Abstract
Megaplatypus mutatus (Chapuis) (=Platypus mutatus), an ambrosia beetle native to South America, attacks standing live trees of a wide range of forest and fruit tree species, and it is particularly damaging to commercial poplar plantations. In 2000, M. mutatus was observed for the first time in Italy, in the province of Caserta, near Naples. The development of a pheromone-based monitoring system, for detecting the spread and for managing M. mutatus infestations, is an important goal for both European and South American control and surveillance programs. Using a three component pheromone blend developed in Argentina into commercial funnel traps we were able to asses the level of dispersion of this pest in the Italian Campania region. Insects were captured in all the plantations suspected of being infested based on the presence of active parental and larval galleries. We also provide the first report of the attack followed by completion of the life cycle of M. mutatus in European hazelnut, Corylus avellana L. (Betulaceae), an important nut species native to Europe and Western Asia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ambrosia beetles are an important group of forest insects that typically attack weakened or felled trees. Their name derives from the symbiotic fungus (ambrosia) they inoculate when they penetrate the xylem of their host. Megaplatypus mutatus (=Platypus mutatus) (Chapuis) (Platypodinae, Curculionidae) is an ambrosia beetle native to South America (Wood 1993) that only attacks standing, healthy living trees, digging deeply into the xylem and making large tunnels that are later colonized by the fungus they transport, Raffaelea santoroi (Guerrero) (Bascialli et al. 1996). These galleries subsequently weaken the tree’s stem, causing it to break under extreme stress and representing a serious problem in poplar, Populus deltoides Marshall (Salicaceae), commercial plantations (Alfaro et al. 2007; Achinelli et al. 2005). Furthermore, the dark staining of the tunnels caused by the decaying ambrosia mycelium reduces the quality of wood for export.
The attack is initiated when the male penetrates the bark and digs a tunnel a few centimetres deep. Then it builds a conical structure surrounding the entry hole using boring dust particles. From there it protrudes its abdomen and releases volatiles to attract the females (Santoro 1963; Gonzalez-Audino et al. 2005). A similar behaviour was reported for Platypus apicalis White and P. gracilis Broun in New Zealand (Milligan and Ytsma 1988). In the case of poplars, the black stain caused by the fungus decreases the commercial value of the timber. In the case of fruit trees, the wounds caused by this insect cause loss of water and nutrients, which can affect product yield and quality, and also increase the risk of attacks by microorganisms.
Megaplatypus mutatus was accidentally introduced to Italy in 1998 (EPPO/OEPP 2004, 2007). In 2000, it was detected in Populus canadensis (Mönch) in the Caserta province, in the Campania region. Attacks were also reported in Juglans regia (L.) and European hazelnut, Corylus avellana L. (Tremblay et al. 2000; Allegro and Della Beffa 2001); and later in Malus spp., Pyrus spp., Castanea spp., Prunus spp., Quercus spp. and Eucalyptus spp. (Carella and Spigno 2002). These reports do not indicate if the attacks had produced offspring or had been ineffective in these hosts.
The risk of dispersion and potential damage of M. mutatus to other regions of Europe (Alfaro et al. 2007) is of great concern to European regulatory authorities, who added it to the EPPO/OEPP Alert List in 2004. It was recommended as a quarantine pest in 2007 (Allegro and Griffo 2008; EPPO/OEPP 2004, 2007).
The population dynamics of M. mutatus suggests a bivoltine emergence in Argentina (Gatti et al. 2008b) and this pattern is also observed in Italy (H Funes et al. 2011). Although external temperature affects the duration of the various life stages (Santoro 1963), in central Argentina and southern Italy M. mutatus starts to fly in search of new host by the beginning of spring and by the end of summer. This species exhibits protandry (earlier emergence of males than females).
The development of pheromone-baited traps for surveillance and management programs of M. mutatus in infested poplar plantations is an important goal for both European and South American control efforts. Pheromone research of M. mutatus began in 2002 in Argentina with field experiments testing the attractiveness of pheromones of North American ambrosia beetles Gnathotricus sulcatus ((Scolytinae, (±)-Sulcatol and (−)-sulcatol) and Gnathotricus retusus ((+)-sulcatol) (Borden and McLean 1979) to M. mutatus populations (Alfaro et al. 2007). In 2003 these were evaluated by the Canadian forest service (Pacific Forestry Centre) in collaboration with the Istituto di Sperimentazione per la Pioppicoltura (Casale Monferrato, Italy) in the Caserta region of Italy (Alfaro et al. 2007). The field tests in Argentina and Italy indicated a low level, but consistent attraction of M. mutatus females to (±)-sulcatol.
