Abstract
The present study aimed to investigate the role of four non-native invertebrates in supporting fish biomass as well as their influence on the carbon flow into the Volta Grande reservoir food web. The fish samples were carried out quarterly between October 2015 and July 2016 using gillnets. At the sampled sites, four non-native invertebrates (golden mussel, Asian clam, trumpet snail and Amazonian prawn), which are potential prey for fish in the Volta Grande reservoir, were collected by targeted sampling using a Petersen-type bottom dredger and semi-circular sieves. The gut contents of the fish were collected and analyzed under stereoscope, and samples of muscle tissue of these fish, as well as the four non-native invertebrate species sampled, were submitted for isotopic analysis. Results obtained by the present study, by both gut content and stable isotopic analyses, pointed to a trophic structure where non-native species represent not only a strong component of the fish community, but also their main carbon source. Based on gut contents and isotopic mixing models, we found that together, non-native prey are essential carbon sources for the fish fauna, fuelling more than 40.0% of the biomass in four dominant fish species. The consumption rate of non-native bivalves by the native omnivorous fish Leporinus friderici suggested these filter-feeding organisms potentially constitute an important trophic connection between littoral consumers and pelagic energy sources. In addition, non-native prey were also prominent carbon sources for non-native fish, fuelling more than half of the biomass in peacock bass and silver croaker, suggesting these prey have a fundamental role in maintaining non-native fish populations in this system. Our results may help to understand fundamental ecological issues bringing to light the extent to which these new combinations of species influence the energy flow and ecosystem properties of the Volta Grande reservoir.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding environmental changes caused by the introductions of non-native species has been focus of ecological studies since the late nineteenth century (Goeze 1882). Non-native species can cause reductions in species richness and diversity (Latini and Petrere 2004), predation pressure (Pelicice et al. 2015), competition (White et al. 2006), extinctions (Pyšek et al. 2016), biotic homogenization (Vitule et al. 2012) and changes in biogeochemical cycles (Glibert et al. 2011). In aquatic environments, several studies have investigated changes in abundance and composition of native biota caused by the introduction of non-native predators, especially by fish (Moyle and Light 1996; Bryan et al. 2002; Beisner et al. 2003; Pelicice and Agostinho 2009; Porter-Whitaker et al. 2012; Green et al. 2012; Sharpe et al. 2017). On the other hand, the influences of non-native prey on predators have received less attention (Magoulick and Lewis 2002; Locke et al. 2014; Puntila-Dodd et al. 2019). Evidence has shown that non-native prey can provide food resources for fish (Puntila-Dodd et al. 2019; González-Bergonzoni et al. 2020), especially in disturbed environments such as developed lakes (Twardochleb and Olden 2016) and reservoirs (Rosa et al. 2015). Thus, understanding how non-native prey affects predators is critical, not only to determine the success of the invasion and new trophic interactions, but also to identify their implications at the ecosystem level (Pintor and Byers 2015).
The effects of non-native species introductions may be either direct or indirect. One introduced species will eat and/or will be eaten by resident species, thus creating new trophic links which may have their effects propagated for entire ecosystems (Courchamp et al. 2003; David et al. 2017). In aquatic food webs, for instance, non-native predators may exclude native species through direct predation (Vander Zanden et al. 1999; Latini and Petrere 2004; Pelicice et al. 2015). However, direct predation may also have effects that reach other levels of the food web. The introduction of two non-native piscivorous fish in natural lakes in Brazil (Cichla sp. and Pygocentrus nattereri) has led to the local extinction of small-sized fish species, and the consumption of fish by the native top predator (Hoplias malabaricus) was partly replaced by aquatic invertebrates (Pompeu and Godinho 2001). On the other hand, non-native prey can complement native fish diets due to decreases in their usual prey or by their high caloric value (Dijkstra et al. 2013; Pintor and Byers 2015; Hostert et al. 2019). However, a simple diet shift caused by the non-native prey availability may have prominent influences on the food web trophic dynamics. For example, predation on invasive zebra mussels (Dreissena polymorpha) substantially changed the carbon flow distribution among species in the Lake Oneida food web, reinforcing benthonic pathways to the detriment of the pelagic one (Miehls et al. 2009). Thus, non-native species affect the surrounding species through creation of new trophic links which may be felt across the food web (see David et al. 2017).
In the Neotropical regions, particularly in Brazil, the construction of reservoirs occur for several purposes, particularly for hydroelectric energy exploitation, and are commonplace in large rivers. Reservoirs are often associated with non-native fish introductions which are introduced for several reasons such as biological control, damming, aquarium release, bait, fish stocking, fish farming and sport fishing (Naylor et al. 2001; Casal 2006; Agostinho et al. 2010; Britton and Orsi 2012; Magalhães and Jacobi 2013). In the reservoirs of the upper Paraná River basin, for instance, the South American silver croaker (Plagioscion squamosissimus) and the peacock bass (Cichla spp.), both Amazonian fish, are among the most widely distributed non-native fish (Pelicice et al. 2018). The abundance of these fish, provide attractiveness for fishing, resulting an intense movement of fishing boats between reservoirs which, in turn, contribute to the spread of other species, especially invasive invertebrates (Belz et al. 2012). Of these, four species of non-native invertebrates may be cited as the most widely distributed in the upper Paraná River reservoirs. Two bivalves, the Asian clam (Corbicula fluminea) and golden mussel (Limnoperna fortunei), both originated from Asia and were possibly introduced to South America by ballast water in 1980 and 1990 decades, respectively (Ituarte 1981; Pastorino et al. 1993). Corbicula fluminea was detected in the upper Paraná River basin in 1994 (Itaipu reservoir; Takeda et al. 1994) and L. fortunei in 2002 (near Rosana city; Avelar et al. 2004). The African-Asiatic trumpet snail (Melanoides tuberculata) was found in Brazil for the first time in 1967, in Santos city, São Paulo state, upper Paraná River basin (Vaz et al. 1986), and was also possibly introduced by ballast water. These are the three species of non-native mollusks most widely distributed in Brazilian reservoirs and records have shown that they coexist in several reservoirs in the upper Paraná River basin (Rocha et al. 2011; Miyahira et al. 2020). Finally, the Amazonian prawn (Macrobrachium amazonicum) originated from Amazonas River basin (Odinetz-Colart 1993) and is widely distributed across north and northeastern Brazilian regions and was probably introduced in the upper Paraná River by fish repopulation programs, which used this prawn to feed these fish (Torloni et al. 1993).
According to studies carried out in this region, of these non-native invertebrates, with the exception of M. tuberculata, are consistently consumed by fish and, in some cases, were recorded as the main food items (Cantanhêde et al. 2008; Oliveira et al. 2010; Isaac et al. 2014; Rosa et al. 2015). However, these studies were based only on gut content analysis, thus information about the consumption rate of these prey by fish fauna in the upper Paraná River basin remains scarce. When non-native prey are substantially ingested and assimilated, they may influence several aspects of the food webs such as structure, trophic ecology, resource partitioning, production sources, trophic position and energy flow (Bootsma et al. 1996; Maguire and Grey 2006; Inger et al. 2010; Nilsson et al. 2012; Ozersky et al. 2012; Locke et al. 2014; Wood et al. 2017).
