Abstract
Moyamoya disease (MMD) and atherosclerotic cerebrovascular disease (ACVD) differ in angiographic appearance and probably hemodynamics. Pediatric MMD (PMMD) usually presents with cerebral ischemia, while intracranial hemorrhage is more common in adult MMD (AMMD), suggesting differences in cerebral hemodynamics. We analyzed the cortical flow velocity and direction of recipient arteries using micro-Doppler ultrasonography to evaluate the cortical circulation before and after anastomosis in MMD and ACVD. Twenty-eight patients with adult MMD (AMMD), 7 with pediatric MMD (PMMD), 16 with ACVD, and 12 control patients were studied. A micro-Doppler probe was applied on the cortical recipient artery (A4 or M4) before and after anastomosis. Systolic maximum flow velocity (V max) and blood flow direction were investigated at proximal and distal parts of anastomosed sites in recipient arteries. Pre- and postoperative regional cerebral blood flow was measured by cold xenon-computed tomography (Xe-CT). Before anastomosis, retrograde cortical flow was significantly more common in PMMD patients, and V max in cortical artery was significantly lower in AMMD patients. Bypass surgery changed the direction of blood flow from the anastomosis site to proximal and distal sites of the recipient artery in most patients, but pre-anastomosis flow direction was preserved more frequently in PMMD patients. The rate of V max increase after anastomosis was significantly higher in AMMD than in PMMD (11.6 ± 9.8 vs. 3.9 ± 1.8; P = 0.01). Micro-Doppler ultrasonography identified differences in cortical circulation among AMMD, PMMD, and ACVD. In AMMD, significantly low velocity in the cortical artery was observed before anastomosis, and bypass surgery reversed the flow and significantly increased flow velocity. The data of PMMD showed unique hemodynamics of the cortical artery before anastomosis, characterized by a higher frequency of retrograde flow and preserved velocity. The V max increase rate was significantly higher in patients with postoperative cerebral hyperperfusion on Xe-CT, and further study is warranted to validate the clinical use of intraoperative micro-Doppler monitoring to predict postoperative hyperperfusion.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Moyamoya disease (MMD) and atherosclerotic cerebrovascular disease (ACVD) are both occlusive cerebrovascular disease, but the cerebral hemodynamics has been shown to differ not only between MMD and ACVD [2, 5, 19] but also between adult MMD (AMMD) and pediatric MMD (PMMD) [15, 25, 29]. ACVD causes occlusion or severe stenosis of carotid or intracranial artery, resulting in ischemic attack. MMD is characterized by chronic progressive steno-occlusive changes affecting the terminal portions of internal carotid arteries, accompanied by the development of collateral networks at the base of the brain, called “moyamoya vessels” [18, 26, 27]. AMMD typically presents with intracranial hemorrhage. In contrast, PMMD usually exhibits transient ischemic attack (TIA) or cerebral infarction. However, the differences between the diseases in terms of hemodynamics and postoperative circulatory changes are not fully known.
Imaging techniques have elucidated the circulatory dynamics and postoperative changes by measuring cerebral blood flow (CBF) before and after surgery. Several studies have conducted intraoperative measurements using laser Doppler flowmeter, infrared brain surface monitoring, indocyanine green (ICG) angiography, and transcranial Doppler ultrasonography (TCD). Micro-Doppler ultrasonography demonstrates microvascular flow wave pattern, velocity, and direction by directly applying a probe on a microvessel. Some systems record flow volume as mathematical values [14]. Micro-Doppler ultrasonography is generally used to evaluate the flow of aneurysm and its trunks during clipping [9]; feeding arteries, nidus, and draining veins of arteriovenous malformation [1]; and patency of microvascular anastomoses [16]. This method is simple and reliable.
There are few hemodynamic studies on occlusive cerebrovascular diseases using micro-Doppler ultrasonography, which compare MMD and ACVD. In the present study, we examined the cortical hemodynamics and surgical effects in AMMD, PMMD, and ACVD patients by applying a micro-Doppler probe on cortical micro-arteries before and after superficial temporal artery–middle cerebral artery (STA–MCA) anastomosis.
