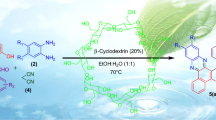
Abstract
An efficient and green method has been developed for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones derivatives by employing 20 mol% β-cyclodextrin via a one-pot multicomponent reaction of aldehydes, malononitrile and phthalhydrazide in H2O–EtOH (4:1) at 100 °C for first time under neutral condition. The catalyst could be recovered and reused for four consecutive cycles without appreciable loss in catalytic activity.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
In recent years, environmental protection laws and global warming effects have impelled the researchers to utilize renewable sources for chemical processes with minimum of waste or zero discharge. Therefore, the development of eco-friendly processes is one of the great challenges for organic chemists. On the other hand, heterocycles containing phthalazine moiety are of interest because they show some pharmacological and biological activities [1–3]. 1H-Pyrazolo[1,2-b]phthalazine-5,10-diones derivatives constitute one of the privileged heterocyclic scaffolds known to exhibit important biological activities such as anticonvulsant [4], cardiotonic [5], vasorelaxant [6], cytotoxic [7, 8], antimicrobial [9], antifungal [10], anticancer [11], and anti-inflammatory activities [12]. In addition, the titled compounds, pyrazolo[1,2-b]phthalazine-diones, were also found to have analgesic, antihypoxic, and anti-pyretic activities [13].
In view of the importance of these heterocycles, number of methods have been developed for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones derivatives including PTSA in 1-butyl-3-methylimidazolium bromide {[bmim]Br} as solvent at 100 °C [14]. Recently, Malek Taher Meghsoodlou and co-workers reported the copper(II) acetate monohydrate as an efficient catalyst for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones derivatives under solvent free condition at 80 °C [15]. Some other methods for synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones has been also reported in the literature involving the use of dicationic 4,4′bipyridinium dichloride ordered mesoporous silica nanocomposite one-pot multicomponent method under solvent free condition [16], triethylamine (0.02 g, 20% mol) as catalyst in EtOH under ultrasonication [17], 1-butyl-3-methylimidazolium hydroxide ([Bmim]OH) under Microwave irradiation [18], CuI nanoparticles as catalyst under solvent-free conditions [19], Indium chloride (InCl3) as catalyst under solvent-free conditions [20], Fe3O4 nanoparticles coated by (3-aminopropyl)-triethoxysilane as catalyst under solvent-free conditions [21], NiCl2 as catalyst in ethanol under refluxing [22], N, N, N′, N′-tetrabromobenzene-1,3-disulfonamide [TBBDA] and poly(N-bromo-N ethylbenzene-1,3-disulfonamide) [PBBS] at 80–100 °C under solvent free conditions [23], Al-KIT-6 reflux in ethanol [24], Nanomagnetic basic catalyst of caesium carbonate supported on hydroxyapatite-coated NiO·5ZnO·5Fe2O4 magnetic nanoparticles (NiO·5ZnO·5Fe2O4@HAP-Cs2CO3) [25], protic ionic liquid at 80 °C [26], NaHCO3 at 120 °C under solvent free condition [27], CAN in solvent PEG-400 [28], and deep eutectic mixture (choline chloride: malonic acid) [29] as a homogeneous catalyst at reflux temperature. These reported methodologies have shown good results in many instances. However, some of synthetic strategies also have limitations in terms of metal catalyst, expensive reagents, long reaction time, environmental hazard, harsh reaction conditions, tedious workup procedure, unsatisfactory yield, and use of homogeneous catalyst which are difficult in separation from reaction mixture and reuse. However, the development of green, mild and simpler procedures to eliminate the use and generation of hazardous substances is the foremost goal of green chemistry.
