Abstract
Eucalyptus globulus has great economic importance in the Iberian Peninsula and is now the most widespread tree species on the Portuguese mainland. We aimed to evaluate the establishment capacity of E. globulus from plantations into natural habitats and to understand its association with the climate, plantation characteristics and host site characteristics. We surveyed 50 E. globulus plantations across Portugal looking at the natural establishment (occurrence and density) of E. globulus in habitats adjacent to plantations. Eucalypts presence was only recorded in 8% of the plots. Site characteristics (such as habitat type, vegetation cover and disturbance) were the most important variables influencing the natural recruitment of E. globulus in comparison to climate or plantation variables. Forest and grassland were the most resistant habitats to eucalyptus invasion while highly disturbed areas can be considered hubs for eucalypt density. The high importance of site characteristics reflects that most of the variability in eucalypt establishment occurs at small spatial scales. Thus, monitoring and management efforts should focus on those sites with higher establishment probability (i.e. open areas) trying to promote native vegetation and reduce disturbance levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forest plantations account for 264 million hectares globally (7% of the total forest area), of which approximately 25% consist of non-native species (FAO 2010). The use of non-native species in forest plantations (mostly pine and eucalypt) is primarily related to the wide range of ecological requirements of these species, their rapid growth and high productivity. In many countries, these species play a significant role towards the economy, particularly in areas where native species do not perform well (Dodet and Collet 2012; Richardson 1998). However, the prolific nature of the non-native tree species selected and their widespread use increases the risk of these species invading the natural and semi-natural habitats surrounding a plantation (Dodet and Collet 2012; Essl et al. 2010; Richardson 1998; Richardson and Rejmánek 2011; Van Der Meer et al. 1999). Forest plantations are one of the main sources of plant invaders worldwide and the expansion of non-native trees into habitats outside plantations is a relevant concern for managers and conservationists (Dodet and Collet 2012; Richardson 1998). In order to manage forest plantations sustainably (minimizing ecological risks and maintaining economical productivity) it is important to study the main factors driving the natural establishment of non-native plant species and determine the capacity of a species to colonize outside the planted areas (Richardson and Rejmánek 2011).
Eucalyptus species are among the most cultivated forest species worldwide. They are mainly planted in temperate, tropical and subtropical regions, where they cover approximately 20 million ha (Rejmánek and Richardson 2011). Eucalyptus globulus Labill. (Tasmanian blue gum), native to southeast Australia, is currently one of the most important tree species planted worldwide, with the majority of plantations occurring in the Iberian Peninsula. Eucalyptus globulus was first introduced into Portugal in the mid-19th century and its use was dramatically promoted by the pulp and paper industries (Alves et al. 2007). Today, E. globulus is the most widespread tree species on the Portuguese mainland, representing 26% of its forest cover (ICNF 2013) with great economic importance to the western region (Potts et al. 2004).
Based on the Australian Weed Risk Assessment (WRA), E. globulus has been reported as a species with “high” invasion risk (Daehler et al. 2004; Gassó et al. 2009; Gordon et al. 2012; Marchante et al. 2014), and is a particular problem in seven regions globally: North, Central and South America, Europe, New Zealand, Pacific Islands and Indian Oceans Islands (Rejmánek and Richardson 2013). WRAs are based upon biological traits and expert knowledge of the species (Pheloung et al. 1999), with most risk assessments considering the history of invasiveness elsewhere (e.g. its distribution in areas where introduced). WRAs are rarely based solely on quantitative information collected from the field and, in this instance, several quantitative field studies have not found eucalypts to be invasive (Callaham et al. 2013; da Silva et al. 2011; Fernandes et al. 2016; Larcombe et al. 2013; Lorentz and Minogue 2015). In general, Eucalyptus have rarely spread considerable distances from planting sites, their regeneration is sporadic (Callaham et al. 2013; Fernandes et al. 2016; Larcombe et al. 2013; Rejmánek and Richardson 2011) and seedlings have a high rate of mortality (Calviño-Cancela and Rubido-Bará 2013; da Silva et al. 2011; Lorentz and Minogue 2015; Rejmánek and Richardson 2011). These factors all contribute to the limited invasiveness of Eucalyptus species (Rejmánek and Richardson 2011). Studies on Pinus and Acacia have found that species invasiveness is related to repeated introductions, planting intensity and a long residence time (Gibson et al. 2011; Procheş et al. 2012; Wilson et al. 2009), which are however, the characteristics of many Eucalyptus species (including E. globulus in Portugal). In general, Eucalyptus are viewed as less invasive than several other widely cultivated trees such as pines and acacias (Rejmánek and Richardson 2011) but a paper by Rejmánek and Richardson (2011) opened with the question: “Are eucalyptus inherently less invasive, or are they just a ticking time bomb?”. Understanding the factors driving the natural establishment of E. globulus should help to evaluate the invasive potential of this species (Richardson and Rejmánek 2011).
Among the abiotic factors that determine biological invasions, the effect of climate is probably the most studied (Thuiller et al. 2005) and is considered to be the main driver of plant distribution from continental to regional scales (Milbau et al. 2009; Pearson et al. 2004). For example, summer droughts in the Mediterranean limits plant performance and prevents the establishment success of many non-native plants (Chytrý et al. 2008b). Although E. globulus is tolerant to a wide range of environmental conditions, water availability and low temperatures (mainly occurrence of temperatures below 0 °C) are the main climatic factors limiting its growth in Portugal (Almeida et al. 1994; Alves et al. 2012; Catry et al. 2015). Thus, given the Mediterranean character of most of the Portuguese territory (with associated drought stress during summer months), we hypothesize that E. globulus establishment is primarily affected by precipitation.
The establishment success of a non-native species depends on the characteristics of the species itself, the host community and the interaction between them (Lonsdale 1999; Rejmánek et al. 2005; Richardson and Pyšek 2006). However our knowledge about the potential natural establishment of E. globulus in different habitats is still scarce. Understanding why some sites are more susceptible to the establishment of a given plant species than others has been a central topic in biological invasion research (Chytrý et al. 2008b). In turn, the susceptibility of the receiving community can change in function of competitive and facilitative interactions, nutrient availability, or disturbance levels (e.g. Davis and Pelsor 2001; Davis et al. 2000; Traveset and Richardson 2014). The role of disturbance has been long recognized as an important driver of biological invasions (Elton 1958) and many studies have observed that disturbance, specifically the removal of native vegetation, favours the establishment of non-native plants (e.g. Alston and Richardson 2006; Davis and Pelsor 2001; Davis et al. 2000; Higgins and Richardson 1998; Mitchell et al. 2006). Disturbance has also been suggested to be a prerequisite for the establishment of eucalyptus (da Silva et al. 2011; Lorentz and Minogue 2015; Wevill and Read 2010).
The number of propagules arriving in a new environment has also been observed as one of the mechanisms that strongly contributes to the success of a non-native species colonizing an area (e.g. Catford et al. 2011, 2009; Krivánek et al. 2006; Lockwood et al. 2009; Pyšek et al. 2009; Rouget and Richardson 2003) and this is also true for Eucalyptus populations (Gordon et al. 2012; Rejmánek and Richardson 2011). For forestry species, the expansion and the intensity of the planted area determines the magnitude of the propagule pressure. The propagule pressure of eucalypts has the potential to be very high considering the quantity of trees planted, the distribution and area in which plantations occupy and the prolific seed production (Rejmánek and Richardson 2011). There is however, evidence to suggest that reproductive output is highly variable in E. globulus plantations (Barbour et al. 2008; Larcombe et al. 2014).
