Abstract
Purpose
Triple-negative breast cancers (TNBC) are aggressive tumours that exhibit abundant lymphoid infiltrates which modulate tumour behaviour. Recent findings suggest that TNBC with higher densities of plasma cells are associated with a favourable prognosis, and tertiary lymphoid structures (TLS) have prognostic significance. Here, we studied the phenotype and function of plasma cells in TNBCs by assessing their association with IgG Kappa light chain expression, B cells, and TLS.
Methods
A retrospective analysis of 269 TNBC cases was performed. Tumour-infiltrating CD38+ plasma cells, CD20+ B cells, and TLS were evaluated on conventional haematoxylin–eosin-stained and immunohistochemical-stained sections of TNBC. We then selected TNBC cases demonstrating the highest and lowest densities of plasma cells, and examined their association with TLS, B cells, as well as immunoglobulin expression using Opal-Vectra multiplex immunofluorescence (IF).
Results
TNBC with high density of plasma cells showed significantly higher numbers of IgG Kappa+ CD38+ cells (p = 0.0089, p < 0.0001), and higher numbers of TLS (p < 0.0001), compared to TNBC with low density of plasma cells. TNBC with high density of plasma cells also showed higher numbers of CD20+ B cells in the tumour core (p < 0.0001), invasive margin (p < 0.0001), as well as stromal (p = 0.015) compartments.
Conclusion
TNBC with high density of plasma cells are associated with higher numbers of IgG Kappa+ CD38+ cells, CD20+ B cells, and TLS. Further studies to characterize the function of plasma cell infiltrates and how they may interact with other tumour-infiltrating lymphocytes and TLS in TNBC may help improve existing immunotherapy strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Triple-negative breast cancers (TNBC), defined by the absence of oestrogen receptor (ER), progesterone receptor (PR) and c-erbB2 (HER2) receptors, constitute 11% to 15% of breast cancers [1,2,3]. Among the molecular subtypes of breast cancer, TNBC are associated with higher histologic grade, higher incidence of metastases and recurrence, poorer prognosis, and poor response to hormonal or targeted therapy [4,5,6]. Immune cells within cancers, also known as Tumour-Infiltrating Lymphocytes (TIL), influence tumour infiltration, progression, and metastasis [7]. TNBC is an immunogenic tumour exhibiting abundant lymphoid cell infiltrates. Different immunophenotypes of TIL predict favourable responses to chemotherapy and are variably correlated with better or worse clinical prognosis in TNBC [8,9,10,11,12,13,14]. The majority of studies on TILs in TNBC have focused largely on the role of T cells. Fewer studies have investigated the role of B cells, although it is known that the humoral immune system is involved in tumour regression [15, 16]. With a growing body of evidence demonstrating successful monoclonal antibody-based cancer immunotherapies [17,18,19], further study into the role of the humoral immune system in TNBC is imperative to develop novel therapeutic strategies.
Tumour-associated B cells are present and activated in one quarter of breast tumours [20,21,22]. Compared to other breast cancer subtypes, TNBC show significantly elevated numbers of CD20+ B cells [23]. Plasma cells, which are specialized antibody-secreting, terminally differentiated CD38+ B cells, have been described as a component of tumour inflammatory infiltrates [24,25,26]. While some studies have reported poorer recurrence-free survival1, disease-free survival (DFS), and overall survival (OS) [27, 28] in breast cancers with higher frequencies of plasma cells, other studies found better DFS in TNBC with higher frequencies of plasma cells [29]. Notably, a high expression of plasma cell genes and B-cell genes has been associated with favourable prognosis in breast cancers [29,30,31]. Our group recently found that TNBC with high intratumoural CD20+ B-cell and high CD38+ plasma cell counts had better prognosis. Futhermore, CD38+ plasma cells is an independent prognostic marker of TNBC, and expression level of IgG genes is associated with improved survival outcomes [29].