Cyclodextrins are cyclic oligosaccharides possessing hydrophobic cavity, which binds substrate selectively and catalyze chemical reactions with high selectivity. They catalyze reaction by supramolecular catalysis involving reversible formation of host–guest complexes by noncovalent bonding as seen in enzymes [30, 31]. β-Cyclodextrin is a cyclic heptamer composed of seven glucose units jointed head to tail by α-1,4-links. It is widely accepted that the binding forces involved in the inclusion complex formation are Vander Waals interactions, hydrophobic interactions between guest molecules and β-CD [32]. Because of such behaviour β-CD is used as catalyst for varieties of organic reactions in aqueous medium [33–40]. To the best of our knowledge, the use of β-cyclodextrin for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones in aqueous medium has not been reported.
2 Results and Discussion
In continuation of our work on β-CD and development of newer methodologies [41–50], we disclosed a newer methodology for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones derivatives as one-pot synthesis involving three component reaction of substituted aldehydes, malononitrile, and phthalhydrazide in the presence of β-cyclodextrin as a recyclable catalyst in aqueous media H2O–EtOH (8:2) at 100 °C. The general scheme for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones is depicted in Scheme 1.
The catalytic role of β-CD for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones has been compared with various reported catalysts and found the present methodology developed green protocol for this transformation as well as the results found decent conversion of products within 2.5–5 h with good to excellent yields up to 82–93% and results are summarized in Table 1.
In order to optimize the reaction conditions and test the performance of β-CD as a catalyst for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones, we studied 4-chlorobenzaldehyde, malononitrile, and phthalhydrazide as a model reaction. The reaction proceeds in the absence of β-CD the reaction is sluggish to give lower yields (20%) with longer reaction times at higher temperature. The best result was obtained for 20 mol% of β-CD affording desired product without column chromatographic purification with 93% of yield within 2.5 h. The results indicate that a catalyst plays a critical role in this reaction. Encouraged by the initial success, we applied the optimal protocol to a variety of aldehydes. Generally, all the reactions were performed using 20 mol% of β-CD in H2O–EtOH (4:1) at 100 °C and all the reaction completed within 2.5–5 h to give the desired products in good to excellent yields.
Temperature plays an important role, as surprisingly, at the low temperature there was a formation of trace amounts of product and required longer reaction times. As the temperature increases from room temperature to 110 °C, the yields were found to increase while the reaction time decreases. We obtained the best results at 100 °C (Table 2, entry 5), hence all the reactions were performed at the temperature of 100 °C.
The amount of catalyst was evaluated in the model reaction (Scheme 2) at 100 °C in H2O–EtOH (4:1). The best result was obtained for 20 mol% of β-CD (Table 3, entry 5), affording 93% of yield within 2.5 h and the use of excess catalyst had no impact either on the rate of reaction or on the yield of product (Table 3, entries 6 and 7).
Solvents also play an important role in the studied transformation; we carried out the above reaction in various solvents in order to check catalytic response of β-CD. As shown in Table 4, when the reaction was carried out under solvent free conditions, the targeted product was obtained with very low yields. To find the best solvent for this transformation, the present one-pot three component reaction was screened in H2O, CH3CN, THF, DMF, EtOH, H2O–EtOH (1:1), H2O–EtOH (4:1) mixture. Among all these solvents, H2O–EtOH (4:1) was found to play an effective role in this transformation affording highest yields (Table 4, entry 8). Therefore, H2O–EtOH (4:1) was selected as the solvent system for this transformation.
After optimizing the reaction conditions, we extended this process to other substrates. The scope of the reactions is illustrated with respect to various aldehydes and the results are summarized in Table 5, entries 5a–5o. Both electron-rich and electron-deficient aromatic aldehydes such as 4-methyl, 4-cyano, and 4-nitro, 4-chlorobenzaldehydes reacted efficiently with phthalhydrazide and malononitrile to furnish the corresponding 1H-pyrazolo[1,2-b]phthalazine-5,10-diones derivatives in good yields (entries 5d, 5f, 5h and 5n, Table 5). The substituents present on the aromatic ring had shown some effect on the conversion. In all cases, the conversion was completed within 2.5–5 h with good to excellent yields of desired products without forming any by-products. The nature of functional group on the aromatic ring of aldehyde exerted a slight influence on the reaction time. The rate of reaction decreases in the case of arylaldehyde carrying an electron donating group in comparison to the unsubstituted. Similarly, hetero aromatic aldehydes like Thiophene-2-carboxaldehyde and Pyridine-3- carboxaldehyde reacted well to furnish the corresponding 1H-pyrazolo[1,2-b]phthalazine-5,10-diones in good yield (entry 5l and 5m, Table 5).
