Abstract
Plantations of Eucalyptus globulus Labill. have expanded rapidly over the last four decades across temperate parts of the globe. There has been growing concern about the potential invasiveness of this species. Portugal grows more E. globulus than any other country (by area). This study investigates poorly understood factors that may influence wilding establishment and spread from Portuguese plantations. We assessed: site-scale factors influencing the presence and abundance of E. globulus wildlings along plantation edges, as well as the size and reproductive state of these wildlings; and the dispersal distances and developmental state of wildlings away from the plantations edge. We found 1630 E. globulus wildlings in 129 survey plots (10 × 10 m) established along the edge of 67 plantations. Half of the surveyed wildlings were taller than 1.3 m. The presence and abundance of E. globulus wildlings was mainly influenced by factors such as soil cover, the age of mother-trees and their reproductive output. Around 8% of the surveyed wildlings were reproductive, including wildlings less than 1.3 m tall. The proportion of reproductive wildlings was mainly influenced by wildling size, the location relative to the plantation edge and the annual number of frost days. In 49 survey transects outside plantations we found 641 E. globulus wildlings up to a distance of 76 m. Establishment distance was associated with natural drainage lines and the dominant wind direction. We discuss the relevance of the results in terms of the potential invasiveness of E. globulus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Global species distributions are a consequence of diversification, dispersal and the rearrangement of land and sea over the geological time. Most taxa today are confined to geographic regions that are delineated by natural barriers to dispersal (Darwin 1859; Elton 1958). However, movement by humans has allowed many species to overcome such barriers, either through premeditated or inadvertent human will (Meyerson and Mooney 2007; Hulme 2009). Some of these exotic species have been a matter of great concern because of their ability to spread, causing negative ecological and economic impacts (Pimentel et al. 2001; Vilà et al. 2011).
Eucalyptus L’Hér. (Myrtaceae) is a large, diverse plant genus, comprising over 800 species, largely native to Australia (Nicolle 2018). Since the end of the 18th century several eucalypt species were introduced to different regions of the world (Potts 2004; Silva-Pando and Pino-Pérez 2016). Some eucalypt species were well adapted to the exotic environments and became widely exploited for timber, firewood, windbreaks, soil retention and, more recently, paper pulp production (Jacobs 1979; Potts 2004). Currently, it is estimated that eucalypt plantations cover around 20 million ha outside the native range (Rejmánek and Richardson 2011).
There has been a growing concern about the invasive potential of some eucalypt species. Several eucalypt species are considered to be naturalised (Pyšek et al. 2017) and a few species are regarded as invasive in different regions of the world (Rejmánek and Richardson 2013). Naturalised plant species have been defined as species able to generate reproductive offspring, normally near the parents, to form self-perpetuating populations, while invasive plant species are able to originate offspring distant from the parents, frequently causing negative ecological and economic impacts (Richardson et al. 2000; Blackburn et al. 2011).
A prior literature review, developed in the frame of another research initiative, revealed around 30 publications (papers and theses), assessing aspects of eucalypt invasiveness using wildling surveys or sowing experiments, mostly published in the last decade. These studies investigated the invasiveness of dozens of eucalypt species in different countries, such as Brazil (e.g. Miolaro et al. 2017), South Africa (e.g. Forsyth et al. 2004), USA (e.g. Callaham Jr et al. 2013), Spain (e.g. Calviño-Cancela et al. 2018), Portugal (e.g. Fernandes et al. 2016) and Australia (e.g. Calviño-Cancela and van Etten 2018). Eucalyptus globulus Labill. is one of the most extensively cultivated eucalypt species (Rejmánek and Richardson 2011), and is probably the most widely studied eucalypt species in terms of invasiveness. Also, it is considered to be the most widely occurring invasive eucalypt species (in seven out of 15 geographic regions of the world; see Rejmánek and Richardson 2013).
Eucalyptus globulus is native to SE Australia, including the Bass Strait Islands and Tasmania (Jordan et al. 1994). It was introduced throughout the world in the 19th century and plantations expanded rapidly in several countries in the late 20th century, especially to feed the paper and pulp markets (Potts et al. 2004). Portugal grows more E. globulus than any other country (by area), covering more than 800,000 ha, around 9% of the country (ICNF 2013; Deus et al. 2018a). The species has a wide environmental tolerance and is able to naturally establish in different regions of Portugal (Catry et al. 2015).
Studies assessing factors associated with E. globulus wildling establishment have shown that climate (Larcombe et al. 2013; Catry et al. 2015; Fernandes et al. 2017, 2018), plantation-related variables such as plantation size and age (Águas et al. 2017; Fernandes et al. 2018), fire (Larcombe et al. 2013; Calviño-Cancela et al. 2018), post-fire management (Águas et al. 2014), soil cover and disturbances (Kirkpatrick 1977; Fernandes et al. 2018), and the invaded land cover type (Calviño-Cancela and Rubido-Bará 2013; Fernandes et al. 2016, 2018) can all be important. Despite previous work investigating E. globulus invasiveness, knowledge gaps persist.
Little is known about the demographics of E. globulus wildling populations. Understanding the developmental stage of wildlings (e.g. size and transition to adult foliage) provides information about demography such as the number of cohorts and their survival rates, and is also crucial for making conclusions about effective recruitment because small E. globulus wildlings experience high levels of mortality (Calviño-Cancela and Rubido-Bará 2013; Fernandes et al. 2017). Few studies assess the height of naturally established wildlings (e.g. Águas et al. 2014; Fernandes et al. 2018) or the transition to adult foliage (e.g. Calviño-Cancela and van Etten 2018). Moreover, to our knowledge, no study investigated the occurrence of wildlings bearing reproductive organs. The onset of sexual maturity is of particular importance, because it is key for determining naturalisation and invasion (Richardson et al. 2000), and has not been studied in exotic E. globulus.
Few studies investigated the natural establishment of E. globulus wildlings along the edges of plantations. The edges of plantations are of particular importance because they represent the point of potential escape for wildings from cultivation, and observations on E. globulus plantations in Australia show that flowering seems to be more abundant in the trees located along the plantation edges (Barbour et al. 2008), meaning that propagule pressure is probably higher at these locations. Moreover, forest edges form transition zones between two adjacent ecosystems, featuring particular biotic (e.g. species diversity) and abiotic (e.g. microclimate) conditions (Murcia 1995). However, among the few studies investigating wildling establishment along plantation edges, the role of site-scale factors such as soil cover, local topography or slope aspect, was either not investigated (Águas et al. 2017) or inconclusive (Larcombe et al. 2013). It is known that fine-scale factors such as soil moisture, vegetation cover and slope are important in native eucalypt recruitment in Australia (Bailey et al. 2012; Wilson and Gibbons 2014). A recent study conducted in Portugal, using survey plots outside E. globulus plantations, also emphasised the importance of site-scale factors such as soil cover and soil disturbance in E. globulus recruitment (Fernandes et al. 2018).