Tertiary lymphoid structures (TLS) are highly organized aggregates of lymphoid cells that resemble lymph nodes [32]. It has been proposed that TLS help coordinate antitumour responses involving the combined actions of both cytotoxic T cells and antibody-producing plasma cells [24]. They have been shown to be prognostic and predictive markers for invasive breast cancer [33]. To understand how higher numbers of plasma cells effect a better prognosis in TNBC, we aimed to study the phenotype and function of plasma cells in TNBCs by assessing their association with B cells and TLS.
Materials and methods
Patients and tumours
The Centralized Institutional Review Board of SingHealth provided ethical approval for the use of patient materials in this study (CIRB Ref: 2011/433/F). A total of 269 archival formalin-fixed paraffin-embedded (FFPE) TNBC specimens from patients diagnosed between 2003 and 2010 at the Department of Anatomical Pathology, Division of Pathology, Singapore General Hospital were analysed. Samples were obtained before patients underwent chemotherapy or radiotherapy; receipt of neoadjuvant therapy was an exclusion criteria. The age of patients ranged between 28 and 89 years (median 55 years); length of follow-up ranged from 1 to 213 months (mean 101, median 97), with recurrence and death occurring in 29 and 24% of these women, respectively. Tumours were typed, staged, and graded according to World Health Organization, American Society of Clinical Oncology—College of American Pathologists (ASCO-CAP) guidelines [34].
Tissue microarray (TMA) construction
For each FFPE tissue block, tumour regions were selected for TMA construction from tissue areas containing > 50% tumour. For each case, two or three representative 1 mm diameter tumour cores were transferred from FFPE tissue blocks to recipient TMA blocks using a MTA-1 Manual Tissue Arrayer (Beecher Instruments, Sun Prairie, WI, USA). TMAs were constructed as previously described [35].
Immunohistochemical analysis of TMAs
TMA sections of 4 μm thickness were incubated with antibodies specific for CD20, CD38, IgG Kappa, IgA, Granzyme B and PD-1, according to previously published protocols [29, 35, 36]. Appropriate positive and negative controls were included. Tumour-infiltrating lymphocytes expressing CD20 (B cells) or CD38 (plasma cells) were identified and manually scored within the stromal and intratumoural regions separately, on TMA cores. Plasma cells and B cells were presented as the percentage of the intratumoural or stromal areas occupied by the respective cell population, based on published methods [37, 38]. Tumours were then divided into “high density” and “low density” when the percentage of the intratumoural or stromal areas occupied by cells labelled for either CD38 (plasma cells) or CD20 (B cells) was above or on/below the median, respectively. The cut-off median percentages used were compatible with accepted clinical pathological practices: 5% for intratumoural CD38+ plasma cells and CD20+ B cells, and 1% for stromal CD38+ plasma cells and CD20+ B cells. Stromal and intratumoural plasma cells were also scored on haematoxylin–eosin (H&E) whole tumour sections in areas demonstrating the highest density of plasma cells and presented as an absolute number. The manual scoring was performed by two pathologists (JI & NC) who were blinded to the clinicopathological information. Scoring was performed independently. Where discordant, the cases were reviewed and a consensus score was given.
Co-localization of immunoglobulins, Granzyme B, and PD-1 with CD38+ plasma cells
Within our cohort of 269 TNBC cases, we selected 30 cases with the highest densities of CD38+ plasma cells (“high CD38+ plasma cell group”), and 30 cases with the lowest densities of CD38+ plasma cells (“low CD38+ plasma cell group”). The markers IgA and IgG kappa, Granzyme B, and PD1 were chosen to assess the phenotype and function of tumour-infiltrating CD38+ plasma cells in TNBC: The immunoglobulins IgA and IgG kappa are secreted by plasma cells and reflect the function of plasma cells. Granzyme B-positive B cells have been found to act as B-regulatory cells in cancer immunosurveillance by mediating cytotoxic pathways [24]. PD-1 positive B-cell subsets also modulate antitumour immune responses [39]. CD38, IgG Kappa, IgA, Granzyme B and PD-1 were stained on whole tissue sections of these cases using Opal-Vectra multiplex immunofluorescence (Perkin Elmer, Waltham, Massachusetts, USA) [8, 40, 41]. Whole slide tumour sections were examined by a pathologist (JY), who assessed it for areas showing the highest densities of CD38+ plasma cells which co-expressed IgG Kappa, IgA, Granzyme B, or PD-1, respectively. These areas were then selected as regions of interest for multiplex immunofluorescence image analysis. For each case, 9 images were selected (×200 magnification, 669 μm × 500 μm). These images were analysed with the inForm 2.3 software (Perkin Elmer, Waltham, Massachusetts, USA), which scored the number of CD38+ plasma cells showing co-expression of IgG Kappa, PD-1, IgA, and Granzyme B [42]. In order to validate the scoring by the inForm software, manual scoring of CD38+ plasma cells on TMA cores was performed by a pathologist (JI, NC), as described in the section above.