Table 5 Synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones in the presence of β-CD in H2O–EtOH (4:1) at 100 °C.
Inspired by the above results, we synthesized 1,1′-(1,3-phenylene)bis(3-amino-5,10-dioxo-5,10-dihydro-1H-pyrazolo[1,2b]phthalazine-2-carbonitrile) derivative from isopthalaldehyde, malononitrile and phthalhydrazide at the same reaction conditions (Scheme 3). The compound was characterized by 1H-NMR, 13C-NMR, Mass and the results obtained are summarized in Table 5, entry 5o.
Next, the green and sustainability factors such as atom economy and E-factor of present method were calculated for the model reaction between 4-chlorobenzaldehyde, malononitrile, and phthalhydrazide to afford the 3-amino-1-(4-chlorophenyl)-5,10-dioxo-5,10-dihydro-1-H pyrazolo[1,2-b]phthalazine-2-carbonitrile (Table 5, entry 5d). The results indicate that the present method follows green chemistry principles with smaller E-factor as 0.13 and high atom economy (AE) as 91% (see the Supporting Information for calculations).
To account for the very efficient catalysis by β-CD of this multicomponent reaction, wherein supramolecular catalyzed reactions are involved, it is proposed that β-CD with its seven free primary –OH groups acting synergistically behaves as an efficient host and supramolecular catalyst Fig. 1. In the first step, the aldehyde binds to the β-CD cavity. Activation of a proton from malononitrile by β-CD catalyzes its Knoevenagel condensation with the carbonyl group to give the ylidenemalononitrile(I). The co-operative enzyme-like binding of these intermediates which ensure their tighter fit into the cavity facilitates further reactions, namely Michael addition of phthalhydrazide(II) to ylidenemalononitrile (by activation and of a proton from phthalhydrazide), its undergo cyclization to form desire product.
1H NMR study of the complex: to gain further evidence we propose a supramolecular catalysis for the formation of pyranopyrazole product by inclusion complex of phthalhydrazide β-CD and 4-cyanobenzaldehyde Fig. 2. The formation of inclusion complex can be explained by 1H NMR studies. The 1H NMR spectra of the catalyst β-CD and its inclusion complex with one of the reactant 4-cyanobenzaldehyde is recorded in D2O and are given in Fig. 3 and Table 6, it is clear that there is upfield shift from 3.8959 to 3.8482 (0.0477) of H3 and 3.7886–3.7204 (0.0682) of H5 protons of β-CD-4-cyanobenzaldehyde complex compared to β-CD, indicating the complex formation. These observations clearly demonstrate that the aldehyde is ideally located for the condensation with malononitrile and phthalhydrazide in the CD cavity. On the basis of the above results and previous studies [51, 52], we suggest that the internal hydrophobic cavity of β-CD forms inclusion complex with aldehyde more effectively and enhances the rate of reaction with increase in the yield of product and decreases the reaction time.
2.1 General Procedure for Synthesis of 1H-Pyrazolo[1,2-b]phthalazine-5,10-diones
The mixture of carbonyl compound aldehyde (2 mmol), phthalhydrazide (2 mmol) and malononitrile (2 mmol) were added in β-cyclodextrin (0.4 mmol) solution containing H2O–EtOH (4:1, 10 mL). The resulting mixture was stirred at 100 °C. After completion of the reaction (monitored by TLC), the reaction mixture was poured into 20 mL water. Filtered the product and washed with hot water. The crude product was recrystallized with 30% aqueous ethanol to afford the desired product. The filtered aqueous layer was cooled at 5 °C to recovery of β-CD by filtration. The recovered β-CD was reused for 3–4 consecutive runs in this reaction without any significant loss in yield and activity.
