Abstract
Estimates of gaseous carbon (C) fluxes in wetlands are heavily based on temperature. However, isolating specific effects of temperature on anaerobic C processing from other controls (C quality and nutrients) has proven difficult. Here, we test the hypothesis that temperature sensitivity of soil organic matter (SOM) decomposition is more influenced by C quality than nutrient availability in subtropical freshwater, sawgrass (Cladium jamaicense)-based peats. Carbon age (characterized by depth: 0–10 and 10–20 cm) was used as a surrogate of C quality while two sites were selected with contrasting levels of nutrient (P) availability. In anaerobic laboratory incubations temperature was increased in 5 °C steps to assess the proportion of C available at a given temperature (i.e. thermo-labile C) as productions of gaseous (CO2 and CH4) and dissolved organic C (DOC) fractions. Thermo-labile C increased 3.1–3.6 times from 15 °C to 30 °C in all soils. Disproportionate increase in the production of gaseous forms versus DOC as well as CH4:CO2 was observed with warming. Observed Q10 values followed the trend of CH4 (~14) ≫ CO2 (~2.5) > DOC (~1.7) and temperature sensitivity was more dependent on C quality than nutrient availability over the entire temperature range. Spectral analysis indicated more bio-available DOC production at higher temperature. Regression analysis also indicated that C quality primarily influenced SOM decomposition at lower temperature, while at higher temperature nutrient limitation dominantly controlled SOM decomposition. These findings confirm the role of C quality in temperature sensitivity of warm peat soils, but also indicate an increased importance of nutrient limitation at higher temperature.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wetlands are a globally important soil carbon (C) reservoir, accounting for about one-third of the total pool of soil C in the world (Bridgham et al. 1995; Mitsch and Gosselink 2007). In addition to being C sinks, wetlands are also the single largest source of methane (CH4) (Matthews and Fung 1987; Bergamaschi et al. 2007; Bloom et al. 2010), but the size of this source remains highly uncertain. Biogeochemical processes of anaerobic systems are known to be susceptible to temperature fluctuations (Gorham 1991; Melton et al. 2013), therefore, the C cycle in wetlands is also believed to be climate sensitive and is a critical component of the global climate system. Despite this understanding, the interaction of temperature and anaerobic C cycling remain poorly represented in ecosystem and climate models (Sulman et al. 2009).
There is a growing consensus that temperature accelerates the rates of soil organic matter (SOM) decomposition (Kirschbaum 1995; Davidson and Janssens, 2006; Conant et al. 2011), thus, a positive feedback of soil ecosystem to increasing temperature may accelerate the loss of C stocks of peats under future climate conditions. Wetlands are vulnerable to losing stored C to the atmosphere as carbon dioxide (CO2) and CH4 (Yavitt et al. 2005; Mitsch et al. 2010), and dissolved organic C (DOC) to receiving waters (Freeman et al. 2001; Moore and Dalva 2001). Despite this understanding, there is still high uncertainty in current C fluxes within wetland components (Bridgham et al. 2006) and in its variation across different scales (Li et al. 2007). In particular, there is high uncertainty in C emissions from tropical wetlands, where spatial coverage fluctuates seasonally, fluxes vary significantly across wetland types, and systems remain poorly studied, especially related to microbial and enzymatic processes that drive biogeochemical cycles, compared to those of temperate counterparts (Strack 2008).
Tropical wetlands emit nearly 60% of the total CH4 from all natural wetlands combined (Megonigal et al. 2005); therefore, studies of SOM decomposition in warmer tropical or subtropical wetlands are particularly important. In warmer systems, it is generally assumed that temperature is not a dominant influence on anaerobic C processing. However, recent discoveries suggested that temperature not only affects the total C released from subtropical peatlands, but also the form of C released with higher CH4 production at higher temperatures (Inglett et al. 2012).
The mechanisms underlying the temperature sensitivity of SOM decomposition are still a matter of debate. It is known that C quality is an important factor regulating CO2 and CH4 production where CH4 production has been found to be more sensitive to recalcitrance of C compounds than is CO2 (Bridgham et al. 1996). Higher CH4 emission has been reported from soils with higher labile C fraction (Yavitt and Lang 1990; Waldrop et al. 2010), and recalcitrant C fraction (Paré and Bedard-Haughn 2013). Inglett et al. (2012) demonstrated that vegetation type and associated differences in SOM quality play an important role in the temperature sensitivity of SOM decomposition in subtropical peats, while other studies using C age (or depth) as a surrogate for C quality (Lomander et al. 1998; Fang et al. 2005; Jinbo et al. 2006; Karhu et al. 2010) have reported that deep soils (i.e. older SOM) were more temperature sensitive due to their relatively high recalcitrance. Increased SOM decomposition at higher temperatures has been reported with higher sensitivity of labile C fraction (Liski et al. 2000; Rey and Jarvis 2006), recalcitrant C fraction (Leifeld and Fuhrer 2005; Hartley et al. 2008), or with no specific soil C fraction (Giardina and Ryan 2000; Conen et al. 2006). Furthermore, Inglett et al. (2012) observed conflicting relationships between temperature sensitivity of peat soil decomposition and two measures of C quality, namely the ligno-cellulose index (LCI) and β (derived labile C).
Complicating the effect of C quality on temperature sensitivity of decomposition is the effect of nutrients on decomposition (Billings and Ballantyne 2013). It is widely accepted that nutrient availability is one of the dominant controls on decomposition in terrestrial (Wardle 1998; Hobbie and Vitousek 2000; Torn et al. 2005) and wetland (DeBusk and Reddy 1998, 2005; White and Reddy 2000; Newman et al. 2001; Neff and Hooper 2002; Penton and Newman 2007) ecosystems. Despite this, the effect of nutrients (e.g., N or P supply) on the temperature sensitivity of SOM decomposition is largely unknown, and the simultaneous assessment of temperature effects on CO2, CH4, and DOC production rates in soils of differing C quality and nutrient availability has not been reported yet. Uncertainty in this area results from the difficulty of distinguishing the intrinsic temperature sensitivity of a given process from its apparent sensitivity realized under natural conditions.
We conducted this study to assess temperature sensitivity of organic matter decomposition in relation to C age (a surrogate for C quality) and nutrient loading in subtropical wetlands using sawgrass (Cladium jamaicense)-based freshwater peats of the Florida Everglades. We hypothesized that C quality (as characterized by age of SOM) would have a greater effect on temperature sensitivity of SOM decomposition than nutrient level and that the quality of mineralized C would change as a function of temperature. The specific objectives of this experiment were to: (i) determine temperature sensitivity of C mineralization pathways and the quantity of mineralized C (thermo-labile C) in peats of contrasting nutrient (P)-level and C quality (surface vs. deep soil or young vs. old SOM), (ii) compare the influence of C quality and nutrient (P)-level on temperature sensitivity of SOM decomposition at low and high temperature ranges, and (iii) determine the changes in C quality/availability as a function of temperature.
Materials and methods
Site description and sample collection
Soil samples were collected from two locations in Water Conservation Area 2A (WCA-2A) in the northern Everglades (Fig. S1). Historically this peatland received elevated nutrient inflows from the Everglades Agricultural Area (EAA) to the north. A distinct P enrichment gradient in soil was notable between the high-nutrient inflow site and the low-nutrient interior marsh of the WCA-2A (Craft and Richardson 1993; DeBusk et al. 2001). Having a single vegetation type (Cladium jamaicense) helped us to eliminate potentially confounding effects associated with different types of peat as evidenced by uniform isotopic signature (Inglett and Reddy 2006) and consistent C:N ratio (Table 1) as well as similar hydrology (all Cladium ridge) among all soil samples. Six intact soil cores (0–20 cm) were collected on 13 March, 2012 from two sites along P-gradient using PVC tubes (10 cm id). Soil cores were sectioned in the field in 10 cm increments, with the top section treated as surface (0–10 cm) soil while the bottom section as subsurface (10–20 cm) soil here. Thus, we have used C age characterized by young (surface soil) and old (subsurface soil) SOM as a surrogate for C quality and sites with nutrient loadings as an indicator of contrasting nutrient levels (or status).
