Abstract
Phosphotungstic acid (PTA) is used an efficient catalyst for the von Pechmann condensation reaction of phenols and β-keto esters under solvent-free conditions. This method was compared with those of the reactions in the different solvents and catalysts. The methodology presented offers significant improvements for the synthesis of coumarins with regard to yield of products, simplicity in operation and green aspects by avoiding toxic conventional catalysts and solvents.
Graphical Abstract
Phosphotungstic acid (PTA) is used an efficient catalyst for the von Pechmann condensation reaction of phenols and β-keto esters under solvent-free conditions. This method was compared with those of the reactions in the different solvents and catalysts. The methodology presented offers significant improvements for the synthesis of coumarins with regard to yield of products, simplicity in operation and green aspects by avoiding toxic conventional catalysts and solvents.

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The application of clean catalytic technologies, especially those with the use of heterogeneous catalysts, is becoming increasingly important for the development of environmentally benign chemical processes [1]. In this context, catalysis by acids can be considered as one of the most important areas of catalysis. Although the acid catalysis is widely employed in chemical industry, it suffers from the traditional use of hazardous mineral acids resulting in pollution and corrosion problems [1, 2]. Heteropoly compounds are green catalysts that function in a variety of reaction fields and are efficient bifunctional catalysts, harmless to the environment with respect to corrosiveness, safety, quantity of waste and separability [3]. Among the Keggin-type Heteropolyacids are more active and possess stronger Bronsted acidity than the usual mineral acids such as H2SO4, HCl, HNO3 [4] and conventional solid acids such as SiO2–Al2O3, H3PO4–SiO2, zeolites including HX, HY, H-ZSM-5, Amberlyst-15 and Nafion-H [5]. Among heteropoly acids, phosphotungstic acids are the most widely used catalysts [6, 7] owing to their high acid strength, thermal stabilities, and low reducibilities.
The synthesis of coumarin and their derivatives has attracted considerable attention from organic and medicinal chemists for many years as a large number of natural products contain this heterocyclic nucleus. They are widely used as additives in food, perfumes, agrochemicals, cosmetics, pharmaceuticals [8] and in the preparations of insecticides, optical brightening agents, dispersed fluorescent and tunable laser dye [9]. Coumarin and its derivatives have varied bioactivitives such as antimicrobial [10], antithrombotics [11], anticoagulants, antipsoriasis activity [12], anticancer [13], anti-HIV [14], antioxidant activity, antiproliferative activity [15], inhibitory activity on viral proteases [16], estrogen-like effects [17] and central nervous system modulating activities [18]. Coumarins also act as intermediates for the synthesis of furocoumarins, chromenes, coumarones and 2-acylresorcinols [19]. Thus, the synthesis of this heterocyclic nucleus is of much current interest.
Coumarins have been synthesized by several routes including Pechmann [20], Perkin [21], Knoevenagel [22], Reformatsky [23], Wittig reactions [24], and by flash vacuum pyrolysis [25]. Among these, the Pechmann reaction is the most widely used method, as the reaction involves the use of simple starting materials, that is, phenols and β-ketoesters, in the presence of acidic condensing agents. The use of various reagents such as H2SO4, P2O5, FeCl3, ZnCl2, POCl3, AlCl3, PPA, HCl, phosphoric acid, trifluoroacetic acid, montmorillonite and other clays are all well documented in the literature [26]. Most of these methods suffer from severe drawbacks including the use of a large amount of catalysts, sometimes long reaction times and very often temperatures to the extent of 150 °C. Some of the recent achievements in the efficient construction of this nucleus include the development of cation exchange resins [27], several solid acid catalysts and metal nitrates [28], heteropolyacids [29, 30] supported polyaniline catalysts, the use of microwave irradiation, the use of ionic liquids as efficient catalysts [31] and very recently the use of Pt-catalysed hydroarylation of propiolic acids with phenols [32].
The drive towards clean technology has encouraged the application of solvent-free conditions [33]. A move away from the use of solvents in organic synthesis has led in some cases to improved results and more benign synthetic procedures. Our approach reduces the use of organic solvents, which are potentially toxic, hazardous and uses simple and mild conditions, with inherently lower costs. Owing the importance of PTA and in continuation with our work on to develop environmentally friendly reactions [34], herein, we report an simple, efficient and high yielding protocol for the synthesis of coumarin derivatives using phosphotungstic acid as a catalyst under solvent-free conditions (Scheme 1).