Association of tertiary lymphoid structures in TNBC with CD38+ plasma cell
Out of our cohort, we selected TNBC cases showing the highest (n = 10) and lowest (n = 10) densities of plasma cells and performed H&E and dual CD3/CD20 immnuohistochemical (IHC) stains on whole tissue sections. TLS were scored on whole tissue sections of tumours by experienced pathologists (JI, NC) who were blinded to the previous CD38 score and clinicopathological parameters. TLS were defined as the presence of aggregates of immune cells on HE slides; perivascular lymphatic aggregates were excluded. Morphologic TLS identification on HE slides was confirmed by IHC-stained slides which contained CD20+ B-cell rich follicles surrounded by or adjacent to a CD3+ T-cell zone [43, 44]. We scored the absolute number of TLS on whole tumour sections, and also scored TLS located within the tumour-associated stroma (in direct contact or within 0.5 mm of the tumour [43]. TLS-positive tumours were defined as having ≥ 1 TLS, and TLS-negative tumours were defined by absence of TLS (< 1). We determined if there was a significant association between number of TLS and high versus low densities of plasma cells in TNBC.
Results
IgG Kappa+ CD38+ plasma cells are significantly higher in TNBC with high CD38+ plasma cells
We investigated the “functional” characteristics of intratumoural plasma cells by examining if there was co-localizing expression of immunoglobulins (IgG Kappa, IgA), PD-1 and cytotoxic protein Granzyme B using multiplex IF. The representative images of PD-1+ CD38+ plasma cells, IgG Kappa+ CD38+ plasma cells a,nd IgA+ CD38+ plasma cells are shown in Fig. 1a–c.
Representative images demonstrate a PD-1+ CD38+ plasma cells, b IgG Kappa+ CD38+ plasma cells, c IgA+ CD38+ plasma cells, and d CD138 + CD38 + plasma cells. a The white arrows indicate the PD-1 + (red) and CD38+ (green) subset of plasma cells. b The white arrows indicate the IgG Kappa chain+ (red) and CD38 + (green) subset of plasma cells. c The white arrows indicate the IgA+ (red) and CD38+ (green) subset of plasma cells. d The white arrows indicate the CD138+ (red) and CD38+ (green) subset of plasma cells, while the surrounding tumour cells are highlighted by both Pan-Cytokeratin (pink) and CD138 (red)
IgG Kappa+ CD38+ plasma cells (Fig. 2a), PD-1+ CD38+ plasma cells (Fig. 2b), and IgA+ CD38+ plasma cells (Fig. 2c) accounted for 10.00%, 8.7%, and 2.6% of all CD38+ plasma cells, respectively. Granzyme B, which is not known to be expressed on plasma cells, were not detected on CD38+ plasma cells. IgG Kappa+ CD38+ plasma cells were significantly higher in TNBC with high CD38+ plasma cells, compared to TNBC with low CD38+ plasma cells (p = 0.0089, p < 0.0001). There was no difference in the proportion of IgA+ CD38+ plasma cells and PD1+ CD38+ plasma cells in the CD38+ high versus the CD38+ low plasma cell groups (Figs. 2b, c).