Upon collection, the soil samples were transported to the Wetland Biogeochemistry Laboratory, University of Florida and stored at 20 °C in dark for up to 24 h before they were prepared for analyses. Our storage temperature was close to the in-situ soil temperature (15–17 °C) in the field so that the temperature or length of storage did not shift microbial structure and existing enzymes which can result in the alteration of the available substrate pool (Turner and Romero 2009). Large detrital pieces and fine roots were removed from the soil samples manually and the samples were gently homogenized avoiding disturbance to microbial functions and alteration of their microsite distribution (Teh and Silver 2006). Care was taken during sample collection, storage, and processing to ensure minimum exposure of the soil samples to oxygenated air. Field moist (saturated) soils were used in anaerobic microcosm experiments as well as for determination of soil pH, microbial biomass carbon (MBC), nitrogen (MBN), and phosphorus (MBP) and labile organic C (LOC) content. Subsamples of all soils were dried at 70 °C for 72 h and ground using a mortar and pestle for analysis of total nutrients.
Biogeochemical measurements
Soil pH was determined in DDI water using a 2:1 soil:water ratio after the equilibration of soil suspension at 25 °C for 1 h on a mechanical shaker. SOM was estimated by loss on ignition (LOI) by heating the soils at 550 °C for 5 h (Nelson and Sommers 1996). Total C and N were determined using method 3010 (USEPA 1993) on a Carlo-Erba NA-1500 CNS analyzer (Haak-Buchler Instruments, Saddlebrook, NJ). Stable C isotopic ratios (δ13C) were determined using a Finnigan MAT Delta PlusXL isotopic ratio mass spectrometer (Finnigan Corp., San Jose, CA) (Inglett and Reddy 2006). Total P was measured by ashing method of Andersen (1976) involving combustion at 550 °C followed by hydrochloric acid extraction of the ash and analysis of P by ascorbic acid colorimetric method (Method 365.4, USEPA 1993).
Microbial biomass C and N (MBC and MBN) were determined by chloroform fumigation followed by 0.5 M K2SO4 extraction. Fumigated and non-fumigated extracts were filtered using Whatman 41 followed by determination of total dissolved organic C and N using a Shimadzu TOC analyzer and TKN digestion and colorimetric analysis, respectively (Sparling et al. 1990). Microbial biomass P (MBP) was determined by chloroform fumigation followed by 0.5 M NaHCO3 extraction, persulfate digestion, and colorimetric analysis (Brookes et al. 1982). Microbial biomass C, N, and P was then calculated as the difference in concentration between the fumigated and the non-fumigated control. No extraction efficiency factor was used for MBP while, an extraction efficiency factor of 0.37 and 0.42 was used for MBC and MBN, respectively.
Labile OC (LOC) was characterized by three different extraction methods, namely cold-water extractable C (CWEC), hot-water extractable C (HWEC), and salt-extractable (0.5 M K2SO4) C (SEC) (Fang et al. 2005; Jinbo et al. 2006; Liu et al. 2006; Dodla et al. 2012). A sequential extraction process was used for the determination of CWEC and HWEC (Ghani et al. 2003). The CWEC and HWEC were determined by extraction with DDI water using a 1:10 soil:water ratio at 20 °C for 30 min and at 80 °C for 16 h, respectively, on an end-over-end shaker at 30 rpm followed by centrifugation for 20 min at 3500 rpm, filtered (0.2 µm filter) and analyzed in the same manner as for MBC. The non-fumigated K2SO4-extract C concentrations were reported as SEC. The CWEC fraction generally represents the hydrophilic fraction of C including carbohydrates, amino sugars, and low molecular weight organic acids (Fröberg et al. 2003). Likewise, SEC fractions are known to be positively associated with soluble carboxyl C compounds like organic acids and negatively associated with aromatic alkyl C compounds (Dodla et al. 2012). Carbonyl C (i.e. ketonic and aldehyde compounds), in addition to polysaccharides, also known to contribute to the HWEC fraction resulting from the ability of hot water to hydrolyze and cleave esters of various organic materials (Siskin and Katritzky 1991; Stange et al. 2001).
Anaerobic microcosms
Four replicates of twelve field samples were anaerobically incubated in 50 mL serum tubes by flooding 5 g dry weight equivalent of homogenized soils with 10 mL of N2 purged distilled de-ionized (DDI) water (1:2 soil: water ratio). The serum tubes were sealed with butyl rubber stoppers, crimped with aluminum seals (Wheaton, Millville, NJ), and purged with ultrapure N2 gas through a stopcock-septa assembly at the top for approximately 10 min. Four soil-free controls were included to account for background concentrations of different C fractions, which were negligible, compared to that produced from the soil.
The experiment was conducted by sequentially increasing incubation temperature by 5 °C at each step over a range of 15–30 °C to assess the proportion of C considered available at a given temperature (i.e. thermo-labile C). In other words, we considered 15 °C as our control temperature and employed sequential warming in equal intervals such that our warmed temperatures are 5, 10, and 15 °C higher than the control condition. Duration of the whole experiment was around four months. It is important to note that the length of incubations tended to decrease with each sequential warming treatment (i.e. length of incubations were 42, 30, 27, and 21 days for 15, 20, 25, and 30 °C temperatures, respectively) resulting from quicker adjustment (i.e. equilibration) of warming response of SOM decomposition with increasing temperature.
Production estimates of decomposition products were determined from these serum tubes by periodic sampling of headspace CO2 and CH4 (every third day), and DOC (every fifth-seventh day) in the water column. Linear rates of CO2, CH4, and DOC productions were obtained after cessation of the lag phase (~1–2 days for CO2, and ~3–5 days for CH4) using regression analysis applied to the linear portion of the cumulative concentrations of CO2 and CH4 over time. Gaseous C production rates from each temperature incubations were calculated from 12–15 points (~82–85% of observations at each step, 0.87 ≥ R 2 ≤ 0.96, p < 0.0001), while DOC production rates were calculated from 3–4 points (~75–80% of observations at each step, 0.82 ≥ R2 ≤ 0.91, p = 0.002). We assumed all mineralized C passes through the dissolved phase, and thus use the term total dissolved organic C (TDOC) to represent the total OC decomposed (the sum of CO2, CH4, and DOC) measured at any given temperature. The amount of CO2 and CH4 in the dissolved phase was also calculated using Henry’s law (Yaws and Yang 1992). During the experimental period, an equivalent amount of N2 purged DDI water was replaced to compensate for the amount of DOC samples removed each time. We also monitored the pH of DOC on a weekly basis, which didn’t change significantly over the course of the experiment resulting from flushing of the serum tubes with ultrapure N2 after each step of temperature ramping.
CO2 and CH4 from the anaerobic microcosms were measured on a Shimadzu 8A gas chromatograph (GC) (Shimadzu Scientific Instruments Inc., Columbia, MD) fitted with a thermal conductivity detector (TCD) and a flame ionization detector (FID), respectively. Calibration curves for both gases were prepared using 1% standard gas mixtures (Scotty Specialty Gases, Plumsteadville, PA). DOC samples were determined in a similar manner as for HWEC/CWEC.
Temperature sensitivity
Temperature sensitivity of any biological reaction can be estimated by determining Q10 function which is calculated as a value or factor in which a reaction rate is altered over a 10 °C temperature range. Q10 for C fractions (CO2, CH4, and DOC) were calculated using Eq. 1:
where k is derived from an exponential relationship (R 2 of the fit was always ≥0.90) between the SOM decomposition rate (expressed as production of DOC, CO2, and CH4) and temperature as follows:
where Y is the rate of production (at each temperature), β and k are derived values and t is the temperature (°C). The y-intercept of Eq. 2 (i.e. base respiration or β) can provide an index of the inherent lability of the C substrates undergoing decomposition such that a higher β value equates to the presence of more bio-available C substrates while a lower β value indicates the prevalence of relatively recalcitrant C substrates (Fierer et al. 2005). Thus, the term β served as a derived C quality parameter in parallel to the water (CWEC and HWEC) and salt extracted C (SEC) fractions.
