Abstract
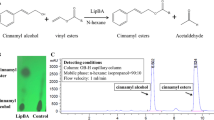
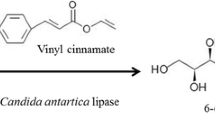
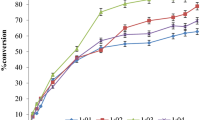
Using Novozym 435 as catalyst, the syntheses of ethyl ferulate (EF) from ferulic acid (4-hydroxy 3-methoxy cinnamic acid) and ethanol, and octyl methoxycinnamate (OMC) from p-methoxycinnamic acid and 2-ethyl hexanol were successfully carried out in this study. A conversion of 87% was obtained within 2 days at 75 °C for the synthesis of EF. For the synthesis of OMC at 80 °C, 90% conversion can be obtained within 1 day. The use of solvent and high reaction temperature resulted in better conversion for the synthesis of cinnamic acid derivatives. Some cinnamic acid esters could also be obtained with higher conversion and shorter reaction times in comparison to other methods reported in the literature. The enzyme can be reused several times before significant activity loss was observed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B Guyot B Bosquette M Pina J Graille (1997) ArticleTitleEsterification of phenolic acids from green coffee with an immobilized lipase from Candida antarctica in solvent-free medium Biotechnol. Lett. 19 529–532 Occurrence Handle10.1023/A:1018381102466 Occurrence Handle1:CAS:528:DyaK2sXktF2msr4%3D
NS Hatzakis D Daphnomili I Smonou (2003) ArticleTitleFerulic acid esterase from Humicola insolens catalyzes enantioselective transesterification of secondary alcohols J. Mol. Catal. B: Enzymatic 21 309–311 Occurrence Handle10.1016/S1381-1177(02)00228-X Occurrence Handle1:CAS:528:DC%2BD3sXivVGqsQ%3D%3D
H Kikuzaki M Hisamoto K Hirose K Akiyama H Taniguchi (2002) ArticleTitleAntioxidant properties of ferulic acid and its related compounds Agric. Food Chem. 50 2161–2168 Occurrence Handle10.1021/jf011348w Occurrence Handle1:CAS:528:DC%2BD38XhsFKjt74%3D
BM Lue S Karboune FK Yeboah S Kermasha (2005) ArticleTitleLipase catalyzed esterification of cinnamic acid and oleyl alcohol in organic solvent media J. Chem. Technol. Biot. 80 462–468 Occurrence Handle1:CAS:528:DC%2BD2MXjtVeqtbc%3D
K Priya A Chadha (2003) ArticleTitleSynthesis of hydrocinnamic esters by Pseudomonas cepacia lipase Enzyme Microb. Technol. 32 485–490 Occurrence Handle10.1016/S0141-0229(02)00340-X Occurrence Handle1:CAS:528:DC%2BD3sXmsFWguw%3D%3D
T Pyysalo H Torkkeli E Honkanen (1997) ArticleTitleThe thermal decarboxyboxylation of some substituted cinnamic acids Lebens-Wissensch. Technol. 10 438–441
P Schuler BJF Hudson (1990) Food Antioxidants Elsevier London 113–127
MM Soumanou UT Bornscheuer (2003) ArticleTitleImprovement in lipase-catalyzed synthesis of fatty acid methyl esters from sunflower oil Enz. Microbial Technol. 33 97–103 Occurrence Handle10.1016/S0141-0229(03)00090-5 Occurrence Handle1:CAS:528:DC%2BD3sXkvVKju7g%3D
H Stamatis V Sereti FN Kolisis (1999) ArticleTitleStudies on the enzymatic synthesis of lipophilic derivatives of natural antioxidants J. Am. Oil Chem. Soc. 76 1505–1510 Occurrence Handle1:CAS:528:DC%2BD3cXks1egtw%3D%3D
SN Timasheff (1993) ArticleTitleThe control of protein stability and associations by weak interactions with water: how do solvents affect these processes? Annu. Rev. Biophys. Biomol. Struct. 22 67–97 Occurrence Handle10.1146/annurev.bb.22.060193.000435 Occurrence Handle1:CAS:528:DyaK3sXlsFCjurk%3D Occurrence Handle8347999
E Topakas H Stamatis P Biely D Kekos BJ Macris P Christakopoulos (2003) ArticleTitlePurification and characterization of a feruloyl esterase from Fusarium oxysporum catalyzing esterification of phenolic acids in ternary water-organic solvent mixtures J. Biotechnol. 102 33–44 Occurrence Handle10.1016/S0168-1656(02)00363-2 Occurrence Handle1:CAS:528:DC%2BD3sXisFSlsr8%3D Occurrence Handle12668312
DB Volkin A Staubli R Langer AM Klibanov (1991) ArticleTitleEnzyme thermoinactivation in anhydrous organic solvent Biotechnol. Bioeng. 37 843–853 Occurrence Handle10.1002/bit.260370908 Occurrence Handle1:CAS:528:DyaK3MXhvVWit7c%3D
A Zaks AM Klibanov (1988) ArticleTitleThe effect of water on enzyme action in organic media J. Biol. Chem. 263 8017–8021 Occurrence Handle1:CAS:528:DyaL1cXksFSktrg%3D Occurrence Handle3131337
Author information
Authors and Affiliations
Corresponding author
Additional information
Revisions requested 10 January 2006; Revisions received 17 January 2006
Rights and permissions
About this article
Cite this article
Lee, GS., Widjaja, A. & Ju, YH. Enzymatic Synthesis of Cinnamic Acid Derivatives. Biotechnol Lett 28, 581–585 (2006). https://doi.org/10.1007/s10529-006-0019-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10529-006-0019-2