Seeds of E. globulus do not have any long-distance dispersal adaptation, being normally dispersed by gravity and wind (Cremer 1977; Booth 2017). Nevertheless, seeds of E. globulus have been recorded 80 m away from plantations in NW Spain (Calviño-Cancela and Rubido-Bará 2013). Previous studies assessing the dispersal of wildlings from plantations (Calviño-Cancela and Rubido-Bará 2013; Larcombe et al. 2013; Fernandes et al. 2016) have not considered the importance of the primary vectors of dispersal, wind and water. Wind is probably the primary dispersal vector of eucalypt seeds (Cremer 1977; Calviño-Cancela and Rubido-Bará 2013), and the orientation of the plantation in relation to the prevailing wind may be an important factor determining the dispersal distances (McBride et al. 1988). Water has also been implicated in the dispersal of eucalypt seeds (Kirkpatrick 1977; Forsyth et al. 2004), and Larcombe et al. (2013) observed E. globulus wildlings established 175 m from a plantation down slope in a drainage line. Therefore, the presence of drainage lines (and presumably ephemeral running water) may be associated with increased wildling dispersal distances.
The goal of this study is to contribute to the understanding of E. globulus invasiveness, by addressing some important, but poorly explored topics. We surveyed wilding establishment along the edges of E. globulus plantations, and assessed dispersal into adjacent areas in Central Portugal. In particular, this study aimed to: a) assess the local site-scale factors influencing presence and abundance of E. globulus wildlings; b) quantify the size, foliage development (adult/juvenile), and proportion of reproductive wildlings; c) investigate the effective dispersal distance of wildlings from plantations, including the distribution of developmental classes; and d) assess factors affecting dispersal distance.
Materials and methods
Study region
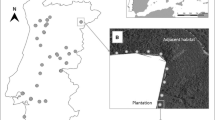
The study was conducted in Central Portugal (Fig. 1). Climate is typically Mediterranean, with hot, dry summers and moist winters. Mean annual precipitation is 652 mm, registering minimum values in July (4 mm) and maximum values in November (107 mm). Mean annual temperature is 17 °C, ranging from 10 °C in January to 24 °C in August (climatic data retrieved from IPMA 2018). The topography is irregular, featuring predominantly rugged terrain of hills and valleys, and river plains to a lesser extent. Sampling units, composed of survey plots and survey transects associated to E. globulus plantations, were located within latitudes 39°32′02″N and 38°47′04″N (distributed within a latitudinal distance of 83 km), and longitudes 7°46′52″W and 9°00′08″W (longitudinal distance of 106 km). Altitude of the sampling units ranged between 30 and 307 m. Plantations of E. globulus are widespread in this region, occupying c. 11% (c. 92,000 ha) of the area delimited by the referred coordinates.
Field surveys
Survey along plantation edges
We performed a survey along the edges of E. globulus plantations aiming to assess wildling development and the influence of site-scale factors on the establishment of E. globulus wildlings. All E. globulus plantations were owned by a Portuguese pulp company (Altri Florestal S.A.). The age of plantations ranged between 4 and 42 years (mean = 23.4; SD = 8.1). Since E. globulus reaches sexual maturity in plantations at 3–4 years of age (Potts and Gore 1995; Potts et al. 2008), plantations were composed of potentially reproductive individuals. The maximum stem age of the surveyed plantation was 12 years-old, meaning that flowering could have started 8–9 years before. In Portugal, plantations are typically managed by coppicing every 10–12 years for 2–3 rotations.
The survey design was similar to a study performed in Australia by Larcombe et al. (2013). The edge (perimeter) of a plantation was defined as a virtual line crossing the boles of the outer trees. The perimeter of the plantations was divided in different edges according to the edge aspect. In this way, a single plantation could comprise distinct edges, which were assessed separately using different transects. In each transect, the survey was performed on 10 × 10 m plots disposed along the plantation edge. In this way, the perimeter of the plantation divided the plot into two halves, i.e. two adjacent 5 × 10 m subplots, one inside (inside subplot) and one outside (outside subplot) the plantation. We aimed to accomplish a paired-plot survey within each transect, featuring a plot where wildlings were present (presence plot) combined with a plot where wildlings were absent (absence plot).
A presence plot was established in the first opportunity to frame a minimum of five wildlings within the plot area, regardless of their size and location (inside and/or outside subplots). The corresponding absence plot was established in the first available wildling-free plot area in either direction along the plantation edge, at a minimum distance of 50 m from the presence plot and 15 m away from any other wildling. Minimum distance between presence plots was 200 m. Due to the recurrent presence of E. globulus wildlings, it was not possible to couple an absence plot to every presence plot. Therefore, the survey included more presence than absence plots. We discarded all plantation edges distanced less than 50 m from another E. globulus plantation to reduce the chances of registering wildlings originating from other plantations. In the outside subplot, we subtracted the area of any surface unsuitable for wildling establishment (e.g. road).
In the presence plots, for each subplot (inside and outside), E. globulus wildlings were counted and categorised according to four size classes: ≤ 0.5 m (size 1); 0.5–1.3 m (size 2); 1.3–3.0 m (size 3); > 3 m (size 4). For each size class, we also registered the number of reproductive wildlings, i.e. wildlings bearing reproductive organs such as flowers or fruits. Wildlings were distinguished from planted individuals by their irregular location and size, within the regularly spaced and similarly sized planted E. globulus trees. Wildlings were also distinguished from resprouts by the presence of a lignotuber. Eucalyptus globulus is a proficient resprouter, able to regenerate through epicormic strands and from basal buds (Catry et al. 2013).
In each survey plot, including presence and absence plots, we collected the following data: (a) number of capsules on each of the ten planted eucalypts closest to the plot centre (visual estimate using binoculars), within a maximum distance of 25 m (categorised as 0: no capsules; 1: between 1 and 10 capsules; 2: 11 and 100 capsules; 3: 101 and 1000 capsules; 4: > 1000 capsules); (b) edge aspect, i.e. direction of exposure of the plantation edge (four cardinal directions); (c) plot topography, i.e. topographic format of the plot surface (flat; valley; slope; ridge); (d) plot aspect, direction of terrain inclination (flat; four cardinal directions, later reclassified north and west to wet, and south and east to dry,); (e) plot slope (degrees of inclination); and (f) the percentage of the plot covered by four soil cover types: bare ground, litter, herbs and shrubs.
A total of 103 edge transects were performed in 67 E. globulus plantations, including 129 presence plots and 53 absence plots. Field work was conducted between January and May 2014.
Survey outside plantations
We surveyed E. globulus wildlings established outside plantations using transects perpendicular to the plantation edges. This survey aimed to record the distance of wildling dispersal from the plantation together with wildling development. The establishment of a perpendicular transect was triggered by the detection of one E. globulus wildling at a distance greater than 10 m from the plantation edge. Transects had a maximum length of 80 m, corresponding to the maximum seed dispersal distance registered in E. globulus plantations in NW Spain (Calviño-Cancela and Rubido-Bará 2013). Minimum distance between transects was 100 m to avoid registering the same wildlings twice. Transects were established in areas with no other E. globulus plantations nearby, to avoid recording wildlings from other plantations.
