Abstract
Clasmatodendrosis is an irreversible astroglial degenerative change, which includes extensive swelling and vacuolization of cell bodies and disintegrated and beaded processes. This study was designed to elucidate whether clasmatodendrosis may be one of the autophagy-related degeneration of astrocytes. In this study, clasmatodendritic astrocytes were observed only in the stratum radiatum in the CA1 region. Vacuoles in clasmatodendritic astrocytes showed LAMP-1 immunoreactivity. In addition, both LC3-II and Beclin-1 expression were detected in most of clasmatodendritic astrocytes as well as a few non-vacuolized astrocytes. Clasmatodendritic astrocytes also showed p65/RelA-Ser529 phosphorylation in the nuclei. The neutralization of TNF-α by sTNFp55R infusion reduced clasmatodendritic astrocytes with nuclear p65/RelA-Ser529 phosphorylation. Therefore, these findings suggest that clasmatodendrosis may be autophagic astroglial death in response to epileptic seizures through TNF-α-mediated p65/RelA-Ser529 phosphorylation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Astrocytes play a role in maintenance of homeostasis in the brain by regulating local ion concentrations, pH, and clearance of neurotransmitters released into the synaptic cleft (Eulenburg and Gomeza 2010). Furthermore, astrocytes release many neuroactive substances, such as chemical transmitters, cytokines, neuropeptides, and growth factors (Ben Achour and Pascual 2010; Theodosis et al. 2008). Xu et al. (2007) have reported that local application of glutamate or GS inhibitors induced glutamate release from astrocytes accompanied by an appearance of large vesicles ex vivo. Thus, they suggest that increase in local glutamate concentration may result in formation and exocytotic fusion of glutamate-containing large vesicles in astrocytes, which play a role in epileptic seizures. However, astroglial vacuolization may also be considered as irreversible coagulative necrotic changes (Sugawara et al. 2002). This fundamental change of irreversibly injured astrocytes was first report by Alzheimer in 1910, which includes extensive swelling and vacuolization of cell bodies and disintegrated and beaded processes, and termed “clasmatodendrosis” by Cajal (Penfield 1928). This irreversible astroglial changes is directly relevant to dysfunction of these cells. Only a few reports on astroglial clasmatodendrosis describe it as an acute astroglial response because of energy failure and acidosis coupled to mitochondrial inhibition in Alzheimer disease and brain ischemia (Friede and van Houten 1961; Kraig and Chesler 1990; Hulse et al. 2001). In contrast to these previous studies describing clasmatodendrosis as an acute phenomenon, we have found hypertrophic vacuolized astroglia in the stratum radiatum of the CA1 region within the rat hippocampus only in the chronic epileptic state, but not in the acute or latent period after status epilepticus (SE) (Kim et al. 2008). Furthermore, we have recently reported that aquaporins (AQPs), water channels, were differently expressed in vacuolized and non-vacuolized astroglia in the hippocampus of chronic epileptic rats (Kim et al. 2009c), and suggested that altered AQP expressions in astroglia might play a role in inhibition of extracellular lactic acidosis induced by repeated spontaneous seizures.
