Abstract
Objectives
To improve the production of 2,3-butanediol (2,3-BD) in Klebsiella pneumoniae, the genes related to the formation of lactic acid, ethanol, and acetic acid were eliminated.
Results
Although the cell growth and 2,3-BD production rates of the K. pneumoniae ΔldhA ΔadhE Δpta-ackA strain were lower than those of the wild-type strain, the mutant produced a higher titer of 2,3-BD and a higher yield in batch fermentation: 91 g 2,3-BD/l with a yield of 0.45 g per g glucose and a productivity of 1.62 g/l.h in fed-batch fermentation. The metabolic characteristics of the mutants were consistent with the results of in silico simulation.
Conclusions
K. pneumoniae knockout mutants developed with an aid of in silico investigation could produce higher amounts of 2,3-BD with increased titer, yield, and productivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
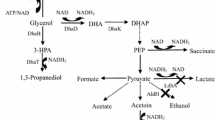
2,3-Butanediol (2,3-BD; C4H10O2) has a wide variety of applications in the bulk and specialty chemical industries. It has a high-heating value of 27,198 J/g, and therefore is considered a very good additive for liquid fuels. It can also be easily converted to methyl ethyl ketone (MEK), commonly used as an industrial solvent, and to 1,3-butadiene, primarily used as a monomer in the manufacture of synthetic rubbers (Winfield 1945; Emerson et al. 1982). Furthermore, 2,3-BD has potential applicability in food, cosmetics, personal care, and agricultural industries as moistening and softening agents (Celinska and Grajek 2009).
Many bacterial strains, including Klebsiella pneumoniae, K. oxytoca, Enterobacter aerognes, Lactococcus lactis, Paenibacillus polymyxa, and Bacillus amyloliquifaciens, produce 2,3-BD as a major end-product during the fermentation of carbohydrates (Celinska and Grajek 2009; Ji et al. 2011). The strains also form various by-products such as lactic acid, ethanol, acetic acid, succinic acid, and formic acid, which lower the yield of 2,3-BD and complicate its recovery process. Among them, K. pneumoniae has shown relatively high performance in the production of 2,3-BD with less formation of by-products (Ma et al. 2009; Cho et al. 2012). Since the mucoidic growth characteristics of K. pneumoniae are unsuitable for industrial applications, we isolated a unique K. pneumoniae strain that forms fewer mucoidic substances (Arasu et al. 2011; Rathnasingh et al. 2012).
In the present study, K. pneumoniae KCTC12133BP, which forms few lipopolysaccharides and mucous-like substances, was engineered to enhance the production of 2,3-BD (Rathnasingh et al. 2012). We primarily aimed to develop several knockout mutants targeted at reducing the formation of by-products with the help of in silico investigation. The performance of the mutants was evaluated by batch fermentation and then, fed-batch fermentation was performed to increase 2,3-BD production through optimization of the agitation speed.
Materials and methods
Bacterial strain and strain development
Klebsiella pneumoniae KCTC12133BP was used as a host for the development of various knockout mutant strains. In-frame deletions were carried out based on homologous recombination by using overlap polymerase chain reaction (PCR) products. The overlap product of the two fragments, which consisted of ~500 bp upstream and downstream of the target gene, was amplified and overlapped by PCR. The fragment was cloned in chloramphenicol-resistant pKGS plasmid (Kim et al. 2013) containing a sacB counter selection marker. The plasmid was transformed into competent cells of the K. pneumoniae strain by electroporation. The colonies were then selected in lysogeny broth (LB) plates at 42 °C with chloramphenicol (25 mg/l), and the integrated cassette was cured by sacB expression under sucrose pressure. Integration and excision were confirmed in all mutants by PCR screening using genome-specific primers.
Culture mediusm and fermentation conditions
The cells were cultivated in M9 medium containing (per liter): 1 g NaCl, 1 g NH4Cl, 0.25 g MgSO4, and 5 g yeast extract. The medium was fortified with 100 mM potassium phosphate buffer (pH 6.8). For the preparation of inocula, the suspended cells from single colonies on LB agar plates were precultured in 20 ml test tubes containing 5 ml LB medium and incubated at 37 °C for 5 h. Then, 0.2 ml of the preculture was transferred into an Erlenmeyer flask containing culture medium (10 g glucose/l) and cultivated to an OD600 of 1.5–2. The tube and flask cultivations were shaken at 150 rpm and 37 °C. 10 % (v/v) of the seed culture was transferred to a bioreactor. Batch fermentations were carried out in a 1.5 l bench top fermenter (FMT DS series Fermentec, Seoul, South Korea) with a 600 ml initial working volume (90 g glucose/l). Fed-batch fermentations were performed in a 6.6 l BioFlo and CelliGen 310 bioreactor (New Brunswick) containing 3 l medium. The pH was allowed to drop from 6.8 to 6.5 and then maintained at 6.5 ± 0.1 by the automatic feeding of 5 M NaOH or 5 M HCl. The temperature was set to 37 °C. The agitation speed was kept at 150 rpm in batch fermentation, whereas it was varied from 150 to 450 rpm in fed-batch fermentation. The bioreactor was continuously flushed with air thorough a 0.2 µm membrane filter at 1 vvm. Fed-batch fermentation was performed by intermittent addition of sterile glucose powder at 50 g/l. Samples were withdrawn periodically to determine the concentrations of cell mass, residual substrate, and metabolites. All bioreactor experiments were performed at least three times independently, and the representative results are shown in the figures.