3 Catalyst Recovery
The catalyst recovery and reusability were studied by four cycles including the use of fresh catalyst for the synthesis of 3-amino-1-(4-chlorophenyl)-5,10-dioxo-5,10-dihydro-1H-pyrazolo[1,2-b]phthalazine-2-carbonitrile (Table 5, entry 5d). In every cycle, the catalyst was almost quantitatively recovered and after third and fourth use of catalyst decreasing yield is not much more significant which is shown in Fig. 4.
4 Conclusion
In summary, we have developed a one-pot, three-component ecofriendly protocol for the synthesis of 1H-pyrazolo[1,2-b]phthalazine-5,10-diones derivatives catalyzed by supramolecular, biodegradable and reusable catalyst β-cyclodextrin. The salient features of the present methods are environmentally benign [low E-factor (0.13) and high atom economy (AE) = 91%], mild neutral reaction conditions, good to excellent yields of products, simple and clean workup of the desired product without column chromatography and easy recovery as well as reuse of the catalyst.
References
Al-Assar F, Zelenin KN, Lesiovskaya EE, Bezhan IP, Chahchir BA (2002) Pharma Chem J 36:598–603
Jain RP, Vederas JC (2004) Bioorg Med Chem Lett 14:3655–3658
Carling RW, Moore KW, Street LJ, Wild D, Isted C, Lesson PD, Thomas SO, cooner D, Mckernan RM, Quirk K, Cook SM, Atach JR, Waftord KA, Thompson SA, Dawson GR, Ferris P, Castro JL (2004) J Med Chem 47:1807–1822
Grasso S, DeSarro G, Micale N, Zappala M, Puia G, Baraldi M, Demicheli C, (2000) J Med Chem 43:2851–2859
Nomoto Y, Obase H, Takai H, Teranishi M, Nakamura J, Kubo K (1990) Chem Pharm Bull 38:2179–2183
Watanabe N, Kabasawa Y, Takase Y, Matsukura M, Miyazaki K, Ishihara H, Kodama K, Adachi H (1998) J Med Chem 41:3367–3372
Chebanov VA, Muravyova EA, Desenko SM, Musatov VI, Knyazeva IV, Shishkina SV, Shishkina OV, Kappe CO (2006) J Comb Chem 8:427–434
Dondoni A, Massi A, Sabbatini S, Bertolasi V (2002) J Org Chem 67:6979–6994
Liu JN, Li J, Zhang L, Song LP, Zhang M, Cao WG, Zhu SZ, Deng HG, Shao M (2012)Tetrahedron Lett 53:2469–2472
Kim JS, Rhee HK, Park HJ, Lee SK, Lee CO, Park Choo H-Y (2008) Bioorg Med Chem 16:4545–4550
El-Sakka SS, Soliman AH, Imam AM (2009) Afinidad 66:167
Ryu CK, Park RE, Ma MY, Nho JH (2007) Bioorg Med Chem Lett 17:2577–2580
Li J, Zhao YF, Yuan XY, Xu JX, Gong P (2006) Molecules 11:574–582
Ghahremanzadeh R, Shakibaei GI, Bazgir A (2008) Synlett 8:1129–1132
Mohamadpour F, Maghsoodlou MT, Heydari R, Lashkari M (2016) Res Chem Intermed 42:7841–7853
Bashti A, Ali Reza Kiasat AR, Mokhtari B (2015) RSC Adv 5:25816–25823
Nabid MR, Tabatabaie SJ, Gahremanzadeh R, Bazgir A (2010) Ultrason Sonochem 17:159–161
Raghuvanshi DS, Singh KN (2011) Tetrahedron Lett 52:5702–5705
Safaei-Ghomi J, Shahbazi-Alavi H, Ziarati A, Teymuri R, Saberi MR (2014) Chin Chem Lett 25:401–405