On the other hand, there are two major known routes for clearance of aberrant components in eukaryotic cells: (i) the ubiquitin–proteasomal pathway and (ii) the autophagy–lysosomal pathway. The ubiquitin–proteasomal pathway is responsible for degradation of short-lived proteins and has been studied intensively during the past decades, including in brain injury (Hu et al. 2000; Ciechanover 2006). The autophagy pathway is an important catabolic process involving the removal of long-lived proteins, and organelles by a complex process involving numerous regulatory proteins and the formation of double-membrane vesicles known as autophagosomes, which ultimately fuse with lysosomes for degradation of contents (Rami 2009). Autophagy is involved not only in the balance between protein synthesis and degradation but also in the execution of cell death (Tsujimoto and Shimizu 2005). Autophagy contributes to the mechanism of non-apoptotic programmed cell death, which is known as autophagic cell death (Clarke 1990; Kitanaka and Kuchino 1999). Autophagy starts with the formation of double-membraned cisternae that subsequently engulf cytoplasmic materials or whole organelles to become double-membrane bubble-like vacuoles known as autophagosomes (APs). After maturation, APs merge with lysosomes for bulk degradation of the cargo contents (Yorimitsu and Klionsky 2005). Hence, the appearance of APs under transmission electron microscopy (EM) is a morphologic hallmark unique to autophagy. Interestingly, Tomimoto et al. (1997) reported that clasmatodendritic astroglia included condensed chromatin, lysosomes, and large membrane-bound osmiophilic cytoplasmic inclusions. Thus, they suggested that clasmatodendritic astroglia incorporate edema fluid and phagocytose cellular debris. Recently, Takenouchi et al. (2009) reported that P2X7R activation negatively regulated autophagic flux through the impairment of lysosomal functions. Similarly, we have reported that P2X7R antagonists, such as BBG and OxATP, increased clasmatodendrosis (Kim et al. 2010b). With respect to these previous reports, clasmatodendrosis may be one of autophagy-related degeneration of astrocytes following SE. Although remarkable progress has been made in pathophysiology, molecular events after SE are still incompletely understood. This study was designed to elucidate whether autophagy involves astroglial vacuolization or clasmatodendrosis in the rat hippocampus following SE. The present data show that clasmatodendrosis may be one of the autophagic phenomenas in response to epileptic seizures through tumor necrotic factor-α (TNF-α)-mediated nuclear factor-kappa B (NF-κb) pathway.
Experimental Procedures
Experimental Animals
This study used the progeny of Sprague–Dawley (SD) rats obtained from Experimental Animal Center, Hallym University, Chunchon, South Korea. The animals were provided with a commercial diet and water ad libitum under controlled temperature, humidity, and lighting conditions (22 ± 2°C, 55 ± 5%, and a 12:12 light/dark cycle with lights). Procedures involving animals and their care were conducted in accord with our institutional guidelines that comply with international laws and policies (NIH Guide for the Care and Use of Laboratory Animals 1996).
Seizure Induction and Drug Treatments
Male SD rats (9–11-weeks old) were intraperitoneally treated with pilocarpine (380 mg/kg, Sigma, St. Louis, MO) at 20 min after intraperitoneal administration of atropine methylbromide (5 mg/kg). Following pilocarpine injection, animals showed acute behavioral features of status epilepticus (including akinesia, facial automatisms, limbic seizures consisting of forelimb clonus with rearing, salivation, masticatory jaw movements, and falling). Diazepam (10 mg/kg) was intraperitoneally administered 2 h after onset of SE and repeated, as needed. Age-matched animals (n = 5) were used as normal control.
Intracerebroventricular Drug Infusion
Four weeks after SE, rats were divided into two groups (n = 7, respectively): Saline- or soluble TNF-α p55 receptor (sTNFp55R 50 μg/ml, Sigma)-treated animals. The dosage of each compound was determined as the highest dose that did not affect seizure threshold in the preliminary study. Animals were anesthetized (Zolretil, 50 mg/kg, I.M. Virbac Laboratories, France) and placed in a stereotaxic frames. For the osmotic pump implantation, holes were drilled through the skull for introducing a brain infusion kit 1 (Alzet, USA) into the right lateral ventricle (1 mm posterior; 1.5 mm lateral; −3.5-mm depth; flat skull position with bregma as reference), according to the atlas of Paxinos and Watson (1997). The infusion kit was sealed with dental cement and connected to an osmotic pump (1007D, Alzet, USA). The pump was placed in a subcutaneous pocket in the dorsal region. Animals received 0.5 μl/h of vehicle or compound for 2 weeks (Siuciak et al. 1996; Pencea et al. 2001; Kim et al. 2010a).