Chemical analysis, electro-antennogram and olfactometer studies performed at the Centro de Investigaciones de Plagas e Insecticidas (CIPEIN) in Buenos Aires, Argentina, showed that male M. mutatus emit a sex pheromone composed mainly by (+)-sulcatol, its related ketone, sulcatone (Gonzalez Audino et al. 2005) and 3-pentanol (Gatti Liguori et al. 2008a). The CIPEIN recently showed the effectiveness of a two-component pheromone blend monitoring M. mutatus infestations in Argentina (Funes et al. 2009).
Although the presence of the galleries is an indication of the probable presence of M. Mutatus, it’s not easy to tell if a gallery is active or just an old empty gallery. Also, in some plant species, the cone of frass does not form on the bark so this sign of infestation cannot be evaluated and last, many galleries that do contain active larvae fail to produce emerging adults as their productivity is highly variable (ranging from 10 to 300 insects approximately (Santoro 1962). In this sense, pheromone traps are more reliable, more efficient, especially cost-efficient, mean of species detection.
The aim of this study was to conduct an operational test of a three-component M. mutatus pheromone blend, and survey the extension of the presence of M. mutatus in the province of Caserta, in the Italian Campania region.
Materials and methods
Selection of experimental fields
An initial visual survey of twenty-six plantations in the in the province of Caserta, searching for signs of infestation (Table 1) was performed in the first week of May 2007 with assistance of inspectors from the Phytosanitary service of region Campania, Italy. These consisted of 13 poplar plantations (P. canadensis (Mönch)), nine fruit tree plantations (three plantations of C. avellana L., three of J. regia L., two of Prunus avium L., one of Prunus persicum L.), and one Eucalyptus plantation. The level of infestation was characterized according to the presence of M. mutatus galleries. In order to monitor emergence of the first adults, emergence traps were installed on entry holes of active galleries, as described by Gatti Liguori et al. (2007). Based on this survey we selected for monitoring nine plantations in four areas of the Caserta province in which visible signs of active infestation were evident (Table 2).
Pheromone traps
The traps were Mastrap A Version ® (Isagro S.R.L, Milan, Italy), with an effective surface of 630 cm2, baited with experimental slow release devices filled with (+)-sulcatol, sulcatone and 3-pentanol. The release devices were polyethylene bags with different effective surfaces in order to obtain different release rates. Optimal surfaces were evaluated in field and behavioural laboratory assays (Funes et al. 2009). The release rates of the different baits were quantified at 28 °C using a wind tunnel with a linear air velocity of 0.5 m/s. The selected surfaces released 10 ± 1, 7 ± 1 and 40 ± 2 mg per day for (+)-sulcatol, sulcatone and 3-pentanol respectively.
Traps were hung from the trees with ropes at 1.5–1.8 m above ground level. Baits were replaced before complete pheromone consumption every 15 days. The number of insects caught was surveyed twice a week between 22 May and 13 July, and once a week afterwards until the end of each trial.
Sexing: males have dark brown abdomen and females clear brown. The elytra of the females are less striated and have careens less salient than in males. Besides, the apical region of females has a gentle slope and at the apex the elytra are rounded.
Pheromones
Sulcatone (6-methyl-5-hepten-2-one) and 3-pentanol were analytical grade (Aldrich Co., Saint Louis, MO, USA); (+)-sulcatol ((+)-6-methyl-5-hepten-2-ol) 99 % was purchased from Pherotech, Vancouver, Canada.
Results
Degree of infestation in Caserta
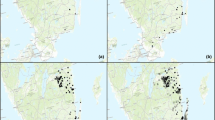
We found signs of probable M. mutatus activity in ten of the 23 (43 %) plantations surveyed. In particular, seven of the 16 poplar plantations (53 %) were suspicious. All three plantations of C. avellana showed signs. We did not find possible infestations in Prunus, Juglans or in the single Eucalyptus plantation surveyed (Table 1). This work presents the first report of attack and successful development (full ontogeny) of the whole life cycle of M. mutatus in C. avellana (Fig. 1).
Insect catch with pheromone traps
Adult M. mutatus were caught in all plantations in which traps were established confirming the presence of flying adults in the survey area (Table 3). In all, we caught 702 M. mutatus over the season, of which, 92 % were females. There were no insects caught in any of the control unbaited traps.
Temporal pattern of catches
Figure 2 shows the time pattern of insects caught in pheromone traps in poplar and hazelnut plantations of Campania region. The trapping season extends during the whole spring and summer with higher catches during summer. Catch level varied by location possibly reflecting population size, although this was not measured.
Discussion
The emergence of M. mutatus from the parental gallery is followed by a dispersive flight searching for a host, in the case of males, or for a mating partner in the case of females.
Our trapping caught a large number of females (92 % of the catches) confirming that the chemical blend used behaves as a true sex pheromone and not as population aggregation pheromone. The role of the sex pheromone had been previously demonstrated in the behavioural bioassays (Gonzalez Audino et al. 2005). M. mutatus does not mass-attack the trees (Santoro 1963) and it is therefore unlikely to have developed aggregation semiochemicals.