Reddy MV, Tae Jeong Y (2013) Tetrahedron Lett 54:3546–3549
Shaterian HR, Mohammadnia M (2014) Res Chem Intermed 40:371–383
Shi-Hua Song, Jun Zhong, Yan-Hong He, Zhi Guan (2012) Tetrahedron Lett 53:7075–7077
Ghorbani-Vaghei R, Noori S, Toghraei-Semiromi Z, Salimi Z (2014) RSC Adv 4:47925–47928
Karthikeyan G, Pandurangan A (2012) J Mol Catal A 361–362:58–67
Maleki B, Nam Chalaki SB, Ashrafi SS, Seresht ER, Moeinpourb F, Khojastehnezhad A, Tayebee R (2015) Appl Organometal Chem 29:290–295
Mulik AG, Chandam DR, Patil DR, Patil PP, Mulik GN, Salunkhe ST, Deshmukh MB (2015) Res Chem Intermed 41:10085–10096
Vafaee A, Davoodnia A, Pordel M, Bozorgmehr MR (2015) Oriental J Chem 31(4):2153–2158
Kidwai M, Chauhan R (2014) J Hetero Chem 51:1689–1696
Chaskar A (2014) Curr Catal 3(3):266–273
Breslowong R, Dong SD (1998) Chem Rev 98:1997–2011
Desper JM, Breslow R (1994) J Am Chem Soc 116:12081–12082
Szejtli J (1998) Chem Rev 98:1743–1754
Rai P, Srivastava M, Yadav S, Singh J, Jagdamba S (2015) Catal Lett 145:2020–2028
Kumar A, Tripathi VD, Kumar P (2011) Green Chem 13:51–54
Kumar A, Shukla RD (2015) Green Chem 17:848–851
Kumar S, Ahmed N (2016) Green Chem 18:648–656
Azath IA, Puthiaraj P, Pitchumani K (2013) ACS Sustain Chem Eng 1:174–179
Sukumari S, Azath IA, Pitchumani K (2012) Synlett 23:2328–2332
Kanagaraj K, Pitchumani K (2013) J Org Chem 78:744–751
Londhe BS, Pratap UR, Mali JR, Mane RA (2010) Bull Korean Chem Soc 31(8):2329–2332
Tayade YA, Patil DR, Wagh YB, Jangle AD, Dalal DS (2015) Tetrahedron Lett 56:666–673
Patil DR, Wagh YB, Ingole PG, Singh K, Dalal DS (2013) New J Chem 37:3261–3266
Deshmukh MB, Wagh ND, Sikder AK, Borse AU, Dalal DS 1014 Ind Eng Chem Res 53:19375–19379
Patil DR, Dalal DS (2012) Chin Chem Lett 23:1125–1128
Patil DR, Ingole PG, Singh K, Dalal DS (2013) J Incl Phenom Macro Chem 76:327–332
Wagh YB, Kuwar A, Patil DR, Tayade YA, Jangale AD, Terdale SS, Trivedi DR, Gallucci J, Dalal DS (2015) Ind Eng Chem Res 54:9675–9682
Wagh YB, Tayade YA, Padvi SA, Patil BS, Patil NB, Dalal DS (2015) Chin Chem Lett 26:1273–1277
Jangale AD, Kumavat PP, Wagh YB, Tayade YA, Mahulikar PP, Dalal DS (2015) Synth Commun 45:376–385
Dalal KS, Tayade YA, Wagh YB, Trivedi DR, Dalal DS, Chaudhari BL (2016) RSC Adv 6:14868–14879
Padvi SA, Tayade YA, Wagh YB, Dalal DS (2016) Chin Chem Lett 27:714–720
Tayade YA, Padvi SA, Wagh YB, Dalal DS (2015) Tetrahedron Lett 56:2441–2447
Kokkirala S, Murthy SN, Venkata D, Nageswar YVD (2011) Eur J Chem 2:272–275
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tayade, Y.A., Dalal, D.S. β-Cyclodextrin as a Supramolecular Catalyst for the Synthesis of 1H-Pyrazolo[1,2-b]phthalazine-5,10-dione Derivatives in Water. Catal Lett 147, 1411–1421 (2017). https://doi.org/10.1007/s10562-017-2032-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-017-2032-6