The survey was conducted by one observer walking along the transect. Each step, previously calibrated, corresponded to 1 m length. At each step, the researcher looked at both sides and recorded the number and developmental stage of wildlings at that specific distance. Since the observer should not move away from the transect in order to have the same detection criteria across transects, we decided not to collect any specific traits which would require closer look, such as plant height and the existence of reproductive organs. In alternative, we characterised the developmental stage of wildlings using the conspicuous juvenile/adult foliage of E. globulus. Eucalyptus globulus is strongly heteroblastic, featuring strikingly different juvenile and adult foliage, making this developmental transition conspicuous and easily detectable (Johnson 1926; Velikova et al. 2008). We used the conservative assumption that wildlings with juvenile foliage are non-reproductive because the onset of sexual reproduction does not normally occur until after the transition to adult leaves (Potts et al. 2008), although juvenile wildlings with reproductive structures were identified in this study (see results; Fig. 2). For each transect, we collected the following data: transect land cover, i.e. predominant land cover along the transect (agriculture; shrubland; pine plantation; riparian zone; montado; paddock); drainage line presence, i.e. occurrence of a natural drainage line running away from the plantation edge (yes; no). Montado is a traditional agroforestry system in Portugal characterised by an open forest of native oaks.
A total of 49 survey transects were performed outside 33 E. globulus plantations (Fig. 1). Field work was conducted between January and May 2014.
Data analysis
Wildling presence
This analysis aimed to investigate the site-scale factors (see "Survey along plantation edges" section) influencing the presence of E. globulus wildlings, regardless of their size, along the edge of plantations. We used wildling presence-absence in the 10 × 10 m survey plots as a binary response variable. This analysis contrasts local conditions that are very adequate (at least five plants in 100 m2) for wildling establishment (recruitment and persistence in presence plots) with local conditions that are inadequate for establishment (absence plots). Presence was preferred to density as a dependent variable, because of poor performance of the density models that were produced at an exploratory stage of the analysis. Explanatory variables included the site-scale variables collected in each subplot (see "Survey along plantation edges" section) and the stem age of the planted E. globulus trees, i.e. the time (years) since planting or coppicing, information that was provided by the plantation owner. The variable stem age was included as a covariate to remove predictable variation on some site-scale factors associated with tree development. For instance, older stems should correspond to lower soil exposure and higher reproductive potential. Also, this factor (stem age) is constant among survey plots located within the same plantation. The reproductive capacity of the plantation was determined using the maximum capsule score from the 10 trees associated with the plot.
We used logistic regression to assess the influence of site-scale factors on the presence of E. globulus wildlings. Due to the hierarchical nature of our sampling design (transects nested within plantations), we used generalised linear mixed models (GLMM) with a binomial distribution and logit link function, using the R package lme4 (Bates et al. 2017). We fitted a model using presence-absence as the dependent variable, with transects nested within plantation as random effects. In order to avoid multi-collinearity between explanatory variables we computed variance inflation factors (VIF) to select the final set of potential predictors with VIF < 4 (Zuur et al. 2009) using the R package usdm (Naimi 2017).
The following fixed effects were included in this order: stem age, bare ground cover, litter cover, shrub cover, herb cover, plot topography, plot slope, maximum capsule score, plot aspect (wet and dry) and edge aspect. We used a backwards step model simplification process where non-significant terms were removed from the model, starting with categorical terms with the highest P-value. The reduced model was compared to the previous model using the likelihood ratio test. If the reduced model was significantly worse than the previous model, the dropped term was reinstated. Alternatively, the reduced model was accepted. This process was repeated until no non-significant terms remained. Residual diagnostics were conducted on simulated scaled residual plots, produced using the R package DHARMa (Hartig 2017). This procedure produces interpretable residuals from hierarchical mixed models. Model fit was assessed by conditional R2 (Nakagawa and Schielzeth 2013), using the R package piecewiseSEM (Lefcheck 2016).
Wildling abundance
We used a paired Wilcoxon test to compare the density (number of wildlings per area) of each wildling size class between the inside and outside subplots, retaining exclusively the presence plots (plots featuring a minimum of five wildlings).
We explored the influence of site-scale factors on the abundance of young plants, as an indicator of site suitability for recruitment. As a response variable, we used wildling density (number of wildlings per area). We restricted this analysis to the abundance of smaller wildlings (size 1; < 50 cm) in the inside subplots of the presence plots. This criterion was used for two reasons: first, younger wildlings should reflect more accurately the site-scale conditions registered at the date of the survey; second, inside subplots are less prone to disturbances (e.g. vehicles; people). Unlike the previous analysis, we discarded all absence plots in order to retain only locations where wildlings find conditions to establish. We used the same set of explanatory variables as in the presence-absence analysis, including stem age and site-scale variables (see "Survey along plantation edges" section).
Since density data were highly skewed and could not be transformed to meet the assumptions of generalised models, we used regression trees as an alternative approach. We chose multivariate regression trees (MRT) to assess wildling density. Among other features, MRT provide a very clear picture of the structure of the data and provide a highly intuitive insight into the kinds of interactions between variables (Crawley 2012). MRT result from the successive splitting of the sites (survey plots) into two groups based on a threshold from one of the explanatory variables, so that each group presents a minimum dissimilarity (De’ath 2002). The minimum number of observations to split (starting with n = 129) was set to 20 and the minimum number of observations in a terminal node was set to 6. The MRT was built using the R packages rpart (Therneau et al. 2018) and rpart.plot (Milborrow 2017).
Occurrence of reproductive wildlings
This analysis aimed to understand the factors influencing the occurrence of reproductive wildlings. We used the proportion of reproductive wildlings in each plot as the response variable. Smaller wildlings (size 1 and 2; < 1.3 m) were excluded from this analysis since only a very small percentage was found to be reproductive (< 0.5% in both size classes). Therefore, our sampling units were the inside and outside subplots featuring size 3 (1.3–3 m) and/or size 4 (> 3 m) wildlings.
We used a larger set of explanatory variables when compared to the previous analyses because the occurrence of reproductive eucalypt wildlings has been poorly investigated. Explanatory variables included: (a) the set of site-scale variables used in previous analyses (see "Survey along plantation edges" section); (b) the subplot location (inside or outside subplots); (c) the number of wildlings of size 3 and size 4 (two variables); (d) plantation-related variables, provided by Altri Florestal S.A., including the plantation age (time since planting), plantation rotation (first; second; third), the stem age (time since planting or coppicing) and site index (height of the dominant trees at ten years old); (e) climate, altitude and soil variables, retrieved from APA (2018), namely: the mean annual temperature (°C; average 1931–60), the mean annual precipitation (mm; average 1931–60), the mean annual number of frost days (average 1941–60), altitude (meters) and the soil type (cambisol; lithosol; luvisol).
In order to assess the factors influencing the proportion of reproductive wildlings, we used a generalised linear model (GLM) with a binomial distribution and logit link function. We computed VIF (> 4) to check for collinearity between variables. Model simplification and validation followed the procedure previously described for the presence model ("Wildling presence" section). Model fit was assessed using the proportion of explained deviance.
Distance of establishment from plantations
This analysis aimed to investigate the factors influencing the distance of establishment of E. globulus wildlings outside plantations. For this analysis, we used linear mixed models (LMM), with the nlme R package (Pinheiro et al. 2018). The dependent variable was the distance of wildling establishment from plantations. Since the dispersal distance of eucalypt wildlings has been less well investigated in eucalypt ecology, we included the same wider set of explanatory variables as in the analysis of reproductive wildlings (see "Occurrence of reproductive wildlings" section), without the site-scale variables. Additionally, we included the variable drainage line presence (see "Survey outside plantations" section) and variables retrieved from a digital terrain map: a) transect aspect, i.e. direction of exposure of that area (flat; four cardinal directions); b) transect slope, i.e. terrain inclination in that area (degrees). Land cover was not included in the analysis because some land cover types were underrepresented. Topographical variables (aspect; slope) were provided by Altri Florestal S.A. and originally retrieved from 1:25,000 cartography produced by the Portuguese Army Geographical Institute (IGeoE). Most transects were performed in downslope areas and transect aspect coincided roughly with the cardinal direction of the transect. The transect ID was included in the model as a random effect. We used VIF (> 4) to discard correlated variables. Model simplification and validation followed the previously described procedure. As in the presence model ("Wildling presence" section), model fit was assessed using the conditional R2. All statistical analyses were performed using R software (R Core Team 2017).