Nevertheless, the importance of propagule pressure on establishment success can vary depending on the local conditions (i.e. characteristics of host community). For instance, a small number of propagules can be sufficient to colonize a community with low resistance (a highly disturbed area), whereas a greater number of propagules may be necessary in locations with highly resistant communities (areas with intense competition and harsh abiotic conditions) (Krivánek et al. 2006; Lockwood et al. 2009; Pyšek et al. 2009; Rouget and Richardson 2003). Similarly, residence time (the time since a species was first introduced into a region) can also influence the success of non-native species (Dawson et al. 2009; Pyšek et al. 2009). Considering that data on these aspects is rarely available for most introduced species, the existence of detailed records of planting history for forestry species offers an opportunity to assess the relative importance of potential surrogates for propagule pressure and residence time in explaining the success of non-native species in colonizing an area.
Recently, Catry et al. (2015) studied the effect of abiotic factors on the natural establishment of E. globulus along roadside transects adjacent to eucalypt plantations in continental Portugal. However, the study did not consider the characteristics of surveyed plantations and other important site variables, such as vegetation cover and disturbance level. In addition, roadsides result from anthropogenic disturbances which are difficult to transpose to other habitat types. As a result, there is still the requirement to study the importance of propagule pressure, residence time and the characteristics of the host community on the natural establishment of E. globulus. In this study, we evaluated how climatic factors, plantation characteristics (i.e. age, size, productivity and fire recurrence of the plantation) and host site characteristics (i.e. habitat type, soil type, aspect, disturbance level and vegetation cover) are associated with the establishment of E. globulus into natural habitats. This study comprises of two specific questions: (1) what is the relative importance of site characteristics; plantation characteristics; and climate, for the natural establishment of Eucalyptus globulus from plantations? (2) once Eucalyptus globulus has established, which factors are associated with their abundance? We assessed the natural colonization of E. globulus in several habitat types adjacent to 50 plantations across mainland Portugal (~ 450 km). A close collaboration with plantation owners offered access to detailed information about E. globulus plantation (e.g. age of the stand, fire history, plantation history) and produced a unique database to estimate the importance of plantation characteristics in forestry species invasions.
Methods
Study species
In Portugal, most Eucalyptus globulus plantations are intensively managed through a coppice system (10–12 year rotations) and the wood is mainly used for pulp production (Soares et al. 2007). Plants reach sexual maturity after 3–4 years (Jordan et al. 1999) and each fruit (a woody capsule) contains approximately 6–25 viable seeds (Hardner and Potts 1995; Mimura et al. 2009). Seeds of E. globulus are small (but relatively large for eucalypts) with no specialized dispersal mechanism (Cremer 1977). Thus, seed dispersal distances are limited to 10–15 m from the edge of a plantation (Calviño-Cancela and Rubido-Bará 2013; Fernandes et al. 2016). Fruits are able to withstand fire which in turn triggers the dehiscence of seeds from the capsules (i.e. serotinous) (Reyes and Casal 2001). E. globulus seeds do not require a period of dormancy and germination is likely to take place within a year after seed release (Calviño-Cancela and Rubido-Bará 2013).
Study area
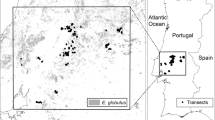
We sampled 50 E. globulus plantations in Portugal, conducting a country-wide field survey to document the establishment (natural regeneration) of E. globulus in natural habitats adjacent to the plantation (with at least one edge in contact) (Fig. 1). The average age of a plantation was 12-years-old, ranging from 8 to 26 years, (Table 1) and the average elevation was 216 m a.s.l (ranging from 14 m to 748 m). The climate is Mediterranean: cool rainy winters and hot dry summers; a mean annual temperature of 15 °C (ranging from 12 to 17 °C); and annual precipitation of 885 mm (ranging from 540 to 1438 mm) (Table 1). The major soil types found among sampled sites were cambisol, luvisol, podzol and lithosol (Table 1).
a Location of the 50 studied Eucalyptus globulus plantations (grey circles) in continental Portugal. b Survey plots (grey circles) adjacent to the boundary edge of Eucalyptus globulus plantations. c Schematic diagram of the plot area (10 m × 5 m) which were established perpendicular to the plantation edge
Survey method
Field surveys were conducted between May 2012 and May 2014, during Spring and Autumn. We performed a series of parallel plots of 50 m2 (10 m × 5 m) to quantify the establishment E. globulus (i.e. number of plants) in the adjacent habitats of the managed forest plantations of E. globulus across Portugal (Fig. 1b, c). Eucalyptus globulus plants counted in this study corresponded to all E. globulus plants that had naturally established from planted trees irrespectively of size. The eucalypt seedlings registered in our study had in most of the cases less than 1.3 m. In total, 1579 plots were surveyed across the 50 plantations of the study area (Fig. 1a; Table 2).
The plots were established with the longer edge perpendicular to the plantation boundary (Fig. 1c). To calculate the confidence level of the survey (the probability of sampling E. globulus at least once given a particular value of plots, β), we applied the following equation (Callaham et al. 2013; Green and Young 1993):
where, in this study, n = the number of plots needed to detect E. globulus plants, m = the mean frequency of occurrence of eucalypt and β = confidence level of the estimate, in the terminology of Green and Young (1993). In practice, it is very difficult to determine m before the end of the sampling and therefore it must be defined a priori. We adopted m = 0.1 for the definition of “rare” (Green and Young 1993). Other studies used similar approaches to detect tree seedlings in the surrounding areas of the plantations (Callaham et al. 2013; Lavi et al. 2005). Before beginning the survey, we calculated that 30 plots per site would result in a confidence level of approximately 0.95. Therefore, the distance between plots at each plantation was determined by dividing the total length of the plantation boundary by 30. For very small plantations (when the plantation perimeter was less than 500 m) a distance of 15 m was used between plots. For larger stands, we sampled more than 30 transects in order to have the similar intensity sampling among plantations with different areas. Areas with very rough terrain such as boulders, ravines, or steep slopes were not considered in the study. Therefore, the number of plots at each site varied according to the size of the sampled plantation and the accessibility of adjacent habitat; the total number of plots ranged from 8 to 58 plots per plantation. Consequently, the values of β varied from site to site (β mean = 0.92 ± 0.09). This method was chosen because it allows a high level of detection of rare events (Green and Young 1993). The geographic coordinates of the centre of each plot was recorded using a hand-held GPS (Garmin, GPSMAP 76CSx).
