Abstract
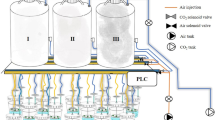
Five in vitro culture systems with different ventilation rates were used to investigate the influence of vessel environment on photosynthesis, dark respiration, ethylene evolution, and rosmarinic acid (RA) production in sweet basil (Ocimum basilicum L.) micropropagated shoots. The systems under comparison were two bioreactors with either temporary (RITA™) or stationary (Growtek™) immersion, and three types of vessels (Magenta™, Microbox ECO 2 ™, and PCCV25™) that are largely used for plant micropropagation. Shoots of green-leaved cv. Genovese and purple-leaved cv. Dark Opal were cultured on a modified Murashige and Skoog medium containing 0.25 mg dm−3 6-benzylaminopurine. The instantaneous rates of photosynthesis, dark respiration, and ethylene production were determined by gas chromatography measuring CO2 and ethylene concentrations in vessel headspaces. The tissue RA content was determined by HPLC in HCl-methanol extracts. The explant growth and morphology were significantly affected by culture conditions and cultivars. The largest biomass production was observed under the photomixotrophic culture conditions provided by Growtek™, whereas the highest RA content in shoot tissues was found in the RITA™ photomixotrophic system, where ethylene accumulated to the greatest extent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CAD:
-
caffeic acid derivative
- PN :
-
net phothosynthetic rate
- PPFD:
-
photosynthetic photon flux density
- RA:
-
rosmarinic acid
- RD :
-
dark respiration rate
References
Afreen, F., Zobayed, S.M.A., Kozai, T.: Photoautotrophic culture of Coffea arabusta somatic embryos: development of a bioreactor for large-scale plantlet conversion from cotyledonary embryos. — Ann. Bot. 90: 21–29, 2002.
Begum, F., Amin, M.N., Azad, M.A.K.: In vitro rapid clonal propagation of Ocimum basilicum L.. — Plant Cell Tissue Organ Cult. 12: 27–35, 2002.
Bertoli, A., Lucchesini, M., Mensuali-Sodi, A., Leonardi, M., Doveri, S., Magnabosco, A., Pistelli L.: Aroma characterisation and UV elicitation of purple basil from different plant tissue cultures. — Food Chem. 141: 776–787, 2013.
Biddington, N.L.: The influence of ethylene in plant tissue culture. — Plant Growth Regul. 11: 173–187, 1992.
Bourgaud, F., Gravot, A., Milesi, S., Gontier, E.: Production of plant secondary metabolites: a historical perspective. — Plant Sci. 161: 839–851, 2001.
Chakrabarty, D., Park, S.Y., Ali, M.B., Shin, K.S., Paek, K.Y.: Hyperhydricity in apple: ultrastuctural and physiological aspects. — Tree Physiol. 26: 377–388, 2005.
Chanemougasoundharam, A., Sarkar, D., Pandey, S.K., Al-Biski, F., Helali, O., Minhas, J.S.: Culture tube closure-type affects potato plantlets growth and chlorophyll contents. — Biol. Plant. 48: 7–11, 2004.
Cho, G.H., Kim, D.I., Pedersen, H., Chin, C.K.: Ethephon enhancement of secondary metabolite synthesis in plant cell cultures. — Biotech. Progr. 4: 184–188, 1988.
Deng, Z., Chu, J., Wang, Q., Wang, L.: Effect of different carbon sources on the accumulation of carbohydrate, nutrient absorption and the survival rate of Chinese Ash (Fraxinus mandshurica) explants in vitro. — Afr. J. agr. Res. 7: 3111–3119, 2012.
Dey, S.: Cost-effective mass cloning of plants in liquid media using a novel Growtek bioreactor. — In: Hvoslef-Eide, A.K., Preil W. (ed.): Liquid Culture Systems for in Vitro Plant Propagation. Pp. 127–141. Springer, Dordrecht 2005.
Diarra, S.T., He, J., Wang, J., Li J.: Ethylene treatment improves diosgenin accumulation in in vitro cultures of Dioscorea zingiberensis via up-regulation of CAS and HMGR gene expression. — Electron. J. Biotechn. 16: http://dx.doi.org/10.2225/vol16-issue5-fulltext-9, 2013.