Tissue Processing
Six weeks after SE, animals were perfused transcardially with phosphate-buffered saline (PBS) followed by 4% paraformaldehyde in 0.1 M phosphate buffer (PB, pH 7.4) under urethane anesthesia (1.5 g/kg, i.p.). The brains were removed and postfixed in the same fixative for 4 h. The brain tissues were cryoprotected by infiltration with 30% sucrose overnight. Thereafter, the entire hippocampus was frozen and sectioned with a cryostat at 30 μm and consecutive sections were contained in six-well plates containing PBS. For stereological study, every sixth section in the series throughout the entire hippocampus was used in some animals (Kim et al. 2008).
Immunohistochemistry
Sections were first incubated with 3% bovine serum albumin in PBS for 30 min at room temperature. Brain tissues were incubated in mixture of mouse anti-GFAP (diluted 1:200, Chemicon, Temecula, CA, USA)/rabbit anti-LAMP-1 (diluted 1:100, Abcam, Cambridge, MA, USA), rabbit anti-EEA-1 (diluted 1:1,000, Calbiochem, San Diego, CA, USA), rabbit anti-Beclin-1 (diluted 1:2,000, Abcam, Cambridge, MA, USA), rabbit anti-LC3-II (diluted 1:200, Abgent, San Diego, CA, USA), rabbit anti-NF-κB p65-Ser276 (diluted 1:100, Abcam, Cambridge, MA, USA), rabbit anti- NF-κB p65-Ser311 (diluted 1:100, Abcam, Cambridge, MA, USA), rabbit anti- NF-κB p65-Ser529 (diluted 1:100, Abcam, Cambridge, MA, USA), or rabbit anti-TNFp55R (diluted 1:1,000, Abcam, Cambridge, MA, USA) antibody overnight at room temperature. After washing three times for 10 min with PBS, sections were also incubated in a mixture of FITC- or Cy3-conjugated secondary antisera (diluted 1:200, Amersham, San Francisco, CA, USA) for 1 h at room temperature. For nuclei counterstaining, we used Vectashield mounting with DAPI (Vector, Burlingame, CA, USA). For negative control, the rat hippocampal tissues were incubated with only the secondary antibody without primary antibody. All negative controls for immunohistochemistry resulted in the absence of immunoreactivity in any structure (data not shown). All images were captured using an AxioImage M2 microscope and AxioVision Rel. 4.8 software, or Bio-Rad MRC 1024 Confocal Microscope System (Bio-Rad Laboratories, 2000 Alfred Nobel Drive, Hercules, CA 94547, USA). Figures were mounted with Adobe PhotoShop 7.0 (San Jose, CA). Manipulation of the images was restricted to threshold and brightness adjustments to the whole image (Kim et al. 2009a, b, c).
Stereology
The hippocampal volumes (V) were estimated according to the formula based on the modified Cavalieri method: V = Σa × t nom × 1/ssf, where a is the area of the region of the delineated subfield measured by AxioVision Rel. 4.8 software, t nom is the nominal section thickness (of 30 μm in this study), and ssf is the fraction of the sections sampled or section sampling fraction (of 1/6 in this study). The subfield areas were delineated with a ×2.5 objective lens. The volumes are reported as mm3 (Bedi 1991; Madeira et al. 1995). The optical fractionator was used to estimate the cell numbers. The optical fractionator (combination of performing counting with the optical disector, with fractionator sampling) is a stereological method based on a properly designed systematic random sampling method that by definition yields unbiased estimates of population number. The sampling procedure is accomplished by focusing through the depth of the tissue (the optical dissector height, h; of 15 μm in all cases for this study). The number of each cell type (C) in each of the subregions is estimated as: C = ΣQ − × t/h × 1/asf × 1/ssf, where Q − is the number of cells actually counted in the dissectors that fell within the sectional profiles of the subregion seen on the sampled sections, and asf is the areal sampling fraction calculated by the area of the counting frame of the dissector, a(frame) (of 50 × 50 μm2 in this study) and the area associated with each x, y movement, grid (x, y step) (of 250 × 250 μm2 in this study) {asf = [a(frame)/a(x, y step)]}. Cells were counted with a ×40 objective lens. Cells were counted regardless the intensity of labeling. Cell counts were performed by two different investigators who were blind to the classification of tissues (Kim et al. 2008).