The small number of male catches (8 %) could be due to the phenomenon described as local enhancement (Krebs et al. 1972). In this case, a recipient receives a message not intended for him and takes advantage of the information acquired. In this case, a male that quickly needs to find a host, interprets the sexual pheromone emitted by another conspecific as an indicator of the presence of a suitable host and directs its flight towards the sexual pheromone source. The dispersal and host-finding phase seems to be the most dangerous period in the life cycle of bark and ambrosia beetles (Atkins 1966; Byers 1995; Byers et al. 1998; Dahlsten 1982; Huber et al. 1999). For this reason, Zhang and Schlyter (2004) and Byers et al. (2000) suggested that natural selection would favour beetles that have evolved multiple mechanisms for quickly finding their host, and avoiding unsuitable hosts and non-host species. Local enhancement seems to be useful in finding the host and not to parasitize tunnels made by other males. However, testing these hypotheses is beyond the scope of this paper.
It is likely that the natural dispersion of M. mutatus towards other areas of the Italic peninsula, and eventually to other parts of Europe, is being delayed by the geography of the infested area, which is surrounded by highlands and mountains where suitable hosts are absent. At the time this study was performed, the southern detection limit was the town of Capua, and a natural dispersion of M. mutatus outside Caserta towards the more southern provinces of Napoli or Benevento, is likely. Further knowledge on the behaviour of M. mutatus in Italy and on the timber pathways in the region is needed to forecast potential movement to other areas.
Attacks on C. avellana indicate the potential of M. mutatus to switch hosts to non-related indigenous trees. This could have deep implications in the dispersion and population dynamics of M. mutatus in Italy and Europe, increasing the threat of potential economic damage, as this pest not only affects poplars destined for high quality timber, but also the yield of other products, such as hazelnuts, one of the most important crops in Italy, and other fruits species in the Mediterranean region. In South America, C. avellana has still not been identified as a host of M. mutatus although there are numerous commercial plantations of this crop.
This work has demonstrated the efficacy of the three-blend pheromone developed in Argentina against M. mutatus introduced to Europe, providing a tool to monitor future dispersal into other parts of the European continent. Our monitoring results provide a useful guide for the timing of chemical or mechanical control in Caserta.
References
Achinelli FG, Liljersthröm G, Aparicio A, Delgado M, Jouanny M, Mastrandrea C (2005) Daños por taladrillo (Megaplatypus mutatus (=Platypus sulcatus)) en plantaciones de álamo (P. deltoides spp.) de Alberti, Buenos Aires: análisis preliminar de la magnitud y distribución de fustes quebrados. Rev Asoc For Arg 59(1):8–11 (in Spanish)
Alfaro R, Humble LM, Gonzalez P, Villaverde R, Allegro G (2007) The threat of the ambrosia beetle Megaplatypus mutatus (Chapuis) [= Platypus mutatus Chapuis] to world poplar resources. Forestry 80:471–479
Allegro G, Della Beffa G (2001) Un nuovo problema entomologico per la pioppicoltura Italiana: platypus mutatus Chapuis (Coleoptera, Platypodidae). Sherwood Foreste ed alberi oggi 66:31–34
Allegro G, Griffo R (2008) I rischi di diffusione di Megaplatypus mutatus. L’Informatore Agrario 13:73–76
Atkins MD (1966) Behavioral variation among scolytids in relation to their habitat. Can Entomol 98:285–288
Bascialli ME, Gimenez RA, Etiennot AE, Toscani H (1996) Manejo de la población de Platypus sulcatus Chapuis, durante tres años en la región del Delta del río Paraná mediante control químico. Investigaciones Agrícolas Sistemas de Recursos Forestales 5:129–140 (in Spanish)
Borden JH, McLean JA (1979) Secondary attraction in Gnathotricus retusus and cross attraction of G. sulcatus (Coleoptera: scolytidae). J Chem Ecol 5:79–88
Byers JA (1995) Host tree chemistry affecting colonization in bark beetles. In: Cardé RT, Bell WJ (eds) Chemical Ecology of Insects 2. Chapman & Hall, New York, pp 154–213
Byers JA, Zhang Q-H, Schlyter F, Birgersson G (1998) Volatiles form nonhost birch trees inhibit pheromone response in spruce bark beetles. Naturwissenschaften 85:557–561