Dias, M.C., Pinto, G.C., Correia, M., Moutinho-Pereira, J., Silva, S., Santos, C.: Photosynthetic parameters of Ulmus minor plantlets affected by irradiance during acclimatization. — Biol. Plant. 57: 33–40, 2013.
Fujiwara, K., Kozai, T., Watanabe, I.: Measurements of carbon dioxide gas concentration in stoppered vessels containing tissue cultured plantlets and estimates of net photosynthetic rate of the plantlets. — J. Agr. Meteorol. 43: 21–30, 1987.
Gaspar, T., Franck, T., Bisbis, B., Kevers, C., Jouve, L., Haussman, J.F., Dommes, J.: Concepts in plant stress physiology. Application to plant tissue cultures. — Plant Growth Regul. 37: 263–285, 2002.
Georgiev, M.I., Weber, J., Maciuk, A.: Bioprocessing of plant cell cultures for mass production of targeted compounds. — Appl. Microbiol. Biotechnol. 83: 809–823, 2009.
Hakkim, F.L., Shankar, C.G., Girua, S.: Chemical composition and antioxidant property of holy basil (Ocimum sanctum L.) leaves, stems, and inflorescence and their in vitro callus cultures. — J. Agr. Food Chem. 55: 9109–9117, 2007.
Hazarika, B.N.: Morpho-physiological disorders in in vitro culture of plants. — Sci. Hort. 108: 105–120, 2006.
Ikemeyer, D., Barz, W.: Comparison of secondary product accumulation in photoautotrophic, photomixitrophic and heterotrophic Nicotiana tabacum cell suspension cultures. — Plant Cell Rep. 8: 479–482, 1989.
Jackson, M. B.: Aeration stress in plant tissue cultures. — Bulg. J. Plant Physiol. 96(Special Issue): 109, 2003.
Jayasinghe, C., Gotoh, N., Aoki, T., Wada, S.: Phenolics composition and antioxidant activity of sweet basil (Ocimum basilicum L.). — J. Agr. Food Chem. 51: 4442–4449, 2003.
Juliani, H.R., Koroch, A.R., Simon, J.E.: Basil: A new source of rosmarinic acid — In: Ho, C.T., Simon, J.E., Shahidi, F., Shao, Y. (ed.): Dietary Supplements. Pp. 129–143. American Chemical Society, Washington 2008.
Karuppusamy, S.: A review on trends in production of secondary metabolites from higher plants by in vitro tissue, organ and cell cultures. — J. med. Plants Res. 3: 1222–1239, 2009.
Kiferle, C., Lucchesini, M., Mensuali-Sodi, A., Maggini, R., Raffaelli, A., Pardossi A.: Rosmarinic acid content in basil plants grown in vitro and in hydroponic. — Cent. Eur. J. Biol. 6: 946–957, 2011.
Kiferle, C., Maggini, R., Pardossi, A. Influence of nitrogen nutrition on growth and accumulation of rosmarinic acid in sweet basil (Ocimum basilicum L.) grown in hydroponic culture. — Aust. J. Crop Sci. 7: 321–327, 2013.
Kintzios, S., Kollias, H., Straitouris, E., Makri, O.: Scale-up micropropagation of sweet basil (Ocimum basilicum L.) in an airlift bioreactor and accumulation of rosmarinic acid. — Biotechnol. Lett. 26: 521–523, 2004.
Kintzios, S., Makri, O., Panagiotopoulos, E., Scapeti, M.: In vitro rosmarinic acid accumulation in sweet basil (Ocimum basilicum L.). — Biotechnol. Lett. 25: 405–408, 2003.
Kobayashi, Y., Fukui, H., Tabata, M.: Effect of carbon dioxide and ethylene on berberine production and cell browning in Thalictrum minus cell cultures. — Plant Cell Rep. 9: 496–499, 1991a.
Kobayashi, Y., Hara, M., Fukui, H., Tabata, M.: The role of ethylene in berberine production by Thalictrum minus cell suspension cultures. — Phytochemistry 30: 3605–3609, 1991b.