Spectral characteristics of DOC
DOC quality was determined using UV–Visible spectroscopic measurements, i.e. spectral slope analysis (Twardowski et al. 2004) and specific UV absorbance (SUVA254). Spectral slope ratio (SR) was used as a proxy for the composition of DOC including fulvic acid to humic acid ratio, molecular weight (MW), and aromaticity (Spencer et al. 2010; Fichot and Benner 2012). Spectral slope (S) was calculated by fitting to an exponential function to the absorption spectrum over 275–295 and 350–400 nm range as follows:
where A = absorbance, a = absorption coefficient (m−1), λ = wavelength (m), λ ref = wavelength (m) and l = path length (m). SR was calculated as the ratio of 275–295 nm slope (S275–295) to 350–400 nm slope (S350–400) (Helms et al. 2008). These ranges of spectra were chosen based on the higher sensitivity of this region to changes in DOM source and processing, and occurrence of lower errors due to higher absorption coefficients at shorter wavelengths (Spencer et al. 2007).
Additionally, SUVA254 was measured by following USEPA Method 415.3 (Potter and Wimsatt 2005). SUVA254 values (L mg C−1 m−1) were determined by dividing the absorption coefficient at λ = 254 nm by the DOC concentration. SUVA254 is also widely used as a proxy for DOC aromaticity (Weishaar et al. 2003).
Statistical analysis
The experiment was carried out as a split–split plot design, with nutrient level (i.e. site) as the whole plot factor, C age (i.e. depth) as the split plot factor, and temperature as the split–split plot factor (SAS 9.3, SAS Institute, Cary, NC, USA). We fitted a linear mixed model using PROC GLIMMIX, where treatment (nutrient level, C age, and temperature) effects were fixed and error terms for whole plot, split plot, and split–split plot were random. Repeated measurements were taken on the same soil samples over four temperature levels and the residual errors across the temperature levels were grouped by nutrient level (or site) with an auto-regressive (AR-1) covariance structure. Tukey’s multiple comparison procedure, as well as the corresponding letter grouping method, were used to separate the treatment means. Regressions were conducted using PROC PHREG procedure. All statistical analyses were done at 5% significance level (α = 0.05).
Because temperature is a variable in the calculation of Q10, redundancy analyses (RDA) were performed using R, version 3.0.1 (R Development Core Team, 2013) to determine which of the explanatory variables (nutrient level/status and C age/C quality) has more influence on Q10 of decomposition products. In this analysis, the projection of a point onto a line for the response variables (Q10 of CO2, CH4, and DOC) at right angle approximates the value of the corresponding variable of the observations, while the angles between lines of response variables and lines of explanatory variables (from centroid positions) represent a two-dimensional approximation of correlations.
Results
The biogeochemical characteristics of the wetland soils used in this study varied among samples collected from different sites and depths (Table 1). The pH of the soils was slightly alkaline ranging from 7.4 ± 0.3 in subsurface soil from low P site to 7.8 ± 0.4 in surface soil from high P site. Total C and N, and soil organic matter (estimated by LOI) values did not differ appreciably among the soils used in this study. The C:N ratios and δ13C values were also not significantly different among all soil samples, which indicated that initial SOM has the same source (i.e. Cladium peat) while the difference in P-concentration was identified from the range of N:P ratios. The surface soil of high P site exhibited significantly higher MBC (9.5 ± 0.1 g kg−1) compared to that in the subsurface soil of low P site (7.8 ± 0.3 g kg−1) while MBN and MBP were significantly different among the four soil types (p < 0.05).
Total P (p < 0.05, Table 1) and NaHCO3 extractable P (p < 0.05, data not shown) were significantly higher in soils from high P sites than low P sites. In contrast, there appeared to be a general trend in that the LOC parameters (SEC, CWEC, and HWEC) in the surface soil layers of these wetland sites were significantly higher than subsurface layers (p < 0.05, Table 1). Interestingly, independent metrics of C quality parameters, i.e. measured (extraction-based LOC) and derived (β) C quality parameters for SOM as well as DOC quality parameters (SR and SUVA), used in this study were found to correlate with each other (Table S1), where SEC, HWEC, β, and initial DOC quality parameters (measured at 15 °C) related more closely to each other (0.86 ≥ R 2 ≤ 0.97) than with HWEC (0.44 ≥ R 2 ≤ 0.54). Additionally, significant (but negative) correlations were observed between SR and SUVA254 (R 2 = 0.73, p < 0.0001) values measured over the entire temperature spectra.
Analysis of variance (ANOVA) indicated that main effects of nutrient level (or status), C age (i.e. depth), and temperature were significant on the decomposition products (CO2, CH4, and DOC) at 5% (α = 0.05) significance level (Table 2). However, stronger effect of C age (p < 0.0001) and temperature (p < 0.0001) were evident on gaseous and dissolved C fractions when compared with that of nutrient level (p < 0.05). These findings suggested an interactive role of C age and temperature for CO2 (p < 0.0001), CH4 (p < 0.0001), and DOC (p < 0.01) production.
The release of C increased with the sequential increase of temperature in all soils (Table S2, Fig. 1). We used the term thermo-labile C here to quantify the fraction of C i.e. labile/available at a particular temperature. On average, as a percentage of total soil C, thermo-labile C increased from 0.72% to 2.57% and 0.45% to 1.39% in surface and subsurface soil, respectively over 15–30 °C temperature range (Fig. 1a). Likewise, thermo-labile C also increased from 0.68% to 2.26% and 0.49% to 1.70% in high P and low P soil, respectively (Fig. 1b). As a percentage of total soil C, production of thermo-labile C was significantly different between surface and subsurface soil (p < 0.05) at four temperature levels but no difference was observed between High P and Low P sites(Fig. 1a,b).
Methanogenesis was the most temperature sensitive process (Q10 ~ 14), followed by respiration (Q10 ~ 2.5), and least for DOC (Q10 ~ 1.7) (Table 3). Correspondence of higher Q10 values with lower β (derived labile parameter) confirmed that recalcitrant C was more temperature sensitive. Most of the variation in Q10 of decomposition products was explained by the C age (or depth) and associated C quality, as shown on the first RDA axis (Fig. 2); where the subsurface soil (i.e. old SOM) had the strongest effect overall as indicated by the small angles between lines of response variables (i.e. Q10 of CO2, CH4, and DOC) and line of the explanatory variable subsurface soil (if drawn from its centroid position).
CO2–C and CH4–C increased as a percentage of TDOC while the proportion of DOC decreased with temperature in all soils (Fig. 3a–f). Higher temperature also affected the form of C released, favoring methanogenesis disproportionately relative to CO2-respiration (Fig. 4a). The differences between surface and subsurface soil were more evident at higher temperatures (e.g., 25 and 30 °C, p < 0.05). In contrast, there were no significant differences in the proportions of C forms in TDOC between High P and Low P soils.
Overall, surface soil from both sites exhibited significantly higher spectral slope ratio i.e. SR (275–295 nm slope to 350–400 nm slope) and significantly lower SUVA254 compared to that in subsurface soil at all temperatures. While, SR showed an increasing trend with the corresponding increase in temperature in all soils (Fig. 4b), SUVA254 values showed a declining trend with warming (Fig. 4c). In surface layer soils, slope ratios increased from 0.86, 0.77 at 15 °C to 1.06, 1.45 at 30 °C in high P and low P site, respectively. In subsurface soil layers, slope ratios increased from 0.50 and 0.46 at 15 °C to 0.68 and 0.62 at 30 °C in high P and low P sites, respectively. On the other hand, SUVA254 decreased from 3.20, 3.53 at 15 °C to 2.17, 2.42 at 30 °C in surface soils from high P and low P site, respectively. In subsurface soil layers, SUVA254 decreased from 3.93, 4.26 at 15 °C to 3.01, 3.31 at 30 °C in soils from high P and low P site, respectively.
To explore the influence of C quality and nutrient (N and P) supply, stepwise multiple regression models were developed (forward process) for the rates of SOM decomposition products. At 15 °C, the importance of C quality parameters (SEC, CWEC, and HWEC) on the production of CO2, CH4, and DOC was evident. While, regression results at 30 °C consistently identified nutrients (i.e. C:P, NaHCO3 extractable P, and K2SO4 extractable N) as dominant variables explaining SOM mineralization rates (Table 4). Multiple regression results for 20 °C were similar to the 15 °C models while those for 25 °C had similar response to that of the 30 °C models (data not shown).