The above mentioned four species of non-native invertebrates have been registered in the Volta Grande reservoir, located on the upper Paraná River basin, Brazil (Morais et al. 2014). In this context, the present study investigates their role in supporting fish biomass and their influence on carbon flow into the Volta Grande reservoir food web. Two hypotheses were tested: (i) since non-native invertebrates are dominant benthonic species in the Volta Grande reservoir, we expect they should be consistently consumed and assimilated by fish; (ii) non-native prey are important carbon sources supporting a large proportion of fish biomass in this system.
Material and methods
Study area
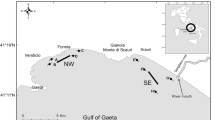
The Volta Grande Hydroelectric Power Plant (HPP) is located in the Grande River between Minas Gerais and São Paulo states (19° 57′, 20° 10′ S and 48° 25′, 47° 35′ W; 500 m above sea level), Brazil. The Volta Grande reservoir has a flooded area of approximately 205 km2, a perimeter of 80 km and volume of 23 × 109 m3 (Rolla et al. 1990). Its formation dates from 1973 and together with ten other reservoirs forms a cascade system on the Grande River which goes from 900 to 300 m above sea level (Braga 2001). In this region, the climate is humid temperate with dry winter and hot summer (Cwa – Koppen), with average annual temperature between 22 and 24 °C, a well defined dry season (May–October) and annual average rainfall of 1550 mm (CPTEC/INPE 2016). Samples were carried out at three equidistant collection sites (Fig. 1). These sites were chosen in order to sample the three compartments of the reservoir, that is, fluvial, intermediate and lacustrine zones (Kimmel and Groeger 1984)..
Field sampling and sample processing
Fish from the three sample sites were collected quarterly between October 2015 and July 2016, with eleven bottoms gillnets per site (10 m length, 1.5–2 m height, mesh size 25–160 mm; total sampled area = 150 m2), totaling twelve sampling events (four at each site). Nets were set in series perpendicularly to the shore. A minimal distance of approximately 20 m was maintained between nets, and depths varied from 1.0 to 7.0 m. All the nets were installed at dusk and collected at dawn the next day, remaining in the environment for about 14 hours. Fish were identified, measured on standard length (SL, cm), weighed with a precision scale (0.1 g) and then dissected to remove their stomach and intestine, which were immediately fixed in 10% formalin solution. Subsequently, the total gut contents were analyzed and identified on a stereomicroscope, with the non-native prey identified at species level and other items at the level of large groups.
In order to obtain tissue samples of the four potential non-native invertebrate prey for fish in Volta Grande reservoir (L. fortunei, C. fluminea, M. tuberculata and M. amazonicum), and the two most locally abundant native mollusks (apple snail Pomacea canaliculata and thiarid snail Aylacostoma tenuilabris), specifically targeted samples were performed. The specimens were collected in the littoral zone of sites; mollusks were manually sampled using a Petersen-type bottom dredger (319 cm2), and prawns were collected with a semicircular hand sieve (80 cm diameter, mesh size 1 mm). The captured specimens were immediately identified, sorted by species and then frozen.
In the field, a fresh dorsal white muscle fillet of 1 to 5 individuals of each fish species, of similar body size, was collected for stable isotope analysis. All samples were stored on ice and transported to the laboratory where they were frozen. Muscle tissue generally has lower contents of lipids and inorganic carbonates, besides producing lower variability in the values of δ13C and δ15N (Pinnegar and Polunin 1999). Regarding the non-native prey, in the laboratory, whole body tissue samples of 4 to 15 specimens of each species were processed for the same purposes as the fish samples. Then all tissue samples were lyophilized for 48 hours, macerated to a fine, homogeneous powder with the aid of a mortar and pestle and stored in Eppendorf tubes.
Stable isotopes analyses
Isotopic analyses for δ13C, δ15N, %C, %N and C/N were conducted by the Isotopic Ecology Laboratory of the Center for Nuclear Energy in Agriculture (CENA) of the University of São Paulo (USP), Campus Piracicaba-SP. To determine the isotopic ratio, the samples were analyzed in continuous flow mode using a Delta Plus Thermo Scientific mass spectrometer, interconnected to a Carlo Erba elemental analyzer (CHN 1110). The data were normalized using four internationally accepted isotope reference standards (International Atomic Energy Agency Standards CH6, CH7, N1 and N2). The δ13C and δ15N data were expressed in relation to the standard reference material: Vienna Pee Dee Belemnite for carbon and atmospheric N for nitrogen calculated by the following formula:\(\delta X = \left[ {\left( {R_{sample} \div R_{s\tan dard} } \right) - 1} \right] \times 10^{3}\). Where, X = 13C or 15N; R = isotopic ratios of 13C/12C or 15N/14N (Barrie and Prosser 1996).
Data analysis
The abundance and biomass of the collected fish were estimated by the catch per unit effort (CPUE) expressed in the following equation:\(CPUE = \left( {C \div E} \right) * 100\). This index was calculated in relation to the number of individuals and biomass. Where, C = number of individuals (abundance) or total weight (biomass) and E = fishing effort (area of the gill nets × fishing time × number of samples − four days samples per site).
The frequency of occurrence (F) and the weight ratio (%P) of the food items found in the stomachs were used to describe the diet of the species (Hyslop 1980). These values were combined to identify the main food items by means of the Food Index (FI) proposed by Kawakami and Vazzoler (1980):\(FI = \left( {F_{i} \times \% P_{i} } \right) \div F_{i} \times \% P_{i}\). Where, FI = Food index of item i; Fi = Frequency of occurrence of item i; %Pi = Relative weight of item i. In addition, in order to estimate the contribution of each food category to the total biomass of fish caught, CPUE biomass values \(\left( {Kg \div 150m^{2} } \right)\) were multiplied by the FI values using the following equation:\(\sum {CPUE_{i} \times FI_{i} }\). Where, CPUEi = CPUE biomass of species i; FIi = Food index of item i. For this estimate, species represented by only one individual in the samples were not contemplated.
In order to evaluate the distribution of the isotopic values of 19 fish species in relation to their potential prey, the δ13C and δ15N composition of the both prey and fish were submitted to a fractionation correction (Post 2002). Of these 19 fish, the proportional isotopic contribution of non-native prey was estimated only for those with at least 5 individuals sampled or who ingested at least 1 of the non-native prey. In order to consider other potential sources of protein, two locally abundant native mollusks (P. canaliculata and A. tenuilabris) and the sampled fish assemblage were also considered as food sources. Thus, the isotopic contribution of non-native prey was estimated for 13 fish species with Bayesian stable isotope mixing models (Moore and Semmens, 2008), specifically using the MixSIAR 3.1.9 package in R 1.2.5033 (Stock and Semmens, 2016). This and other mixing models use Bayesian inferences to help solve linear mixing models, which can determine the diet composition of consumers based on their isotopic signatures and that of their sources (Parnell et al. 2010).