Analytical methods
Cell growth was monitored from the OD600 value and the cell concentration, was then calculated by using a predetermined calibration curve (1 OD600 = 0.54 g dry wt/l). The concentration of d-glucose and metabolites, including 2,3-BD, lactic acid, ethanol, acetic acid, formic acid, acetoin, and succinic acid, were measured using HPLC equipped with a refractive index detector and Aminiex HPX-87H column (300 mm × 7.8 mm, Bio-Rad). The column, at 80 °C, was isocratically eluted using 0.02 M H2SO4 at 0.6 ml/min.
In silico investigation based on genome-scale metabolic model
The genome-scale metabolic model of K. pneumonia, iYL1228, was used to investigate the metabolic characteristics of genetically engineered strains in response to genetic perturbations (Liao et al. 2011). The model was simulated by using an optimization technique involving constraints-based flux analysis, including flux balance analysis (FBA) under the assumption of a pseudo-steady state. Mass balances in the stoichiometric model can be set up as S ij ·v j = 0, in which S ij is a stoichiometric coefficient of a metabolite i in the j th reaction and v j is the metabolic flux [mmol/g DCW/h] of the j th reaction. Then, an objective function, which is usually the maximization of cell growth rate, was maximized or minimized by linear programming, with the constraints of mass balances, experimental measurements, and thermodynamics. In order to improve the accuracy of the in silico investigation, the limits of the uptake and secretion rates for some metabolites, including organic acids, alcohols, and amino acids, were constrained by experimentally measured flux values. To make a 3D mesh plot graph a continuous surface for in silico flux solution spaces, the cell growth rate was maximized while gradually increasing the 2,3-BD production and the by-product formation rates, respectively, from their minimal to maximal flux values.
Results and discussion
Batch fermentation and in silico investigation of K. pneumoniae wild-type and ldhA knockout strains
Klebsiella pneumoniae wild type strain produced 20.9 g 2,3-BD/l along with various by-products, including lactic acid, ethanol, acetic acid, formic acid, succinic acid, and acetoin during batch fermentation (Fig. 1). The formation of the aforementioned by-products should be decreased, after which the redundant metabolic fluxes can be redirected for the synthesis of 2,3-BD. Since the wild-type K. pneumoniae strain formed lactic acid as the main by-product, the deletion of the ldhA gene, encoding lactate dehydrogenase, was first targeted. Before developing the ldhA gene knockout strain, the changes in flux solution spaces between the wild-type and ΔldhA strains were examined.
Batch fermentation results of a K. pneumoniae wild-type and b K. pneumoniae ΔldhA strains, and contour graph of three-dimensional mesh plot graph as a continuous surface for in silico predicted the flux solution spaces of c K. pneumoniae wild-type and d K. pneumoniae ΔldhA strains. Batch fermentations were performed with a working volume of 600 ml at 37 °C and 150 rpm. The aeration rate was 1 vvm and pH was controlled at 6.5 by the automatic feeding of 5 M NaOH or 5 M HCl. Symbols in fermentation profiles denote the concentration of glucose (filled diamonds), 2,3-BD (filled circles), cell mass (filled squares), lactic acid (open circles), ethanol (filled triangles), acetic acid (open triangles), succinic acid (asterisks), formic acid (open diamonds), and acetoin (open squares). The maximal point of in silico cell growth rate is generally considered to be a reasonable position for predicting the actual state of a cell, which is indicated by a small circle, because the top priority of an actual cell is viability and survival. The gradient gray colours in the contour graph indicate the in silico cell growth rate. By-products contain several organic acids (lactic acid, acetic acid, succinic acid, and formic acid) and ethanol
As shown in Fig. 1, deleting the ldhA gene caused the optimal point of the model to shift to a state with decreased formation of by-products but increased production of 2,3-BD. When the ldhA gene was deleted experimentally in K. pneumoniae, the mutant showed ~20 % improvement of the 2,3-BD titer and yield compared with the wild-type strain. On the other hand, the sum of the amount of by-products decreased from 57 % of the total amount of products to 44 % in the ΔldhA strains.