Discussion
Many wetland studies have evaluated the effect of temperature (Gorham 1991; Moore 2001; Mitsch et al. 2013; Melton et al. 2013), C quality (Moore and Knowles 1990; Bridgham and Richardson 1992; Karhu et al. 2010; Inglett et al. 2012), and nutrients (Davis 1991; Craft and Richardson 1993; DeBusk and Reddy 2003) on SOM decomposition. In contrast, relatively few studies have evaluated the interactive effect of C quality and temperature (Hartley and Ineson 2008; Inglett et al. 2012), and to our knowledge, no studies have compared the relative interaction of temperature with C quality and nutrient availability on all decomposition products (i.e. CO2, CH4, and DOC) in wetlands. In this study, we used peat soils from a single vegetation type (Cladium jamaicense) to allow better separation of the effects of C quality (characterized by C age), nutrient availability, temperature, and their interactions on anaerobic C processing (Bridgham et al. 1996).
As observed in other studies, we consistently found that temperature and C quality (i.e. C age) were the dominant factors controlling decomposition rate, while the effect of nutrient (P) level was always secondary to C quality, as evidenced by the ANOVA model (Table 2). The importance of C quality was also reflected in the strong association of C age (or soil depth) with Q10 of decomposition products (Fig. 2), as well as the differences between surface (i.e. young SOM) and deep (i.e. old SOM) soils in the proportional production of CO2–C (Fig. 3a), CH4–C (Fig. 3c), and DOC (Fig. 3e) (as % of TDOC) which was particularly evident at higher temperature.
Our observation of the high temperature dependence of production of the various C forms i.e. CO2, CH4, and DOC is another commonly reported finding (Inglett et al. 2012; Taggart et al. 2012). Overall, production of thermo-labile C increased at higher temperature, where on average TDOC increased approximately 3 fold in all soils from 15 to 30 °C (Fig. 1). Similar amount of increase in the C pool sizes with warming have been reported previously by others (Macdonald et al. 1995; Zogg et al. 1997). Of the forms of these released C, the relative trend in temperature sensitivity (Q10) followed the trend of CH4 ≫ CO2 > DOC (Table 3), which is also similar with that reported by others (Tsutsuki and Ponnamperuma 1987; Updegraff et al. 1995; Neff and Hooper 2002).
Patterns of C forms demonstrate more gaseous C production at higher temperature. For instance, production rates of gaseous C relative to DOC, i.e. (CO2–C + CH4–C):DOC ratios increased 1.8–2.3 times in all soils from 15 to 30 °C. Furthermore, the decreased proportion of CO2–C:CH4–C ratio implied increased C channeling through the methanogenic pathway at higher temperatures (Fig. 4a). Our measured Q10 of CO2 production is in agreement with Waddington et al. (2001), however, our measured Q10 of CH4 production observed in this study was comparatively higher than that reported by Van Hulzen et al. (1999) and Inglett et al. (2012) (Q10 ~ 2–4), but comparable to Gujer and Zehnder (1983), Tsutsuki and Ponnamperuma (1987), Schütz et al. (1990), and Megonigal and Schlesinger (2002) (Q10 ~ 7–16). Higher Q10 of CH4 as compared to CO2 in our system is not a function of electron acceptor competition of being reduced with warming as suggested by Segers (1998). Rather it is more likely the result of a cascading temperature effect on C flow where either fermenters are stimulated (Bridgham et al. 1995), or there is a simultaneous stimulation of fermenters and methanogens at higher temperature (Larionova et al. 2007).
Like some other studies, we also observed that old SOM (i.e. deep soil), here used as a surrogate of recalcitrant C, was more sensitive to elevated temperature (Table 3). It is generally believed that higher temperature allows more decomposition of refractory C through stimulation of enzyme reactions (Sinsabaugh and Linkins 1993; Bosatta and Ågren 1999; Fierer et al. 2005; Wagai et al. 2013; Liang et al. 2015). Additionally, inherent decomposability (β values) were strongly negatively correlated with Q10 of CO2 (P < 0.01), CH4 (P = <0.01), and DOC (P < 0.05), respectively (data not shown). Therefore, the warming-induced increase of C availability allowed greater flow through the methanogenic community, which was observed to be more pronounced with increasing organic matter recalcitrance.
Many biogeochemical reactions accelerate as temperature increases (Kadlec and Reddy 2001) and a number of mechanisms have been proposed to explain apparent increases in C availability with warming. Elevated temperature can stimulate C-decomposition via amplifying cellulose and chitin-degradation (Nie et al. 2013), chemical changes in conformation or C compound solubility (Davidson and Janssens 2006), or shifts in microbial community composition (Waldrop and Firestone 2004; Andrews et al. 2000). Also, isoenzymes of different temperature optima are maintained by both individual microorganisms (Hochachka and Somero 2002) and communities (Grzymski et al. 2008), and warming may increase production of enzymes with greater conformational stability and function (reviewed by Conant et al. 2011; Wallenstein et al. 2011).
In addition to biochemical processes, apparent temperature sensitivity of a reaction is also dependent on biophysical factors. For example, higher temperature can enhance dissolution and diffusion of C substrates to enzyme’s active sites, thus increasing substrate availability to soil microbes (Xu and Saiers 2010; Davidson et al. 2012). Higher temperature could also favor desorption of SOM–humate complexes (Ten Hulscher and Cornelissen 1996; Davidson and Janssens 2006), which could accelerate SOM decomposition by reducing physical protection (Conant et al. 2011; Schmidt et al. 2011). Elevated temperatures may also lead to an increased solubilization of substrates like waxes and lipids from the membranes of the dead microorganisms (Davidson and Janssens 2006). For example, Dodla et al. (2012) and Wang and Wang (2007) demonstrated a primary effect of increased temperature on direct solubilization of C in terms of HWEC and CWEC. In our study, HWEC was 7–12 times greater than CWEC and there was a relatively weak correlation of HWEC with other LOC fractions (SEC and CWEC) that mostly represent carboxyl-C containing compounds like polysaccharides (Table S2), suggesting that elevated temperature may also have increased C solubilization either by hydrolyzing ester linkages (Siskin and Katritzky 1991; Stange et al. 2001) or by desorption of occluded C compounds (Ghani et al. 2003; von Lüzow et al. 2007) in these soils.
As a result of both biogeochemical and biophysical mechanisms, total C mineralized increases as well as the residual DOC pool (Table S1) with warming. However, at higher temperature, proportionally more DOC pool was made available for gaseous C production, suggesting much of this dissolved pool made available (i.e. of better quality) for microbial utilization at higher temperature. In support of this, we observed increases in slope ratio (SR) of DOC from 19–47% over the range of 15–30 °C, however, the increase was 19–26% for most of the soil, with an exceptionally higher increase for the low P surface soil at 30 °C (Fig. 4b). Additionally, we observed decreases in SUVA254 from 22–32% over the range of 15–30 °C (Fig. 4c). Bianchi et al. (2013) observed strong positive correlations of SUVA254 with dissolved lignin and humification index in a riverine study. Additionally, Osburn and Stedmon (2011) further indicated that absorption coefficients measured at 300 nm strongly predicted dissolved lignin concentrations in a marine ecosystem. These observations suggests warming induced increases in SR and decreases in SUVA254 in our study may represent either degradation of complex phenolic compounds (Zhao 2012) or release of other water soluble, extracellular substances excreted by microorganisms resulting from cell death and lysis (i.e. freshly produced or less condensed aromatics of microbial origin) in DOC pool (Birdwell and Engel 2010; Tfaily et al. 2015).