Results
Wildling establishment along plantation edges
A total of 1630 E. globulus wildlings were recorded in the 129 presence plots. Wildlings of size 2 (0.5–1.3 m) were the most frequent (n = 575; 35.3% of total), followed by size 3 (1.3–3 m; n = 481; 29.5%), size 4 (> 3 m; n = 334; 20.5%) and size 1 (< 0.5 m; n = 240; 14.7%). The overall wildling density (all plants in total area of presence plots) was 1461.2 wildlings ha−1, being higher in the inside subplots (1771.7 wildlings ha−1) compared to the outside subplots (1044 wildlings ha−1). Taking into account the factor wildling size, the overall wildling density was higher in the inside subplots for wildlings of size 2 (650.5 vs. 334.0 wildlings ha−1), size 3 (556.7 vs. 262.6 wildlings ha−1) and size 4 (405.0 vs. 157.6 wildlings ha−1), while it was lower for size 1 wildlings (159.5 vs. 289.9 wildlings ha−1), compared to the outside subplots (Fig. 3). The maximum wildling density was 6000 wildlings ha−1, registered in a 5x10 m inside subplot, composed exclusively of size 1 (< 0.5 m) wildlings. A paired Wilcoxon test showed that differences in wildling density between the inside and outside subplots were significant for wildlings of size 2 (P = 0.03), size 3 (P < 0.001) and size 4 (P < 0.001). We assessed the reproductive capacity of 1820 neighbour E. globulus trees in the survey plots, of which 69% possessed capsules.
Among the 1630 E. globulus wildlings found, there were 124 (7.6%) reproductive wildlings, i.e. sexually mature wildlings, possessing flowers or capsules. The overall density of reproductive wildlings was 115.6 wildlings ha−1, being higher in the inside subplots (146.9 wildlings ha−1) compared to the outside subplots (63.0 wildlings ha−1). We found one reproductive size 1 wildling (0.4% of the total size 1 wildlings) and two reproductive size 2 wildlings (0.3%; see Fig. 2). The proportion of reproductive wildlings was higher in size 3 (5%; n = 24) and size 4 wildlings (29%; n = 97).
Factors influencing wildling presence
The GLMM, performed to assess the influence of site-scale factors on wildling presence (Table 1), explained 32% of the variance. The variable stem age, included as a covariate, was retained in the model, but it was not significant. Three variables showed a significant, positive influence on wildling presence: litter cover, plot slope and maximum capsule score. The variable edge aspect also had a significant influence, particularly a negative effect of the west aspect on wildling presence.
Factors influencing wildling abundance
The MRT, used to assess the influence of site-scale factors on the abundance of size 1 (< 0.5 m) wildlings in the inside subplots (see "Wildling abundance" section), created six clusters of plots. Four variables, mostly soil cover-related, were responsible for the five partitions originating the six clusters of plots (Fig. 4). Wildling density was found to be negatively related with shrub cover (first two partitions). The remaining plots were split by stem age of 10 years (third partition), where plots under trees with older stem age registered higher wildling density. The percentage of bare ground created the fourth partition, where plots featuring higher proportion of bare ground had higher wildling density. The fifth partition was created by the percentage of herb cover, being negatively related to wildling density. The whole tree explained 22% of the variance.
Multivariate Regression Tree (MRT) of Eucalyptus globulus wildling density (plants ha−1; only wildlings up to 0.5 m tall–size class 1) using site-scale variables and stem age as explanatory variables (see "Wildling abundance" section). Each splitting node is represented by a box featuring the average wildling density (number above) and the corresponding number of plots (number below). Colour darkness in each box is proportional to the fitted value. The splitting variable and the threshold value responsible for the partition is presented below the box. The length of each branch is proportional to the difference in deviance between each node and the sums of the nodes below
Factors influencing reproductive wildlings
The final GLM produced to explore the factors influencing the proportion of reproductive E. globulus wildlings along the edge of plantations, retained 11 variables, but only seven were significant (P < 0.05). The proportion of reproductive wildlings was positively influenced by the location in the outside subplots, a higher site index, wet aspects, high abundance of size 4 (> 3 m) wildlings and older stem age in the cultivated trees. On the contrary, higher altitude and increased number of frost days negatively affected the proportion of reproductive wildlings (Table 2). The model explained 53% of deviance.
Wildling establishment outside plantations
Most survey transects were established in montado (n = 27; 55%) and shrubland (n = 14; 29%). A few transects were established in paddock areas (n = 5; 10%), and one transect (2%) was established in a pine stand, another in an agriculture area and another in a riparian area. We found a total of 641 E. globulus wildlings along the perpendicular transects, mostly established in montado (53.2%) and shrubland (39.9%). On average, we found 13.1 wildlings per transect (SD = 14.6; min. = 1; max. = 70).
Around 75% of the E. globulus wildlings escaped from plantations were found within a distance of 15 m from the plantation edge, and c. 91% within 30 m. There were 18 wildlings (c. 3%) located beyond 50 m from the edge of plantations. The maximum distance recorded was 76 m. Around 59% of the E. globulus wildlings recorded outside plantations had adult foliage. The proportion of wildlings exhibiting adult foliage increased with the distance from the plantations (Fig. 5).
Factors influencing the distance of establishment
The LMM, computed to understand the factors influencing the dispersal distance of E. globulus from plantations, highlighted the importance of drainage line presence, the soil type and the transect aspect (Table 3). The presence of natural drainage lines along the surveyed area enhanced the distance of wildling establishment. On the other hand, the distance of establishment was negatively influenced by west exposures and, with less importance, by the occurrence of podzols. The fitted model with these three variables explained 18% (Nagelkerke R2) of the observed variance.
Discussion
Wildling establishment along plantation edges
Most previous studies investigating the natural establishment of exotic eucalypts do not consider wildling development, especially the onset of sexual reproduction. This study provides evidence of the effective recruitment of E. globulus wildlings along the edges of plantations. Others have reported a high rate of mortality of E. globulus wildlings in the first months after germination (Fernandes et al. 2017). Calviño-Cancela and Rubido-Bará (2013) found that the survival probability of E. globulus seedlings increases every month, reaching 82% between the 6th and 8th months. We found no previous information regarding the growth rate of naturally established E. globulus wildlings. However, based on recent field observations in Portugal, we are confident that wildlings of size 3 (1.3–3 m) and size 4 (> 3 m), which comprise half of the surveyed wildlings in this study, should be more than six months old, and thus have high chances of survival.