Lactic acid and 2,3-BD are produced from the common pyruvic acid pool; therefore, increasing the metabolic pool of pyruvic acid by deleting the ldhA gene could improve the production of 2,3-BD. In addition, cell growth and glucose consumption rates of the ΔldhA strain were maintained in comparison with the wild-type strain because the knockout of the ldhA gene did not interrupt the redox balance of NAD+/NADH. The NADH that accumulated through the inactivation of lactate dehydrogenase, forming lactic acid with the oxidization of NADH, seemed to be compensated with 2,3-BD dehydrogenase, producing 2,3-BD with the oxidization of NADH.
Production of 2,3-BD at high yield by knockout of ldhA and adhE genes
Klebsiella pneumoniae ΔldhA strain still formed significant amounts of by-products, especially ethanol and acetic acid. In order to prevent the formation of ethanol, the main by-product in the K. pneumoniae ΔldhA strain, the adhE gene, encoding alcohol dehydrogenase, was further deleted. Production in the K. pneumoniae ∆ldhA ∆adhE strain was improved to 0.39 g 2,3-BD per g glucose, which is more than 1.4- and 1.3-fold higher than those obtained from the wild-type and ΔldhA strains, respectively (Fig. 2; Table 1). However, cell growth was severely retarded by deleting the adhE gene which was strongly related to the reduced glucose uptake rate. Alcohol dehydrogenase encoded by the adhE gene has been recognized to synthesize 1 mol ethanol from 1 mol acetyl-CoA by oxidizing 2 mol NADH. By inactivating alcohol dehydrogenase, the pool of NADH is likely to increase. Then, the excess NADH might inhibit pyruvate dehydrogenase complex (PDC), playing a role in the conversion of pyruvic acid into acetyl-CoA (Hansen and Henning 1966). The glycolytic flux is also dependent on the redox balance of NAD+/NADH, which is important for glyceraldehyde-3-phosphate dehydrogenase (GAPDH), serving to breakdown glucose for cell growth and energy generation (Wolfe 2005). Also, the reduced cell growth caused by the deletion of the adhE gene can be supported by the fact that AdhE has a number of important roles in cellular metabolism (Echave et al. 2003).
Batch fermentation results for a K. pneumoniae ΔldhA ΔadhE and b K. pneumoniae ΔldhA ΔadhE Δpta-ackA strains, and contour graph of three-dimensional mesh plot graph as a continuous surface for in silico predicted flux solution spaces of c K. pneumoniae ΔldhA ΔadhE and d K. pneumoniae ΔldhA ΔadhE Δpta-ackA strains. Batch fermentations were performed with a working volume of 600 ml at 37 °C and 150 rpm. The aeration rate was maintained at 1 vvm and pH was controlled at 6.5 by the automatic feeding of 5 M NaOH or 5 M HCl. Symbols in fermentation profiles denote the concentration of glucose (filled diamonds), 2,3-BD (filled circles), cell mass (filled squares), lactic acid (open circles), ethanol (filled triangles), acetic acid (open triangles), succinic acid (asterisks), formic acid (open diamonds), and acetoin (open squares). The maximal point of in silico cell growth rate is generally considered to be a reasonable position for predicting the actual state of a cell, which is indicated by a small circle, because the top priority of an actual cell is viability and survival. The gradient gray colours in the contour graph indicate the in silico cell growth rate. By-products contain several organic acids (lactic acid, acetic acid, succinic acid, and formic acid) and ethanol
The pta-ackA genes in the K. pneumonia ∆ldhA ∆adhE mutant were additionally deleted to prevent the formation of acetic acid, the main by-product in the K. pneumoniae ∆ldhA ∆adhE strain. The K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain produced 38.5 g 2,3-BD/l with less than 2 g acetic acid/l in batch fermentation. The yield of 2,3-BD on glucose was 0.44, 88 % of theoretical yield (0.5 g 2,3-BD per g glucose), which is 1.6- and 1.4-fold higher than those obtained from the wild-type and ΔldhA strains, respectively (Fig. 2; Table 1). This resulted in a very high selectivity of 2,3-BD, comprising >70 % of the total amount of excreted metabolites. Interestingly, the cell growth of the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain was faster than that of the K. pneumoniae ∆ldhA ∆adhE strain. This could be due to the earlier and increased 2,3-BD production in the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain compared to the K. pneumoniae ∆ldhA ∆adhE strain because the impaired NADH/NAD+ ratio could be adjusted to some degree by the enhanced 2,3-BD pathway.