Patients and methods
Patients
Fifty-one patients (52 hemispheres) were monitored by micro-Doppler ultrasonography during STA–MCA double anastomoses between July 2007 and March 2010. The moyamoya disease patients in our recently reported study [10] were not included in the present study. Thirty-five patients were diagnosed as MMD, consisting of 28 with AMMD (median age 42.7 ± 11.5 years, range 20 to 70 years) and 7 with PMMD (younger than 15 years of age) (median age 8.6 ± 1.7 years, range 6 to 11 years). Sixteen patients had ACVD (median age 64.2 ± 10.4 years, range 34 to 72 years): 10 with internal cerebral artery (ICA) occlusion, 1 with severe ICA stenosis, 2 with MCA occlusion, and 3 with MCA stenosis. All patients underwent preoperative cerebral angiography, and MMD patients were classified into angiographic stages according to the classification described by Suzuki and Takaku [26, 27]. In 28 AMMD patients (29 hemispheres), 15 were stage 3, 11 were stage 4, and 3 were stage 5. In PMMD patients, 6 were stage 3, and 1 was stage 5. The clinical characteristics including symptoms, the preoperative regional CBF (rCBF), and the preoperative cerebrovascular reactivity (rCVR) are shown in Table 1. Although the PMMD group consisted of only seven cases, this group was included in the comparison and the results were interpreted with caution. When the preoperative rCBF in adult patients was compared with normal control rCBF data obtained from healthy volunteers (15 healthy volunteers; aged 18–72 years; mean 50 years) [22, 30], rCBF was reduced in AMMD (P < 0.0001) and ACVD patients (P < 0.0001) (Table 1) (F = 25.16, P < 0.0001). The data of PMMD patients could not be compared because control rCBF data of children were not available. Unfortunately, we have no normal control rCVR data from the healthy volunteers (Table 1).
As controls, 12 patients (median age 56.8 years) with unruptured cerebral aneurysms were investigated. The velocity of cortical artery was measured by micro-Doppler ultrasonography during clipping surgery.
Surgery
In MMD patients, STA–MCA double bypass or STA–MCA/STA–ACA bypass was selected based on symptoms and CBF [12]. In ACVD patients, only STA–MCA double bypass was used. STA was anastomosed end-to-side to each of the cortical artery MCA (M4) or ACA (A4) on the brain surface in supra-sylvian fissure and infra-sylvian areas. A silicone stent (400 μm in diameter, 3 to 4 mm in length) that we designed was inserted into the recipient artery by an arteriotomy to facilitate anastomotic procedures [24]. The surgical indication of direct bypass surgery for ACVD was based on the criteria in the Japanese EC–IC Bypass Trial [20]. In MMD patients, indication of surgical treatment was minor completed ischemic stroke or repeated TIAs. The operation was performed more than 2 weeks after the latest ischemic attack.
Intraoperative micro-Doppler monitoring
The monitoring system (Companion III, Nicolet Vascular Inc., Madison, WI, USA) displayed Doppler waveform, direction of blood flow, and maximum systolic flow velocity (V max). The center frequency was 20 MHz. A micro-probe (Monitoring Probe X1-P112, Nicolet Vascular Inc.) 1.5 mm in diameter was carefully applied on a vessel at a 45° angle. Measurable V max range was from 0 to 120 cm/s. V max higher than 120 cm/s was recorded as 120 cm/s.