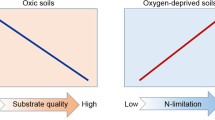
Our observation of increased C bioavailability as a function of temperature is in contrast to many studies in terrestrial systems, where depletion of labile C substrates was identified as the key driver for reduced soil respiration in short-term laboratory incubation studies (Fang et al. 2005; Hartley et al. 2008; Tucker et al. 2013). Within this context, findings of some theoretical models (Kirschbaum, 2004; Eliasson et al., 2005; Knorr et al., 2005) also suggested that rates of microbial respiration will acclimate on a longer time scale resulting from a change in the composition of the remaining SOM pool after the C pools with shorter turnover times are preferentially lost. In our study, as C availability increased (both quantity and apparent quality) under warming, it is likely that C limitation of respiration was less at higher temperatures. Thus, nutrient availability could limit decomposition at higher temperature due to the stoichiometric control of microbial nutrient demand (Allison and Vitousek 2005; Geisseler and Horwath 2009; Sinsabaugh et al. 2009; Hernández and Hobbie 2010; Billings and Ballantyne 2013). In support of this, stepwise multiple regressions demonstrated that at lower temperatures (15 and 20 °C), SOM decomposition was better predicted by C quality, while at higher temperatures (25 and 30 °C), decomposition became more influenced by nutrient parameters (Table 4).
The positive coefficients of NaHCO3 extractable P and the large negative coefficients of C:P in models for the high temperature range were indicative of a potentially P-limited decomposition process, an observation which is not surprising, given the high degree of P-limitation in the Everglades (Craft and Richardson 1993; DeBusk et al. 2001). With regards to anaerobic methanogenic C cycling, studies have also reported that P availability directly limited fermentation and CH4 production in low P Everglades soils (reviewed by Medvedeff et al. 2014). Secondary to P, N was also identified in the high-temperature multiple regression models. Although little studied, N has also received support as a limiting nutrient in the Everglades, either in activities of enzymes (Penton and Newman 2007), N2 fixation (Inglett et al. 2004, Liao and Inglett 2014), or in peat dynamics (Inglett et al. 2011; Wozniak et al. 2012) across the Everglades landscape. The temperature-driven decoupling of the C-limited and nutrient-limited decomposition in our peat-based system is qualitatively similar to the observation in an anoxic marine system. Weston and Joye (2005) also reported a sudden change in the microbial C processing above 20 °C resulting from a variable temperature response of anaerobic microbial metabolisms in the marine sediment.
We acknowledge that our study used only a relative measure of temperature sensitivity (Q10), and although Q10 is a widely used parameter, it could be biased if used over a larger temperature range as the measurement is itself temperature dependent (Lloyd and Taylor 1994; Hamdi et al. 2013). Additionally, we did not include the indirect effect of temperature mediated by the temperature-induced increase in diffusion of soluble substrates and C allocation to aboveground and belowground plant tissues which can alter substrate concentrations at enzyme’s active site and thus confound the temperature effect on the apparent response of the soil microbial process (Davidson et al. 2006). While these extrinsic temperature effects are extremely important on an ecosystem scale response, it is beyond the scope of this paper, and our results highlight the interacting effect of temperature with C age and nutrient status when evaluating the intrinsic sensitivity of SOM decomposition in warmer wetlands.
Studies evaluating the sensitivity of SOM decomposition with sequential methods generally increase temperature over a very short duration, varying from few hours (Fang et al. 2005; Liu et al. 2006; Yuste et al. 2007) to few days (Koch et al. 2007). While these studies quantify the instantaneous effect of temperature on rates of C loss, longer term laboratory (Bradford et al. 2010; Tucker et al. 2013) and field (Oechel et al. 2000; Luo et al. 2001; Melillo et al. 2002) warming studies often report an attenuation of the immediate warming response with time resulting from either labile C limitation (Hartley et al. 2008), microbial physiological acclimation (Allison et al. 2010), or both (Bradford et al. 2008). To that end, our experiment accounted for not only the instantaneous warming response but also the gaseous C production rate after acclimation. Undoubtedly, an alteration of the initial microbial community structure may have contribute to the observed long-term warming response in our study (also see Bradford et al. 2010), but this longer incubation may be justified by the fact that, ultimately, it is the total amount of SOC loss (not the immediate response) that matters for assessing climate change feedbacks.
Conclusions
Our findings add to a growing body of evidence that the effect of temperature on SOM decomposition is highly interactive with C quality and, as in this study, nutrients. Overall, greater influence of C quality than nutrient availability on temperature sensitivity of dissolved and gaseous C (CO2, CH4) production evokes the need to better account for temperature effects on microbial physiological parameters (e.g. C use efficiency) in response to C and nutrients in anaerobic system models. Likewise differential C versus nutrient limitation on SOM decomposition with temperature suggests our observation of nutrient limited decomposition at higher temperatures more broadly applies to tropical ecosystems, given the fact that warmer systems already tend toward a more nutrient (especially P) limited condition than arctic/boreal systems (Fisher et al. 2012; Sistla and Schimel 2012). This indicates that anthropogenic loading of nutrients would further stimulate C losses from tropical and subtropical wetlands in the future warmer world.
Our observation of increasing C quality at higher temperature ranges disagrees with studies in terrestrial systems where short-term attenuation of respiration results from labile C-limited conditions. Rather, in anaerobic wetland soils transient responses (acclimation) of anaerobic C processing may be more associated with adaptation of microbial and enzymatic processes due to microbial nutrient limitation. Based on the results of our multiple regression models, we have identified a need for incorporation of microbial nutrient demand and enzymes to existing physiology-based, C-only models (Allison et al. 2010; German et al. 2012; Sihi et al. 2016). Integration of these microbial parameters in next-generation climate models has been suggested (Schimel 2001; Allison and Martiny 2008; Todd-Brown et al. 2012; Cotrufo et al. 2013; Wieder et al. 2013), and if accomplished, climate-C feedback would be better predicted. Therefore, we believe our findings would be useful, in conjunction with other modeling approaches, for improved understanding of global (terrestrial and wetland) C stocks.