We recorded higher wildling abundance in the inside subplots. A similar trend was found in other studies (Larcombe et al. 2013; Fernandes et al. 2016; Águas et al. 2017). This discrepancy between inside and outside subplots is probably related to the higher frequency of disturbances outside plantations, such as the passage of vehicles and people, or fuel reduction operations for fire prevention, targeting especially larger plants, as hypothesised by other studies (Larcombe et al. 2013; Águas et al. 2017). This discrepancy between inside and outside subplots is more significant for older plants (> 50 cm; sizes 2, 3 and 4). This is consistent with the findings of a similarly study in Australia by Larcombe et al. (2013), and seems to support the idea that disturbances such as multi-year management cycles or stochastic events outside the plantation may periodically reduce abundance.
The role of site-scale factors on wildling establishment
The presence of E. globulus wildlings along the edge of plantations was affected by factors as diverse as soil cover, topography, propagule pressure and aspect. We note that because presence considered wildlings of all sizes, some more dynamic site-scale variables, such as soil cover and maximum capsule score, may not portray the conditions at the time of wildling establishment.
Among the soil cover variables, litter positively affected wildling presence, being the most significant variable. We did not discriminate the type of litter but it included mostly eucalypt leaves, branches and bark, even though litter from other plant species was also common and, in a few locations, quite conspicuous. The amount of litter may reflect indirectly important factors for wildling establishment. For instance, litter is positively related to the residence time of the plantations, which in its turn indicates a greater propagule pressure and thus more chances of wildling establishment. More litter also suggests that the plantation had not been recently managed, therefore favouring wildling persistence. Likewise, more litter should correspond to lower cover of other categories, in particular shrubs or herbs which compete for light and soil resources, and thus may limit the establishment of E. globulus wildlings, which are particularly sensitive to competition (Kirkpatrick 1977). In fact, it is known that E. globulus litter causes negative allelopathic effects on other plant species (Souto et al. 1994, 2001).
Plot slope was also found to affect positively the presence of E. globulus wildlings. Catry et al. (2015) found a similar result in roadsides and hypothesised that, in steeper areas, plant competition is lower due to greater soil erosion, and there is lower likelihood of waterlogging. In our case, it is also likely that areas featuring steeper slopes are managed less frequently or less effectively in terms of vegetation removal, due to technical and operational limitations.
The maximum capsule score (an indicator of reproductive output) was a significant factor positively affecting the presence of E. globulus wildlings, which is consistent with the findings of a similar study in Australia by Larcombe et al. (2013). It is known that propagule pressure has a central importance in plant invasions (Simberloff 2009). In our study, locations where trees produce more flowers and seeds are also likely to have wildlings.
The west exposure of the edge of the plantation, was the only site-scale factor influencing negatively the presence of E. globulus wildlings. Winds in this region are predominantly from northwest, meaning that west edges are more frequently exposed to wind. Therefore, one explanation may be related with higher seed-shadow drift towards the inner part of the stand, resulting in lower wildling density close to the edge of the plantation. Another possible explanation may be that western plots have no shelter against predominant winds, which may act together with higher sunlight exposure and higher temperatures, resulting in higher drieness and therefore worst conditions for wildling establishment (Stoneman 1994; Wang et al. 1998).
The analysis of wildling abundance, using Multivariate Regression Trees (MRT), aimed to detect direct correspondence between local conditions and the abundance of young plants (< 0.5 m; see "Wildling abundance" section). We found that shrub cover, bare ground cover and herb cover, were responsible for four of the five data partitions in the MRT. These results seem to highlight the importance of competition with other vegetation in the emergence and development of E. globulus wildlings, because shrubs and herbs negatively affected wildling density, while bare ground had the opposite effect. The findings are also in line with the known preference of eucalypt wildlings for disturbed, competition-free areas (Kirkpatrick 1977; Fernandes et al. 2018). The positive effect of stem age on wildling abundance should reflect the positive influence of residence time and the increasing propagule pressure originating from the cultivated trees as they grow older (Richardson 1998; Lockwood et al. 2005; Simberloff 2009).
Reproductive offspring
To our knowledge, this is the first study to record and assess the reproductive status of E. globulus wildlings. Reproductive E. globulus wildlings were not found in a similar survey conducted on E. globulus plantations in Australia (Larcombe et al. 2013). Importantly, flowering was identified in juvenile wildlings (Fig. 2), which does not normally occur until after the transition to adult leaves (Potts and Gore 1995). Early flowering is often a target of breeding programs (Flachowsky et al. 2011), and may have been specifically targeted to accelerate production during the development of the E. globulus industry in Portugal. Time to first flowering is under strong genetic control in E. globulus (Jordan et al. 1999) and is highly heritable (Chambers et al. 1997). A short juvenile period was also found for E. grandis in South Africa (Musengi and Archibald 2017) and is a common trait among some of the most invasive plant species (Rejmánek and Richardson 1996; Grotkopp et al. 2002).
Among the factors influencing the proportion of reproductive wildlings, wildling size (height) was the most significant. Larger wildlings (size class 4; > 3 m) were more likely to be reproductive, which is similar to observations in South Africa for E. grandis wildlings (Musengi and Archibald 2017). Stem age was the second most influential factor, also showing a positive influence. Again, older plantations have more chance of accommodating older (larger) wildlings, which in its turn have a greater likelihood of being reproductive.
The number of frost days negatively affected the proportion of reproductive wildlings. Countrywide surveys in Portugal suggest that higher numbers of frost days limits wildling abundance (Catry et al. 2015; Deus et al. 2016). This has been associated with the sensitivity of wildlings to frost and freezing temperatures, but recurrent frost may also supress flowering or it may damage the reproductive structures of plants. Likewise, altitude was found to affect negatively the proportion of reproductive wildlings, despite a short altitudinal range (30–307 m) in our sampling areas. Altitude may have acted as a surrogate of local edapho-climatic conditions as hypothesised by Catry et al. (2015). In fact, a posterior analysis revealed that the sampling areas at higher altitudes are also located towards inland, probably experiencing lower temperatures and higher thermal amplitudes, which may supress flowering.
Being in the outside subplots increased the chances of E. globulus wildlings being reproductive. This probably reflects lower competition for light and nutrients, which may enhance flowering (Williams et al. 2006), meaning that wildlings on the edges of plantations and those dispersed into adjacent open areas may be more likely to be reproductive. This may be corroborated by findings in Australia, where greater abundance of flowering E. globulus trees was found in the edges of plantations, when compared to the trees inside plantations (Barbour et al. 2008), and where lower tree densities in plantations promoted flowering in the related E. nitens (Williams et al. 2006). Site index, which is an indicator of site productivity, also increased the proportion of reproductive wildlings. It is probable that the same site conditions that enhance the growth of cultivated trees (nutrients, moisture), also favour the development of reproductive organs in younger plants.
Wildling establishment outside plantations
Survey transects outside plantations were established in different land cover types, indicating that E. globulus can spread into different habitats. Land cover effect was not analysed, because some land cover types were underrepresented. The unequal distribution of land covers could be indicative of different levels of susceptibility to invasion or an unequal representation of the areas surrounding E. globulus in the study region. Nonetheless, other studies found that some habitats such as native forests and grasslands are more resistant to E. globulus invasion (Calviño-Cancela and Rubido-Bará 2013; Fernandes et al. 2016). In our study, most transects and most of the surveyed wildlings were established in montado, a common agroforestry system in the region characterised by an open forest of native oaks.