Results
A total of 21 fish species belonging to 11 families, of 4 orders, were caught in the Volta Grande reservoir. The most representative orders of the total biomass were Characiformes and Perciformes, with 53.4% and 46.0%, respectively. The most diverse families, in number of species, were Cichlidae (six species), Serrasalmidae and Anostomidae (three species each), and Characidae (two species). The most abundant species were P. squamosissimus, Serrasalmus maculatus, Hoplias intermedius and Satanoperca pappaterra, together responsible for 81.3% of the total biomass collected (Fig. 2). Regarding the numerical abundance, in terms of importance, the main species were S. maculatus, P. squamosissimus, S. pappaterra, Leporinus friderici and Astyanax fasciatus, which accounted for 71.5% of the total number of collected fish. Six non-native fish species (Astronotus crassipinnis, Cichla kelberi, C. piquiti, Metynnis gr. lippincottianus, P. squamosissimus and S. marginatus) were caught, which accounted for 39.0% and 30.0% of the total sample biomass and number of specimens, respectively.
Percentage of Capture per Unit of Effort (CPUE) in abundance (left) and biomass (right) of the main fish species, collected quarterly between October 2015 and July 2016, in the Volta Grande reservoir, Brazil. Black horizontal bars represent values of native fish and grey bars of non-native ones. “Others” category was represented by nine species
A total of 326 fish were submitted for diet analysis. Food items were found in the stomach contents of 248 fish (76.0%) and comprised of 16 species (Table 1). The food items found were grouped into eight categories, three of which were composed by non-native prey (M. amazonicum, L. fortunei and M. tuberculata) and the others by fish (scales, muscle fragments, fins, whole fish and in a high degree of digestion), other invertebrates (mollusks, aquatic and terrestrial insects, crustaceans, annelids and nematodes), periphyton, plants (macrophytes, grass, filamentous algae, cyanobacteria, fruits and seeds), and debris/sediment.
Non-native prey were ingested by 11 fish species (42% of stomachs containing food items), with M. amazonicum being the most consumed (by all 11 species), followed by L. fortunei (by 6 species) and M. tuberculata (by 3 species). No specimen of C. fluminea was found in the stomach content analysis. According to FI, non-native prey were consumed substantially by eight fish species; M. amazonicum was the main prey ingested by P. squamosissimus, C. kelberi, C. piquiti, H. intermedius and A. crassipinnis, whereas L. fortunei was the main prey consumed by L. friderici, Australoheros facetus and Crenicichla britiskii (Fig. 3).
Based on results obtained from the relationship between CPUE biomass and FI (Table 2), we observed that 53.9% of the fish biomass is supported by the non-native prey species (M. amazonicum = 49.7%; L. fortunei = 4.2%; M. tuberculata = 0.05%).
The fish fauna and non-native invertebrate prey of the Volta Grande reservoir were characterized isotopically from a sample of 68 specimens of 19 species and 43 specimens of four species, respectively (Table 3).
Results obtained by isotopic mixing models also suggested that, altogether, non-native prey were essential carbon sources for the Volta Grande fish assemblage possibly supporting more than half of their biomass (Fig. 4). Four dominant fish species, which altogether accounted for 81.3% of the total biomass sampled in this reservoir, derived substantial amounts of their biomass from these prey: the non-native P. squamosissimus (54.2%), S. maculatus (55.3%), H. intermedius (55.4%) and S. pappaterra (47.5%). Beside P. squamosissimus, non-native prey were also prominent carbon sources for other non-native fish such as the predators C. piquiti (64.8%), C. kelberi (60.2%) and the omnivore A. crassipinnis (44.6%). Among the non-native prey species, M. amazonicum stands out as the most important carbon source for native and non-native fish. The non-native C. kelberi, C. piquiti, P. squamosissimus and A. crassipinis, respectively derived 38%, 34%, 33% and 19% of their biomass from this prawn. Regarding native fish, the predators H. intermedius, S. maculatus, and the omnivores Gymnotus carapo, Eigenmannia virescens and C. britskii, derived from this prey 30%, 28%, 39%, 37% and 28% of their biomass, respectively. Corbicula fluminea and L. fortunei were prominent carbon sources for the native omnivore L. friderici which derived 48% (25% and 23%, respectively) of their biomass from these non-native bivalves. Finally, M. tuberculata were important carbon sources for the omnivores S. pappaterra and A. facetus, fueling 28.1% and 21.0% of their biomass, respectively.
Discussion
Results obtained by the present study by both gut content and stable isotopic analyses pointed to a trophic structure where non-native species represent not only a strong component of the fish community, but also their main carbon source. The lack of pre-invasion data do not favor a comparative analysis and, therefore, this work does not support inferences regarding changes, although the non-native species presence constitute a novel scenario. Thus, additional research assessing data prior to invasions are needed to investigate the consequences of the new trophic links created after the arrivals of these species. However, we did find that non-native benthonic prey represented the main energy sources for fish assemblages, pointing their potential in supporting fish stocks in reservoirs where they are available. Additionally, non-native prey are the main source of carbon for non-native fish biomass suggesting that its trophic interaction can favor the maintenance of these fish in the Volta Grande reservoir.
In comparison with the species richness estimated in previous studies, the species richness found here (21) was lower than 47 (ELETROBRAS 1995), 37 (Braga and Gomiero 1997), 26 (Braga 2001) and 30 species (Becker et al. 2015) previously recorded in the Volta Grande reservoir. Although the variation between the richness estimates can be influenced by the application of different sampling methods, among other factors (e.g. absence of riparian vegetation and anthropogenic influences in the surroundings of the reservoir such as sewage, agriculture and livestock activities), can also be related to the great difference in timing among the studies. The reservoir age has been related to the reduction of total fish richness and the increase in non-native species (Loures and Pompeu 2019). Plagioscion squamosissimus, introduced from the Amazon basin, was the dominant fish species in samples; this pattern has also been found in other reservoirs in the upper Paraná River basin. It is the main fish species that inhabits the pelagic areas of the upper Paraná reservoirs (Torloni et al. 1993; Agostinho et al. 1999), including the Volta Grande (Sanches et al. 2014; Becker et al. 2015). This fish has adaptations for lacustrine environments, and its success in these reservoirs may be attributed to reproductive strategy (Agostinho et al. 1999), as its larvae and eggs are pelagic (Fontenele and Peixoto 1978; Nakatani et al. 1993). Two other non-native lacustrine species sampled in Volta Grande reservoir – and also introduced from Amazon basin – are the cichlids C. kelberi, C. piquiti and A. crassipinnis. These non-native fish are also common in reservoirs of the region, but their abundances are usually low (Gomes and Miranda 2001). The abundance of non-native fish in several reservoirs of the upper Paraná River basin suggests a regional tendency toward biotic homogenization in this basin (Vitule et al. 2012). Such community structuring is common in reservoirs and is in part related to their limnological characteristics and introduction of non-native species, which drive the replacement of diverse communities composed mainly by rheophilic fish for lacustrine species (Poff et al. 2007; Agostinho et al. 2016).