Byers JA, Zhang Q-H, Birgersson G (2000) Strategies of a bark beetle, Pytogenes bidentatus, in an olfactory landscape. Naturwissenschaften 87:503–507
Carella D, Spigno P (2002) Lo xilofago Platypus mutatus (Coleoptera: platypodidae) dal pioppo passa ai fruttiferi. Bollettino del Laboratorio di Entomologia Agraria Filippo Silvestri 58:139–141
Dahlsten DL (1982) Relationship between bark beetles and their natural enemies. In: Mitton JB, Sturgeon KB (eds) A system for the study of evolutionary biology. University of Texas Press, Austin, pp 140–182
EPPO/OEPP Pest Risk Analysis Reporting Service (2004) First report of Platypus mutatus in Italy: addition to the EPPO Alert List No. 04 2004/061
European and Mediterranean Plant Protection Organisation (2007) Report of the 39th meeting of the panel on phytosanitary measures (Paris, 2007-03-06/09) 07-13694 http://archives.eppo.org/EPPOStandards/PM1_GENERAL/pm1-02(16)_A1A2_2007.pdf. Accessed Sep 2007
Funes H, Zerba E, González-Audino P (2009) Comparison of three types of traps baited with sexual pheromones for ambrosia beetle M. mutatus in poplar plantations. J Econ Entomol 102:1546–1550
Funes H, Griffo R, Zerba E, Gonzalez-Audino P (2011) Mating disruption of the ambrosia beetle Megaplatypus mutatus in poplar and hazelnut plantations using reservoir systems for pheromones. Entomol Exp Appl 139(3):226–234
Gatti Liguori P, Zerba E, Gonzalez-Audino P (2007) New trap for emergent Megaplatypus mutatus. Can Entomol 139:894–896
Gatti Liguori P, Zerba E, Alzogaray R, Gonzalez-Audino P (2008) 3-Pentanol: a new attractant present in volatile emissions from the Ambrosia Beetle, Megaplatypus mutatus. J Chem Ecol 34:1446–1451
Gatti P, Funes H, Zerba E, González-Audino P (2008b) Bivoltinismo, la amenaza duplicada de Megaplatypus mutatus. VII Congreso Argentino de Entomología
Gonzalez Audino P, Villaverde R, Alfaro R, Zerba E (2005) Identification of volatile emissions from Platypus mutatus (=sulcatus) (Coleoptera: platypodidae) and their behavioral activity. J Econ Entomol 98:1506–1509
Huber DP, Gries R, Borden JH, Pierce HD Jr (1999) Two pheromones of coniferophagous bark beetles found in the bark nonhost angiosperms. J Chem Ecol 25:805–816
Krebs JR, MacRoberts MH, Cullen JM (1972) Flocking and feeding in the great tit Parus major—An experimental study. Ibis 114:507–530
Milligan RH, Ytsma G (1988) Pheromone dissemination by male Platypus apicalis White and P. gracilis Broun (Col. Platypodidae). J App Entomol 106:113–118
Santoro FH (1962) La copula en Platypus sulcatus Chapuis (Coleoptera, Platypodidae). Rev Invest For 3:25–27 (in Spanish)
Santoro FH (1963) Bioecologia de Platypus sulcatus Chapuis (Coleoptera - Platypodidae). Rev Invest For 4:47–78 (in Spanish)
Tremblay E, Espinosa B, Mancini D, Caprio G (2000) Un coleottero proveniente dal Sudamerica minaccia i pioppi. Informatore Agrario 56:89–90
Wood SL (1993) Revision of the genera of Platypodidae (Coleoptera). Great Basin Nat 53:259–281
Zhang Q-H, Schlyter F (2004) Olfactory recognition and behavioural avoidance of angiosperm nonhost volatiles by conifer-inhabiting bark beetles. Agric For Entomol 6:1–19
Acknowledgments
This study received financial support by the Secretary of Science and Technology (SECYT) of Argentina and the Italian Ministry of External Affairs (MAE) through an International Cooperation Program, and from the Servizio Fitosanitario Regionale Se.S.I.R.C.A. Napoli, Regione Campania. We are very grateful to Dr Rene Alfaro from the Canadian Forest Service for his initiatory work in the pheromone detection of M. mutatus and the editing of the manuscript, and the phytosanitary inspectors Eduardo Ucciero, Patrizia Nappa, Vincenzo Orologialo, Tommaso Brosco, Rhino Melenghi, Guiseppe Salzillo, Pasquale Viola, Domenico Esposito and Francesco Del Vecchio for their support in field work. PG had a doctoral grant from the University of San Martin. PGA and EZ are members of the Consejo Nacional Investigaciones Científicas y Técnicas (CONICET) and of University of San Martin (UNSAM).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonzalez-Audino, P., Griffo, R., Gatti, P. et al. Pheromone detection of the introduced forest pest Megaplatypus mutatus (=Platypus mutatus) (Chapuis) (Platypodinae, Curculionidae) in Italy. Agroforest Syst 87, 109–115 (2013). https://doi.org/10.1007/s10457-012-9527-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-012-9527-3