Explanatory variables
We characterized each sampled plot with a broad range of variables as potential surrogates for biotic and abiotic conditions that can determine the susceptibility of the site for eucalypts establishment (Table 1). We used a 1 m2 quadrat to calculate the percentage cover of vegetation (herbs and shrubs) and tree canopy cover (estimated as the sum of the projected crown area inside the plot limits) and used this to calculate the percentage of non-bare ground in each plot. Within each plot we visually classified the disturbance level into three categories: undisturbed or low disturbed (intact vegetation structure with low animal trampling); medium (sporadic to moderate debris or moderate animal/human trampling); and highly disturbed (high human trampling and accumulation of debris, dumping, logging and high soil erosion). We recorded the aspect of the plantation edge for each plot. To facilitate interpretation and decrease the complexity of the models, we transformed south-facing aspects into an unfavourable group where radiation and evapotranspiration are expected to be higher; the remaining aspects were classified as favourable (Beers et al. 1966; Kutiel and Lavee 1999). Plots were established in areas with moderate slope (i.e. 5% in average) in order to minimize the effect of slope in the relation of eucalypt density among sites. Slope and aspect could interact to change radiation levels. However, as plots were established in sites with moderate topography, we regarded aspect as a sufficient indicator of light penetration (González-Moreno et al. 2013b). The habitat type of each sampling plot was recorded in the field: (a) forest, mainly small woods with Quercus robur, Quercus pyrenaica and Quercus faginea); (b) shrubland, Mediterranean garrigue composed by a mix of short and medium shrubs (maximum 2 m of height) as rockrose (Cistus spp.) and others Cistaceae, heather (genus Erica and Calluna), gorse (Ulex spp.), broom (Cytisus spp.) and many aromatic species (e.g. Lavandula stoechas, Rosmarinus officinalis, Thymus vulgaris); and (c) grassland occupied by rich annual herbaceous species. As a result of agricultural land abandonment, shrubland was the most dominant habitat type surrounding forest plantations, The soil type in each plot was calculated from the Portuguese Atlas of Environment (APA 2013; scale 1:1,100,000).
Using data obtained from plantation owners, we characterized plantations based on their age, residence time, area, productivity and fire history (Table 1). Plantation age was defined as the time (in years) since trees were first planted or the time since last harvest, to the date of survey. Residence time was calculated based on the number of plantation rotations and cycles (the first cycle corresponds to the establishment of trees for new plantings, including 2–4 rotations and the next cycles correspond to replanting after 2–4 rotations). The productivity of plantation was categorized into three levels (low, medium and high) based on climate and soil suitability for the growth of E. globulus (Soares et al. 2007; Tomé et al. 2001). Fire history was quantified as the presence or absence of large fire events in the last decade (mostly corresponding to 2003 and 2005).
Finally, to account for climate influences, we selected seven variables that are ecologically most relevant for establishment success (i.e. plant growth and survival; Ibáñez et al. 2009) (Table 1). We selected mean annual temperature, mean minimum temperature of the coldest month, mean maximum temperature of the warmest month (the latter two to inform us about the temperature tolerance limits of E. globulus), temperature seasonality and precipitation seasonality (i.e. coefficient of variation in temperature/precipitation along the year), annual precipitation and mean precipitation of warmest quarter (which provides a proxy for dryness during summer months). Climatic data were downloaded from the WorldClim database and taken as averages from 1950 to 2000 (Hijmans et al. 2005). Each of the plot centre location was intersected with climate data at a 30 arcsec resolution (~ 1 km).
Data analysis
Considering the high number of zeros in the data, we used separate models to analyse occurrence (i.e. presence/absence of eucalypts) and density data (i.e. density models only used data from occupied plots). This approach solves the over-dispersion problem and the potential bias in parameter estimation when modelling zero-inflated data. Furthermore, the results of both models reflect the different stages of plant colonization and they are interpreted as two coupled process (Catford et al. 2011; González-Moreno et al. 2014; Ibáñez et al. 2009). Since there is no reason to expect factors to be of equal importance for both presence and density stages, the coupled modelling process ensures that factors that contribute to high density could be identified regardless of whether sites are presently occupied by eucalypts (Catford et al. 2011). We analysed the association of the occurrence of eucalypts (i.e. presence/absence data in 1579 plots) and subsequent density (i.e. number of plants) with site characteristics, plantation characteristics and climate (Table 1) by multimodel inference (Burnham and Anderson 2002) and variance partitioning techniques (Mood 1969) based on generalized linear mixed models (GLMM) with the identification of plantation as a random effect to account for pseudoreplication. Due to the low number of plots with eucalypts present in grassland (n = 3) and forest (n = 2) habitats, habitat type was not considered in the density model. This approach facilitated model convergence and results were similar if data is restricted to the main habitat type (i.e. shrubland; See Appendix Table 4). The final density dataset has information from 120 plots. Occurrence was modelled by setting a binomial distribution for the response variable and a logit link (presence model) (Quinn and Keough 2002). The density was modelled using a negative binomial distribution, which is typically used for count data when over-dispersion occurs (Gelman and Hill 2007). Preliminary analysis using Moran´s Index, indicated no relevant spatial autocorrelation in the model´s residuals (I < 0.4) (see Appendix Fig. 4).
Prior to modelling we checked the explanatory variables for collinearity using pair-wise Pearson´s correlation test, excluding correlation values higher than 0.6 and then selected ones with best explanatory power (see Appendix Tables 5 and 6). With regard to site variables, shrub and herb cover were correlated with non-bare ground cover. For the final model, we used the percentage of non-bare ground cover and canopy cover site variables. We kept all plantation variables. Climate variables were highly correlated and thus we selected only two non-collinear variables: temperature seasonality and annual precipitation.
Multimodel inference, the model selection method used in this study, provides a framework to ‘evaluate the relative strength of evidence in data for multiple hypotheses represented as models’ (Hobbs and Hilborn 2006). This method provides two approaches to find the best models to use: model selection and model averaging. Thus, multimodel inference method allowed us to identify the best possible models and rank all explanatory variables according to their importance on eucalyptus occurrence and density (Burnham and Anderson 2002). We performed multimodel inference based on all-subsets selection of GLMMs using Akaike’s information criterion corrected for a large number of predictors (AICc) (Burnham and Anderson 2002; González-Moreno et al. 2013a). First, for each group of predictors (site, plantation characteristics and climate) we selected the best model (smallest AICc). The same procedure was then performed combining the best variables of each group to establish the set of best candidate models. All models within four AICc units from the best model (delta < 4) were considered as the set of best models given the selected predictors. This threshold is within the limits adopted in other studies (González-Moreno et al. 2013a; Grueber et al. 2011). Within the best models subset we calculated the Akaike weight of evidence (wi) for each candidate model to rank the predictors in order of importance in their relation to response variables. The wi of each predictor was estimated as the sum of the model AICc weights across all models in which the selected predictor appeared. This means that the predictor with highest wi (i.e. closest to one) gets the highest relative importance (compared to the rest of predictors) to explain the response variables within the given data (Burnham and Anderson 2002). In addition, multimodel inference was used to estimate regression coefficients and their confidence intervals (with adjusted standard error) within the best models subset (Δ < 4). Coefficient of a given selected predictor was calculated as the sum of the predictor´s coefficient multiplied by the weight of evidence, across all possible models where the predictor is present (Burnham and Anderson 2002).