Before anastomosis, the selected recipient arteries (M4 or A4) were investigated with micro-Doppler. The ultrasonic waveform, V max, and flow directions were recorded. After anastomosis, the same parameters were measured at proximal (“a” and “c” in Fig. 1a) and distal (“b” and “d” in Fig. 1a) sides of the anastomoses. Figure 1b demonstrates the data of a representative case.
a Intraoperative view (left) and indocyanine green angiogram (right) of a patient undergoing STA–MCA double anastomoses. Points a to d denote sites of measurement by micro-Doppler ultrasonography after anastomoses: proximal part (a) and distal part (b) of the anastomosed site at infra-sylvian M4 and proximal part (c) and distal part (d) of the anastomosed site at supra-sylvian M4. b Micro-Doppler ultrasonograms of a 40-year-old man with AMMD presented with transient ischemic attack demonstrating Doppler wave form, flow direction, and velocity before and after anastomosis. a to d denote the sites described in a. Upper figures (supra-sylvian area) show that pre-anastomosis retrograde flow of the recipient artery was altered after anastomosis to both retrograde and anterograde flow divided at the anastomosed site. In other words, the flow direction proximal to the anastomosis did not change, while that distal to the anastomosis was reversed. Lower figures (infra-sylvian area) show that pre-anastomosis anterograde flow changed to retrograde flow proximal to the anastomosis, while the flow distal to the anastomosis remained anterograde
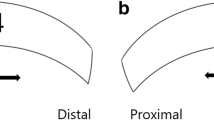
The flow direction of the STA and recipient artery after anastomosis could be divided into three patterns as illustrated in Fig. 2. In pattern A, blood flow of the recipient artery is almost totally supplied by STA and is retrograde proximal to the anastomosis and anterograde distal to the anastomosis. In pattern B, blood flow of the recipient artery is anterograde. In pattern C, blood flow of the recipient artery is retrograde. Patterns B and C imply that the pre-anastomosis flow direction is preserved.
Schemes demonstrating three patterns of post-anastomosis blood flow. A Blood flow of recipient artery is almost totally supplied by STA. The flow proximal to the anastomosis is retrograde and that distal to the anastomosis is anterograde. B Blood flow of recipient artery is anterograde. C Blood flow of recipient artery is retrograde
V max increase rate was calculated as the increase in V max after anastomosis divided by pre-anastomosis V max. The rate was measured at the proximal and distal sites of the anastomosed point. When the flow direction was reversed after anastomosis, we added post-anastomosis V max to pre-anastomosis V max to obtain the increase in V max. When the flow direction was unchanged after anastomosis, we subtracted pre-anastomosis V max from post-anastomosis V max.
Cold xenon-computed tomography (Xe-CT)
Preoperative and postoperative (immediately after surgery under general anesthesia) rCBF were measured by cold Xe-CT. Preoperative regional rCVR was measured by the acetazolamide loading test (17 mg/kg i.v.) compared to resting CBF.
We defined postoperative hyperperfusion on rCBF as greater than 100 % increase in corrected postoperative ipsilateral rCBF over the preoperative value [11]. The corrected postoperative ipsilateral rCBF was calculated based on the rate of reduction in postoperative rCBF relative to preoperative value on the nonoperative side (corrected postoperative ipsilateral rCBF = postoperative ipsilateral rCBF × preoperative contralateral rCBF / postoperative contralateral rCBF).
Statistical analysis
Preoperative rCBF was compared among three groups of adults (AMMD, ACVD, and normal adult control) by one-way analysis of variance (ANOVA), followed by Tukey’s test. Correlations between preoperative rCBF and pre-anastomosis V max and between preoperative rCVR and pre-anastomosis V max were analyzed by Spearman’s rank correlation coefficient (rs). Pearson’s chi-square test was used to compare pre-anastomosis flow direction (anterograde or retrograde) among the three groups. An adjusted residual score greater than 1.96 for a subgroup percentage indicates that the subgroup differed from the other subgroups. Post-anastomosis patterns (A, B, and C) among the groups were compared by Fisher’s exact test followed by Bonferroni adjustment, because the cell of low expected frequency was less than 5. Differences were considered statistically significant when P was less than 0.016.
Kruskal–Wallis rank sum test followed by Steel–Dwass test was used to compare V max and V max increase rate as nonparametric variables among the three groups. Wilcoxon rank sum test was used to compare V max increase rate and postoperative hyperperfusion as nonparametric variables for two groups. Differences were considered statistically significant when P was less than 0.05. All data are presented as mean ± SD.