Quantification of Data and Statistical Analysis
All data obtained from the quantitative measurements were analyzed using Student’s t-test to determine statistical significance. Wilcoxon rank sum test was used for post-hoc comparisons. A P-value less than 0.05 was considered statistically significant (Kim et al. 2009a, b, c).
Results
Characterization of Clasmatodendrosis
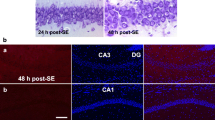
Consistent with our previous studies (Kim et al. 2010a, b), saline-infused animals showed vacuolization of astrocytes only in the stratum radiatum in the CA1 region at 6 weeks after SE (Fig. 1a, b). Vacuolized astrocytes showed clasmatodendrosis that is characterized by round-shaped edematous cell body, short blunt processes, and GFAP tangles in the cytoplasm and nuclear dissolution. Fibrous GFAP-positive fragments surrounded these cells. In addition, vacuoles in clasmatodendritic astrocytes showed LAMP-1 immunoreactivity (Fig. 1a), not EEA-1 immunoreactivity (Fig. 1b). Non-SE-induced animals did not show clasmatodendrosis or reactive gliosis (data not shown). These findings reveal that vacuoles in clasmatodendritic astrocytes are derived from lysosomes, not early endosomes.
Clasmatodendorosis is autophagic astroglial death. (a) Vacuoles in clasmatodendritic astrocytes show LAMP-1 immunoreactivity (arrows). (b) Vacuoles in clasmatodendritic astrocytes do not contain EEA-1 immunoreactivity (arrowheads). (c) Beclin-1 immunoreactivity is observed in the cytoplasm of clasmatodenritic astrocytes (arrows). (d) Vacuoles in clasmatodendritic astrocytes show LC3-II immunoreactivity (arrows). Bar = 12.5 μm
Autophagy is characterized by the formation of double-membraned autophagosomes that fuse with lysosomes to form autolysosomes. Autophagosome formation involves the induction of LC3-II, a marker of early autophagy, expression in autophagosomes (Gozuacik and Kimchi 2004; Codogno and Meijer 2005). In addition, Beclin-1, a Bcl-2-interacting protein, is known to be a promoter of autophagy (Aita et al. 1999, Liang et al. 1999, 2001). Beclin-1 is essential for the formation of autophagosomes (Kihara et al. 2001). Thus, analysis of beclin-1 and LC3-II is the most used method to detect autophagosomes (Kabeya et al. 2000). In this study, both LC3-II and Beclin-1 expression were detected in most of clasmatodendritic astrocytes as well as a few non-vacuolized astrocytes (Fig. 1c, d). These findings indicate that vacuoles in clasmatodendritic astrocytes may be autolysosomes.
p65/RelA-Ser529 NF-κB Phosphorylation in Clasmatodendritic Astrocytes
Nivon et al. (2009) reported that p65/RelA nuclear factor-kappa B (NF-κB) subunit activates autophagy. With respect to this previous study, it is likely that NF-κB signalling may involve in clasmatodendrosis. To confirm this hypothesis, we performed immunohistochemical study for five phospho-p65/RelA NF-κB antibodies. Among them, only p65/RelA-Ser529 phosphorylation was observed in the nuclei of clasmatodendritic astrocytes, which showed watery nuclear dissolution (Fig. 2a–c). These findings indicate that p65/RelA-Ser529 NF-κB phosphorylation may activate autophagy in clasmatodendritic astrocytes. TNF-α is expressed at low levels in the normal brain and it is rapidly upregulated in glia, neurons, and endothelial cells in various pathophysiological conditions, and induces NF-κB phosphorylations (Sriram and O’Callaghan 2007). Therefore, we confirmed the TNFp75 receptor expression in clasmatodendritic astrocytes. Expectedly, TNFp75 receptor expression was strongly observed in clasmatodendritic astrocytes (Fig. 2d). These findings indicate that p65/RelA-Ser529 NF-κB phosphorylation may be mediated by TNF-α.