Variance partitioning techniques indicate the variability explained by the single and shared groups of variables (Mood 1969). We set up the best GLMMs selected in the previous approach for each dependent variable and incorporated site variables, plantation variables and climate variables individually before combining all three variables. Considering that deviance is a good measure of variability explained by a model, we used the deviance explained by each model to identify the pure and mixed effects by simple equation systems (Carrete et al. 2007; González-Moreno et al. 2013a). Following the same approach, we also partitioned the deviance of eucalyptus presence and density accounted by site characteristics within three best site factors explaining each dependent variable. We performed the variance partitioning analysis with the two of the most important non-collinear variables of each group, in order to compare the differences between the results with all variables and with the same number of variables by group. The results did not show significant differences between these two approaches (see Appendix Fig. 5). We only present the results for all variables.
Statistical analysis was performed with the R-CRAN software (R Development Core Team 2009). We used the package MuMIn for some procedures of the multimodel inference method and the VEGAN package as the base code for deviance-partition.
Results
Differences in E. globulus occurrence probability among habitat types
Eucalyptus globulus was recorded in 120 plots, representing 8% of the total plots assessed (Table 2). The survey detected a total of 480 eucalypts (Table 2), ranging from 0 to 20 individuals per plot. Natural regeneration of eucalyptus occurred mainly in shrubland with eucalyptus being recorded in 12% (115 plots) of the plots sampled of this habitat type, accounting for a total of 468 plants (Table 2). Considering the best occurrence model (see Appendix Table 7), forest and grassland were the most resistant habitat to eucalyptus establishment (Table 2).
Partitioning the influence of climate, plantation and site characteristics on the natural regeneration of eucalyptus
The variability of both the occurrence and density of eucalyptus was predominantly explained by site variables, which had a very high single effect on eucalypts occurrence (57%; Fig. 2a-i). In contrast, climate and plantation characteristics had a very low single effect and its explanatory power was shared mainly with site variables (Fig. 2a-i). Considering site variables separately, deviance partitioning-analyses revealed that the percentage cover of non-bare ground of the areas surrounding plantations explained most of the deviance of eucalyptus occurrence (47%; Fig. 2a-ii). The explanatory power of habitat type was partially shared with percentage of non-bare ground cover (3%) and accounts for 7% of single effect. The effect of disturbance on the occurrence of eucalypts was shared with percentage of non-bare ground cover (Fig. 2a-ii).
Deviance partitioning of E. globulus presence (a) and density (b) using generalized linear mixed models i among climate, plantation and site characteristics predictors, and ii among the three best site factors explaining each dependent variable. Each circle corresponds to a group of variables. Numbers within circles are the proportion of deviance explained by each set of predictors alone (non-overlapping parts of circles) or shared. Residuals indicate the deviance not explained by the models
Deviance partitioning indicates that a combined effect of plantation and site variables accounted for the largest proportion (22%) of variability of the eucalyptus density, followed by the combined effect of climate and site variables (18%). Site variables had the largest independent effect (17%) and the independent effect of the others two groups was practically inexistent (Fig. 2b-i). Considering site variables, the variability of eucalyptus density was mainly explained by the single effect of disturbance (17%), followed by the combined effect with non-bare ground cover (13%). The percentage of non-bare ground cover had a low single effect (8%) on eucalyptus density. The total amount of aspect effect was shared with disturbance and non-bare ground cover (Fig. 2b-ii).
The association of climate, plantation and site characteristics with the natural regeneration of eucalypts
The variables best explaining eucalyptus occurrence and density differed. In the case of density data analysis, climate and plantation variables were not selected (Tables 3 and see Appendix Table 8). The major correlates (i.e. higher weight of evidence) for eucalyptus presence were site factors: habitat type and non-bare ground cover. After site factors, productivity region (the best plantation predictor explaining eucalyptus occurrence) was the next in importance to model fit, followed by annual precipitation (the best climate factor) (Table 3). Specifically, we found a higher probability of eucalyptus occurring in shrubland than in forest habitat (Table 2). The probability of eucalyptus occurrence was lower at low productivity regions. Non-bare ground cover showed a significant negative association with eucalyptus occurrence. Finally, annual precipitation had a weak positive relation with eucalyptus occurrence (Table 3).
The variables best explaining eucalyptus density were disturbance and non-bare ground cover (Table 3). Considering the best density model (Table 8), eucalyptus density had a significant negative association with non-bare ground cover and was significantly higher in the highly disturbed plots (Fig. 3). Aspect also had some importance explaining eucalyptus density, with slightly higher densities recorded in northern aspects, although this difference was not significant (Table 3).
Mean (± SE) eucalypts density (no of eucalypts/plot) for low (N = 2), medium (N = 46) and, high (N = 72) disturbance levels. Letters indicate significant differences (p > 0.05) between disturbance levels according to post hoc test corrected for multiple hypothesis based on the best density model selected by AICc criteria
Discussion
Conceptual models to explain the establishment of non-native plants have suggested that a combination of having a large enough propagule pressure and a suitable biotic and abiotic environment favour establishment (Catford et al. 2009; Chytrý et al. 2008a). In this study, climate was used to estimate abiotic suitability (Walther et al. 2009), while site characteristics (including habitat type, vegetation cover and disturbance) represented the main characteristics of the host community (biotic and abiotic suitability) (Rejmánek et al. 2005). Among these variables, we found site characteristics to be the most important group of variables influencing the establishment of E. globulus (occurrence and density) outside planted areas in comparison to climate and plantation characteristics. This pattern reflects that most of the variability in E. globulus establishment occurs at a small spatial scale (site scale; < 1 km) (Milbau et al. 2009). Factors affecting species distribution most likely do so in a hierarchical manner, with different ecological factors affecting plant establishment success more strongly at different spatial scales (Milbau et al. 2009). For example, a study by Pearson et al. (2004) on the impact of climate and land cover changes on species distributions in Britain reported a dominant effect of climate measured at the European scale over land-cover data at the British scale. In accordance with this, climate and plantation characteristics might control variability in the establishment of E. globulus among plantations (regional scale: 2000–200 km), whilst site characteristics influence the establishment of E. globulus within a plantation (site scale: 10–1000 m). At larger scales, such as continental, climate would probably have a more important role in shaping the natural establishment of E. globulus (Catry et al. 2015; Larcombe et al. 2013). Despite the scale of our study, we found a relatively high importance of annual precipitation as a climate predictor and, as such, E. globulus was more likely to be present in areas with high annual precipitation. This finding has also been identified in previous studies (Águas et al. 2014; Catry et al. 2015; Larcombe et al. 2013). For example, Catry et al. (2015) found that the density of eucalypts was lowest below c. 600–700 mm of annual precipitation and sharply increased up to around 1500 mm. They also found that eucalypts density reached the lowest values above 2400 mm. The maximum annual precipitation sampled in our study was 1438 mm with an average of 885 mm and therefore, optimal conditions for the establishment of eucalypts.