Kozai, T.: Autotrophic micropropagation. — In: Bajaj, Y.P.S. (ed.): Biotechnology in Agriculture and Forestry: High Tech and Micropropagation I. Pp. 313–343. Springer-Verlag, Berlin — Heidelberg 1991.
Kozai, T., Kubota, C.: Concepts, definitions, ventilation methods, advantages and disadvantages. — In: Kozai T., Afreen, F., Zobayed, S.M.A. (ed.): Photoautotrophic (Sugar-Free Medium) Micropropagation as a New Propagation and Tansplant Production System. Pp. 9–30. Springer, Berlin 2005.
Kozai, T., Kubota, C., Chun, C., Afreen, F., Ohyama, K.: Necessity and concept of the closed transplant production system. — In: Kubota, C., Chun, C. (ed.): Proceedings for the International Symposium on Transplant Production in Closed System. Pp. 3–19. Kluwer Academic Publisher, Dordrecht 2000.
Lucchesini, M., Bertoli, A., Mensuali-Sodi, A., Pistelli, L.: Establishment of in vitro tissue cultures from Echinacea angustifolia D.C. adult plants for the production of phytochemical compounds. — Sci. Hort. 122: 484–490, 2009.
Lucchesini, M., Mensuali-Sodi, A.: Plant tissue culture-an opportunity for the production of nutraceuticals. — In: Giardi, M.T., Rea, G., Berra, B. (ed.): Bio-Farms for Nutraceuticals: Functional Food and Safety Control by Biosensor. Pp: 185–200. Springer, New York 2010.
Lucchesini, M., Mensuali-Sodi, A., Massai, R., Gucci, R.: Development of autotropy and tolerance to acclimatization of Myrtus communis transplant cultured in vitro under different aeration. — Biol. Plant. 44: 167–174, 2001.
Lucchesini, M., Monteforti, G., Mensuali-Sodi, A., Serra, G.: Leaf ultrastructure, photosynthetic rate and growth of myrtle plantlets under different in vitro culture condictions. — Biol. Plant. 50: 161–168, 2006.
Makri, O., Kintzios, S.: Ocimum sp. (basil): botany, cultivation, pharmaceutical properties, and biotechnology. — J. Herbs Spices med. Plants 13: 123–150, 2007.
Martre, P., Lacan, D., Just, D., Teisson, C.: Physiological effects of temporary immersion on Hevea brasiliensis callus. — Plant Cell Tissue Organ Cult. 67: 25–35, 2001.
Matkowski, A.: Plant in vitro for the production of antioxidants, a review. — Biotech. Adv. 26: 548–560, 2008.
Mensuali-Sodi, A., Lucchesini, M., Maltinti, S., Serra, G., Tognoni, F.: Leaf senescence in tissue culture of Passiflora incarnata L.: the role of ethylene. — In: Ramina, A., Chang, C., Giovannoni, J., Klee, H., Perata, P., Woltering, E. (ed.): Advances in Plant Ethylene Research. Pp. 151–152. Springer, Dordrecht 2007.
Mensuali-Sodi, A., Panizza, M., Tognoni, F.: Studies on lavandin callus cultures: ethylene production in relation to the growth. — Biol. Plant. 31: 247–253, 1989.
Mensuali-Sodi, A., Panizza, M., Tognoni, F.: Quantification of ethylene losses in different container-seal systems and comparison of biotic and abiotic contributions to ethylene accumulation in cultured tissues. — Physiol. Plant. 84: 472–476, 1992.
Mingozzi M, Morini S., Lucchesini M., Mensuali-Sodi A.: Effects of leaf soluble sugars content and net photosynthetic rate of quince donor shoots on subsequent morphogenesis in leaf explants. — Biol. Plant. 55: 237–242, 2011.
Morgan, P.W., Drew, M.C.: Ethylene and plant responses to stress. — Physiol. Plant. 100: 620–630, 1997.
Mosaleeyanon, K., Zobayed, S.M.A., Afreen, F., Kozai, T.: Relationships between net photosynthetic rate and secondary metabolite contents in St. John’s wort. — Plant. Sci. 169: 523–537, 2005.
Moschopoulou, G., Kintzios, S.: Achievement of thousand-fold accumulation of rosmarinic acid in immobilized cells of sweet basil (Ocimum basilicum L.) by ten-fold increase of the volume of the immobilization matrix. — J. biol. Res. Thessalon. 15: 59–65, 2011.