Results
Doppler records of pre-anastomosis recipient arteries
Flow directions of cortical arteries were significantly different (P = 0.036) among ACVD, AMMD, and PMMD by Pearson’s chi-square test (Fig. 3). Retrograde flow was recorded significantly more frequently in PMMD than in AMMD and ACVD patients, with adjusted residual of +2.4 (>1.96). Retrograde flow in recipient artery was found in 57 % of PMMD patients, 29 % of AMMD patients, and 19 % of ACVD patients (Fig. 3).
Comparison of pre-anastomosis blood flow direction in recipient arteries among AMMD, PMMD, and ACVD groups. The numbers of the column denote patient numbers. A significant difference among three patient groups was observed (Pearson’s chi-square test: P = 0.036). Black star: retrograde flow was recorded significantly more frequently in PMMD patients, with adjusted residual +2.4 (>1.96)
Pre- and post-anastomoses V max of cortical recipient arteries (M4 or A4) are summarized in Table 2. Mean pre-anastomosis V max of cortical recipient arteries (M4 or A4) were 9.0 ± 6.8 cm/s in AMMD patients, 14.2 ± 8.2 cm/s in PMMD patients, and 12.9 ± 7.9 cm/s in ACVD patients. Control patients had mean cortical V max of 13.7 ± 3.0 cm/s. Kruskal–Wallis rank sum test revealed significant difference among four groups (P = 0.001) (Fig. 4). Furthermore, Steel–Dwass test showed significantly lower V max in AMMD patients compared to controls (P = 0.007) (Fig. 4) and a tendency of lower V max in AMMD compared to PMMD (P = 0.07) and to ACVD (P = 0.058).
Systolic maximum flow velocity (V max) in control, AMMD, PMMD, and ACVD groups before direct bypass. V max in the AMMD group was significantly lower than the control. Kruskal–Wallis rank sum test revealed significant difference among the four groups (P = 0.001) Furthermore, Steel–Dwass test showed significantly lower V max in AMMD patients compared to controls (asterisk: P = 0.007), and a tendency of lower V max in AMMD compared to PMMD (P = 0.07) and to ACVD (P = 0.058)
Comparing the flow velocity and rCBF before surgery in adult patients, both flow velocity and rCBF were significantly reduced in AMMD, while flow velocity was preserved but rCBF was significantly reduced in ACVD (Fig. 4 and Table 1). Flow velocity appears to reflect rCBF in AMMD but not in ACVD. Taken together, flow velocity did not reflect rCBF directly in adult patients with occlusive cerebrovascular diseases in the present study.
Rank correlation analysis showed no correlation between preoperative rCBF and pre-anastomosis V max in all subjects (rs = 0.17, P = 0.24) and in AMMD (rs = 0.2, P = 0.29), PMMD (rs = 0.0, P = 1.0), and ACVD (rs = 0.14, P = 0.62). Likewise, no correlation was observed between preoperative rCVR and pre-anastomosis V max in all subjects (rs = 0.18, P = 0.23) and in AMMD (rs = 0.26, P = 0.21), PMMD (rs = 0.66, P = 0.16), and ACVD (rs = 0.14, P = 0.63). Therefore, flow velocity did not reflect rCBF and rCVR in the present study.
No correlation was observed between pre-anastomosis V max and the angiographic stage of MMD. In AMMD patients, V max (cm/s) were 10.1 ± 8.4 in stage 3, 7.5 ± 4.1 in stage 4, and 9.2 ± 5.9 in stage 5. In PMMD patients, V max were 15.0 ± 7.9 in stage 3 and 10.0 ± 11.3 in stage 5.
Doppler records of recipient arteries after anastomosis
After bypass surgeries, patterns B and C were found in PMMD patients more frequently (21.4 %) than in AMMD (1.9 %) and ACVD patients (3.6 %). The rate of patterns B and C tended to be higher in PMMD than in AMMD, although the difference did not reach statistical significance [P = 0.027 (>0.016)] by Fisher’s exact test followed by Bonferroni adjustment. The post-anastomosis flow direction did not depend on the slope direction of STA.