p65/RelA NF-κB phosphorylation and TNFp75 receptor expression in clasmatodendritic astrocytes. (a) p65/RelA-Ser276 NF-κB phosphorylation. (b) p65/RelA-Ser311 NF-κB phosphorylation. (c) p65/RelA-Ser529 NF-κB phosphorylation. Only p65/RelA-Ser529 NF-κB phosphorylation is observed in the watery nucleus of clasmatodendritic astrocytes (arrow). (d) TNFp75 receptor expression in clasmatodendritic astrocytes (arrows). Bar = 5 (c) and 10 (a, b, and d) μm
Effects of sTNFp55R on Clasmatodendrosis
To directly confirm the role of TNF-α in clasmatodendrosis, we performed the neutralization of TNF-α by sTNFp55R infusion. In saline-infused animals, 51% of astrocytes showed clasmatodendrosis in the stratum radiatum of the CA1 region, accompanied by accumulation of nuclear p65/RelA-Ser529 phosphorylation (Fig. 3a, c). In sTNFp55R-infused animals, 17% of astroglia showed typical clasmatodendrosis with nuclear p65/RelA-Ser529 phosphorylation (Fig. 3b, c, P < 0.05 vs. saline-infused animals). These findings indicate that TNF-α may induce p65/RelA-Ser529 NF-κB phosphorylation resulting in autophagic astroglial death (clasmatodendrosis).
Effect of sTNFp55R infusion on clasmatodendrosis (a, b). As compared with saline-infusion (a, arrows), sTNFp55R infusion reduces the fraction of clasmatodendritic astrocytes in total astrocytes, and accumulation of nuclear p65/RelA-Ser529 phosphorylation (b, arrows). Bar = 15 μm. (c) The quantitative analysis of the fraction of clasmatodendritic astrocytes in the total astrocytes (mean ± SEM). Significant differences from saline-infused animals, *P < 0.05
Discussion
We have recently reported that SE results in caspase-3-independent/AIF-dependent apoptosis-like degeneration of astrocytes in the molecular layer of rat dentate gyrus (Kang et al. 2006; Kim et al. 2008, 2010b, c). SE also induces astroglial death through severe vasogenic edema in the piriform cortex (Kim et al. 2010c). Although both the dentate gyrus and the piriform cortex show acute astroglial loss in responses to SE, the CA1 region shows clasmatodendrosis as the chronic responses to SE (Kim et al. 2010b). Therefore, the vulnerability of astrocytes to SE shows the regional-specific manners like neurons. In addition, it is likely that the underlying mechanism of clasmatodendrosis is relevant to chronic necrotic responses. Indeed, vacuolar degeneration is considered as early stage of necrosis, because these cells often show necrotic features, such as eosinophilic cytoplasm, mitochondrial/nuclear membrane alterations, or TUNEL negativity (Petito et al. 1990; Struys-Ponsar et al. 1994; Tomimoto et al. 1997; Deloncle et al. 2001; Sugawara et al. 2002). Consistent with these previous studies, we have also reported that clasmatodendritic astrocytes in the CA1 region have edematous eosinophilic cytoplasm and large-sized vacuoles at 6 weeks after SE (Kim et al. 2010b). Autophagy is an intracellular pathway that is activated in response to cell stress. It is a phenomenon where the cytoplasmic organelles in the cell are engulfed by double membrane vesicles called the autophagosomes and delivered to the lysosomes where the organelles are broken down by lysosomal proteases and the amino acids recycled back into the cell machinery to aid cell survival (Luo and Rubinsztein 2010). In addition, the autophagic process leads to non-apoptotic programmed cell death, which is known as autophagic cell death (Bursch et al. 1996; Tsujimoto and Shimizu 2005). Indeed, the activation of autophagy induced cell death in a myocardial ischemia and reperfusion model (Matsui et al. 2008), and autophagy also leads to autophagic cell death in a renal ischemia and reperfusion injury (Suzuki et al. 2008). In this study, clamsmatodendritic astrocytes contained autophagic markers (Beclin-1 and LC3-II). Beclin-1 is indicative of autophagy induction that is essential for the formation of autophagosomes (Kihara et al. 2001). In addition, LC3-II is found on the outer and to a lesser extent the inner membrane of the double membrane of the autophagosome. In this study, furthemrore, vacuoles in clasmatodendritic astrocytes contained LAMP-1 immunoreactivity, not EEA-1 immunoreactivity. Furthermore, some vacuoles in clasmatodendritic astrocytes showed LC3-II immunoreactivity. These findings indicate that vacuoles in clasmatodendritic astrocytes may be lysosomes, not early endosomes, and that clasmatodendrosis may be typical autophagic astroglial death.