Multiplex IHC analysis on whole slide section by inForm software showed that tumours bearing high CD38+ plasma cells also harboured significantly higher proportion of a IgG kappa+ /CD38+ plasma cells, compared to the patients harbouring low CD38+ plasma cells. The proportion of b PD-1+ /CD38+ plasma and c IgA+ /CD38+ plasma cells are comparable between two groups of patients. d inForm software scoring further validates that our manual scoring whereby tumours bearing high CD38+ (manual scoring) plasma cells indeed harboured significantly higher cell count of CD38+ plasma cells (inForm scoring)
Additionally, TNBC with high CD38+ plasma cells on manual scoring showed significantly higher cell count of CD38+ plasma cells on inForm scoring (Fig. 2d). This demonstrates that the scoring of CD38+ plasma cells by the inForm software was comparable and consistent with manual scoring.
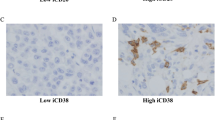
TNBC with high CD38+ plasma cells showed higher numbers of CD20+ B cells
We investigated CD38+ plasma cell and CD20+ B-cell expression on whole tissue sections of TNBC with high and low CD38+ plasma cell counts. Manual whole slide scoring confirmed significantly higher numbers of CD38+ plasma cells (Fig. 3a) and CD20+ B cells (Fig. 3b) in TMA cores of TNBC with high density of plasma cells, compared to those with low CD38+ plasma cells (analysed on TMA). These significant trends were also observed on the tumour centre and the tumour invasive margin/front, which validates the reproducibility of our scoring on TMA with that on whole tissue sections.
a Manual scoring on whole slide sections shows that tumours bearing high CD38+ plasma cells indeed harboured significantly higher cell count of CD38+ plasma cells when we assess the tumour centre and advancing invasive tumour margin/front. b Manual scoring on whole slide sections shows that tumours bearing high CD38+ plasma cells also harboured significantly higher cell count of CD20+ B cells regardless of the location of the CD20+ B cells. c Number of CD38+ plasma cells is significantly higher in the tumour centre compared to adjacent stroma/normal tissue. d Number of CD20+ B cells is significantly higher in the advancing invasive tumour front compared to adjacent stroma/normal tissue
Within the adjacent normal stroma, higher stromal B cells were observed in TNBC with high CD38+ plasma cells, compared to tumours with low CD38+ plasma cells (Fig. 3b). Interestingly, within whole slide sections, higher numbers of intratumoural plasma cells were present in the tumour centre compared to the adjacent stroma (Fig. 3c). Absolute numbers of intratumoural CD20+ B cells were lower than adjacent stromal CD20+ B cells (Fig. 3d).
TNBCs with high CD38+ plasma cells showed higher numbers of tertiary lymphoid structures
Since TLS have been reported as predictors of pathological complete response in TNBC3, we examined TLS in a limited cohort of TNBC demonstrating the highest (n = 10) and lowest (n = 10) densities of plasma cells. Figure 4a–d illustrates an example of TLS identified on a whole slide section with H&E stain, as well as CD3, CD20, and CD38 immunohistochemical staining, respectively. In our limited cohort, 90% of TNBC with the highest density of plasma cells harboured TLS, while 60% of TNBC with the lowest density of plasma cells harboured TLS. Across all examined TNBC cases, the mean number of TLS present was 7.15 and the median number of TLS present was 4. In TNBC with the highest density of plasma cells, the mean and median number of TLS present was 13. In TNBC with the lowest density of plasma cells, the mean number of TLS was 1.3 and the median number of TLS was 1. TNBC with high density of plasma cells showed significantly higher numbers of TLS, compared to TNBC with low density of plasma cells (p < 0.0001) (Fig. 4e).
a Tertiary lymphoid structure identified on whole slide section of TNBC, H&E ×200 magnification. Immunohistochemical staining demonstrates the organization of (b) CD3 + T cells and c CD20+ B cells within the tertiary lymphoid structure, ×200 magnification. d CD38+ plasma cells within and around the same tertiary lymphoid structure. e TNBC with higher number of CD38+ cells show significantly higher numbers of tertiary lymphoid structures
Dicussion
We demonstrated that TNBC with high CD38+ plasma cell count showed significantly higher numbers of IgG Kappa + CD38+ cells, CD20+ B cells, and TLS. Collectively, our findings lend weight to the argument that CD38+ plasma cells have cooperative interactions with other TILs and TLS as part of the adaptive antitumour immune response to restrain TNBC progression.