Habitat type and the percentage of non-bare ground cover were the most important factors explaining the occurrence of eucalypts. Previous studies have also found habitat type to be the foremost factor explaining differences in plant invasions (Chytrý et al. 2008a; Gassó et al. 2012). Moreover, it has been suggested that invasive species traits related to successful establishment are habitat dependent (González-Moreno et al. 2013b; Lake and Leishman 2004; Lloret et al. 2005). The probability of Eucalyptus occurrence was higher in shrubland than in forest and grassland. Shrubs can have a nurse effect on seedlings of eucalypts, protecting them from harsh environmental conditions (Gómez-Aparicio et al. 2008). Shrubland may also provide greater light availability during later stages of seedling development. E. globulus are classified as shade-intolerant species, requiring a high light regime to develop (Correia et al. 1989). Native forests dominated by pedunculated oak, as other broad leaved trees, have low light irradiance regimes at ground level compared to the open vegetation of shrubland (Barbier et al. 2008). Therefore, the lowest probability of eucalypts occurrence found in forest patches is likely to be caused by a negative shading effect. A study by Calviño-Cancela and Rubido-Bará (2013) reported lower E. globulus seedling emergence under native forest patches (pedunculated oak) than in shrubland. This species is also reported as highly sensitive to drought, particularly at the seedling stage (Stoneman 1994). Eucalypts produce large quantities of seeds which are very small with no obvious endosperm. Thus, the newly emerged seedlings (sustained by cotyledon photosynthesis) need their roots to penetrate a suitable wet substrate very quickly in order to survive (Rejmánek and Richardson 2011). Grasslands normally show high seasonal fluctuation in water soil and nutrients availability (Rundel et al. 2014) which could negatively affect seedling survival (Rejmánek and Richardson 2011). High mortality during the initial phase of seedling growth, due to unsuitable environmental conditions or competition with other plants, has been recognized as one of the most important factors limiting eucalypts colonization (Callaham et al. 2013; da Silva et al. 2011; Rejmánek and Richardson 2011).
The survival of Eucalyptus seedlings is generally low in dense vegetation sites (Rejmánek and Richardson 2011). This is in agreement with the significant negative association found in our study between percentage non-bare ground cover (mainly determined by understory vegetation cover) and the probability of eucalypts occurrence and density. Non-bare ground cover alone explained 47% of the variability in eucalypts occurrence. Therefore, the higher probability of eucalypts establishment is mainly related to the horizontal structure of the community. Vegetation structure could be the responsible of the low establishment rate found in comparison to other studies. In Catry et al. (2015), eucalypts presence was found in 60% of the roadside transects adjacent to eucalypt plantations. On the contrary, E. globulus had established in only 8% of the total plots. Roadsides usually have a high percentage of exposed soil with little or no vegetation. This characteristic seems to increase the suitability of the community to the establishment of eucalypts, increasing the window of opportunity for eucalypt seeds to reach suitable sites.
The productivity of the region was, after site variables, the most important factor explaining the occurrence of eucalypts and the best plantation predictor. This factor is based on climate and soil suitability for E. globulus growth (Soares et al. 2007; Tomé et al. 2001). Thus, as climate factors, productivity can be also considered as a proxy for abiotic suitability for the growth of eucalypts at regional scale. The partition analyses revealed that plantation characteristics (e.g. plantation age and area) had a low explanatory strength and shared its explained deviance with site characteristics. The very short rotations (10–12 years) and intensive management of these plantations could explain these results. Thus, the plantations might not have enough time decrease to produce a significant propagule pool. The importance of plantation characteristics might increase in abandoned plantations (not considered in this study), as the propagule pressure builds with time. Thus, future research in abandoned eucalypt plantations is needed to better understand the role of residence time in E. globulus colonization.
The occurrence and density of eucalypts were modulated by different variables. In contrast to occurrence, the best models for density data only included site characteristics. These results indicate that once eucalypts have a high probability of establishment then the main aspect determining eucalypts success is the characteristics of the host community. This is in accordance with Milbau et al. (2009), who suggested that small-scale factors are better predictors at finer scales if conditions at higher levels are satisfied. Within site characteristics, disturbance level was the most important factor explaining eucalypts density. Disturbance, specifically native vegetation removal, is an important driver of invasive species expansion (e.g. Alston and Richardson 2006; Davis and Pelsor 2001; Davis et al. 2000; Elton 1958; Higgins and Richardson 1998; Mitchell et al. 2006). Our study found that an increase in the level of disturbance resulted in an increase in the density of eucalypts. This result agrees with previous findings by Lorentz and Minogue (2015) and Silva et al. (2011), where litter and plant removal improved the survival of eucalypts seedlings. In our study, the density of eucalypts found in all highly disturbed plots close to plantations (mean ± SD = 293 ± 673 plants ha−1) is similar to the mean density (277 plants ha−1) calculated by Catry et al. (2015) in roadside transects adjacent to eucalypt plantations in Portugal. However, the observed average eucalypts density in both studies is very low compared to well-known invasive forestry species such as pines for example (Richardson et al. 1994) and also with native maritime pine planted in Portugal (892 plants ha−1) (Fernandes et al. 2016).
Conclusions
In the present study we show that the establishment of E. globulus is limited to the natural habitats adjacent to managed plantations, compared to roadsides (Catry et al. 2015). This was particularity true when the habitat type surrounding the sampled plantations was forest or grassland. The percentage of bare ground had a significant effect on the establishment of eucalypts and our findings suggest that an increase in bare ground increases the susceptibility to eucalypts establishment. Therefore, our results suggest that by reducing the disturbance of habitats adjacent to eucalypt plantations during management activities the establishment success of E. globulus would be greatly reduced. In summary, our study suggests that eucalypts establishment is highly limited by biotic interactions (i.e. competition with other plants), low light and water availability. Nowadays, the expansion of trees into habitats outside of a plantation is a concern for managers and conservationists. These small-scale factors are essential to precise predictions at more local scales. Other higher scale factors, namely climate and productivity region, had lower explanatory strength on the establishment probability of eucalypts. As a cultivated tree, the current distribution of E. globulus in Portugal has resulted from human activity, targeting environmental conditions highly suitable for the species. Thus, monitoring efforts should focus on the site factors determining the colonization (i.e. ground cover, habitat type and disturbance). Industrial E. globulus plantations are grown with very short rotations and with regular basis management, so there are multiple opportunities for detection and control over the life of a plantation. Taking into account our results, we also suggest the establishment of “sentinel sites” using remote sensing tools (e.g. Google Earth) as described by Visser et al. (2014) or in situ visits during conventional management. This monitoring could provide an early-warning system for prevention management and, over time, it will produce very valuable data contributing to our understanding of eucalypts colonization dynamics.