Murashige, T., Skoog, F.: A revised medium for rapid growth and bioassays with tobacco tissue cultures. — Physiol. Plant. 15: 473–493, 1962.
Nguyen, P.M., Kwee, E.M., Niemeyer, E.D.: Potassium rate alters the antioxidant capacity and phenolic concentration of basil (Ocimum basilicum L.) leaves. — Food Chem. 23: 1235–1241, 2010.
Panizza, M., Mensuali-Sodi, A., Tognoni, F.: Role of ethylene in axillary shoot proliferation of lavandin-interaction with benzyladenine and polyamines. — J. exp. Bot. 44: 387–394, 1993.
Park, S.U., Uddin, M.R., Xu, H., Kim, Y.K., Lee, S.Y.: Biotechnological applications for rosmarinic acid production in plant. — Afr. J. Biotechnol. 7: 4959–4965, 2008.
Patnaik, J., Sahoo, S., Debata, B.K.: Somaclonal variation in cell suspension culture-derived regenerants of Cymbopogon martini (Roxb.) Wats var. motia. — Plant Breed. 118: 351–354, 1999.
Petersen, M., Simmonds, M.S.J.: Molecules of interest: rosmarinic acid. — Phytochemistry 62: 121–125, 2003.
Pospíšilová, J., Haisel, D., Synková, H., Čatský, J., Wilhelmová, N., Plzáková, Š., Procházková, D., Šrámek, F.: Photosynthetic pigments and gas exchange during ex vitro acclimation of tobacco plants as affected by CO2 supply and abscisic acid. — Plant Cell Tissue Organ Cult. 61: 125–133, 2000.
Rady, M.R., Nazif, N.M.: Rosmarinic acid content and RAPD analysis of in vitro regenerated basil (Ocimum basilicum L.) plants. — Fitoterapia 6: 525–533, 2005.
Saher, S., Piqueras, A., Hellin, E., Olmos, E.: Hyperhydricity in micropropagated carnation shoots: the role of oxidative stress. — Physiol. Plant. 120: 152–161, 2004.
Sahoo, Y., Patfnaik, S.K., Chand, P.K.: In vitro propagation of an aromatic medicinal herb Ocimum basilicum L. (sweet basil) by axillary shoot proliferation. — In vitro cell. dev. Biol. Plant. 33: 293–296, 1997.
Savio, L.E. B., Astarita, L.V., Santarém, E.R.: Secondary metabolism in micropropagated Hypericum perforatum L. grown in non-aerated liquid medium. — Plant Cell Tissue Organ Cult. 108: 465–472, 2012.
Steyn, W.J., Wand, S.J.E., Holcroft, D.M., Jacobs, G.: Anthocyanins in vegetative tissues: a proposed unified function in photoprotection. — New Phytol. 55: 349–361, 2002.
Xiao, Y., Niu, G., Kozai, T.: Development and application of photoautotrophic micropropagation plant system. — Plant Cell Tissue Organ Cult. 105: 149–158, 2011.
Yasmin, S., Mensuali-Sodi, A., Perata, P., Pucciariello, C.: Ethylene influences in vitro regeneration frequency in the FR13A rice harbouring the SUB1A gene. — Plant Growth Regul. 72: 97–103, 2014.
Zhao, J., Davis, L.C., Verpoorte, R.: Elicitor signal transduction leading to production of plant secondary metabolites. — Biotechnol. Adv. 23: 283–333, 2005.
Ziv, M.: Bioreactor technoloy for plant micropropagation. — Hort. Rev. 24: 1–30, 2000.
Zobayed, S.M.A., Saxena, P.K.: Production of St. John’s wort plants under controlled environment for maximizing biomass and secondary metabolites. — In vitro cell. dev. Biol. Plant. 40: 108–114, 2004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kiferle, C., Lucchesini, M., Maggini, R. et al. In vitro culture of sweet basil: gas exchanges, growth, and rosmarinic acid production. Biol Plant 58, 601–610 (2014). https://doi.org/10.1007/s10535-014-0434-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-014-0434-5