References
Bergamaschi P, Frankenberg C, Meirink JF, Krol M, Dentener F, Wagner T, Platt U, Kaplan JO, Körner S, Heimann M, Dlugokencky EJ, Goede A (2007) Satellite chartography of atmospheric methane from SCIAMACHY on board ENVISAT: 2. Evaluation based on inverse model simulations. J Geophys Res 112(D2)
Allison SD, Martiny JB (2008) Resistance, resilience, and redundancy in microbial communities. Proc Natl Acad Sci 105:11512–11519
Allison SD, Vitousek PM (2005) Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol Biochem 37:937–944
Allison SD, Wallenstein MD, Bradford MA (2010) Soil-carbon response to warming dependent on microbial physiology. Nat Geosci 3:336–340
Andersen J (1976) An ignition method for determination of total phosphorus in lake sediments. Water Res 10:329–331
Andrews JA, Matamala R, Westover KM, Schlesinger WH (2000) Temperature effects on the diversity of soil heterotrophs and the δ13C of soil-respired CO2. Soil Biol Biochem 32:699–706. doi:10.1016/S0038-0717(99)00206-0
Bianchi TS et al (2013) Enhanced transfer of terrestrially derived carbon to the atmosphere in a flooding event. Geophys Res Lett 40:116–122
Billings SA, Ballantyne F (2013) How interactions between microbial resource demands, soil organic matter stoichiometry, and substrate reactivity determine the direction and magnitude of soil respiratory responses to warming. Glob Change Biol 19:90–102
Birdwell JE, Engel AS (2010) Characterization of dissolved organic matter in cave and spring waters using UV–Vis absorbance and fluorescence spectroscopy. Org Geochem 41:270–280
Bloom AA, Palmer PI, Fraser A, Reay DS, Frankenberg C (2010) Large-scale controls of methanogenesis inferred from methane and gravity spaceborne data. Science 327:322–325
Bosatta E, Ågren GI (1999) Soil organic matter quality interpreted thermodynamically. Soil Biol Biochem 31:1889–1891
Bradford MA, Davies CA, Frey SD, Maddox TR, Melillo JM, Mohan JE, ReynoldsJF Treseder KK, Wallenstein MD (2008) Thermal adaptation of soil microbial respiration to elevated temperature. Ecol Lett 11:1316–1327
Bradford MA, Watts BW, Davis CA (2010) Thermal adaptation of heterotrophic soil respiration in laboratory microcosms. Glob Change Biol 16:1576–1588
Bridgham SD, Richardson CJ (1992) Mechanisms controlling soil respiration (CO2 and CH4) in southern peatlands. Soil Biol Biochem 24:1089–1099. doi:10.1016/0038-0717(92)90058-6
Bridgham SD, Johnston CA, Pastor J, Updegraff K (1995) Potential feedbacks of northern wetlands on climate change. BioScience 45:262–274
Bridgham S, Pastor J, Janssens J, Chapin C, Malterer T (1996) Multiple limiting gradients in peatlands: a call for a new paradigm. Wetlands 16:45–65. doi:10.1007/BF03160645
Bridgham S, Megonigal JP, Keller J, Bliss N, Trettin C (2006) The carbon balance of North American wetlands. Wetlands 26:889–916. doi:10.1672/0277-5212(2006)26[889:TCBONA]2.0.CO;2
Brookes P, Powlson D, Jenkinson D (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329
Conant RT et al (2011) Temperature and soil organic matter decomposition rates—synthesis of current knowledge and a way forward. Glob Change Biol 17:3392–3404
Conen F, Leifeld J, Seth B, Alewell C (2006) Warming mobilises young and old soil carbon equally. Biogeosci Discuss 3:1355–1366
Cotrufo MF, Wallenstein MD, Boot CM, Denef K, Paul E (2013) The microbial efficiency-matrix stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: do labile plant inputs form stable soil organic matter? Glob Change Biol 19:988–995
Craft C, Richardson C (1993) Peat accretion and phosphorus accumulation along a eutrophication gradient in the northern Everglades. Biogeochemistry 22:133–156
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440:165–173
Davidson EA, Janssens IA, Luo Y (2006) On the variability of respiration in terrestrial ecosystems:moving beyond Q10. Glob Change Biol 12:154–164
Davidson EA, Samanta S, Caramori SS, Savage K (2012) The dual arrhenius and Michaelis-Menten kinetics model for decomposition of soil organic matter at hourly to seasonal time scales. Glob Change Biol 18:371–384
Davis SM (1991) Growth, decomposition, and nutrient retention of Cladium jamaicense Crantz and Typha domingensis Pers. in the Florida Everglades. Aquat Bot 40:203–224. doi:10.1016/0304-3770(91)90059-E
DeBusk W, Reddy K (1998) Turnover of detrital organic carbon in a nutrient-impacted Everglades marsh. Soil Sci Soc Am J 62:1460–1468
DeBusk W, Newman S, Reddy K (2001) Spatio–temporal patterns of soil phosphorus enrichment in Everglades Water Conservation Area 2A. J Environ Qual 30:1438–1446
DeBusk W, Reddy K (2003) Nutrient and hydrology effects on soil respiration in a northern Everglades marsh. J Environ Qual 32:702–710
DeBusk WF, Reddy KR (2005) Litter decomposition and nutrient dynamics in a phosphorus enriched everglades marsh. Biogeochemistry 75:217–240
Dodla SK, Wang JJ, DeLaune RD (2012) Characterization of labile organic carbon in coastal wetland soils of the Mississippi River deltaic plain: Relationships to carbon functionalities. Sci Total Environ 435:151–158
Eliasson PE, McMurtrie RE, Pepper DA, Strömgren M, Linder S, Ågren GI (2005) The response of heterotrophic CO2 flux to soil warming. Glob Change Biol 11:167–181
Fang C, Smith P, Moncrieff JB, Smith JU (2005) Similar response of labile and resistant soil organic matter pools to changes in temperature. Nature 433:57–59
Fichot CG, Benner R (2012) The spectral slope coefficient of chromophoric dissolved organic matter (S275–295) as a tracer of terrigenous dissolved organic carbon in river-influenced ocean margins. Limnol Oceanogr 57:1453–1466
Fierer N, Craine JM, McLauchlan K, Schimel JP (2005) Litter quality and the temperature sensitivity of decomposition. Ecology 86:320–326
Fisher JB, Badgley G, Blyth E (2012) Global nutrient limitation in terrestrial vegetation. Glob Biogeochem Cycles 26:GB3007 doi:10.1029/2011GB004252
Freeman C, Evans C, Monteith D, Reynolds B, Fenner N (2001) Export of organic carbon from peat soils. Nature 412:785–785
Fröberg M, Berggren D, Bergkvist B, Bryant C, Knicker H (2003) Contributions of Oi, Oe and Oa horizons to dissolved organic matter in forest floor leachates. Geoderma 113:311–322
Geisseler D, Horwath WR (2009) Relationship between carbon and nitrogen availability and extracellular enzyme activities in soil. Pedobiologia 53:87–98. doi:10.1016/j.pedobi.2009.06.002
German DP, Marcelo KR, Stone MM, Allison SD (2012) The Michaelis-Menten kinetics of soil extracellular enzymes in response to temperature: a cross-latitudinal study. Glob Change Biol 18:1468–1479
Ghani A, Dexter M, Perrott K (2003) Hot-water extractable carbon in soils: a sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil Biol Biochem 35:1231–1243
Giardina CP, Ryan MG (2000) Evidence that decomposition rates of organic carbon in mineral soil do not vary with temperature. Nature 404:858–861
Gorham E (1991) Northern peatlands: role in the carbon cycle and probable responses to climatic warming. Ecol Appl 1:182–195
Grzymski JJ et al (2008) Metagenome analysis of an extreme microbial symbiosis reveals eurythermal adaptation and metabolic flexibility. Proc Natl Acad Sci. doi:10.1073/pnas.0802782105
Gujer W, Zehnder A (1983) Conversion processes in anaerobic digestion. Water Sci Technol 15:127–167
Hamdi S, Moyano F, Sall S, Bernoux M, Chevallier T (2013) Synthesis analysis of the temperature sensitivity of soil respiration from laboratory studies in relation to incubation methods and soil conditions. Soil Biol Biochem 58:115–126
Hartley IP, Ineson P (2008) Substrate quality and the temperature sensitivity of soil organic matter decomposition. Soil Biol Biochem 40:1567–1574
Hartley IP, Hopkins DW, Garnett MH, Sommerkorn M, Wookey PA (2008) Soil microbial respiration in arctic soil does not acclimate to temperature. Ecol Lett 11:1092–1100
Helms JR, Stubbins A, Ritchie JD, Minor EC, Kieber DJ, Mopper K (2008) Absorption spectral slopes and slope ratios as indicators of molecular weight, source, and photobleaching of chromophoric dissolved organic matter. Limnol Oceanogr 53:955
Hernández D, Hobbie S (2010) The effects of substrate composition, quantity, and diversity on microbial activity. Plant Soil 335:397–411. doi:10.1007/s11104-010-0428-9