Our survey protocol was biased towards finding E. globulus wildlings outside plantations, because the survey transects were triggered by the existence of at least one wildling at a minimum distance of 10 m. However, a recent study conducted in Central and North Portugal, using random survey transects outside E. globulus plantations, found wildlings in nearly one-third of the transects (Fernandes et al. 2016), indicating that this is not a rare event.
In our study, we found E. globulus wildlings established up to a distance of 76 m, similarly to observations by Fernandes et al. (2016) in Portugal. These dispersal distances are consistent with the maximum seed dispersal distance from E. globulus plantations recorded in NW Spain (80 m; Calviño-Cancela and Rubido-Bará 2013). The sharp decrease in the number of E. globulus wildlings after 15 m was also observed in other studies investigating the escape of E. globulus (Larcombe et al. 2013; Fernandes et al. 2016) and other Eucalyptus spp. (Callaham Jr et al. 2013). Seeds of E. globulus do not have any particular adaptation for seed dispersal and most seeds are dispersed by gravity and wind (Cremer 1965, 1977). Calviño-Cancela and Rubido-Bará (2013) found that practically all E. globulus seeds (98.6%) fall within 15 m from the edge of plantations. Nevertheless, in this study, we found a larger proportion of wildlings at greater distances than in previous studies, which can be explained by our survey protocol. Instead of using survey plots along the transects outside plantations (e.g. Fernandes et al. 2016), we opted for a visual detection on both sides of the transect. As a result, we recorded more E. globulus wildlings than we would if we opted for survey plots. Our survey protocol should also explain why the proportion of adult E. globulus wildlings also increased with distance from the plantation, because larger wildlings are more easily detectable, while smaller wildlings likely remained unnoticed when distanced more than a few meters from the transect. It is important to stress that many of the surveyed wildlings established outside plantations possessed adult foliage, meaning that they overcame the most critical stage of survival and reached adulthood. This transition is recognised as a critical step in plant invasions (Richardson et al. 2000). Escaped wildlings can act as a stepping-stone for posterior spread in the absence of management (Moody and Mack 1988; Richardson et al. 1994).
The factors influencing the distance of establishment of E. globulus from plantations highlighted the importance of the main vectors of seed dispersal: wind and water. Wind is considered the main vector of dispersal of eucalypts (Cremer 1977). The distance of wildling establishment was inferior in west exposure when compared to east, in line with the dominant winds in this region, which are predominantly from northwest. The same trend was observed in California, where the greatest dispersal distances of E. globulus wildlings coincided with the direction of the prevailing winds (McBride et al. 1988).
Water is also acknowledged as an important dispersal vector of eucalypt seeds (Kirkpatrick 1977; Forsyth et al. 2004). In our study, the presence of a natural drainage line was found to increase the distance of establishment of E. globulus from plantations suggesting that hydrochory may play an important role in wildling dispersal. In Australia, Larcombe et al. (2013) suggested that a drainage line might be responsible for the establishment of an E. globulus wildling at a distance of 175 m from a plantation. Apart from wind and water, ants may also contribute to the secondary dispersal of E. globulus seeds in Portugal (Deus et al. 2018b). Our analysis revealed that podzols affected negatively the distance of establishment from plantations. Soil itself has no influence on seed dispersal but it may affect wildling establishment. A study conducted in Portugal reported the opposite effect, finding that podzols positively influenced wildling abundance (Catry et al. 2015). We suspect that the effect of soil type maybe an artefact of the analysis related to other important factors such as slope and land cover.
Conclusions
The survey along the edges of Eucalyptus globulus plantations in Central Portugal provided evidence of an effective wildling recruitment since many wildlings overcame the most critical period for survival. The abundance of young E. globulus wildlings was mostly affected by soil cover variables symptomatic of the harmful impact of plant competition. Wildling presence was influenced by a wider range of site-scale factors including slope, aspect of the plantation edge and propagule pressure. Around 8% of the surveyed wildlings reached sexual maturity, supporting the hypothesis that the species is naturalised. A few very young wildlings were reproductive suggesting that the species can have a very short juvenile period. The proportion of reproductive wildlings was related to different factors ranging from plant development (positively related to wildling size), plantation-related (positively related to stem age and site productivity) and climatic variables (negatively related to the number of frost days).
This study also provides evidence of an effective recruitment of E. globulus outside plantations, in different habitats, since more than half of the surveyed wildlings outside plantations were adult. Wildlings were found up to 76 m of distance. Greater dispersal distances coincided roughly with the direction of the prevailing winds and the presence of natural drainage lines.
This study tackled important knowledge gaps about the demography, phenology and ecology of E. globulus in the introduced range, which may help to identify the factors and situations enhancing E. globulus recruitment and escape from plantations. The development of reproductive wildlings can increase propagule pressure from plantation edges and outside plantations they can act as a stepping-stone for posterior spread in the absence of management. Particular caution is needed in downslope aligned with predominant winds and with the presence of drainage lines capable of significantly enhancing seed dispersal. Further research is needed to evaluate the reproductive capacity of E. globulus wildlings escaped from plantations, the parental relationships among naturally established populations outside plantations, the rate of expansion and the ecological succession of invaded areas.
References
Águas A, Ferreira A, Maia P et al (2014) Natural establishment of Eucalyptus globulus Labill. in burnt stands in Portugal. For Ecol Manage 323:47–56. https://doi.org/10.1016/j.foreco.2014.03.012
Águas A, Larcombe MJ, Matias H et al (2017) Understanding the naturalization of Eucalyptus globulus in Portugal: a comparison with Australian plantations. Eur J Forest Res 136:433–446. https://doi.org/10.1007/s10342-017-1043-6
APA (2018) SNIAmb: Sistema Nacional de Informação de Ambiente [Portuguese National System of Environmental Information]. Agência Portuguesa do Ambiente (APA). https://sniamb.apambiente.pt/content/geo-visualizador. Accessed Sep 2018
Bailey TG, Davidson NJ, Close DC (2012) Understanding the regeneration niche: microsite attributes and recruitment of eucalypts in dry forests. For Ecol Manage 269:229–238. https://doi.org/10.1016/j.foreco.2011.12.021