Regarding the species diets, we observed a poorly-diversified food spectrum with a marked presence of non-native invertebrate prey. Among these, the abundance of M. amazonicum and L. fortunei in gut contents suggests they were consistently ingested by both native and non-native fish species. Besides demonstrating the food plasticity of these fish, the results indicate a high availability of non-native prey in the sampled sites. Similarly, high amount of these prey species were consumed by other fish fauna in the upper Paraná River reservoirs such as Itaipú (Oliveira et al. 2010) and Ilha Solteira (Rosa et al. 2015). In the latter one, L. fortunei were also the main prey ingested by L. friderici. In temperate reservoirs, non-native bivalves such as zebra mussels (Dreissena polymorpha) and C. fluminea have also been extensively exploited by omnivores such as Aplodinotus grunniens and Lepomis microlophus (Robinson and Wellborn 1988; Magoulick and Lewis 2002; Bartsch et al. 2005). According to the authors, these fish have pharyngeal teeth adapted to shell fragmentation, similarly to those observed in South American Leporinus and Megaleporinus species (Occhi and Oliveros 1974) which are shifting their diets preferences to non-native bivalves (Penchaszadeh et al. 2000; García and Protogino 2005; Oliveira et al. 2010; Isaac et al. 2014; Cataldo 2015). Regarding the M. amazonicum, we found that carnivorous fish such as native H. intermedius and non-native P. squamosissimus, C. piquiti and C. kelberi were their main consumers, as found previously in the Ilha Solteira reservoir (Rosa et al. 2015). However, high intake rates of L. fortunei and M. amazonicum may also be associated with the relative abundance of these organisms in the analyzed system. Evidence has shown that non-native species can be more successful than native ones in systems with altered functions (Levine and D’Antonio 1999; Menke and Holway 2006; Crooks et al. 2011), such as reservoirs (Havel et al. 2005), where they can reach extremely high densities (Boltovskoy et al. 2006). In this sense, the high densities of L. fortunei and M. amazonicum in Volta Grande and other upper Paraná River reservoirs (Bialetzki et al. 1997; Takeda et al. 2003; Silva 2010; Morais et al. 2014) represent new food resources for fish, as verified by the present and previous studies (Almeida et al. 1997; Cantanhêde et al. 2008; Oliveira et al. 2010; Rosa et al. 2015).
Based on gut contents and isotopic mixing models, we found that together, non-native prey are essential carbon sources for the fish fauna, fueling more than 40.0% of the biomass in four dominant fish species, which altogether accounted for > 80.0% of the total fish sampled biomass, thus accepting our hypothesis. However, such values may be overestimated since unsampled resources (e.g. plants and detritus) were not considered, although they should represent less important sources of protein. Thus, we suggest that non-native invertebrates are highly incorporated into the Volta Grande food web, representing a strong base for supporting the fish assemblage. Furthermore, consumers of different trophic levels derived substantial amounts of their biomass from these sources, suggesting that assimilation of non-native prey items can influence the carbon flow of an entire food web. Similarly, strong influences in energy sources supporting North and South American food webs have been noted after D. polymorpha (Miehls et al. 2009; Locke et al. 2014) and L. fortunei invasions (González-Bergonzoni et al. 2020), respectively. In fact, non-native species often pose threats to biodiversity (Vitousek et al. 1996; Strayer 2010); however, in some cases they can provide benefits to the ecosystems that lose their original functions due to anthropic disturbances (Schlaepfer et al. 2011; Carroll and Peterson 2013). Evidence found in North American lakes have shown that non-native prey (the Chinese mystery snail, Bellamya chinensis), provide a prey substitute in developed lakes where native snail populations were depressed and, furthermore, B. chinensis’s influence extended to higher trophic-level consumers (Twardochleb and Olden 2016). In this context, rivers regulated (e.g. reservoir ecosystems), whose native prey communities can change due to new environmental conditions (Growns and Growns 2001), may have some of these effects alleviated from non-native prey acting as alternative energy sources. Of course, their contribution in diminishing the native species abundance by competition cannot be neglected.
The consumption rate of non-native bivalves by the native omnivorous fish L. friderici suggested that these filter-feeding organisms are important as carbon sources and can have implications for energy flow. Bivalves and snails are reliable integrators of the basal resources of the pelagic and benthonic energy pathways, respectively (Post 2002; Locke et al. 2014). Some studies have shown the importance of benthonic pathways in the maintenance of known pelagic fish (Vander Zanden and Vadeboncoeur 2002; Karlsson and Bystrom 2005). However, we observed the opposite: in the upper Paraná River, L. friderici is known isotopically for deriving its biomass from C3 vascular plants (Manetta et al. 2003). In the Volta Grande reservoir, however, this fish derived prominent parts of its biomass from filter-feeding mussels, which usually derive their biomass from phytoplankton. Thus, these non-native bivalves seem to connect L. friderici to the pelagic pathway by carbon transference from this energy source. Similarly, the connection of typically benthonic consumers to the pelagic pathways was previously verified in Lake Opinicon (Canada) for the northern map turtle Graptemys geographica (Bulté and Blouin-Demers 2008) and pumpkinseed Lepomis gibbosus (Locke et al. 2014) due to predation on D. polymorpha. Thus, since L. fortunei and C. fluminea occur in abundance in the littoral zone of the Volta Grande reservoir (Brito et al. 2018), they potentially constitute an important trophic connection between littoral consumers and pelagic energy sources.
Non-native prey also were prominent carbon sources for non-native fish, fueling more than 50.0% of the biomass in C. kelberi, C. piquiti and P. squamosissimus. This trophic relationship may be related to the invasional meltdown hypothesis proposed by Simberloff and Von Holle (1999). The authors suggest that interactions among non-native species can accelerate the process of replacing native communities, ranging from a simple interaction (e.g. one species helps another; present study) to mutualistic interactions (Simberloff 2006). Although the assessment of this phenomenon was not the focus here, we showed evidence of strong interactions among non-native species in the Volta Grande reservoir. We suggest that these non-native prey species have a fundamental role in maintaining non-native fish populations which are commercially important in region. However, a specific approach that accounts for how these non-native prey facilitate the non-native fish’s initial establishment and invasion process is necessary to accurately characterize the occurrence of the phenomenon.
The main findings of the present study, although they apply to one reservoir, may have implications across the upper Paraná River basin. The analyzed non-native prey, which are essential in fueling fish biomass, occur in high densities in many reservoirs of the region (Bialetzki et al. 1997; Takeda et al. 2004; Pamplin and Rocha 2005; França et al. 2005; Suriani et al. 2005; Pareschi et al. 2008; Pestana et al. 2010; Silva 2010; Rosa et al. 2015; Morais et al. 2014). We suggest that, besides dam construction and species introduction pathways, non-native vs. native and non-native vs. non-native interactions may be an interesting metric to improve the investigations about biotic homogenization in the upper Paraná River basin. Ultimately, our results may help to understand fundamental ecological issues (sensu Sutherland et al. 2013) bringing to light the extent to which these new species combinations influence the energy flow and properties of ecosystems.
Data availability
Request from authors.
Code availability
MixSIAR 3.1.9 package in R 1.2.5033.