Hobbie SE, Vitousek PM (2000) Nutrient limitation of decomposition in Hawaiian forests. Ecology 81:1867–1877
Hochachka P, Somero G (2002) Biochemical adaptation, mechanism and process in physiological evolution. Oxford University Press, Oxford, p 478
Inglett PW, Reddy KR (2006) Investigating the use of macrophyte stable C and N isotopic ratios as indicators of wetland eutrophication: Patterns in the P-affected Everglades. Limnol Oceanogr 51:2380–2387
Inglett P, Reddy K, McCormick P (2004) Periphyton chemistry and nitrogenase activity in a northern Everglades ecosystem. Biogeochemistry 67:213–233
Inglett P, Rivera-Monroy V, Wozniak J (2011) Biogeochemistry of nitrogen across the Everglades landscape. Crit Rev Environ Sci Technol 41:187–216
Inglett K, Inglett P, Reddy K, Osborne T (2012) Temperature sensitivity of greenhouse gas production in wetland soils of different vegetation. Biogeochemistry 108:77–90
Jinbo Z, Changchun S, Wenyan Y (2006) Land use effects on the distribution of labile organic carbon fractions through soil profiles. Soil Sci Soc Am J 70:660–667
Kadlec RH, Reddy K (2001) Temperature effects in treatment wetlands. Water Environ Res 73:543–557
Karhu K, Fritze H, Tuomi M, Vanhala P, Spetz P, Kitunen V, Liski J (2010) Temperature sensitivity of organic matter decomposition in two boreal forest soil profiles. Soil Biol Biochem 42:72–82
Kirschbaum MUF (1995) The temperature dependence of soil organic matter decomposition, and the effect of global warming on soil organic C storage. Soil Biol Biochem 27:753–760
Kirschbaum MUF (2004) Soil respiration under prolonged soil warming: are rate reductions caused by acclimation or substrate loss? Glob Change Biol 10:1870–1877
Knorr W, Prentice IC, House JI, Holland EA (2005) Long-term sensitivity of soil carbon turnover to warming. Nature 433:298–301
Koch O, Tscherko D, Kandeler E (2007) Temperature sensitivity of microbial respiration, nitrogen mineralization, and potential soil enzyme activities in organic alpine soils. Glob Biogeochem Cycles 21:GB4017
Larionova A, Yevdokimov I, Bykhovets S (2007) Temperature response of soil respiration is dependent on concentration of readily decomposable C. Biogeosciences 4:1073–1081
Leifeld J, Fuhrer J (2005) The temperature response of CO2 production from bulk soils and soil fractions is related to soil organic matter quality. Biogeochemistry 75:433–453
Li W, Dickinson RE, Fu R, Niu GY, Yang ZL, Canadell JG (2007) Future precipitation changes and their implications for tropical peatlands. Geophys Res Lett 34:L01403. doi:10.1029/2006GL028364
Liang J et al (2015) Methods for estimating temperature sensitivity of soil organic matter based on incubation data: a comparative evaluation. Soil Biol Biochem 80:127–135
Liao X, Inglett PW (2014) Dynamics of periphyton nitrogen fixation in short-hydroperiod wetlands revealed by high-resolution seasonal sampling. Hydrobiologia 722:263–277
Liski J, Ilvesniemi H, Mäkelä A, Westman CJ (2000) Temperature dependence of old soil organic matter. Ambio 29:56–57
Liu HS, Li LH, Ha XG, Huang JH, Sun JX, Wang HY (2006) Respiratory substrate availability plays a crucial role in the response of soil respiration to environmental factors. Appl Soil Ecol 32:284–292
Lloyd J, Taylor JA (1994) On the temperature dependence of soil respiration. Funct Ecol 8:315–323
Lomander A, Kätterer T, Andrén O (1998) Carbon dioxide evolution from top- and subsoil as affected by moisture and constant and fluctuating temperature. Soil Biol Biochem 30:2017–2022. doi:10.1016/S0038-0717(98)00076-5
Luo YQ, Wan SQ, Hui DF, Wallace LL (2001) Acclimatization of soil respiration to warming in a tall grass prairie. Nature 413:622–625
MacDonald NW, Zak DR, Pregitzer KS (1995) Temperature effects on kinetics of microbial respiration and net nitrogen and sulfur mineralization. Soil Sci Soc Am J 59:233–240. doi:10.2136/sssaj1995.03615995005900010036x
Matthews E, Fung I (1987) Methane emission from natural wetlands: global distribution, area, and environmental characteristics of sources. Glob Biogeochem Cycles 1:61–86
Medvedeff CA, Inglett KS, Inglett PW (2014) Evaluation of direct and indirect phosphorus limitation of methanogenic pathways in a calcareous subtropical wetland soil. Soil Biol Biochem 69:343–345
Megonigal JP, Schlesinger W (2002) Methane-limited methanotrophy in tidal freshwater swamps. Global Biogeochem Cycles 16:1088
Megonigal J, Mines M, Visscher P (2005) Anaerobic metabolism: linkages to trace gases and aerobic processes. Biogeochemistry 8:317–424
Melillo JM, Steudler PA, Aber JD et al (2002) Soil warming and carbon-cycle feedbacks to the climate system. Science 298:2173–2176
Melton JR et al (2013) Present state of global wetland extent and wetland methane modelling: conclusions from a model inter-comparison project (WETCHIMP). Biogeosciences 10:753–788. doi:10.5194/bg-10-753-2013
Mitsch WJ, Gosselink JG (2007) In: Wetlands, 4th edn. Wiley, Hoboken
Mitsch WJ, Nahlik A, Wolski P, Bernal B, Zhang L, Ramberg L (2010) Tropical wetlands: seasonal hydrologic pulsing, carbon sequestration, and methane emissions. Wetlands Ecol Manage 18:573–586
Mitsch WJ et al (2013) Wetlands, carbon, and climate change. Landscape Ecol 28:583–597
Moore PD (2001) Wetlands. New York, NY
Moore T, Dalva M (2001) Some controls on the release of dissolved organic carbon by plant tissues and soils. Soil Sci 166:38–47
Moore T, Knowles R (1990) Methane emissions from fen, bog and swamp peatlands in Quebec. Biogeochemistry 11:45–61
Neff JC, Hooper DU (2002) Vegetation and climate controls on potential CO2, DOC and DON production in northern latitude soils. Glob Change Biol 8:872–884
Nelson DW, Sommers LE (1996) Total carbon, organic carbon, and organic matter. Methods of soil analysis Part 3, pp 961–1010
Newman S, Kumpf H, Laing J, Kennedy W (2001) Decomposition responses to phosphorus enrichment in an Everglades (USA) slough. Biogeochemistry 54:229–250
Nie M, Pendall E, Bell C, Gasch CK, Raut S, Tamang S, Wallenstein MD (2013) Positive climate feedbacks of soil microbial communities in a semi-arid grassland. Ecol Lett 16:234–241. doi:10.1111/ele.12034
Oechel WC, Vourlitis GL, Hastings SJ, Zulueta RC, Hinzman L, Kane D (2000) Acclimation of ecosystem CO2 exchange in the Alaskan Arctic in response to decadal climate warming. Nature 406:978–981
Osburn CL, Stedmon CA (2011) Linking the chemical and optical properties of dissolved organic matter in the Baltic-North Sea transition zone to differentiate three allochthonous inputs. Mar Chem 126:281–294. doi:10.1016/j.marchem.2011.06.007
Paré MC, Bedard-Haughn A (2013) Soil organic matter quality influences mineralization and GHG emissions in cryosols: a field-based study of sub-to high Arctic. Glob Change Biol 19:1126–1140
Penton CR, Newman S (2007) Enzyme activity responses to nutrient loading in subtropical wetlands. Biogeochemistry 84:83–98. doi:10.1007/s10533-007-9106-2
Potter B, Wimsatt J (2005) Determination of total organic carbon and specific UV absorbance at 254 nm in source water and drinking water. EPA Document: EPA
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org/
Rey A, Jarvis P (2006) Modelling the effect of temperature on carbon mineralization rates across a network of European forest sites (FORCAST). Glob Change Biol 12:1894–1908
SAS software, Version (9.3) of the SAS System for Windows. Copyright © (2010) SAS Institute Inc., Cary, NC, USA
Schimel J (2001) 1.13-Biogeochemical models: implicit versus explicit microbiology. Global biogeochemical cycles in the climate system, pp 177–183
Schmidt MWI et al (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:49–56
Schütz H, Seiler W, Conrad R (1990) Influence of soil temperature on methane emission from rice paddy fields. Biogeochemistry 11:77–95
Segers R (1998) Methane production and methane consumption: a review of processes underlying wetland methane fluxes. Biogeochemistry 41:23–51
Sihi D, Gerber S, Inglett PW, Inglett KS (2016) Comparing models of microbial-substrate interactions and their response to warming. Biogeosciences 13:1–20
Sinsabaugh RL, Linkins AE (1993) Statistical modeling of litter decomposition from integrated cellulase activity. Ecology 74:1594–1597. doi:10.2307/1940087