With the deletion of the aforementioned four genes, the metabolic characteristics of the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain were also investigated through changes in the in silico flux solution space (Fig. 2). Although the knockout of adhE and pta-ackA genes significantly increased the pool of pyruvic acid (an precursor for 2,3-BD synthesis), which was then redirected into 2,3-BD synthesis, the overall 2,3-BD productivity (0.53 g/l.h) of the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain was relatively lower than those of the wild-type (1.44 g/l.h) and ∆ldhA (1.51 g/l.h) strains (Table 1). This was mainly a result of the decreased cell growth rate, which was <2 mmol/g DCW.h. In order to improve the 2,3-BD productivity of the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain, we increased cell growth by optimizing the agitation speed in fed-batch fermentation.
Improvement of 2,3-BD productivity by changing agitation speed in fed-batch fermentation
The effects of agitation speed ranging from 150 to 450 rpm on the cell growth and 2,3-BD production were examined in fed-batch fermentation with aeration at 1 vvm. As shown in Fig. 3, the cell growth and 2,3-BD productivity increased considerably as the agitation speed was increased up to 350 rpm. 2,3-BD productivity at 250 and 350 rpm were 2- and 3- fold higher than that obtained at 150 rpm, respectively. K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain produced 91 g 2,3-BD/l with a productivity of 1.62 g/l.h with a yield of 0.45 g 2,3-BD/g glucose at 350 rpm (Table 1). The concentrations of ethanol and acetic acid did not change regardless of the agitation speed. However, the increase of the agitation speed up to 350 rpm generated more acetic acid (10 g/l) and succinic acid (15.8 g/l). It is well known that pyruvate oxidase (PoxB), which converts pyruvic acid directly to acetic acid and CO2, makes a significant contribution to cell growth under aerobic conditions. Thus, the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain might form more acetic acid via the PoxB pathway with increased agitation speed (Abdel-Hamid et al. 2001). The accumulation of succinic acid can be explained by the fact that succinic acid is an intermediate of the tricarboxylic acid (TCA) cycle, and its activity is dramatically stimulated when sufficient O2 is supplemented for cell growth (Song and Lee 2006).
Fed-batch fermentations of K. pneumoniae ΔldhA ΔadhE Δpta-ackA strain at different agitation speeds. Fermentation profiles of a 150 rpm, b 250 rpm, c 350 rpm, and d 450 rpm. Fed-batch fermentations were performed with a working volume of 3 l at 37 °C and the aeration rate was at 1 vvm. The pH was controlled at 6.5 by the automatic feeding of 5 M NaOH or 5 M HCl. Symbols in fermentation profiles denote the concentration of glucose (filled diamonds), 2,3-BD (filled circles), cell mass (filled squares), lactic acid (open circles), ethanol (filled triangles), acetic acid (open triangles), succinic acid (asterisks), formic acid (open diamonds), and acetoin (open squares)
A further increase of the agitation speed up to 450 rpm increased the cell growth rate and final cell density. The glucose uptake and cell growth rates tend to be improved at faster agitation speeds by activating aerobic and respiratory pathways, including the TCA cycle for energy generation and the amino acids and fatty acids for cell metabolism. However, more CO2 is released through the aerobic and respiratory pathways, causing decrease in the production of 2,3-BD. As expected, the titer of acetic acid rose to 21.2 g/l at 450 rpm. A significant amount of acetoin (12.9 g acetoin/l) was observed at 450 rpm as well. Acetoin is mainly due to an excess of O2 (Hugenholtz and Kleerebezem 1999; Oliver et al. 2013). Furthermore, α-acetolactate synthase, the first enzyme in the 2,3-BD synthesis pathway, is rapidly inactivated under a high O2 supply (Kosaric et al. 1992). These diminished the titer of 2,3-BD from 91 g/l (350 rpm) to 83.4 g/l (450 rpm) (Fig. 3).
In summary We have developed 2,3-BD over-producing K. pneumonia mutants by systematically disrupting the ldhA, adhE, and pta-ackA genes. The mutants diminished the formation of by-products, including lactic acid, ethanol, and acetic acid, and their metabolic characteristics were deciphered by in silico investigation. The fed-batch fermentation of the K. pneumoniae ∆ldhA ∆adhE ∆pta-ackA strain with the optimization of agitation speed achieved production of 91 g 2,3-BD/l with a yield of 0.45 g per g glucose and a 2,3-BD productivity of 1.62 g/l.h, which should be useful for industrial production of 2,3-BD.