Barbour RC, Otahal Y, Vaillancourt RE et al (2008) Assessing the risk of pollen-mediated gene flow from exotic Eucalyptus globulus plantations into native eucalypt populations of Australia. Biol Conserv 141:896–907. https://doi.org/10.1016/j.biocon.2007.12.016
Bates D, Maechler M, Bolker B et al. (2017) lme4: Linear Mixed-Effects Models using ‘Eigen’ and S4. https://CRAN.R-project.org/package=lme4. Accessed Feb 2018
Blackburn TM, Pyšek P, Bacher S et al (2011) A proposed unified framework for biological invasions. Trends Ecol Evol 26:333–339
Booth TH (2017) Going nowhere fast: a review of seed dispersal in eucalypts. Aust J Bot. https://doi.org/10.1071/bt17019
Callaham Jr MA, Stanturf JA, Hammond WJ et al. (2013) Survey to evaluate escape of Eucalyptus spp. seedlings from plantations in Southeastern USA. Int J For Res: Article ID 946374. https://doi.org/10.1155/2013/946374
Calviño-Cancela M, Rubido-Bará M (2013) Invasive potential of Eucalyptus globulus: seed dispersal, seedling recruitment and survival in habitats surrounding plantations. For Ecol Manage 305:129–137. https://doi.org/10.1016/j.foreco.2013.05.037
Calviño-Cancela M, van Etten EJB (2018) Invasive potential of Eucalyptus globulus and Pinus radiata into native eucalypt forests in Western Australia. For Ecol Manage 424:246–258. https://doi.org/10.1016/j.foreco.2018.05.001
Calviño-Cancela M, Lorenzo P, González L (2018) Fire increases Eucalyptus globulus seedling recruitment in forested habitats: effects of litter, shade and burnt soil on seedling emergence and survival. For Ecol Manage 409:826–834. https://doi.org/10.1016/j.foreco.2017.12.018
Catry FX, Moreira F, Tujeira R et al (2013) Post-fire survival and regeneration of Eucalyptus globulus in forest plantations in Portugal. For Ecol Manage 310:194–203. https://doi.org/10.1016/j.foreco.2013.08.036
Catry FX, Moreira F, Deus E et al (2015) Assessing the extent and the environmental drivers of Eucalyptus globulus wildling establishment in Portugal: results from a countrywide survey. Biol Invasions 17:3163–3181. https://doi.org/10.1007/s10530-015-0943-y
Chambers PGS, Potts BM, Tilyard P (1997) The genetic control of flowering precocity in Eucalyptus globulus ssp. globulus. Silvae Genet 46:207–214
Crawley MJ (2012) The R book, 2nd edn. Wiley, Chichester
Cremer KW (1965) How Eucalypt fruits release their seed. Aust J Bot 13:11–16. https://doi.org/10.1071/BT9650011
Cremer KW (1977) Distance of seed dispersal in eucalypts estimated from seed weights. Aust For Res 7:225–228
Darwin C (1859) On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life. John Murray, London
De’ath G (2002) Multivariate regression trees: a new technique for modeling species-environment relationships. Ecology 83:1105–1117. https://doi.org/10.1890/0012-9658(2002)083%5b1105:MRTANT%5d2.0.CO;2
Deus E, Silva JS, Catry FX et al (2016) Google Street View as an alternative method to car surveys in large-scale vegetation assessments. Environ Monit Assess 188:1–14. https://doi.org/10.1007/s10661-016-5555-1
Deus E, Silva JS, Castro-Díez P et al (2018a) Current and future conflicts between eucalypt plantations and high biodiversity areas in the Iberian Peninsula. J Nat Conserv. https://doi.org/10.1016/j.jnc.2018.06.003
Deus E, Silva JS, Marchante H et al (2018b) Are post-dispersed seeds of Eucalyptus globulus predated in the introduced range? Evidence from an experiment in Portugal. Web Ecol 18:67–79. https://doi.org/10.5194/we-18-67-2018
Elton CS (1958) The ecology of invasions by animals and plants. Methuen, London. https://doi.org/10.1007/978-94-009-5851-7
Fernandes P, Antunes C, Pinho P et al (2016) Natural regeneration of Pinus pinaster and Eucalyptus globulus from plantation into adjacent natural habitats. For Ecol Manage 378:91–102. https://doi.org/10.1016/j.foreco.2016.07.027
Fernandes P, Máguas C, Correia O (2017) Combined effects of climate, habitat, and disturbance on seedling establishment of Pinus pinaster and Eucalyptus globulus. Plant Ecol. https://doi.org/10.1007/s11258-017-0706-1
Fernandes P, Máguas C, Correia O et al (2018) What drives Eucalyptus globulus natural establishment outside plantations? The relative importance of climate, plantation and site characteristics. Biol Invasions 20:1129–1146. https://doi.org/10.1007/s10530-017-1614-y
Flachowsky H, Le Roux P-M, Peil A et al (2011) Application of a high-speed breeding technology to apple (Malus × domestica) based on transgenic early flowering plants and marker-assisted selection. N Phytol 192:364–377. https://doi.org/10.1111/j.1469-8137.2011.03813.x
Forsyth GG, Richardson DM, Brown PJ et al (2004) A rapid assessment of the invasive status of Eucalyptus species in two South African provinces: working for water. S Afr J Sci 100:75–77
Grotkopp E, Rejmánek M, Rost TL (2002) Toward a causal explanation of plant invasiveness: seedling growth and life-history strategies of 29 pine (Pinus) species. Am Nat 159:396–419. https://doi.org/10.1086/338995
Hartig F (2017) DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models. R package version 0.1.5. https://CRAN.R-project.org/package=DHARMa. Accessed Feb 2018
Hulme PE (2009) Trade, transport and trouble: managing invasive species pathways in an era of globalization. J Appl Ecol 46:10–18. https://doi.org/10.1111/j.1365-2664.2008.01600.x
ICNF (2013) IFN6–Área dos usos do solo e das espécies florestais de Portugal continental: resultados preliminares [6th National Forest Inventory—Area of the land-uses and the tree species in mainland Portugal: preliminary results]. Instituto da Conservação da Natureza e das Florestas, Lisbon
IPMA (2018) Climatological normals 1981-2010 (provisional)—District of Santarém. http://www.ipma.pt/pt/oclima/normais.clima/1981-2010/018/. Accessed Jan 2018
Jacobs MR (1979) Eucalypts for planting. Food and Agriculture Organization of the United Nations, Rome
Johnson ED (1926) A comparison of the juvenile and adult leaves of Eucalyptus globulus. N Phytol 25:202–212. https://doi.org/10.1111/j.1469-8137.1926.tb06691.x
Jordan GJ, Borralho NMG, Tilyard P et al (1994) Identification of races in Eucalyptus globulus spp globulus based on growth traits in Tasmania and geographic distribution. Silvae Genet 43:292–298
Jordan GJ, Potts BM, Wiltshire RJE (1999) Strong, independent, quantitative genetic control of the timing of vegetative phase change and first flowering in Eucalyptus globulus ssp. globulus (Tasmanian Blue Gum). Heredity (Edinb) 83:179–187
Kirkpatrick JB (1977) Eucalypt invasion in Southern California. Aust Geogr 13:387–393. https://doi.org/10.1080/00049187708702717
Larcombe MJ, Silva JS, Vaillancourt RE et al (2013) Assessing the invasive potential of Eucalyptus globulus in Australia: quantification of wildling establishment from plantations. Biol Invasions 15:2763–2781. https://doi.org/10.1007/s10530-013-0492-1
Lefcheck JS (2016) piecewiseSEM: piecewise structural equation modelling in R for ecology, evolution, and systematics. Methods Ecol Evol 7:573–579. https://doi.org/10.1111/2041-210X.12512
Lockwood JL, Cassey P, Blackburn T (2005) The role of propagule pressure in explaining species invasions. Trends Ecol Evol 20:223–228. https://doi.org/10.1016/j.tree.2005.02.004