References
Águas A, Ferreira A, Maia P, Fernandes PM, Roxo L, Keizer J, Silva JS, Rego FC, Moreira F (2014) Natural establishment of Eucalyptus globulus Labill. in burnt stands in Portugal. For Ecol Manag 323:47–56. https://doi.org/10.1016/j.foreco.2014.03.012
Almeida MH, Chaves MM, Silva JC (1994) Cold acclimation in eucalypt hybrids. Tree Physiol 14:921–932. https://doi.org/10.1093/treephys/14.7-8-9.921
Alston KP, Richardson DM (2006) The roles of habitat features, disturbance, and distance from putative source populations in structuring alien plant invasions at the urban/wildland interface on the Cape Peninsula, South Africa. Biol Conserv 2:183–198. https://doi.org/10.1016/j.biocon.2006.03.023
Alves AM, Pereira JS, Silva JMN (2007) A introdução e a expansão do eucalipto em Portugal. In: Alves AM, Pereira JS, Silva JMN (eds) O Eucaliptal Em Portugal. Impactes Ambientais E Investigação Científica. ISAPress, Lisboa, pp 13–24
Alves AM, Pereira JS, Correia AV (2012) Silvicultura—a gestão dos ecossistemas florestais. Fundação Calouste Gulbenkian, Lisbon
Barbier S, Gosselin F, Balandier P (2008) Influence of tree species on understory vegetation diversity and mechanisms involved—a critical review for temperate and boreal forests. For Ecol Manag 254:1–15. https://doi.org/10.1016/j.foreco.2007.09.038
Barbour R, Otahal Y, Vaillancourt R, Potts B (2008) Assessing the risk of pollen-mediated gene flow from exotic Eucalyptus globulus plantations into native eucalypt populations of Australia. Biol Conserv 141:896–907. https://doi.org/10.1016/j.biocon.2007.12.016
Beers TW, Dress PE, Wensel LC (1966) Aspect transformation in site productivity research. J For 64:691–692
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Callaham MA, Stanturf JA, Hammond WJ, Rockwood DL, Wenk ES, O’Brien JJ (2013) Survey to evaluate escape of Eucalyptus spp. seedlings from plantations in Southeastern USA. Int J For Res. https://doi.org/10.1155/2013/946374
Calviño-Cancela M, Rubido-Bará M (2013) Invasive potential of Eucalyptus globulus: seed dispersal, seedling recruitment and survival in habitats surrounding plantations. For Ecol Manag 305:129–137. https://doi.org/10.1016/j.foreco.2013.05.037
Carrete M, Grande JM, Tella JL, Sánchez-Zapata JA, Donázar JA, Díaz-Delgado R, Romo A (2007) Habitat, human pressure, and social behavior: partialling out factors affecting large-scale territory extinction in an endangered vulture. Biol Conserv 136:143–154. https://doi.org/10.1016/j.biocon.2006.11.025
Catford JA, Jansson R, Nilsson C (2009) Reducing redundancy in invasion ecology by integrating hypotheses into a single theoretical framework. Divers Distrib 15:22–40. https://doi.org/10.1111/j.1472-4642.2008.00521.x
Catford JA, Vesk PA, White MD, Wintle BA (2011) Hotspots of plant invasion predicted by propagule pressure and ecosystem characteristics. Divers Distrib 17:1099–1110. https://doi.org/10.1111/j.1472-4642.2011.00794.x
Catry FX, Moreira F, Deus E, Silva JS, Águas A (2015) Assessing the extent and the environmental drivers of Eucalyptus globulus wildling establishment in Portugal: results from a countrywide survey. Biol Invasions 17:3163–3181. https://doi.org/10.1007/s10530-015-0943-y
Chytrý M, Jarošík V, Pyšek P, Hájek O, Knollová I, Tichý L, Danihelka J (2008a) Separating habitat invasibility by alien plants from the actual level of invasion. Ecology 89:1541–1553. https://doi.org/10.1890/07-0682.1
Chytrý M, Maskell LC, Pino J, Pyšek P, Vilà M, Font X, Smart SM (2008b) Habitat invasions by alien plants: a quantitative comparison among Mediterranean, subcontinental and oceanic regions of Europe. J Appl Ecol 45:448–458. https://doi.org/10.1111/j.1365-2664.2007.01398.x
Correia MJ, Torres F, Pereira JS (1989) Water and nutrient supply regimes and the water relations of juvenile leaves of Eucalyptus globulus. Tree Physiol 5:459–471. https://doi.org/10.1093/treephys/5.4.459
Cremer KW (1977) Distance of seed dispersal in eucalypts estimated from seed weights. Aust Res 7:225–228
da Silva PHM, Poggiani F, Sebbenn AM, Mori ES (2011) Can Eucalyptus invade native forest fragments close to commercial stands? For Ecol Manag 261:2075–2080. https://doi.org/10.1016/j.foreco.2011.03.001
Daehler CC, Denslow JS, Ansari S, Kuo H (2004) A risk-assessment system for screening out invasive pest plants from Hawaii and other Pacific Islands. Conserv Biol 18:360–368. https://doi.org/10.1111/j.1523-1739.2004.00066.x
Davis MA, Pelsor M (2001) Experimental support for a resource-based mechanistic model of invasibility. Ecol Lett 4:421–428. https://doi.org/10.1046/j.1461-0248.2001.00246.x
Davis MA, Grime JP, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–534. https://doi.org/10.1046/j.1365-2745.2000.00473.x
Dawson W, Burslem DFRP, Hulme PE (2009) Factors explaining alien plant invasion success in a tropical ecosystem differ at each stage of invasion. J Ecol 97:657–665. https://doi.org/10.1111/j.1365-2745.2009.01519.x
Dodet M, Collet C (2012) When should exotic forest plantation tree species be considered as an invasive threat and how should we treat them? Biol Invasions 14:1765–1778. https://doi.org/10.1007/s10530-012-0202-4
Elton CS (1958) The ecology of invasions by animals and plants. Methuen, London
Essl F, Moser D, Dullinger S, Mang T, Hulme PE (2010) Selection for commercial forestry determines global patterns of alien conifer invasions. Divers Distrib 16:911–921. https://doi.org/10.1111/j.1472-4642.2010.00705.x
FAO (2010) Global forest resource assessment. Department, Food and Agriculture Organization of the United Nations, Rome
Fernandes P, Antunes C, Pinho P, Máguas C, Correia O (2016) Natural regeneration of Pinus pinaster and Eucalyptus globulus from plantation into adjacent natural habitats. For Ecol Manag 378:91–102. https://doi.org/10.1016/j.foreco.2016.07.027
Gassó N, Basnou C, Vilà M (2009) Predicting plant invaders in the Mediterranean through a weed risk assessment system. Biol Invasions 12:463–476. https://doi.org/10.1007/s10530-009-9451-2
Gassó N, Pino J, Font X, Vilà M (2012) Regional context affects native and alien plant species richness across habitat types. Appl Veg Sci 15:4–13. https://doi.org/10.1111/j.1654-109X.2011.01159.x
Gelman A, Hill J (2007) Data analysis using regression and multilevel/hierarchical models, 1st edn. Cambridge University Press, New York
Gibson MR, Richardson DM, Marchante E, Marchante H, Rodger JG, Stone GN, Byrne M, Fuentes-Ramírez A, George N, Harris C, Johnson SD, Le Roux JJ, Miller JT, Murphy DJ, Pauw A, Prescott MN, Wandrag EM, Wilson JRU (2011) Reproductive biology of Australian acacias: important mediator of invasiveness? Divers Distrib 17:911–933. https://doi.org/10.1111/j.1472-4642.2011.00808.x