p65/RelA NF-κB phosphorylation activates autophagy (Nivon et al. 2009). In addition, TNF-α is one of inducer for NF-κB phosphorylations (Sriram and O’Callaghan 2007). However, little data are available to explain the relationship between specific p65/RelA NF-κB subunit phosphorylation and TNF-α in autophagy. In this study, only p65/RelA-Ser529 phosphorylation was observed in the nuclei of clasmatodendritic astrocytes (Fig. 2). Furthermore, sTNFp55R infusion reduced clasmatodendrosis accompanied by reduction in accumulation of nuclear p65/RelA-Ser529 phosphorylation. p65/RelA-Ser529 NF-κB subunit is phosphorylated by casein kinase II (CKII), and enhances nuclear transcriptional activity. Interestingly, activation of P2X7 receptor (a purinergic receptor) attenuates clasmatodendrosis (Kim et al. 2010b), which reduces the DNA-binding activity of NF-κB (Budagian et al. 2003). Taken together, our findings indicate that TNF-α may be one of inducers for autophagic astroglial death through p65/RelA-Ser529 phosphorylation.
In conclusion, p65/RelA-Ser529 phosphorylation induced by TNF-α plays important roles in clasmatodendrosis, an autophagic astroglial death.
References
Aita VM, Liang XH, Murty VV, Pincus DL, Yu W, Cayanis E, Kalachikov S, Gilliam TC, Levine B (1999) Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics 59:59–65
Bedi KS (1991) Early-life undernutrition causes deficits in rat dentate gyrus granule cell number. Experientia 47:1073–1074
Ben Achour S, Pascual O (2010) Glia: the many ways to modulate synaptic plasticity. Neurochem Int 57:440–445
Budagian V, Bulanova E, Brovko L, Orinska Z, Fayad R, Paus R, Bulfone-Paus S (2003) Signaling through P2X7 receptor in human T cells involves p56lck, MAP kinases, and transcription factors AP-1 and NF-kappa B. J Biol Chem 278:549–560
Bursch W, Ellinger A, Kienzl H, Török L, Pandey S, Sikorska M, Walker R, Hermann RS (1996) Active cell death induced by the anti-estrogens tamoxifen and ICI 164 384 in human mammary carcinoma cells (MCF-7) in culture: the role of autophagy. Carcinogenesis 17:1595–1607
Ciechanover A (2006) The ubiquitin proteolytic system: from a vague idea, through basic mechanisms, and onto human diseases and drug targeting. Neurology 66:S7–S19
Clarke PG (1990) Developmental cell death: morphological diversity and multiple mechanisms. Anat Embryol 181:195–213
Codogno P, Meijer A (2005) Autophagy and signaling: their role in cell survival and cell death. Cell Death Differ 12:1509–1518
Deloncle R, Huguet F, Fernandez B, Quellard N, Babin P, Guillard O (2001) Ultrastructural study of rat hippocampus after chronic administration of aluminum l-glutamate: an acceleration of the aging process. Exp Gerontol 36:231–244
Eulenburg V, Gomeza J (2010) Neurotransmitter transporters expressed in glial cells as regulators of synapse function. Brain Res Rev 63:103–112
Friede RL, Van Houten WH (1961) Relations between postmortem alterations and glycolytic metabolism in the brain. Exp Neurol 4:197–204
Gozuacik D, Kimchi A (2004) Autophagy as a cell death and tumor suppressor mechanism. Oncogene 23:2891–2906
Hu BR, Martone ME, Jones YZ, Liu CL (2000) Protein aggregation after transient cerebral ischemia. J Neurosci 20:3191–3199
Hulse RE, Winterfield J, Kunkler PE, Kraig RP (2001) Astrocytic clasmatodendrosis in hippocampal organ culture. Glia 33:169–179
