
Moritz C (1994) Applications of mitochondrial DNA analysis in conservation: a critical review. Mol Ecol 3:401–411
Nei M (1987) Molecular evolutionary genetics. Columbia Univ. Press, New York
Newton AC, Allnutt TR, Gillies ACM, Lowe AJ, Ennos RA (1999) Molecular phylogeography, intraspecific variation and the conservation of tree species. Trends Ecol Evol 14:140–145
Plaziat J-C, Cavagnetto C, Koeniguer J-C, Baltzer F (2001) History and biogeography of the mangrove ecosystem, based on a critical reassessment of the paleontological record. Wetlands Ecol Manage 9:161–179
Robertson AI, Alongi DM (1992) Tropical mangrove ecosystems (coastal and estuarine studies; vol. 41), American Geophysical Union
Ronquist F, Huelsenbeck JP (2003) MRBAYES 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Rozas J, Sanchez-Delbarrio JC, Messeguer X, Rozas R (2003) DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 19:2496–2497
Ryder OA (1986) Species conservation and systematics: the dilemma of subspecies. Trends Ecol Evol 1:9–10
Schaal BA, Hayworth DA, Olsen KM, Rauscher JT, Smith WA (1998) Phylogeographic studies in plants: problems and prospects. Mol Ecol 7:465–474
Schwarzbach AE, Ricklefs RE (2001) The use of molecular data in mangrove plant research. Wetlands Ecol Manage 9:195–201
Sheue CR, Liu HY, Yong JWH (2003) Kandelia obovata (Rhizophoraceae), a new mangrove species from Eastern Asia. Taxon 52:287–294
Shi S, Zhong Y, Huang Y, Du Y, Qiu X, Chang H (2002) Phylogenetic relationships of the Rhizophoraceae in China based on sequences of the chloroplast gene matK and the internal transcribed spacer regions if nuclear ribosomal DNA and combined data set. Biochem Syst Ecol 30:309–319
Sun M, Wong KC, Lee JSY (1998) Reproductive biology and population genetic structure of Kandelia candel (Rhizophoraceae), a viviparous mangrove species. Am J Bot 85(11):1631–1637
Taberlet P, Gielly L, Pautou G, Bouvet J (1991) Universal primers for amplification of three non-coding regions of chloroplast DNA. Plant Mol Biol 17:1105–1109
Tan F, Huang Y, Ge X, Su G, Ni X, Shi S (2005) Population genetic structure and conservation implications of Ceriops decandra in Malay Peninsula and North Australia. Aquat Bot 81:175–188
Templeton AR (2001) Using phylogeographic analyses of gene trees to test species status and processes. Mol Ecol 10:779–791
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Tomlinson PB (1986) The botany of mangroves. Cambridge of University Press, Cambridge
Voris HK (2000) Maps of Pleistocene sea levels in Southeast Asia: shorelines, river systems and time durations. J Biogeogr 27:1153–1167
Zhong Y, Shi S, Tang X, Huang Y, Tan F, Zhang X (2000) Testing relative evolutionary rates and estimating divergence time among six genera of Rhizophoraceae using cpDNA and nrDNA sequences. Chinese Sci Bull 45:1011–1015
Acknowledgements
We are grateful to two anonymous reviewers for helpful comments on the manuscript. We thank Dr. C. R. Sheue for providing samples of West Bengal and Ms. Angele Kok Shoo Tan and Mr. Zhi-Yi Shi for the assistance in the sampling in Malaysia and thank Mr. Bing-Hong Huang for the laboratory experiment. This research was supported by an Academia Sinica Thematic Grant (2001–2004) and a grant (NSC92-2311-B003-005) from the National Science Council, R.O.C.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liao, PC., Havanond, S. & Huang, S. Phylogeography of Ceriops tagal (Rhizophoraceae) in Southeast Asia: the land barrier of the Malay Peninsula has caused population differentiation between the Indian Ocean and South China Sea. Conserv Genet 8, 89–98 (2007). https://doi.org/10.1007/s10592-006-9151-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-006-9151-8