Wee et al. recently described a new technique to integrate an automated multispectral imaging technique that simultaneously detects and quantitates RNA and immune cell protein markers in fixed sections of solid tumours [45]. Our group successfully reproduced this technique and applied it to our TNBC cohort, demonstrating the utility of integrated multiplex immunofluorescence approach to concurrently detect multiple protein biomarkers of immune cells associated with a tumour. This technique may have potential future applications for diagnostic use in solid tumours.
We found a significant association between higher numbers of IgG Kappa + plasma cells in TNBC with high CD38+ plasma cell counts. IgG Kappa, an immunoglobulin gene, is a predictive marker of metastasis-free survival and response to chemotherapy in solid tumours, including breast cancers [46,47,48]. Our findings support the results published by our group that showed that mRNA levels of the IgG Kappa gene, IGKC, was correlated with the density of plasma cells in TNBC [29]. We propose that these tumour-infiltrating plasma cells are not just passive bystanders, but instead “functional” participants that secrete antibodies, such as IgG Kappa, which is known to bind to tumour antigens and mediate antibody-dependent cellular cytotoxicity against tumour [48]. This would explain the association of higher expression of plasma cell and B-cell genes with favourable prognosis in TNBC [30, 31].
Our results also indicate that the plasma cells within the TNBC microenvironment are distributed predominantly within the intratumoural and invasive front of the tumour, as opposed to the adjacent stroma. Furthermore, the absolute numbers of intratumoural B cells are lower than adjacent stromal B cells. In our previous study, we reported that TNBC patients with high densities of intratumoural plasma cells showed significantly better DFS compared to TNBC patients with low intratumoural plasma cells [29]. Furthermore, the addition of intratumoural CD38+ plasma cell density to clinicopathological features significantly increased the prognostic value for both DFS and OS, compared to clinicopathological features alone [29]. The density of stromal plasma cells had no bearing on survival outcomes in TNBC, while high stromal B cells showed significantly better OS and DFS [29]. Together, our findings suggest that the compartments that plasma cells and B cells occupy within TNBC appear to influence tumour progression, although this mechanism is still not well understood. More studies are needed to elucidate the interaction between plasma cells and B cells, and to understand how it creates an activated immune microenvironment that contributes to superior patient survival.
TLS present in human solid tumours are essential to shape a favourable immune microenvironment to control tumour development [24, 32]. Their functions include T-cell priming, B-cell activation, and differentiation into plasma cells and antibody production [49], with resultant antitumour responses [32, 49]. Past studies have shown the detection of TLS on HE slides alone leads to underestimation, as compared to detection of TLS on IHC-stained tissues, which has greater intra- and inter-observer agreement [43]. As such, in our study we identified TLS on both HE slides and CD3/CD20 IHC-stained slides. With this method, we found a significantly higher number of TLS present in TNBC with high density of plasma cells compared to TNBC with low density of plasma cells. Though only demonstrated in a limited cohort, our findings corroborate other studies that showed the close association of TLS with plasma cells in tumours of the ovary [25], breast, metastatic melanoma [50], and non-small-cell lung cancer [51]. A group has previously reported that TLS is an important predictor of pathological complete response in TNBC [44]. Our findings suggest that there may be an interaction between plasma cells and TLS in TNBC. Furthermore, findings from the current study and our past study [29] have indicated that TNBC with higher densities of plasma cells were also associated with higher densities of B cells. Hence, an association of TLS with other TILs in TNBC may exist, although evidence for this is not yet well-established. More research using a larger cohort should be conducted, not only to determine the proportion of TNBC harbouring TLS and their respective frequencies, but also the association and interaction between TLS and other TILs in forming an anti-tumoural microenvironment for TNBC.
In summary, our results demonstrate that TNBC with high numbers of CD38+ plasma cells are associated with higher numbers of IgG Kappa+ CD38+ cells, CD20+ B cells, and TLS. We propose that optimal antitumour immunity may involve closely integrated antibody-mediated humoral mechanisms in addition to T-cell-mediated antitumour immunity. Though the findings of our study are limited by its retrospective design and relatively small cohort, we have highlighted the important role of the humoral immune system and more specifically plasma cells in TNBC. Given the significance of TLS in supporting antitumour immunity, further understanding of its organization and biology may help improve existing immunotherapy strategies.