References
Abdel-Hamid AM, Attwood MM, Guest JR (2001) Pyruvate oxidase contributes to the aerobic growth efficiency of Escherichia coli. Microbiology 147:1483–1498
Arasu MV, Kumar V, Ashok S, Hyohak S, Rathnasingh C, Lee HJ, Seung D, Park S (2011) Isolation and characterization of the new Klebsiella pneumoniae J2B strain showing improved growth characteristics with reduced lipopolysaccharide formation. Biotech Bioproc Eng 16:1134–1143
Celinska E, Grajek W (2009) Biotechnological production of 2,3-butanediol: current state and prospects. Biotechnol Adv 27:715–725
Cho JH, Rathnasingh C, Song H, Chung BW, Lee HJ, Seung D (2012) Fermentation and evaluation of Klebsiella pneumoniae and K. oxyotca on the production of 2,3-butanediol. Bioprocess Biosyst Eng 35:1081–1088
Echave P, Tamarit J, Cabiscol E, Ros J (2003) Novel antioxidant role of alcohol dehydrogenase E from Escherichia coli. J Biol Chem 32:30193–30198
Emerson RR, Flickinger MC, Tsao GT (1982) Kinetics of dehydration of aqueous 2,3-butanediol to methyl ethyl ketone. Ind Eng Chem Prod Res Dev 21:473–477
Hansen HG, Henning U (1966) Regulation of pyruvate dehydrogenase activity in Escherichia coli K12. Biochim Biophys Acta 122:355–358
Hugenholtz J, Kleerebezem M (1999) Metabolic engineering of lactic acid bacteria: overview of the approaches and results of pathway rerouting involved in food fermentations. Curr Opin Biotech 10:492–497
Ji XJ, Huang H, Du J, Zhu JG, Ren LJ, Li S, Nie ZK (2009) Development of an industrial medium for economical 2,3-butanediol production through co-fermentation of glucose and xylose by Klebsiella oxytoca. Bioresour Technol 100:5214–5218
Ji XJ, Huang H, Ouyang PK (2011) Microbial 2,3-butanediol production: a state-of-the-art review. Biotechnol Adv 29:351–364
Kim DK, Rathnasingh C, Song H, Lee HJ, Seung D, Chang YK (2013) Metabolic engineering of a novel Klebsiella oxytoca strain for enhanced 2,3-butaneeiol production. J Biosci Bioeng 116:186–192
Kosaric N, Magee RJ, Blaszczyk R (1992) Redox potential measurement for monitoring glucose and xylose conversion by K. pneumoniae. Chem Biochem Eng Q 6:145–152
Liao YC, Huang TW, Chen FC, Charusanti P, Hong JS, Chang HY, Tsai SF, Palsson BO, Hsiung CA (2011) An experimentally validated genome-scale metabolic reconstruction of Klebsiella pneumoniae MGH 78578, iYL1228. J Bacteriol 193:1710–1717
Ma C, Wang A, Qin J, Li L, Ai X, Jiang T, Tang H, Xu P (2009) Enhanced 2,3-butanediol production by Klebsiella pneumoniae SDM. Appl Microbiol Biotechnol 82:49–57
Oliver JW, Machado IM, Yoneda H, Atsumi S (2013) Cyanobacterial conversion of carbon dioxide to 2,3-butanediol. Proc Natl Acad Sci USA 110:1249–1254
Rathnasingh C, Kim DK, Song H, Park S, Lee HJ, Seung D (2012) Isolation and characterization of a new mucoid-free Klebsiella pneumoniae strain for 2,3-butanediol production. Afr J Biotechnol 11:11252–11261
Song H, Lee SY (2006) Production of succinic acid by bacterial fermentation. Enzyme Microb Tech 39:352–361
Winfield ME (1945) The catalytic dehydration of 2,3-butanediol to 1,3-butadiene. J Council Sci Ind Res 18:412–423
Wolfe AJ (2005) The acetate switch. Microbiol Mol Biol Rev 69:12–50
Acknowledgments
This work was supported by the Industrial Strategic Technology Development Program (No. 10050407) funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea) and the Advanced Biomass R&D Center of Global Frontier Project (No. ABC-2010-0029728) funded by the Ministry of Science, ICT and Future Planning (MSIP, Korea).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rathnasingh, C., Park, J.M., Kim, Dk. et al. Metabolic engineering of Klebsiella pneumoniae and in silico investigation for enhanced 2,3-butanediol production. Biotechnol Lett 38, 975–982 (2016). https://doi.org/10.1007/s10529-016-2062-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2062-y