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720–5728
Kang TC, Kim DS, Kwak SE, Kim JE, Won MH, Kim DW, Choi SY, Kwon OS (2006) Epileptogenic roles of astroglial death and regeneration in the dentate gyrus of experimental temporal lobe epilepsy. Glia 54:258–271
Kihara A, Kabeya Y, Ohsumi Y, Yoshimori T (2001) Beclin-phosphatidylinositol 3-kinase complex functions at the trans-Golgi network. EMBO Rep 2:330–335
Kim DS, Kim JE, Kwak SE, Choi KC, Kim DW, Kwon OS, Choi SY, Kang TC (2008) Spatiotemporal characteristics of astroglial death in the rat hippocampo-entorhinal complex following pilocarpine-induced status epilepticus. J Comp Neurol 511:581–598
Kim JE, Kwak SE, Kang TC (2009a) Upregulated TWIK-related acid-sensitive K+ channel-2 in neurons and perivascular astrocytes in the hippocampus of experimental temporal lobe epilepsy. Epilepsia 50:654–663
Kim JE, Kim DW, Kwak SE, Ryu HJ, Yeo SI, Kwon OS, Choi SY, Kang TC (2009b) Pyridoxal-5′-phosphate phosphatase/chronophin inhibits long-term potentiation induction in the rat dentate gyrus. Hippocampus 19:1078–1089
Kim JE, Ryu HJ, Yeo SI, Seo CH, Lee BC, Choi IG, Kim DS, Kang TC (2009c) Differential expressions of aquaporin subtypes in astroglia in the hippocampus of chronic epileptic rats. Neuroscience 163:781–789
Kim JE, Ryu HJ, Yeo SI, Kang TC (2010a) P2X7 receptor regulates leukocyte infiltrations in rat frontoparietal cortex following status epilepticus. J Neuroinflammation 7:65
Kim JE, Ryu HJ, Yeo SI, Kang TC (2010b) P2X7 receptor differentially modulates astroglial apoptosis and clasmatodendrosis in the rat brain following status epilepticus. Hippocampus doi:10.1002/hipo.20850
Kim JE, Yeo SI, Ryu HJ, Kim MJ, Kim DS, Jo SM, Kang TC (2010c) Astroglial loss and edema formation in the rat piriform cortex and hippocampus following pilocarpine-induced status epilepticus. J Comp Neurol 518:4612–4628
Kitanaka C, Kuchino Y (1999) Caspase-independent programmed cell death with necrotic morphology. Cell Death Differ 6:508–515
Kraig RP, Chesler M (1990) Astrocytic acidosis in hyperglycemic and complete ischemia. J Cereb Blood Flow Metab 10:104–114
Liang XH, Jackson S, Seaman M, Brown K, Kempkes B, Hibshoosh H, Levine B (1999) Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature 402:672–676
Liang XH, Yu J, Brown K, Levine B (2001) Beclin 1 contains a leucine-rich nuclear export signal that is required for its autophagy and tumor suppressor function. Cancer Res 61:3443–3449
Luo S, Rubinsztein DC (2010) Apoptosis blocks Beclin 1-dependent autophagosome synthesis: an effect rescued by Bcl-xL. Cell Death Differ 17:268–277
Madeira MD, Sousa N, Santer RM, Paula-Barbosa MM, Gundersen HJ (1995) Age and sex do not affect the volume, cell numbers, or cell size of the suprachiasmatic nucleus of the rat: an unbiased stereological study. J Comp Neurol 361:585–601
Matsui Y, Kyoi S, Takagi H, Hsu CP, Hariharan N, Ago T, Vatner SF, Sadoshima J (2008) Molecular mechanisms and physiological significance of autophagy during myocardial ischemia and reperfusion. Autophagy 4:409–415
Nivon M, Richet E, Codogno P, Arrigo AP, Kretz-Remy C (2009) Autophagy activation by NFkappaB is essential for cell survival after heat shock. Autophagy 5:766–783