2 Experimental
The melting points of the products were determined by open capillaries on a Buchi apparatus and are uncorrected. The IR spectra were recorded on a Nicolet Impact-410 FT-IR Spectrophotometer using KBr pellets. 1H NMR and 13C NMR spectra were recorded on a Buckner AC-300F 300 MHz spectrometer in CDCl3 using TMS as an internal standard with 1H resonant frequency of 300 MHz and 13C resonant frequency of 75 MHz. The Mass spectra were recorded on an Autospec EI-MS. The elemental analysis was carried out by using Heraus CHN rapid analyzer. All the compounds gave C, H and N analysis within ±0.4% of the theoretical values. The homogeneity of the compounds was described by TLC on aluminum silica gel 60 F254 (Merck) detected by UV light (254 nm) and iodine vapours.
2.1 Materials
Phosphotungstic acid, H3PW12O40 (PTA hydrate), phenols and β-keto ester, were procured from Aldrich, India Ltd. Solvents was procured from M/s Loba Chemie, Mumbai with >99.9 purity.
2.2 General Experimental Procedure for the Synthesis of Coumarins
The mixture of phenols (1 mmol) and the β-keto ester (1 mmol) and Phosphotungstic acid (2 mol%) was refluxed at 90 °C with stirring for the indicated time (Table 4), and reaction monitored by TLC. After completion of reaction, the mixture got solidified within hour. The resulting solidified mixture was diluted with ethyl acetate (1 ml) and the catalyst was separated through a Büchner funnel. The filtrate obtained was washed with water (two times) and evaporated the solvent under reduced pressure yielded the crude product, which was purified by recrystallization. All synthesized coumarin derivatives were characterized using analytical techniques like IR, 1H NMR, 13C NMR and mass spectroscopy. Also the melting points were measured for all synthesized coumarin derivatives and were compared with the corresponding reported melting points.
2.3 The Spectral Data for Selected Products
2.3.1 4-Methyl Coumarin (1a)
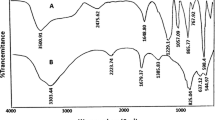
Colorless solid, m. p. 84–85, IR (KBr): υ 2,973, 1,685, 1,607, 1,460 cm−1; 1H NMR (300 MHz, CDCl3) δ ppm: 2.70 (s, 3H, CH3), 6.52 (s, 1H, C = CH), 7.20–7.7 (m, 4H, Ar–H); 13C NMR (300 MHz, CDCl3) δ ppm 26.7, 113.4, 120.3, 125.7, 128.1, 152.4, 156.1,169.0; ESI-MS: 160 (M+); Anal. Calcd for C10H8O2: C, 74.99; H, 5.04; Found C, 74.80; H, 5.10.
2.3.2 4,7-Dimethyl Coumarin (1b)
Colorless solid, m. p. 131–132, IR (KBr): υ 2,973, 1,686, 1,607, 1,460 cm−1; 1H NMR (300 MHz, CDCl3) δ ppm: 2.64 (s, 3H, CH3), 2.82 (s, 3H, CH3), 6.43 (s, 1H, C = CH), 7.0–7.70 (m, 3H, Ar–H), OH not observd; 13C NMR (300 MHz, CDCl3) δ ppm 20.1, 23.8, 104.3, 120.0, 126.7, 135.9, 150.9, 167.0; ESI-MS: 174 (M+); Anal. Calcd for C11H10O2: C, 75.84; H, 5.79; Found C, 75.87; H, 5.74.
2.3.3 7-Hydroxy–4-Methyl Coumarin (1d)
Colorless solid, m. p. 186–187, IR (KBr): υ 3,467, 2,973, 1,690, 1,607, 1,460 cm−1; 1H NMR (300 MHz, CDCl3) δ ppm: 2.58 (s, 3H, CH3), 6.37 (s, 1H, C = CH), 6.8–7.78 (m, 3H, Ar–H), OH not observd; 13C NMR (300 MHz, CDCl3) δ ppm 23.6, 106.7, 109.7, 121.0, 129.0, 157.4, 159.2,172.0; ESI-MS: 185 (M+); Anal. Calcd. for C11H8O3: C, 68.18; H, 4.58; Found C, 68.22; H, 4.54.
2.3.4 5,7-Dihydroxy–4-Methyl Coumarin (1g)
Colorless solid, m. p. 283–284, IR (KBr): υ 3,467, 3,160, 1,670, 1,607, 1,460 cm−1; 1H NMR (300 MHz, DMSO) δ ppm: 2.60 (s, 3H, CH3), 6.27 (d, 1H, Ar–H), 6.32 (d, 1H, Ar–H),6.40 (s, 1H, C = CH), OH not observd; 13C NMR (300 MHz, DMSO) δ ppm 23.9, 100.7, 108.8, 153.2, 156.2, 158.1,166.0; ESI-MS: 192 (M+); Anal. Calcd. for C11H8O4: C, 62.50; H, 4.21; Found C, 63.78; H, 4.31.