References
Agostinho AA, Miranda LE, Bini LM, Gomes LC, Thomaz SM, Suzuki HI (1999) Patterns of colonization in neotropical reservoirs, and prognoses on aging. In: Tundisi JG, Straskraba M (eds) Theoretical reservoir ecology and its applications. Backhuys Publishers, Leiden, pp 227–265
Agostinho AA, Pelicice FM, Gomes LC, Júlio HF Jr (2010) Reservoir fish stocking: when one plus one may be less than two. Nat Conserv 8:103–111. https://doi.org/10.4322/natcon.00802001
Agostinho AA, Gomes LC, Santos NCL, Ortega JCG, Pelicice FM (2016) Fish assemblages in Neotropical reservoirs: colonization patterns, impacts and management. Fish Res 173:26–36. https://doi.org/10.1016/j.fishres.2015.04.006
Almeida VLL, Hahn NS, Vazzoler AEAM (1997) Feeding patterns in five predatory fishes of the high Parana River floodplain (PR, Brazil). Ecol Freshw Fish 6:123–133. https://doi.org/10.1111/j.1600-0633.1997.tb00154.x
Avelar WEP, Martim SL, Vianna MP (2004) A new occurrence of Limnoperna fortunei (Dunker, 1857) (Bivalvia, Mytilidae) in the state of São Paulo. Brazil Braz J Biol 64(4):739–742. https://doi.org/10.1590/S1519-69842004000500002
Barrie A, Prosser SJ (1996) Automated analysis of light–element stable isotopes by isotope ratio mass spectrometry. In: Boutton TW, Yamsahi S (eds) Mass spectrometry of soils. Marcel Dekker, New York, pp 1–46
Bartsch MR, Bartsch LA, Gutreuter S (2005) Strong effects of predation by fishes on an invasive macroinvertebrate in a large floodplain river. J N Am Benthol Soc 24:168–177. https://doi.org/10.1899/0887-3593(2005)024%3c0168:SEOPBF%3e2.0.CO;2
Becker B, Galhardo BOS, Macedo DR, Hughes RM, Callisto M, Santos GB (2015) Influence of limnological zones on the spatial distribution of fish assemblages in three Brazilian reservoir. J Limnol 75(1):156–168. https://doi.org/10.4081/jlimnol.2015.1266
Beisner BE, Ives AR, Carpenter SR (2003) The effects of an exotic fish invasion on the prey communities of two lakes. J Anim Ecol 72:331–342. https://doi.org/10.1046/j.1365-2656.2003.00699.x
Belz CE, Darrigran G, Mader-Netto OS, Boeger WA, Junior PRR (2012) Analysis of four dispersion vectors in inland waters: the case of the invading bivalves in South America. J Shellfish Res 31:777–784. https://doi.org/10.2983/035.031.0322
Bialetzki A, Nakatani K, Baumgartner G, Bond Buckup G (1997) Occurrence of Macrobrachium amazonicum (Heller 1862) (Decapoda, Palaemonidae) in Leopoldo’s inlet (Ressaco do Leopoldo), upper Paraná river, Porto Rico, Paraná, Brazil. Rev Bras Zool 14:379–390. https://doi.org/10.1590/S0101-81751997000200011
Boltovskoy D, Correa N, Cataldo D, Sylvester F (2006) Dispersion and ecological impacts of the invasive freshwater bivalve Limnoperna fortunei in Río de la Plata watershed and beyond. Biol Invasions 8:947–963. https://doi.org/10.1007/s10530-005-5107-z
Bootsma HA, Hecky RE, Hesslein RH, Turner GF (1996) Food partitioning among lake Malawi nearshore fishes as revealed by stable isotopes analyses. Ecology 77(4):1286–1290. https://doi.org/10.2307/2265598
Braga FMS (2001) Reprodução de peixes (OSTEICHTHYES) em afluentes do reservatório de Volta Grande, rio Grande, sudeste do Brasil. Iheringia Ser Zoo 91:67–74. https://doi.org/10.1590/S0073-47212001000200009
Braga FMS, Gomiero LM (1997) Análise da pesca experimental realizada no reservatório de Volta Grande, rio Grande (MG-SP). B Inst Pesca 24:131–138
Brito SL, López CM, Souza GCT, Rocha SF, Marques MMGSM, Guarda VLM, Greco MKB, Carvalho MD (2018) Índice plâncton-bentônico para avaliação da qualidade de água no rio Grande-MG/SP. Luz PM (org) Ecologia, Evolução e Diversidade. Atena Editora, Ponta Grossa, pp 33–49
Britton JR, Orsi ML (2012) Non-native fish in aquaculture and sport fishing in Brazil: economic benefits versus risks to fish diversity in the upper River Paraná Basin. Rev Fish Biol Fish 22:555–565. https://doi.org/10.1007/s11160-012-9254-x
Bryan SD, Robinson AT, Sweetser MG (2002) Behavioral responses of a small native fish to multiple introduced predators. Environ Biol Fish 63:49–56. https://doi.org/10.1023/A:1013899125938
Bulté G, Blouin-Demers G (2008) Northern map turtles (Graptemys geographica) derive energy from the pelagic pathway through predation on zebra mussels (Dreissena polymorpha). Freshw Biol 53:497–508. https://doi.org/10.1111/j.1365-2427.2007.01915.x
Cantanhêde G, Hahn NS, Gubiani EA, Fugi R (2008) Invasive molluscs in the diet of Pterodoras granulosus (Valenciennes, 1821) (Pisces, Doradidae) in the Upper Paraná River floodplain, Brazil. Ecol Freshw Fish 17(1):47–53. https://doi.org/10.1111/j.1600-0633.2007.00258.x
Carroll JM, Peterson BJ (2013) Comparisons in demographic rates of bay scallops in eelgrass and the introduced alga, Codium fragile, in New York. Mar Biol 160:1451–1463. https://doi.org/10.1007/s00227-013-2197-5
Casal CMV (2006) Global documentation of fish introductions: the growing crisis and recommendations for action. Biol Invasions 8:3–11. https://doi.org/10.1007/s10530-005-0231-3
Cataldo D (2015) Trophic relationships of Limnoperna fortunei with adult fishes. In: Boltovskoy D (ed) Limnoperna fortunei. The ecology, distribution and control of a swiftly spreading invasive fouling mussel, London, Springer, pp 231–248
Centro de Previsão de Tempo e Estudos Climáticos/Instituto Nacional de Pesquisas Espaciais (2016) http://www.cptec.inpe.br/clima/monit/monitor_brasil.shtml. Accessed 13 Oct 2016
Courchamp F, Chapuis J-L, Pascal M (2003) Mammal invaders on islands: impact, control and control impact. Biol Rev 78(3):347–383. https://doi.org/10.1017/S1464793102006061
Crooks JA, Chang AL, Ruiz GM (2011) Aquatic pollution increases the relative success of invasive species. Biol Invasions 13:165–176. https://doi.org/10.1007/s10530-010-9799-3
David P, Thébault E, Anneville O, Loeuille N (2017) Impacts of invasive species on food webs: a review of empirical data. Adv Ecol Res 56:1–60. https://doi.org/10.1016/bs.aecr.2016.10.001
Dijkstra JA, Lambert WJ, Harris LG (2013) Introduced species provide a novel temporal resource that facilitates native predator population growth. Biol Invasions 15:911–919. https://doi.org/10.1007/s10530-012-0339-1
Eletrobrás, (1995) Seminário sobre fauna aquática e o setor elétrico brasileiro, reuniões temáticas preparatórias: caderno 4-Estudos e Levantamentos. Comase, Rio de Janeiro