Mean V max increase rates in AMMD, PMMD, and ACVD patients were 11.6 ± 9.8, 3.9 ± 1.8, and 9.0 ± 12.0, respectively, in the first anastomosis and 9.2 ± 9.7, 4.7 ± 3.4, and 5.5 ± 5.2 in the second anastomosis. The increase rates did not depend on the slope direction of STA. Kruskal–Wallis rank sum test detected a significant difference (P = 0.006) in V max increase rate among three groups in the first anastomosis, and Steel-Dwass test identified a significant difference between AMMD and PMMD (P = 0.01) (Fig. 5). The V max increase rate of the second anastomosis was significantly lower than that of the first anastomosis (Wilcoxon rank sum test P = 0.029). Kruskal-Wallis rank sum test detected no significant difference in V max increase rate among angiographic stages in AMMD (P = 0.36) and PMMD (P = 0.15).
V max increase rates in AMMD, PMMD, and ACVD groups in the first and second anastomoses. Kruskal–Wallis rank sum test detected a significant difference (P = 0.006) in V max increase rate among the three groups in the first anastomosis, and V max increase rate in the first anastomosis was significantly higher in the AMMD group than in the PMMD group (Steel–Dwass test; asterisk: P = 0.01)
Cerebral hyperperfusion on Xe-CT and V max increase rate
Postoperative hyperperfusion in ACA or MCA territories, defined as 100 % or greater increase over preoperative rCBF, was observed in six hemispheres of AMMD patients (20.7 %). No cerebral hyperperfusion was observed in PMMD patients, while three hemispheres of ACVD patients (18.8 %) showed hyperperfusion. In all patients, the V max increase rate was significantly higher in patients with cerebral hyperperfusion in the operative hemisphere on Xe-CT (12.2 ± 11.8) than in those without cerebral hyperperfusion (7.8 ± 8.4) (Wilcoxon rank sum test: P = 0.02) (Fig. 6).
Discussion
Cortical hemodynamics of AMMD, PMMD, and ACVD patients
Flow direction and velocity of cortical micro-arteries measured by micro-Doppler ultrasonography were different among AMMD, PMMD, and ACVD patients. The preoperative flow velocity in recipient arteries in AMMD patients was significantly lower than that in control. PMMD patients had unique hemodynamics characterized by a high frequency of retrograde cortical blood flow and comparable flow velocity as the control group. In ACVD patients, the frequency of retrograde flow was the lowest, and flow velocity was comparable to that of the control.
Several studies have demonstrated that hemodynamics differ among the three groups. While rCBF in AMMD has been found to be preserved or higher than in ACVD, rCVR decreases more severely in AMMD patients [19]. In our previous studies in AMMD, calculated rCBF and perfusion pressure in the MCA territory indicated high cerebral arterial vascular resistance at the proximal side and very low cerebral vascular resistance at the distal side [23]. There is a general agreement that PMMD patients have impaired rCVR. These may explain why the common clinical presentation of PMMD is ischemic stroke and/or TIA [15].
Most of the recent reports on Doppler ultrasonographic study of cerebrovascular diseases are related to TCD. Ruan et al. [25] observed high velocity and low resistance index in common carotid artery and ICA and either low or extremely high velocity in stenotic ACA, MCA, and posterior cerebral artery. According to a report of TCD data from the ICA to the distal part of basal MCA in MMD, the vascular resistance through the stenotic lesion was lower in later stage, and severe stenosis resulted in low velocity in the distal MCA [29]. The hemodynamics of distal and cortical vessels have been found to differ from those of major cerebral arteries in terms of rCVR and flow velocity.
TCD is mainly used to study main trunks, while ICG angiography has been used to study cortical hemodynamics. Czabanka et al. [5] demonstrated using ICG angiography that cortical microvascular diameter was increased, and arterial microvascular transit time (indirectly representing velocity) was prolonged in AMMD patients compared with ACVD and control patients. Their results suggest decreased microvascular resistance in AMMD. There is increasing evidence that CBF in AMMD is preserved by compensatory lowering of vascular resistance and increase of moyamoya vessel and microvascular diameter, leading to lowered velocity in peripheral arteries. The present results of significantly lower V max in AMMD patients may be compatible with the above and our previous findings [23].