2.3.5 7-Hydroxy–4,8-Dimethyl Coumarin (1k)
Colorless solid, m. p. 217–218, IR (KBr): υ 3,460, 3,148, 1,685, 1,607, 1,460 cm−1; 1H NMR (300 MHz, DMSO) δ ppm: 1.90 (s, 3H, CH3), 2.47 (s, 3H, CH3), 6.36 (s, 1H, C = CH), 6.57 (d, 1H, Ar–H), 7.12 (d, 1H, Ar–H), OH not observd; 13C NMR (300 MHz, DMSO) δ ppm 14.3, 24.6, 109.7, 112.8, 119.2, 125.0, 152.2, 155.2, 169.0; ESI-MS: 190 (M+); Anal. Calcd. for C11H10O3: C, 69.45; H, 5.35; Found C, 69.37; H, 5.40.
3 Results and Discussion
First, the optimization of the reaction, the effect of solvent, temperature and catalyst were carried, by selecting phenol and ethyl acetoacetate as a model. After stirring for 2–3 h, reaction did not proceed as monitored by TLC at room temperature in solvent-free conditions. Subsequently, the mixture was heated to reflux at different temperatures ranging from 60 to 120 °C, with an increment of 10 °C each time. The yield of product 1a was increased and the reaction was raised from 60 to 90 °C (Table 1, entries 1–4). However, no significant increase in the yield of product 1a was observed as the reaction temperature was raised from 100 to 120 °C (Table 1, entries 5–7). Therefore, 90 °C was chosen as the reaction temperature for all further reactions.
To evaluate the effect of catalyst concentration, Pechmann condensation of phenol and ethyl acetoacetate in equimolar ratio (1:1) was carried out in presence of different amounts of catalyst (1, 2, 5, 10 mol%) at 90 °C under solvent-free conditions and the isolated yields of the product were shown in Table 2. From this we concluded that 2 mol% of PTA to be optimum amount of the catalyst for this reaction. Use of higher amount of catalysts (5 and 10 mol%) neither improves the yield nor reaction time further.
After optimizing the temperature and amount of catalyst, the reaction is carried out in solvents like toluene, THF, acetonitrile 1,4-dioxane and also without solvent. It is observed that, reaction in solvents takes more time and also the yield are low compared to the solvent-free condition (Table 3). Considering the importance of green chemistry, the solvent-free reaction conditions are the advantageous aspect of the present method, since it avoids the use of environmental hazardous and toxic solvents. The efficiency of PTA catalyst was demonstrated by synthesizing the range of coumarins using series of monohydric and polyhydric phenols and ethyl acetoacetate (Table 4). The catalyst was found to be equally effective for phenols bearing either electron-donating or electron-withdrawing substituents.
Furthermore, in order to show the excellent catalytic activity of the catalyst, we carried out the synthesis 7-hydroxy-4-methyl coumarin (entry 1d in Table 4) catalyzed by other several catalyst under the same reaction conditions (Table 5). It shows that the yield of the desired product in the presence of PTA is higher than that in presence of other catalyst. From these results we concluded that, the present method was superior to reported methods regarding yields and reaction time. Also, the work-up of present method was easy and it includes the pouring of reaction mixture on ice-water to precipitate the solid, which could be collected by filtration to give the corresponding coumarin product with better yield. All the synthesized products were characterized by elemental analysis, mp, IR, 1H NMR, 13NMR and MS spectra.
3.1 Regeneration of Catalyst
To examine the reusability, the catalyst recovered by filtration from the reaction mixture phenol and ethylacetoacetate after dilution with ethyl acetate was reused as such for subsequent experiments (up to four cycles) under similar reaction conditions. The observed fact that yields of the product remained comparable in these experiments (Table 6 ), established the recyclability and reusability of the catalyst without significant loss of activity.
4 Conclusions
In conclusion, we have developed an efficient, facile and environmentally acceptable synthetic methodology for the synthesis of coumarin derivatives using phosphotungstic acid as a catalyst under solvent-free condition. The attractive features of this procedure are the mild reaction conditions, high conversions, ease separation and recyclability of the catalyst, inexpensive and environmentally friendly catalyst, excellent yields, all of which make it a useful and attractive strategy for the preparation of various coumarin derivatives simply by changing different substrates.