Gómez-Aparicio L, Zamora R, Castro J, Hódar JA (2008) Facilitation of tree saplings by nurse plants: microhabitat amelioration or protection against herbivores? J Veg Sci 19:161–172. https://doi.org/10.3170/2008-8-18347
González-Moreno P, Pino J, Carreras D, Basnou C, Fernández-Rebollar I, Vilà M (2013a) Quantifying the landscape influence on plant invasions in Mediterranean coastal habitats. Landsc Ecol 28:891–903. https://doi.org/10.1007/s10980-013-9857-1
González-Moreno P, Pino J, Gassó N, Vilà M (2013b) Landscape context modulates alien plant invasion in Mediterranean forest edges. Biol Invasions 15:547–557. https://doi.org/10.1007/s10530-012-0306-x
González-Moreno P, Diez JM, Ibáñez I, Font X, Vilà M (2014) Plant invasions are context-dependent: multiscale effects of climate, human activity and habitat. Divers Distrib 20:720–731. https://doi.org/10.1111/ddi.12206
Gordon DR, Flory SL, Cooper AL, Morris SK (2012) Assessing the invasion risk of Eucalyptus in the United States using the Australian weed risk assessment. Int J For Res 2012:1–7. https://doi.org/10.1155/2012/203768
Green RH, Young RC (1993) Sampling to detect rare species. Ecol Appl 3:351–356
Grueber CE, Nakagawa S, Laws RJ, Jamieson IG (2011) Multimodel inference in ecology and evolution: challenges and solutions. J Evol Biol 24:699–711. https://doi.org/10.1111/j.1420-9101.2010.02210.x
Hardner CM, Potts BM (1995) Inbreeding depression and changes in variation after selfing in Eucalyptus globulus ssp. globulus. Silvae Genet 44:46–54
Higgins SI, Richardson DM (1998) Pine invasions in the southern hemisphere: modelling interactions between organism, environment and disturbance. Plant Ecol 135:79–93
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978. https://doi.org/10.1002/joc.1276
Hobbs NT, Hilborn R (2006) Alternatives to statistical hypothesis testing in ecology: a guide to self teaching. Ecol Appl 16(1):5–19
Ibáñez I, Silander JA Jr, Allen JM, Treanor SA, Wilson A (2009) Identifying hotspots for plant invasions and forecasting focal points of further spread. J Appl Ecol 46:1219–1228. https://doi.org/10.1111/j.1365-2664.2009.01736.x
ICNF (2013) IFN6—Áreas dos usos do solo e das espécies florestais de Portugal continental. Resultados preliminares, Instituto da Conservação, da Natureza e das Florestas
Jordan G, Potts BM, Wiltshire R (1999) Strong, independent quantitative genetic control of vegetative phase change and first flowering in Eucalyptus globulus ssp. globulus. Heredity (Edinb) 83:179–187
Krivánek M, Pysek P, Jarosík V (2006) Planting history and propagule pressure as predictors of invasion by woody species in a temperate region. Conserv Biol 20:1487–1498. https://doi.org/10.1111/j.1523-1739.2006.00477.x
Kutiel P, Lavee H (1999) Effect of slope aspect on soil and vegetation properties along an aridity transect. Israel J Plant Sci 47(3):169–178
Lake JC, Leishman M (2004) Invasion success of exotic plants in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biol Conserv 117:215–226. https://doi.org/10.1016/S0006-3207(03)00294-5
Larcombe MJ, Silva JS, Vaillancourt RE, Potts BM (2013) Assessing the invasive potential of Eucalyptus globulus in Australia: quantification of wildling establishment from plantations. Biol Invasions 15:2763–2781. https://doi.org/10.1007/s10530-013-0492-1
Larcombe MJ, Barbour RC, Vaillancourt RE, Potts BM (2014) Assessing the risk of exotic gene flow from Eucalyptus globulus plantations to native E. ovata forests. For Ecol Manag 312:193–202. https://doi.org/10.1016/j.foreco.2013.10.005
Lavi A, Perevolotsky A, Kigel J, Noy-Meir I (2005) Invasion of Pinus halepensis from plantations into adjacent natural habitats. Appl Veg Sci 8(1):85–92
Lloret F, Medail F, Brundu G, Camarda I, Moragues E, Rita J, Lambdon P, Hulme PE (2005) Species attributes and invasion success by alien plants on Mediterranean islands. J Ecol 93:512–520. https://doi.org/10.1111/j.1365-2745.2005.00979.x
Lockwood JL, Cassey P, Blackburn TM (2009) The more you introduce the more you get: the role of colonization pressure and propagule pressure in invasion ecology. Divers Distrib 15:904–910. https://doi.org/10.1111/j.1472-4642.2009.00594.x
Lonsdale WM (1999) Global patterns of plant invasions and the concept of invasibility. Ecology 80:1522–1536. https://doi.org/10.1890/0012-9658(1999)080[1522:GPOPIA]2.0.CO;2
Lorentz KA, Minogue PJ (2015) Potential Invasiveness for Eucalyptus species in Florida. Invasive Plant Sci Manag 8:90–97. https://doi.org/10.1614/IPSM-D-14-00030.1
Marchante H, Morais M, Freitas H, Marchante E (2014) Guia prático para a identificação de Plantas Invasoras em Portugal. Imprensa da Universidade de Coimbra, Coimbra
Milbau A, Stout JC, Graae BJ, Nijs I (2009) A hierarchical framework for integrating invasibility experiments incorporating different factors and spatial scales. Biol Invasions 11:941–950. https://doi.org/10.1007/s10530-008-9306-2
Mimura M, Barbour RC, Potts BM, Vaillancourt RE, Watanabe KN (2009) Comparison of contemporary mating patterns in continuous and fragmented Eucalyptus globulus native forests. Mol Ecol 18:4180–4192. https://doi.org/10.1111/j.1365-294X.2009.04350.x
Mitchell CE, Agrawal AA, Bever JD, Gilbert GS, Hufbauer RA, Klironomos JN, Maron JL, Morris WF, Parker IM, Power AG, Seabloom EW, Torchin ME, Vazquez DP (2006) Biotic interactions and plant invasions. Ecol Lett 9:726–740. https://doi.org/10.1111/j.1461-0248.2006.00908.x
Mood AM (1969) Macro-analysis of the American educational system. Oper Res 17:770–784. https://doi.org/10.1287/opre.17.5.770
Pearson RG, Dawson TP, Liu C (2004) Modelling species distributions in Britain: a hierarchical integration of climate and land-cover data. Ecography (Cop.) 27:285–298. https://doi.org/10.1111/j.0906-7590.2004.03740.x
Pheloung PC, Williams PA, Halloy SR (1999) A weed risk assessment model for use as a biosecurity tool evaluating plant introductions. J Environ Manag 57:239–251. https://doi.org/10.1006/jema.1999.0297
Potts BM, Vaillancourt RE, Jordan GJ, Dutkowski GW, Costa e Silva J, McKinnon GE, Steane DA, Volker PW, Lopez GA, Apiolaza LA, Li Y, Marques C, Borralho NMG (2004) Exploration of the Eucalyptus globulus gene pool. In: Borralho NMG, Pereira JS, Marques C, Coutinho J, Madeira M, Tomé M (eds) Eucalyptus in a changing world – IUFRO conference. RAIZ, Instituto Investigação de Floresta e Papel, Aveiro, pp 46–61
Procheş Ş, Wilson JRU, Richardson DM, Rejmánek M (2012) Native and naturalized range size in Pinus: relative importance of biogeography, introduction effort and species traits. Glob Ecol Biogeogr 21:513–523. https://doi.org/10.1111/j.1466-8238.2011.00703.x