Fontenele O, Peixoto JT (1978) Análise dos resultados de introdução da pescada do Piauí, Plagioscion squamosissimus (Heckel 1840, nos açudes do nordeste. Boletim Técnico DNOCS 36:85–112
França RS, Suriani AL, Lucca JV, Rocha O (2005) A invasão dos reservatórios do Baixo Rio Tietê (SP) pelas espécies exóticas Melanoides tuberculata e Corbicula fluminea e o impacto destes sobre os moluscos nativos. Rocha O, Espíndola ELG, Fenerich-Verani N, Verani JR, Rietzler AC (orgs) Espécies invasoras em águas doces - Estudos de caso e propostas de manejo. Editora Universidade Federal de São Carlos, São Carlos, pp 307–317
García ML, Protogino LC (2005) Invasive freshwater mollusks are consumed by native fishes in South America. J Appl Ichthyol 21:34–38. https://doi.org/10.1111/j.1439-0426.2004.00570.x
Glibert PM, Fullerton D, Burkholder JM, Cornwell J, Kana TM (2011) Ecological stoichiometry, biogeochemical cycling, invasive species, and aquatic food webs: San Francisco Estuary and comparative systems. Rev Fish Sci 19:358–417. https://doi.org/10.1080/10641262.2011.611916
Goeze E (1882) Pflanzengeographie für Gärtner und Freude des Gartenbaues. Ulmer, Stuttgart
Gomes LC, Miranda LE (2001) Riverine characteristics dictate composition of fish assemblages and limit fisheries in reservoirs of the upper Paraná River basin. Regul Rivers Res Mgmt 17:67–76
González-Bergonzoni I, Silva I, Mello FT, D’Anatro A, Boccardi L, Stebniki S, Brugnoli E, Tesitore G, Vidal N, Naya DE (2020) Evaluating the role of predatory fish controlling the invasion of the Asian golden mussel Limnoperna fortunei in a subtropical river. J Appl Ecol 57:717–728. https://doi.org/10.1111/1365-2664.13573
Green SJ, Akins JL, Maljković A, Côté IM (2012) Invasive lionfish drive atlantic coral reef fish declines. PLoS ONE 7(3):e32596. https://doi.org/10.1371/journal.pone.0032596
Growns IO, Growns JE (2001) Ecological effects of flow regulation on macroinvertebrate and periphytic diatom assemblages in the Hawkesbury – Nepean River, Australia. Regul Rivers Res Mgmt 17:275–293. https://doi.org/10.1002/rrr.622
Havel JE, Lee CE, Vander Zanden MJ (2005) Do reservoirs facilitate invasions into landscapes? Bioscience 55:518–252. https://doi.org/10.1641/0006-3568(2005)055[0518:DRFIIL]2.0.CO;2
Hostert LE, Pintor LM, Byers JE (2019) Sex, size and prey caloric value affects diet specialization and consumption of an invasive prey by a native predator. Curr Zool 65(5):499–507. https://doi.org/10.1093/cz/zoy076
Hyslop EJ (1980) Stomach contents analysis: a review of methods and their application. J Fish Biol 17(4):411–429. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Inger R, McDonald RA, Rogowski D, Jackson AL, Parnell A, Preston SJ, Harrod C, Goodwin C, Griffths D, Dick JTA, Elwood RW, Newton J, Bearhop S (2010) Do non-native invasive fish support elevated lamprey populations? J Appl Ecol 47:121–129. https://doi.org/10.1111/j.1365-2664.2009.01761.x
Isaac A, Fernandes A, Ganassin MJM, Hahn NS (2014) Three invasive species occurring in the diets of fishes in a Neotropical floodplain. Braz J Biol 74:16–22. https://doi.org/10.1590/1519-6984.18312
Ituarte CF (1981) Primera noticia acerca de la presencia de pelecípodos asiáticos en el área rioplatense. Neotropica 27:79–82
Karlsson J, Bystrom P (2005) Littoral energy mobilization dominates energy supply for top consumers in subarctic lakes. Limnol Oceanogr 50:538–543. https://doi.org/10.4319/lo.2005.50.2.0538
Kawakami E, Vazzoler G (1980) Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Bol Inst Oceanogr 29(2):205–207. https://doi.org/10.1590/S0373-55241980000200043
Kimmel BL, Groeger AW (1984) Factors controlling primary production in lakes and reservoirs: a perspective. Lake Reserv Manag 1:277–281. https://doi.org/10.1080/07438148409354524
Latini AO, Petrere M Jr (2004) Reduction of a native fish fauna by alien species: an example from Brazilian freshwater tropical lakes. Fish Manag Ecol 11(2):71–79. https://doi.org/10.1046/j.1365-2400.2003.00372.x
Levine JM, D’Antonio CM (1999) Elton revisited: a review of evidence linking diversity and invasibility. Oikos 87:15–26. https://doi.org/10.2307/3546992
Locke SA, Bulté G, Marcogliese DJ, Forbes MR (2014) Altered trophic pathway and parasitism in a native predator (Lepomis gibbosus) feeding on introduced prey (Dreissena polymorpha). Oecologia 175:315–324. https://doi.org/10.1007/s00442-014-2898-6
Loures RC, Pompeu PS (2019) Temporal changes in fish diversity in lotic and lentic environments along a reservoir cascade. Freshw Biol 00:1–15. https://doi.org/10.1111/fwb.13372
Magalhães ALB, Jacobi CM (2013) Asian aquarium fishes in a Neotropical biodiversity hotspot: Impending establishment, spread and impacts. Biol Invasions 15(10):2157–2163. https://doi.org/10.1007/s10530-013-0443-x
Magoulick DD, Lewis LC (2002) Predation on exotic zebra mussels by native fishes: effects on predator and prey. Freshw Biol 47:1908–1918. https://doi.org/10.1046/j.1365-2427.2002.00940.x
Maguire C, Grey J (2006) Determination of zooplankton dietary shift following a zebra mussel invasion, as indicated by stable isotope analysis. Freshw Biol 51:1310–1319. https://doi.org/10.1111/j.1365-2427.2006.01568.x
Manetta GL, Benedito-Cecilio E, Martinelli M (2003) Carbon sources and trophic position of the main species of fishes of Baía River, Paraná River floodplain, Brazil. Braz J Biol 63(2):283–290. https://doi.org/10.1590/S1519-69842003000200013
Menke SB, Holway DA (2006) Abiotic factors control invasion by Argentine ants at the community scale. J Anim Ecol 75:368–376. https://doi.org/10.1111/j.1365-2656.2006.01056.x
Miehls ALJ, Mason DM, Frank KA, Krause AE, Peacor SD, Taylor WW (2009) Invasive species impacts on ecosystem structure and function: a comparison of Oneida Lake, New York, USA, before and after zebra mussel invasion. Ecol Model 220:3194–3209. https://doi.org/10.1016/j.ecolmodel.2009.07.013
Miyahira IC, Pereira LS, dos Santos LN (2020) Non-native freshwater molluscs in the Neotropics: what can be learned from Brazilian reservoirs? Aquat Invasions 15(3):455–472. https://doi.org/10.3391/ai.2020.15.3.06
Moore JW, Semmens BX (2008) Incorporating uncertainty and prior information into stable isotope mixing models. Ecol Lett 11:470–480. https://doi.org/10.1111/j.1461-0248.2008.01163.x