References
Foulkes WD, Smith IE, Reis-Filho JS (2010) Triple-negative breast cancer. N Engl J Med 363(20):1938–1948. https://doi.org/10.1056/NEJMra1001389
Thike AA, Yian Cheok P, Richelia Jara-Lazaro A, Tan B, Tan P, Hoon TP (2009) Triple-negative breast cancer: clinicopathological characteristics and relationship with basal-like breast cancer. Mod Pathol 23(1):123–133. https://doi.org/10.1038/modpathol.2009.145
Rakha EA, Reis-Filho JS, Ellis IO (2008) Basal-like breast cancer: a critical review. J Clin Oncol. https://doi.org/10.1200/JCO.2007.13.1748
Dent R, Trudeau M, Pritchard KI et al (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13(15):4429–4434. https://doi.org/10.1158/1078-0432.CCR-06-3045
Cleator S, Heller W, Coombes RC (2007) Triple-negative breast cancer: therapeutic options. Lancet Oncol. https://doi.org/10.1016/S1470-2045(07)70074-8
Hudis CA, Gianni L (2011) Triple-negative breast cancer: an unmet medical need. Oncologist 16(Supplement 1):1–11. https://doi.org/10.1634/theoncologist.2011-S1-01
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell. https://doi.org/10.1016/j.cell.2011.02.013
Yeong J, Thike AA, Lim JCT et al (2017) Higher densities of Foxp3+ regulatory T cells are associated with better prognosis in triple-negative breast cancer. Breast Cancer Res Treat 163(1):21–35. https://doi.org/10.1007/s10549-017-4161-4
Bottai G, Raschioni C, Losurdo A et al (2016) An immune stratification reveals a subset of PD-1/LAG-3 double-positive triple-negative breast cancers. Breast Cancer Res 18:121. https://doi.org/10.1186/s13058-016-0783-4
Denkert C, von Minckwitz G, Brase JC et al (2015) Tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy with or without carboplatin in human epidermal growth factor receptor 2-positive and triple-negative primary breast cancers. J Clin Oncol. https://doi.org/10.1200/JCO.2014.58.1967
Ono M, Tsuda H, Shimizu C et al (2012) Tumor-infiltrating lymphocytes are correlated with response to neoadjuvant chemotherapy in triple-negative breast cancer. Breast Cancer Res Treat. https://doi.org/10.1007/s10549-011-1554-7
Miyashita M, Sasano H, Tamaki K et al (2015) Prognostic significance of tumor-infiltrating CD8+ and FOXP3+ lymphocytes in residual tumors and alterations in these parameters after neoadjuvant chemotherapy in triple-negative breast cancer: a retrospective multicenter study. Breast Cancer Res 17(1):124. https://doi.org/10.1186/s13058-015-0632-x
Loi S, Michiels S, Salgado R et al (2014) Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: results from the FinHER trial. Ann Oncol. https://doi.org/10.1093/annonc/mdu112
DeNardo DG, Brennan DJ, Rexhepaj E et al (2011) Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov 1(1):54–67. https://doi.org/10.1158/2159-8274.CD-10-0028
Schwartz M, Zhang Y, Rosenblatt JD (2016) B cell regulation of the anti-tumor response and role in carcinogenesis. J Immunother Cancer. https://doi.org/10.1186/s40425-016-0145-x
Zhang Y, Gallastegui N, Rosenblatt JD (2015) Regulatory B cells in anti-tumor immunity. Int Immunol. https://doi.org/10.1093/intimm/dxv034
Weiner LM, Dhodapkar MV, Ferrone S (2009) Monoclonal antibodies for cancer immunotherapy. Lancet. https://doi.org/10.1016/S0140-6736(09)60251-8.Monoclonal
Topalian SL, Weiner GJ, Pardoll DM (2011) Cancer immunotherapy comes of age. J Clin Oncol. https://doi.org/10.1200/JCO.2011.38.0899
Weiner LM, Murray JC, Shuptrine CW (2012) Antibody-based immunotherapy of cancer. Cell. https://doi.org/10.1016/j.cell.2012.02.034