Paxinos G, Watson C (1997) The rat brain in stereotaxic coordinates, 3rd edn. Academic, San Diego
Pencea V, Bingaman KD, Wiegand SJ, Luskin MB (2001) Infusion of brain-derived neurotrophic factor into the lateral ventricle of the adult rat leads to new neurons in the parenchyma of the striatum, septum, thalamus, and hypothalamus. J Neurosci 21:6706–6717
Penfield W (1928) Neuroglia and microglia: the interstitial tissue of the central nervous system. In: Cowdry EV (ed) Special cytology, the form and function of the cell in health and disease. Hoeber, New York, pp 1033–1068
Petito CK, Morgello S, Felix JC, Lesser ML (1990) The two patterns of reactive astrocytosis in postischemic rat brain. J Cereb Blood Flow Metab 10:850–859
Rami A (2009) Review: autophagy in neurodegeneration: firefighter and/or incendiarist? Neuropathol Appl Neurobiol 35:449–461
Siuciak JA, Boylan C, Fritsche M, Altar CA, Lindsay RM (1996) BDNF increases monoaminergic activity in rat brain following intracerebroventricular or intraparenchymal administration. Brain Res 710:11–20
Sriram K, O’Callaghan JP (2007) Divergent roles for tumor necrosis factor-alpha in the brain. J Neuroimmune Pharmacol 2:140–153
Struys-Ponsar C, Florence A, Gauthier A, Crichton RR, de Aguilar P (1994) Ultrastructural changes in brain parenchyma during normal aging and in animal models of aging. J Neural Transm Suppl 44:111–132
Sugawara T, Lewén A, Noshita N, Gasche Y, Chan PH (2002) Effects of global ischemia duration on neuronal, astroglial, oligodendroglial, and microglial reactions in the vulnerable hippocampal CA1 subregion in rats. J Neurotrauma 19:85–98
Suzuki C, Isaka Y, Takabatake Y, Tanaka H, Koike M, Shibata M, Uchiyama Y, Takahara S, Imai E (2008) Participation of autophagy in renal ischemia/reperfusion injury. Biochem Biophys Res Commun 368:100–106
Takenouchi T, Nakai M, Iwamaru Y, Sugama S, Tsukimoto M, Fujita M, Wei J, Sekigawa A, Sato M, Kojima S, Kitani H, Hashimoto M (2009) The activation of P2X7 receptor impairs lysosomal functions and stimulates the release of autophagolysosomes in microglial cells. J Immunol 182:2051–2062
Theodosis DT, Poulain DA, Oliet SH (2008) Activity-dependent structural and functional plasticity of astrocyte-neuron interactions. Physiol Rev 88:983–1008
Tomimoto H, Akiguchi I, Wakita H, Suenaga T, Nakamura S, Kimura J (1997) Regressive changes of astroglia in white matter lesions in cerebrovascular disease and Alzheimer’s disease patients. Acta Neuropathol 94:146–152
Tsujimoto Y, Shimizu S (2005) Another way to die: autophagic programmed cell death. Cell Death Differ 2:1528–1534
Xu J, Peng H, Kang N, Zhao Z, Lin JH, Stanton PK, Kang J (2007) Glutamate-induced exocytosis of glutamate from astrocytes. J Biol Chem 282:24185–24197
Yorimitsu T, Klionsky DJ (2005) Autophagy: molecular machinery for self-eating. Cell Death Differ 2:1542–1552
Acknowledgments
This work was supported by a grant No. 2009-0093812 and 2010K000808 from the National Research Foundation of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ryu, H.J., Kim, JE., Yeo, SI. et al. p65/RelA-Ser529 NF-κB Subunit Phosphorylation Induces Autophagic Astroglial Death (Clasmatodendrosis) Following Status Epilepticus. Cell Mol Neurobiol 31, 1071–1078 (2011). https://doi.org/10.1007/s10571-011-9706-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-011-9706-1