Morais L, Farias RL, Martins I, Barbosa JEL, Molozzi J, Anacléto MJ, Callisto M (2014) Bioindicadores bentônicos de qualidade ambiental em reservatórios da CEMIG. In: Callisto M, Alves CBM, Lopes JM, Castro MA (org) Condições ecológicas em bacias hidrográficas de empreendimentos hidrelétricos, Companhia Energética de Minas Gerais, Belo Horizonte, pp 161–184
Moyle PB, Light T (1996) Fish invasions in California: do abiotic factors determine success? Ecology 77:1666–1670. https://doi.org/10.2307/2265770
Nakatani K, Latini JD, Baumgartner G, Baumgartner MST (1993) Distribuição espacial e temporal das larvas de curvina Plagioscion squamosissimus (Heckel, 1840) (Osteichthyes, Sciaenidae), no reservatório de Itaipu. Rev UNIMAR 15:191–209
Naylor RL, Williams SL, Strong DR (2001) Aquaculture—A gateway for exotic species. Science 294:1655–1656. https://doi.org/10.1126/science.1064875
Nilsson E, Solomon CT, Wilson KA, Willis TV, Larget B, Vander Zanden MJ (2012) Effects of an invasive crayfish on trophic relationships in north-temperate lake food webs. Freshw Biol 57:10–23. https://doi.org/10.1111/j.1365-2427.2011.02688.x
Occhi RH, Oliveros OB (1974) Estudio anátomo-histológico de la cavidade bucofaríngea de Leporinus obtusidens (Pisces, Tetragonopteridae). Phys Sección B Las Aguas Continentales y sus Organismos 33:77–90
Odinetz-Colart O (1993) Ecologia e potencial pesqueiro do camarão-canela, Macrobrachium amazonicum, na Bacia Amazônica. In: Ferreira EJ, Santos GM, Leão ELM, Oliveira LA (eds) Bases Científicas para Estratégias de Preservação e desenvolvimento da Amazônia (2). INPA, Manaus, pp 147–166
Oliveira CRC, Fugi R, Brancalhão KP, Agostinho AA (2010) Fish as potential controllers of invasive mollusks in a neotropical Reservoir. Nat Conservação 8(2):140–144. https://doi.org/10.4322/natcon.00802006
Ozersky T, Evans DO, Barton DR (2012) Invasive mussels alter the littoral food web of a large lake: stable isotopes reveal drastic shifts in sources and flow of energy. PLoS ONE 7(12):e51249. https://doi.org/10.1371/journal.pone.0051249
Pamplin PAZ, Rocha O (2005) Distribuição batimétrica e sazonal de moluscos na represa Bariri, com ênfase nas espécies exóticas, Melanoides tuberculata (Gastropoda Thiaridae) e Corbicula fluminea (Bivalvia, Corbiculidae). In: Rocha O, Espíndola ELG, Fenerich-Verani N, Verani JR, Rietzler AC (orgs) Espécies invasoras em águas doces - Estudos de caso e propostas de manejo. Editora Universidade Federal de São Carlos, São Carlos, SP, pp 281–305
Pareschi DC, Matsumura-Tundisi T, Medeiros GR, Luzia AP, Tundisi JG (2008) First occurrence of Limnoperna fortunei (Dunker, 1857) in the Rio Tietê watershed (São Paulo State, Brazil). Braz J Biol 68(4):1107–1114
Parnell A, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS ONE 3:e9672. https://doi.org/10.1371/journal.pone.0009672
Pastorino G, Darrigran G, Martin S, Lunaschi L (1993) Limnoperna fortunei (Dunker, 1857) (Mytilidae), nuevo bivalvo invasor en aguas del rio de La Plata. Neotropica 39(101–102):34
Pelicice FM, Agostinho AA (2009) Fish fauna destruction after the introduction of non-native predator (Cichla kelberi) in a neotropical reservoir. Biol Invasions 11:1789–1801. https://doi.org/10.1007/s10530-008-9358-3
Pelicice FM, Latini JD, Agostinho AA (2015) Fish fauna disassembly after the introduction of a voracious predator: main drivers and the role of the invader’s demography. Hydrobiologia 746(1):271–283. https://doi.org/10.1007/s10750-014-1911-8
Pelicice FM, Azevedo-Santos VM, Esguícero ALH, Agostinho AA, Arcifa MS (2018) Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotrop Ichthyol 16(2):1–18. https://doi.org/10.1590/1982-0224-20170150
Penchaszadeh PE, Darrigran G, Angulo C, Averbuj A, Brogger M, Dogliotti A, Pirez N (2000) Predation of the invasive freshwater mussel Limnoperna fortunei (Dunker, 1857) (Mytilidae) by the fish Leporinus obtusidens Valenciennes, 1846 (Anostomidae) in the Río de la Plata, Argentina. J Shellfish Res 19:229–231
Pestana D, Ostrensky A, Tscha MK, Boeger W (2010) Prospecção do molusco invasor Limnoperna fortunei (Dunker, 1857) nos principais corpos hídricos do estado do Paraná, Brasil. Zoologia 50(34):553–559
Pinnegar JK, Polunin NVC (1999) Differential fractionation of δ13C and δ15N among fish tissues: implications for the study of trophic interactions. Funct Ecol 13:225–231. https://doi.org/10.1046/j.1365-2435.1999.00301.x
Pintor LM, Byers JE (2015) Individual variation in predator behavior and demographics affects consumption of non-native prey. Behav Ecol 26:797–804. https://doi.org/10.1093/beheco/arv013
Poff NL, Olden JD, Merritt DM, Pepin DM (2007) Homogenization of regional river dynamics by dams and global biodiversity implications. PNAS USA 104(14):5732–5737. https://doi.org/10.1073/pnas.0609812104
Pompeu PS, Godinho AL (2001) Mudança na dieta da traíra Hoplias malabaricus (Bloch) (Erythrinidae, Characiformes) em lagoas da bacia do rio doce devido à introdução de peixes piscívoros. Rev Bras Zool 18(4):1219–1225. https://doi.org/10.1590/S0101-81752001000400016
Porter-Whitaker AE, Rehage JS, Liston SE, Loftus WF (2012) Multiple predator effects and native prey responses to two non-native Everglades cichlids. Ecol Freshw Fish 21:375–385. https://doi.org/10.1111/j.1600-0633.2012.00557.x
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83:703–718. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2
Puntila-Dodd R, Loisa O, Riipinen K, Fowler AE (2019) A taste for aliens: Contribution of a novel prey item to native fishes’ diet. Biol Invasions 21:2907–2917. https://doi.org/10.1007/s10530-019-02021-w
Pyšek P, Blackburn TM, Garcia-Berthou E, Perglová I, Rabitsch W (2016) Displacement and local extinction of native and endemic species. In: Vilà M, Hulme PE (Eds) Impact of biological invasions on ecosystem services. Invading Nature - Springer Series in Invasion Ecology 12, Berlin, pp 157–175. https://doi.org/10.1007/978-3-319-45121-3_10
Robinson JV, Wellborn GA (1988) Ecological resistance to the invasion of a freshwater clam, Corbicula fluminea: fish predation effects. Oecologia 77:445–452. https://doi.org/10.1007/BF00377258
Rocha O, Espíndola ELG, Rietzler AC, Fenerich-Verani N, Verani JR (2011) Animal invaders in São Paulo state reservoirs. Oecol Aust 15(3):631–642. https://doi.org/10.4257/oeco.2011.1503.14