Pyšek P, Křivánek M, Jarošík V (2009) Planting intensity, residence time, and species traits determine invasion success of alien woody species. Ecology 90:2734–2744
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
R Development Core Team (2009) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Available from: http://www.r-project.org/
Rejmánek M, Richardson DM (2011) Eucalypts. In: Simberloff D, Rejmánek M (eds) Encyclopedia of biological invasions. University of California Press, Berkeley, pp 203–209
Rejmánek M, Richardson DM (2013) Trees and shrubs as invasive alien species—2013 update of the global database. Divers Distrib 19:1093–1094. https://doi.org/10.1111/ddi.12075
Rejmánek M, Richardson DM, Pyšek P (2005) Plant invasions and invasibility of plant communities. In: Van der Maarel E (ed) Vegetation ecology. Blackwell, Oxford, pp 332–355
Reyes O, Casal M (2001) The influence of seed age on germinative responseto the effects of fire in Pinus pinaster, Pinus radiata and Eucalyptus globulus. Ann For Sci 58:439–447. https://doi.org/10.1051/forest:2001137
Richardson DM (1998) Forestry trees as invasive aliens. Conserv Biol 12:18–26. https://doi.org/10.1111/j.1523-1739.1998.96392.x
Richardson DM, Pyšek P (2006) Plant invasions: merging the concepts of species invasiveness and community invasibility. Prog Phys Geogr 30:409–431. https://doi.org/10.1191/0309133306pp490pr
Richardson DM, Rejmánek M (2011) Trees and shrubs as invasive alien species—a global review. Divers Distrib 17:788–809. https://doi.org/10.1111/j.1472-4642.2011.00782.x
Richardson DM, Williams PA, Hobbs RJ (1994) Pine invasions in the southern hemisphere: determinants of spread and invadability. J Biogeogr 21:511. https://doi.org/10.2307/2845655
Rouget M, Richardson DM (2003) Inferring process from pattern in plant invasions: a semimechanistic model incorporating propagule pressure and environmental factors. Am Nat 162:713–724. https://doi.org/10.1086/379204
Rundel PW, Dickie IA, Richardson DM (2014) Tree invasions into treeless areas: mechanisms and ecosystem processes. Biol Invasions 16:663–675. https://doi.org/10.1007/s10530-013-0614-9
Soares P, Tomé M, Pereira JS (2007) A produtividade do eucaliptal. In: Alves AM, Pereira JS, Silva JMN (eds), O Eucaliptal Em Portugal—Impactes Ambientais E Investigação Científica. Lisbon, pp 27–60
Stoneman GL (1994) Ecology and physiology of establishment of eucalypt seedlings from seed: a review. Aust For 57:11–29
Thuiller W, Richardson DM, Pysek P, Midgley GF, Hughes GO, Rouget M (2005) Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob Change Biol 11:2234–2250. https://doi.org/10.1111/j.1365-2486.2005.001018.x
Tomé M, Ribeiro F, Soares P (2001) Modelo Globulus 2.1. In: Relatórios Técnico- Científicos Do GIMREF. Departamento Engenharia Florestal, Instituto Superior de Agronomia, Lisbon, p. 96
Traveset A, Richardson DM (2014) Mutualistic interactions and biological invasions. Annu Rev Ecol Evol Syst 45:89–113. https://doi.org/10.1146/annurev-ecolsys-120213-091857
Van Der Meer PJ, Dignan P, Saveneh AG (1999) Effect of gap size on seedling establishment, growth and survival at three years in mountain ash (Eucalyptus regnans F. Muell.) forest in Victoria, Australia. For Ecol Manag 117:33–42. https://doi.org/10.1016/S0378-1127(98)00471-X
Visser V, Langdon B, Pauchard A, Richardson DM (2014) Unlocking the potential of Google Earth as a tool in invasion science. Biol Invasions 16:513–534. https://doi.org/10.1007/s10530-013-0604-y
Walther G-R, Roques A, Hulme PE, Sykes MT, Pysek P, Kühn I, Zobel M, Bacher S, Botta-Dukát Z, Bugmann H, Czúcz B, Dauber J, Hickler T, Jarosík V, Kenis M, Klotz S, Minchin D, Moora M, Nentwig W, Ott J, Panov VE, Reineking B, Robinet C, Semenchenko V, Solarz W, Thuiller W, Vilà M, Vohland K, Settele J (2009) Alien species in a warmer world: risks and opportunities. Trends Ecol Evol 24:686–693. https://doi.org/10.1016/j.tree.2009.06.008
Wevill T, Read J (2010) Fine-scale patterns in the distribution of semi-arid tree species at Wyperfeld National Park, southeastern Australia—the potential roles of resource gradients vs disturbance. J Arid Environ 74:482–490. https://doi.org/10.1016/j.jaridenv.2009.10.009
Wilson JRU, Dormontt EE, Prentis PJ, Lowe AJ, Richardson DM (2009) Something in the way you move: dispersal pathways affect invasion success. Trends Ecol Evol 24:136–144. https://doi.org/10.1016/j.tree.2008.10.007
Acknowledgements
This research was funded by Fundação para a Ciência e a Tecnologia (FCT) and the Navigator Company in the frame of Patrícia Fernandes PhD scholarship from FCT (SFRH/BDE/51709/2011) and short-term mission from COST Actions TD1209 (Alien Challenge) supported by the European Cooperation in Science and Technology. PGM was partially funded by CABI, the Severo Ochoa Program (SEV-2012-0262), the Spanish Ministerio de Ciencia e Innovación projects RIXFUTUR (CGL2009-7515) and IMPLANTIN (CGL 2015-65346-R). We would like to thank the Navigator Company for providing access to plantations, maps and data. Namely, we would like to thank: André Carvalho, Eduardo Mendes, Luís Alarico, João Bandeira, Américo Campos, Francisco José Inácio, Isidro Costa, João Pires, Joaquim Fonseca, Jorge Lourenço, José Carlos Pereira, Leonel Almeida and Ricardo Mendes. We also express our gratitude to Sara Coelho, Elsa Gonçalves, Andreia Anjos and Márcia Vieira for the help during field surveys and to Kate Pollard for the fruitful comments and final editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Figs. 4 and 5 and Tables 4, 5, 6, 7 and 8.
Deviance partitioning of E. globulus presence and density using generalized linear mixed models among climate (X1), propagule pressure (X2) and site characteristics (X3) predictors. Each group of predictor included only the two of the most important non-collinear variables explaining each dependent variables. Each circle corresponds to a group of variables. Numbers within circles are the proportion of deviance explained by each set of predictors alone (non-overlapped part of circles) or shared. Residuals indicate the deviance non-explained by the models
Rights and permissions
About this article
Cite this article
Fernandes, P., Máguas, C., Correia, O. et al. What drives Eucalyptus globulus natural establishment outside plantations? The relative importance of climate, plantation and site characteristics. Biol Invasions 20, 1129–1146 (2018). https://doi.org/10.1007/s10530-017-1614-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-017-1614-y