Marsigliante S, Biscozzo L, Marra A et al (1999) Computerised counting of tumour infiltrating lymphocytes in 90 breast cancer specimens. Cancer Lett. https://doi.org/10.1016/S0304-3835(98)00379-6
Nelson BH (2010) CD20+ B Cells: the other tumor-infiltrating lymphocytes. J Immunol. https://doi.org/10.4049/jimmunol.1001323
Coronella-Wood JA, Hersh EM (2003) Naturally occurring B-cell responses to breast cancer. Cancer Immunol Immunother. https://doi.org/10.1007/s00262-003-0409-4
Brown JR, Wimberly H, Lannin DR, Nixon C, Rimm DL, Bossuyt V (2014) Multiplexed quantitative analysis of CD3, CD8, and CD20 predicts response to neoadjuvant chemotherapy in breast cancer. Clin Cancer Res. https://doi.org/10.1158/1078-0432.CCR-14-1622
Kroeger DR, Milne K, Nelson BH (2016) Tumor-infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-cell responses, and superior prognosis in ovarian cancer. Clin Cancer Res. https://doi.org/10.1158/1078-0432.CCR-15-2762
Berntsson J, Nodin B, Eberhard J, Micke P, Jirström K (2016) Prognostic impact of tumour-infiltrating B cells and plasma cells in colorectal cancer. Int J Cancer 139(5):1129–1139. https://doi.org/10.1002/ijc.30138
Ito T, Saga S, Nagayoshi S et al (1986) Class distribution of immunoglobulin-containing plasma cells in the stroma of medullary carcinoma of breast. Breast Cancer Res Treat 7(2):97–103
Mohammed ZMA, Going JJ, Edwards J, Elsberger B, Mcmillan DC (2013) The relationship between lymphocyte subsets and clinico-pathological determinants of survival in patients with primary operable invasive ductal breast cancer. Br J Cancer. https://doi.org/10.1038/bjc.2013.493
Wei H, Fu P, Yao M, Chen Y, Du L (2016) Breast cancer stem cells phenotype and plasma cell-predominant breast cancer independently indicate poor survival. Pathol Res Pract. https://doi.org/10.1016/j.prp.2016.01.008
Yeong J, Lim JCT, Lee B et al (2018) High densities of tumor-associated plasma cells predict improved prognosis in triple negative breast cancer. Front Immunol 9:1209
Alistar A, Chou JW, Nagalla S, Black MA, D’Agostino R, Miller LD (2014) Dual roles for immune metagenes in breast cancer prognosis and therapy prediction. Genome Med. https://doi.org/10.1186/s13073-014-0080-8
Hanker LC, Rody A, Holtrich U et al (2013) Prognostic evaluation of the B cell/IL-8 metagene in different intrinsic breast cancer subtypes. Breast Cancer Res Treat 137(2):407–416. https://doi.org/10.1007/s10549-012-2356-2
Dieu-Nosjean MC, Goc J, Giraldo NA, Sautès-Fridman C, Fridman WH (2014) Tertiary lymphoid structures in cancer and beyond. Trends Immunol. https://doi.org/10.1016/j.it.2014.09.006
Lee M, Heo S-H, Song IH et al (2019) Presence of tertiary lymphoid structures determines the level of tumor-infiltrating lymphocytes in primary breast cancer and metastasis. Mod Pathol 32(1):70–80. https://doi.org/10.1038/s41379-018-0113-8
Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ (2012) WHO classification of tumours of the breast, 4th edn. IARC, France
Thike AA, Chong LYZ, Cheok PY et al (2014) Loss of androgen receptor expression predicts early recurrence in triple-negative and basal-like breast cancer. Mod Pathol. https://doi.org/10.1038/modpathol.2013.145
Thike AA, Iqbal J, Cheok PY, Tse GM-K, Tan PH (2013) Ductal carcinoma in situ associated with triple negative invasive breast cancer: evidence for a precursor–product relationship. J Clin Pathol 66(8):665–670. https://doi.org/10.1136/jclinpath-2012-201428