References
Clark JH (2001) Pure Appl Chem 73:103
Kozhevnikov IV (2002) Catalysts for fine chemicals, catalysis by polyoxometalates, vol 2. Wiley, Chichester
Misono M, Ono I, Koyano G, Aoshima A (2000) Pure Appl Chem 72:1305
Misono M, Mizuno N, Okuhara T (1996) Adv Catal 41:113
Misono M, Mizuno N (1998) Chem Rev 98:199
Chen X, She J, Shang ZC, Wu J, Wu HF, Zhang PZ (2008) Synthesis 3478
Chen X, She J, Shang ZC, Wu J, Zhang PZ (2008) Synthesis 3931
O’Kennedy R, Thornes RD (1997) Coumarins: biology applications and mode of action. Wiley, Chichester
Zabradnik M (1992) The production and application of fluorescent brightening agents. Wiley, New York
Costova IN, Nikolov NM, Chipilska LN (1993) J Ethnopharm 39:205
Mitra AK, De A, Karchaudhuri N, Misra SK, Mukhopadhyay AK (1998) J Indian Chem Soc 75:666
Bravic G, Gaultier J, Hauw CCR (1968) Acad Sci Paris 267:1790
Wang CJ, Hsieh YJ, Chu CY, Lin YL, Tseng TH (2002) Cancer Lett 183:163
Palmer CJ, Josephs JL (1995) J Chem Soc Perkin Trans 1:3135
Taniguchi M, Xiao YQ, Liu XH, Yabu A, Hada Y, Guo LQ, Yamazoe Y, Baba K (1999) Chem Pharm Bull 47:713
Nettleton DE (1996) Drugs Future 34:1257
Jacquot Y, Rojaz C, Refouvelet B, Robert JF, Leclercq G, Xicluna A (2003) Mini-Rev Med Chem 3:387
Noeldner M, Hauer H, Chatterjee SS (1996) Drugs Future 21:779
Sethna SM, Kong NP (1945) Chem Rev 36:1
Von Pechmann H, Duisberg C (1884) Chem Ber 17:929
Johnson JR (1942) Org React 1:210
Brufola G, Fringuelli F, Piermatti O, Pizzo F (1996) Heterocycles 43:1257
Shirner RL (1942) Org React 1:1
Yavari I, Hekmat-Shoar R, Zonouzi A (1998) Tetrahedron Lett 39:2391
Cartwright GA, McNab W (1997) J Chem Res (S) 296
Li TS, Zhang ZH, Yang F, Fu CG (1998) J Chem Res (S) 38
John EVO, Israelstam SS (1961) J Org Chem 26:240
Reddy BM, Patil MK, Lakshmann P (2006) J Mol Catal A 256:290
Torviso R, Mansilla D, Belizán A, Alesso E, Moltrasio G, Vázquez P, Pizzio L, Blanco M, Cáceres C (2008) Appl Catal A: Gen 339:53
Romanelli GP, Bennaedi D, Ruiz DM, Baronetti G, Thomas HJ, Autino JC (2004) Tetrahedron Lett 45:8935
Singh V, Kaur S, Sapehiyia V, Singh J, Kad GL (2005) Catal Commun 6:57
Oyamada J, Kitamura T (2006) Tetrahedron 62:6918
Dittmer DC (1997) Chem Ind 779
Hosamani KM, Hiremath VB, Keri RS, Harisha RS, Hallagudi SB (2008) Can J Chem 86(11):1030
Valizadeh H, Shockravi A (2005) Tetrahedron Lett 46:3501
Singh V, Singh J, Kaur KP, Kad GL (1997) J Chem Res 58
Potdar MK, Mohile SS, Salunkhe MM (2001) Tetrahedron Lett 42:9285
Gibbs SA, De SK (2005) Synthesis 1231
Acknowledgments
This research work is financially supported by the Department of Science & Technology (DST), New Delhi-110 016. (Ref. No SR/S1/OC-08/2005 dated 05-09-2005). Dr. K. M. Hosamani is highly indebted to the British Commonwealth Scholarship Commission, UK (Ref. No. INCF-2008-68).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keri, R.S., Hosamani, K.M. & Seetharama Reddy, H.R. A Solvent-Free Synthesis of Coumarins Using Phosphotungstic Acid as Catalyst. Catal Lett 131, 321–327 (2009). https://doi.org/10.1007/s10562-009-9940-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-009-9940-z