Sinsabaugh RL, Hill BH, Shah JJF (2009) Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 462:795–798
Siskin M, Katritzky AR (1991) Reactivity of organic compounds in hot water: geochemical and technological implications. Science 254:231–237
Sistla SA, Schimel JP (2012) Stoichiometric flexibility as a regulator of carbon and nutrient cycling in terrestrial ecosystems under change. New Phytol 196:68–78. doi:10.1111/j.1469-8137.2012.04234.x
Sparling GP, Feltham CW, Reynolds J, West AW, Singleton P (1990) Estimation of soil microbial C by a fumigation-extraction method: use on soils of high organic matter content, and a reassessment of the kEC-factor. Soil Biology and Biochemistry 22(3):301–307
Spencer RG et al (2007) Diurnal variability in riverine dissolved organic matter composition determined by in situ optical measurement in the San Joaquin River (California, USA). Hydrol Process 21:3181–3189
Spencer RG, Hernes PJ, Ruf R, Baker A, Dyda RY, Stubbins A, Six J (2010) Temporal controls on dissolved organic matter and lignin biogeochemistry in a pristine tropical river, Democratic Republic of Congo. J Geophys Res Biogeosci 2005–2012:115
Stange RR Jr, Raph J, Peng J, Sims JJ, Midland SL, McDonald RE (2001) Acidolysis and hot water extraction provide new insight into the composition of the induced “lignin-like” material from squash fruit. Phytochemistry 57:1005–1011
Strack M (2008) Peatlands and climate change. IPS, International Peat Society
Sulman B, Desai A, Cook B, Saliendra N, Mackay D (2009) Contrasting carbon dioxide fluxes between a drying shrub wetland in Northern Wisconsin, USA, and nearby forests. Biogeosciences 6:1115–1126
Taggart M, Heitman JL, Shi W, Vepraskas M (2012) Temperature and water content effects on carbon mineralization for sapric soil material. Wetlands 32:939–944. doi:10.1007/s13157-012-0327-3
Teh YA, Silver WL (2006) Effects of soil structure destruction on methane production and carbon partitioning between methanogenic pathways in tropical rain forest soils. J Geophys Res Biogeosci 2005–2012:111
Ten Hulscher TE, Cornelissen G (1996) Effect of temperature on sorption equilibrium and sorption kinetics of organic micropollutants—a review. Chemosphere 32:609–626
Tfaily MM et al (2015) Utilization of PARAFAC-modeled excitation-emission matrix (EEM) fluorescence spectroscopy to identify biogeochemical processing of dissolved organic matter in a Northern Peatland. Photochem Photobiol 91:684–695
Todd-Brown KE, Hopkins FM, Kivlin SN, Talbot JM, Allison SD (2012) A framework for representing microbial decomposition in coupled climate models. Biogeochemistry 109:19–33
Torn MS, Vitousek PM, Trumbore SE (2005) The influence of nutrient availability on soil organic matter turnover estimated by incubations and radiocarbon modeling. Ecosystems 8:352–372
Tsutsuki K, Ponnamperuma FN (1987) Behavior of anaerobic decomposition products in submerged soils: Effects of organic material amendment, soil properties, and temperature. Soil Sci Plant Nutr 33:13–33
Tucker CL, Bell J, Pendall E, Ogle K (2013) Does declining carbon-use efficiency explain thermal acclimation of soil respiration with warming? Glob Change Biol 19:252–263
Turner BL, Romero TE (2009) Short-term changes in extractable inorganic nutrients during storage of tropical rain forest soils. Soil Sci Soc Am J 73:1972–1979
Twardowski MS, Boss E, Sullivan JM, Donaghay PL (2004) Modeling the spectral shape of absorption by chromophoric dissolved organic matter. Mar Chem 89:69–88
Updegraff K, Pastor J, Bridgham SD, Johnston CA (1995) Environmental and substrate controls over carbon and nitrogen mineralization in northern wetlands. Ecol Appl 5:151–163
USEPA (1993) Methods for chemical analysis of water and wastes. Environmental Monitoring Support Lab, Cincinnati
Van Hulzen J, Segers R, Van Bodegom P, Leffelaar P (1999) Temperature effects on soil methane production: an explanation for observed variability. Soil Biol Biochem 31:1919–1929
von Lüzow M, Kögel-Knabner I, Ekschmitt K, Flessa H, Guggenberger G, Matzner E et al (2007) Soil fractionation methods: relevance to functional pools and stabilization mechanisms. Soil Biol Biochem 39:2183–2207
Waddington J, Rotenberg P, Warren F (2001) Peat CO2 production in a natural and cutover peatland: implications for restoration. Biogeochemistry 54:115–130
Wagai R, Kishimoto-Mo AW, Yonemura S, Shirato Y, Hiradate S, Yagasaki Y (2013) Linking temperature sensitivity of soil organic matter decomposition to its molecular structure, accessibility, and microbial physiology. Glob Change Biol 19:1114–1125
Waldrop M, Firestone M (2004) Altered utilization patterns of young and old soil C by microorganisms caused by temperature shifts and N additions. Biogeochemistry 67:235–248
Waldrop MP, Wickland KP, White Iii R, Berhe AA, Harden JW, Romanovsky VE (2010) Molecular investigations into a globally important carbon pool: Permafrost-protected carbon in Alaskan soils. Glob Change Biol 16:2543–2554
Wallenstein M, Allison SD, Ernakovich J, Steinweg JM, Sinsabaugh R (2011) Controls on the temperature sensitivity of soil enzymes: a key driver of in situ enzyme activity rates. In: Soil enzymology. Springer, pp 245–258
Wang Q, Wang S (2007) Soil organic matter under different forest types in Southern China. Geoderma 142:349–356
Wardle DA (1998) Controls of temporal variability of the soil microbial biomass: a global-scale synthesis. Soil Biol Biochem 30:1627–1637
Weishaar JL, Aiken GR, Bergamaschi BA, Fram MS, Fujii R, Mopper K (2003) Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ Sci Technol 37:4702–4708. doi:10.1021/es030360x
Weston NB, Joye SB (2005) Temperature-driven decoupling of key phases of organic matter degradation in marine sediments. Proc Natl Acad Sci USA 102:17036–17040. doi:10.1073/pnas.0508798102
White J, Reddy K (2000) Influence of phosphorus loading on organic nitrogen mineralization of Everglades soils. Soil Sci Soc Am J 64:1525–1534
Wieder WR, Bonan GB, Allison SD (2013) Global soil carbon projections are improved by modelling microbial processes. Nat Clim Change 3:909–912
Xu N, Saiers JE (2010) Temperature and hydrologic controls on dissolved organic matter mobilization and transport within a forest topsoil. Environ Sci Technol 44:5423–5429. doi:10.1021/es1002296
Yavitt JB, Lang GE (1990) Methane production in contrasting wetland sites: response to organic-chemical components of peat and to sulfate reduction. Geomicrobiology J 8:27–46
Yavitt J, Williams C, Wieder R (2005) Soil chemistry versus environmental controls on production of CH4 and CO2 in northern peatlands. Eur J Soil Sci 56:169–178
Yaws CL, Yang HC (1992) Henry’s law constant for compounds in water. In: Yaws CL (ed) Thermodynamic and physical property data. Gulf Publ. Co., Houston, pp 181–206
Yuste JC, Baldocchi DD, Gershenson A, Misson L, Wong S (2007) Microbial soil respiration and its dependency on carbon inputs, soil temperature and moisture. Glob Change Biol 13:2018–2035
Zhao L (2012) Spectral characteristics of dissolved organic matter released during the metabolic process of small medusa. Spectrosc Spect Anal 32:1584–1587
Zogg GP, Zak DR, Ringelberg DB, White DC, MacDonald NW, Pregitzer KS (1997) Compositional and functional shifts in microbial communities due to soil warming. Soil Sci Soc Am J 61:475–481
Acknowledgements
The authors would like to thank Matt Norton, Yu Wang, Biswanath Dari, and Swati Goswami for their laboratory and field assistance. We would like to give a special thanks to Dr. Mihai Giurcanu for assisting in statistical analysis. The project was supported by National Science Foundation (NSF) Grant DEB 0841596. We also thank two anonymous reviewers for their constructive comments which helped us significantly improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Edward Brzostek.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sihi, D., Inglett, P.W. & Inglett, K.S. Carbon quality and nutrient status drive the temperature sensitivity of organic matter decomposition in subtropical peat soils. Biogeochemistry 131, 103–119 (2016). https://doi.org/10.1007/s10533-016-0267-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-016-0267-8