McBride JR, Sugihara N, Amme D (1988) The effects of Eucalyptus establishment on native plant communities. In: Boyd D (ed) Focused environmental study: restoration of Angel Island natural areas affected by Eucalyptus. California Department of Parks and Recreation, California, pp 43–85
Meyerson LA, Mooney HA (2007) Invasive alien species in an era of globalization. Front Ecol Environ 5:199–208. https://doi.org/10.1890/1540-9295(2007)5%5b199:IASIAE%5d2.0.CO;2
Milborrow S (2017) rpart.plot: Plot ‘rpart’ Models: an enhanced version of ‘plot.rpart’. R package version 2.1.2. https://CRAN.R-project.org/package=rpart.plot Accessed Feb 2018
Miolaro LG, Gonçalves AN, Mendes JCT et al (2017) Spontaneous regeneration of eucalypts from seed production areas. Biol Invasions 10:1–5. https://doi.org/10.1007/s10530-017-1397-1
Moody ME, Mack RN (1988) Controlling the spread of plant invasions: the importance of nascent foci. J Appl Ecol 25:1009–1021. https://doi.org/10.2307/2403762
Murcia C (1995) Edge effects in fragmented forests: implications for conservation. Trends Ecol Evol 10:58–62. https://doi.org/10.1016/S0169-5347(00)88977-6
Musengi K, Archibald S (2017) Demographics of Eucalyptus grandis and implications for invasion. Koedoe 59:1–12. https://doi.org/10.4102/koedoe.v59i1.1437
Naimi B (2017) usdm: Uncertainty Analysis for Species Distribution Models. R package version 1.1-18. https://CRAN.R-project.org/package=usdm. Accessed Feb 2018
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Nicolle D (2018) Classification of the eucalypts (Angophora, Corymbia and Eucalyptus). Version 3. http://www.dn.com.au/Classification-Of-The-Eucalypts.pdf. Accessed Feb 2018
Pimentel D, McNair S, Janecka J et al (2001) Economic and environmental threats of alien plant, animal, and microbe invasions. Agric Ecosyst Environ 84:1–20. https://doi.org/10.1016/S0167-8809(00)00178-X
Pinheiro J, Bates D, DebRoy S et al. (2018) nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1-137. https://CRAN.R-project.org/package=nlme. Accessed Oct 2018
Potts BM (2004) Genetic improvement of eucalypts. In: Burley J, Evans J, Youngquist JA (eds) Encyclopedia of forest science. Elsevier Science, Oxford, pp 1480–1490
Potts BM, Gore P (1995) Reproductive biology and controlled pollination of Eucalyptus—a review. School of Plant Science, University of Tasmania, Hobart
Potts BM, Vaillancourt RE, Jordan G et al (2004) Exploration of the Eucalyptus globulus gene pool. In: Borralho N, Pereira JS, Marques C, Coutinho J, Madeira M, Tomé M (eds) Eucalyptus in a changing world—IUFRO conference, Aveiro, 11–15 October 2004 2004. RAIZ, Instituto Investigação de Floresta e Papel, pp 46–61
Potts BM, McGowen MH, Williams DR et al (2008) Advances in reproductive biology and seed production systems of Eucalyptus: the case of Eucalyptus globulus. South For 70:145–154. https://doi.org/10.2989/SOUTH.FOR.2008.70.2.10.538
Pyšek P, Pergl J, Essl F et al (2017) Naturalized alien flora of the world: species diversity, taxonomic and phylogenetic patterns, geographic distribution and global hotspots of plant invasion. Preslia 89:203–274. https://doi.org/10.23855/preslia.2017.203
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing. http://www.R-project.org. Accessed Nov 2017
Rejmánek M, Richardson DM (1996) What attributes make some plant species more invasive? Ecology 77:1655–1661
Rejmánek M, Richardson DM (2011) Eucalypts. In: Simberloff D, Rejmánek M (eds) Encyclopedia of biological invasions. University of California Press, California, pp 203–209
Rejmánek M, Richardson DM (2013) Trees and shrubs as invasive alien species—2013 update of the global database. Divers Distrib 19:1093–1094. https://doi.org/10.1111/ddi.12075
Richardson DM (1998) Forestry trees as invasive aliens. Conserv Biol 12:18–26. https://doi.org/10.1111/j.1523-1739.1998.96392.x
Richardson DM, Williams PA, Hobbs RJ (1994) Pine invasions in the Southern Hemisphere: determinants of spread and invadability. J Biogeogr 21:511–527. https://doi.org/10.2307/2845655
Richardson DM, Pyšek P, Rejmánek M et al (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107
Silva-Pando FJ, Pino-Pérez R (2016) Introduction of Eucalyptus into Europe. Aust For 79:283–291. https://doi.org/10.1080/00049158.2016.1242369
Simberloff D (2009) The role of propagule pressure in biological invasions. Annu Rev Ecol Evol Syst 40:81–102. https://doi.org/10.1146/annurev.ecolsys.110308.120304
Souto XC, Gonzales L, Reigosa MJ (1994) Comparative analysis of allelopathic effects produced by four forestry species during decomposition process in their soils in Galicia (NW Spain). J Chem Ecol 20:3005–3015. https://doi.org/10.1007/BF02098405
Souto XC, Bolaño JC, González L et al (2001) Allelopathic effects of tree species on some soil microbial populations and herbaceous plants. Biol Plant 44:269–275. https://doi.org/10.1023/A:1010259627812
Stoneman GL (1994) Ecology and physiology of establishment of eucalypt seedlings from seed: a review. Aust For 57:11–29. https://doi.org/10.1080/00049158.1994.10676109
Therneau T, Atkinson B, Ripley B (2018) rpart: Recursive Partitioning and Regression Trees. R package version 4.1-13. https://CRAN.R-project.org/package=rpart. Accessed Feb 2018
Velikova V, Loreto F, Brilli F et al (2008) Characterization of juvenile and adult leaves of Eucalyptus globulus showing distinct heteroblastic development: photosynthesis and volatile isoprenoids. Plant Biol 10:55–64. https://doi.org/10.1055/s-2007-964964
Vilà M, Espinar JL, Hejda M et al (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708. https://doi.org/10.1111/j.1461-0248.2011.01628.x
Wang D, Bachelard EP, Banks JCG (1998) Growth and water relations of seedlings of two subspecies of Eucalyptus globulus. Tree Physiol 4:129–138. https://doi.org/10.1093/treephys/4.2.129
Williams DR, Potts BM, Neilsen WA et al (2006) The effect of tree spacing on the production of flowers in Eucalyptus nitens. Aust For 69:299–304. https://doi.org/10.1080/00049158.2006.10676250
Wilson N, Gibbons P (2014) Microsite factors influencing Eucalyptus regeneration in temperate woodlands. Ecol Manage Restor 15:155–157. https://doi.org/10.1111/emr.12108
Zuur A, Ieno EN, Walker N et al (2009) Mixed effects models and extensions in ecology with R. Statistics for biology and health. Springer, New York. https://doi.org/10.1007/978-0-387-87458-6
Acknowledgements
This research was produced under the WildGum Project (PTDC/AGRFOR/2471/2012), funded by the Portuguese Foundation for Science and Technology (FCT). ED and AA were supported by a Ph.D. Grant provided by FCT (PB/BD/113936/2015; SFRH/BD/76899/2011). FXC was supported by a postdoctoral FCT Grant (SFRH/BPD/93373/2013). MJL ‘s travel to Portugal was funded by the WildGum project and a Maxwell Ralf Jacobs Scholarship Grant. The authors thank Altri Florestal S.A. for providing information, logistic support and allowing the access to the surveyed plantations.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deus, E., Silva, J.S., Larcombe, M.J. et al. Investigating the invasiveness of Eucalyptus globulus in Portugal: site-scale drivers, reproductive capacity and dispersal potential. Biol Invasions 21, 2027–2044 (2019). https://doi.org/10.1007/s10530-019-01954-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-019-01954-6