Rolla ME, Rosa SG, Freitas OMC, Gomes MCS, Junqueira MV, Souza MLG (1990) Composição físico-química e biológica e do sedimento do reservatório de Volta Grande, Minas Gerais / São Paulo. Acta Limnol Bras 3:201–218
Rosa DM, Santos GB, Gomes PLA, Campos MCS, Dias JHP (2015) Occurrence of Limnoperna fortunei (Dunker, 1857) in the fish diet from a south-eastern Brazilian reservoir. J Appl Ichthyol 31(1):188–191. https://doi.org/10.1111/jai.12623
Sanches BO, Becker B, Gomes PLA, Hughes RM, Santos GB (2014) Ichthyofauna of four CEMIG Reservoirs: assemblage characteristics and conservation perspectives. In: Calisto M, Hughes RM, Lopes JM, de Castro MA (eds) Ecological conditions in hydropower basins. Companhia Energética de Minas Gerais, Belo Horizonte, pp 181–207
Schlaepfer MA, Sax DF, Olden JD (2011) The potential conservation value of non-native species. Conserv Biol 25:428–437
Sharpe DMT, De León LF, González R, Torchin ME (2017) Tropical fish community does not recover 45 years after predator introduction. Ecology 98:412–424. https://doi.org/10.1002/ecy.1648
Silva CJ (2010) Biologia e ecologia dos camarões de água doce Macrobrachium amazonicum (Heller 1862) e Macrobrachium jelskii (Miers 1778) (Crustacea: Caridea: Palaemonoidea) no Rio Grande, região de Planura, MG. Dissertation, Universidade Estadual Paulista
Simberloff D (2006) Invasional meltdown 6 years later: important phenomenon, unfortunate metaphor, or both? Ecol Lett 9:912–919. https://doi.org/10.1111/j.1461-0248.2006.00939.x
Simberloff D, Von Holle M (1999) Positive interactions of nonindigenous species: invasional meltdown? Biol Invasions 1:21–32. https://doi.org/10.1023/A:1010086329619
Stock BC, Semmens BX (2016) Unifying error structures in commonly used biotracer mixing models. Ecology 97(10):2562–2569. https://doi.org/10.1002/ecy.1517
Strayer DL (2010) Alien species in fresh waters: ecological effects, interactions with other stressors, and prospects for the future. Freshw Biol 55(1):152–174. https://doi.org/10.1111/j.1365-2427.2009.02380.x
Suriani AL, França RS, Lucca JV, Rocha O (2005) A invasão de moluscos exóticos nas represas do Médio Rio Tietê (SP). In: Rocha O, Espíndola ELG, Fenerich-Verani N, Verani JR, Rietzler AC (orgs) Espécies invasoras em águas doces - Estudos de caso e propostas de manejo. Editora Universidade Federal de São Carlos, São Carlos, pp 295–305
Sutherland WJ, Freckleton RP, Godfray HCJ et al (2013) Identification of 100 fundamental ecological questions. J Ecol 101:58–67. https://doi.org/10.1111/1365-2745.12025
Takeda AM, Mansur MCD, Fujita DS, Bibian JPR (2003) Ocorrência da espécie invasora de mexilhão dourado, Limnoperna fortunei (Dunker, 1857), em dois pequenos reservatórios próximos a Curitiba. PR Acta Biol Leopondensia 25(1):251–254
Takeda AM, Fujita DS, Fontes HM (2004) Perspectives on exotic bivalves proliferation in the Upper Paraná River Floodplain. In: Agostinho AA, Rodrigues L, Gomes LC, Magela TS, Miranda LE (eds) Structure and functioning of the Paraná River and its floodplain. EDUEM, Maringá, pp 97–100
Torloni CEC, Santos JJ, Carvalho Junior AA, Corrêa ARA (1993) A pescada-do-piaui Plagioscion squamosissimus (Heckel, 1840) (Osteichthyes, Perciformes) nos reservatórios da Companhia Energética de São Paulo-Cesp. CESP Série Pesquisa e Desenvolvimento 84:1–23
Twardochleb LA, Olden JD (2016) Non-native Chinese mystery snail (Bellamya chinensis) supports consumers in urban lake food webs. Ecosphere 7(5):e01293. https://doi.org/10.1002/ecs2.1293
Vander Zanden MJ, Vadeboncoeur Y (2002) Fishes as integrators of benthic and pelagic food webs in lakes. Ecology 83:2152–2161. https://doi.org/10.1890/0012-9658(2002)083[2152:FAIOBA]2.0.CO;2
Vander Zanden MJ, Casselman JM, Rasmussen JB (1999) Stable isotope evidence for the food web consequences of species invasions in lakes. Nature 401:464–467. https://doi.org/10.1038/46762
Vaz JF, Teles HMS, Correa MA, Leite SPS (1986) Ocorrência no Brasil de Thiara (Melanoides) tuberculata (O.F. Müller, 1774) (Gastropoda, Prosobranchia), primeiro hospedeiro intermediário de Clonorchis sinensis (Cobbold, 1875) (Trematoda, Plathyhelmintes). Rev Saúde Públ 20:318–322. https://doi.org/10.1590/S0034-89101986000400008
Vitousek PM, D’Antonio CM, Loope LL, Westbrooks R (1996) Biological invasions as global environmental change. Am Sci 84(5):468–478
Vitule JRS, Skóra F, Abilhoa V (2012) Homogenization of freshwater fish faunas after the elimination of a natural barrier by a dam in Neotropics. Divers Distrib 18(2):111–120. https://doi.org/10.1111/j.1472-4642.2011.00821.x
White EM, Wilson JC, Clarke AR (2006) Biotic indirect effects: a neglected concept in invasion biology. Divers Distrib 12(4):443–455. https://doi.org/10.1111/j.1366-9516.2006.00265.x
Wood KA, Hayes RB, England J, Grey J (2017) Invasive crayfish impacts on native fish diet and growth vary with fish life stage. Aquat Sci 79:113–125. https://doi.org/10.1007/s00027-016-0483-2
Acknowledgements
Authors are grateful to Fundação de Desenvolvimento Científico e Cultural (FUNDECC) and Companhia Energética de Minas Gerais (CEMIG) for the financial support provided by means of the notice FUNDECC/CEMIG EAVG 1/2015. Thanks to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the scholarship provided. To Helena Ayres for the manuscript review. Paulo Santos Pompeu was awarded a productivity research Grant by CNPq/Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq No. 303548/2017-7) and a Minas Gerais State Researcher Grant by Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG PPM-00608/15). Finally, we thank the Editor and two anonymous reviewers for their valuable comments.
Funding
This work was funded by Fundação de Desenvolvimento Científico e Cultural (FUNDECC) and Companhia Energética de Minas Gerais (CEMIG) for the financial support provided by means of the notice FUNDECC/CEMIG EAVG 1/2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
N/A.
Consent for publication
The authors declare they consent to participate.
Consent for publication
The authors declare they consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rosa, D.M., de Sene, A.M., Moreira, M.Z. et al. Non-native prey species supporting fish assemblage biomass in a Neotropical reservoir. Biol Invasions 23, 2355–2370 (2021). https://doi.org/10.1007/s10530-021-02510-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-021-02510-x