Salgado R, Denkert C, Demaria S et al (2015) The evaluation of tumor-infiltrating lymphocytes (TILS) in breast cancer: recommendations by an International TILS Working Group 2014. Ann Oncol. https://doi.org/10.1093/annonc/mdu450
Herbst RS, Soria JC, Kowanetz M et al (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature. https://doi.org/10.1038/nature14011
Sun X, Zhang T, Li M, Yin L, Xue J (2019) Immunosuppressive B cells expressing PD-1/PD-L1 in solid tumors: a mini review. QJM An Int J Med. https://doi.org/10.1093/qjmed/hcz162
Garnelo M, Tan A, Her Z et al (2017) Interaction between tumour-infiltrating B cells and T cells controls the progression of hepatocellular carcinoma. Gut. https://doi.org/10.1136/gutjnl-2015-310814
Lim JCT, Yeong JPS, Lim CJ et al (2018) An automated staining protocol for seven-colour immunofluorescence of human tissue sections for diagnostic and prognostic use. Pathology. https://doi.org/10.1016/j.pathol.2017.11.087
Fiore C, Bailey D, Conlon N et al (2012) Utility of multispectral imaging in automated quantitative scoring of immunohistochemistry. J Clin Pathol. https://doi.org/10.1136/jclinpath-2012-200734
Buisseret L, Desmedt C, Garaud S et al (2017) Reliability of tumor-infiltrating lymphocyte and tertiary lymphoid structure assessment in human breast cancer. Mod Pathol 30(9):1204–1212. https://doi.org/10.1038/modpathol.2017.43
Lee HJ, Park IA, Song IH et al (2016) Tertiary lymphoid structures: prognostic significance and relationship with tumour-infiltrating lymphocytes in triple-negative breast cancer. J Clin Pathol 69(5):422–430
Wee YTF, Alkaff SMF, Lim JCT et al (2018) An integrated automated multispectral imaging technique that simultaneously detects and quantitates viral RNA and immune cell protein markers in fixed sections from Epstein-Barr virus-related tumours. Ann Diagn Pathol 37:12–19. https://doi.org/10.1016/j.anndiagpath.2018.09.002
Schmidt M, Micke P, Gehrmann M, Hengstler JG (2012) Immunoglobulin kappa chain as an immunologic biomarker of prognosis and chemotherapy response in solid tumors. Oncoimmunology. https://doi.org/10.4161/onci.21653
Chen Z, Gerhold-Ay A, Gebhard S et al (2012) Immunoglobulin kappa C predicts overall survival in node-negative breast cancer. PLoS ONE. https://doi.org/10.1371/journal.pone.0044741
Whiteside TL, Ferrone S (2012) For breast cancer prognosis, immunoglobulinkappa chain surfaces to the top. Clin Cancer Res. https://doi.org/10.1158/1078-0432.CCR-12-0566
Teillaud JL, Dieu-Nosjean MC (2017) Tertiary lymphoid structures: An anti-tumor school for adaptive immune cells and an antibody factory to fight cancer? Front Immunol. https://doi.org/10.3389/fimmu.2017.00830
Erdag G, Schaefer JT, Smolkin ME et al (2012) Immunotype and immunohistologic characteristics of tumor-infiltrating immune cells are associated with clinical outcome in metastatic melanoma. Cancer Res. https://doi.org/10.1158/0008-5472.CAN-11-3218
Lohr M, Edlund K, Botling J et al (2013) The prognostic relevance of tumour-infiltrating plasma cells and immunoglobulin kappa C indicates an important role of the humoral immune response in non-small cell lung cancer. Cancer Lett. https://doi.org/10.1016/j.canlet.2013.01.036
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Seow, D.Y.B., Yeong, J.P.S., Lim, J.X. et al. Tertiary lymphoid structures and associated plasma cells play an important role in the biology of triple-negative breast cancers. Breast Cancer Res Treat 180, 369–377 (2020). https://doi.org/10.1007/s10549-020-05548-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05548-y