Young MMD patients exhibit rapid clinical progression and significantly more frequent ischemic attacks [13, 28]. This study showed no significant correlation between angiographic stage and cortical velocity. Previous studies have proposed that angiographic stage is not directly related to clinical presentation, hemodynamics, or treatment risk [4, 8]. We propose that the unique hemodynamic pattern in PMMD, characterized by a high frequency of retrograde cortical flow and preserved velocity, may be entirely distinct from the hemodynamics seen in AMMD. The direction of blood flow depends on the pressure gradient. Retrograde flow in PMMD patients might be caused by insufficient anterograde flow or the development of leptomeningeal anastomoses.
Effect of STA–MCA anastomosis on cortical hemodynamics
In general, flow directions after bypass surgery depend on the arterial pressure of cortical recipients and donor artery (STA). The present results suggest that STA–MCA anastomosis affects the cortical circulation and clinical course differently in various patient groups. In most patients, the post-anastomosis flow direction in the recipient artery was anterograde distal to the anastomosed point and retrograde proximal to the anastomosis and was dependent on the flow from STA against the pre-anastomosis flow (pattern A). Patterns B and C, in which pre-anastomosis flow direction was preserved, were rarely seen in AMMD and ACVD patients, but were found in 21 % of bypasses in PMMD patients. The present results concerning the flow direction in the recipient artery after bypass may reflect the preoperative lower velocity in AMMD and retrograde flow with preserved velocity in PMMD.
Our micro-Doppler study demonstrated a significantly higher rate of increase in velocity in AMMD patients but not in ACVD patients after direct bypass surgery. Using ICG angiography, Awano et al. [2] demonstrated that the STA–MCA anastomosis supplies blood flow to a greater area in AMMD patients than in patients with ACVD. In our previous studies in AMMD, STA–MCA anastomosis partially normalized cerebral circulation by increasing perfusion pressure and rCBF at the anastomosed area, which resulted in decreasing moyamoya vessels [23]. The above data together with our results indicate that after direct bypass surgery, blood flows into the dilated vessels and perfuses a wide area, signifying abnormally high cortical perfusion, especially in AMMD patients. On the other hand, the rate of increase in V max did not correlate with the angiographic stage of MMD. One reason may be that the staging does not directly reflect circulation or postoperative changes as mentioned above.
Recent reports have indicated that STA–MCA anastomosis sometimes results in focal cerebral hyperperfusion in MMD patients. Preoperative reduced rCVR is known to be a risk factor of postoperative cerebral hyperperfusion, and adult onset or hemorrhagic onset patients had significantly higher risk of symptomatic cerebral hyperperfusion [6]. A few studies reported postoperative hyperperfusion in ACVD patients and indicated preoperative steal phenomenon as a predictor [30]. Several methods have been proposed for the detection of cerebral hyperperfusion during operation or early after operation [10]. Gesang et al. [7] indicated that laser Doppler flowmeter is useful for postoperative real-time monitoring during the high-risk period. Intraoperative measurements such as thermography, infrared brain surface monitoring, and ICG fluorescence angiography predict post-anastomosis cortical hemodynamics [2, 10, 17, 21]. It is generally known that cortical velocity does not directly influence CBF, especially in normal individuals, who may show variable cortical velocities due to responses to cerebrovascular resistance and diameter [3, 5]. On the contrary, in cerebrovascular occlusive diseases, especially in MMD, cerebrovascular resistance decreases and distal vascular diameter dilates to the maximum [23]. Under these conditions, since the cortical velocity fluctuates minimally, micro-Doppler ultrasound may be useful to reflect local CBF. In the present study, the V max increase rate in patients with postoperative cerebral hyperperfusion on Xe-CT was elevated significantly. However, the variability was large probably due to the small number of hyperperfusion cases. The hand-held microprobe may be a source of error in quantitative analysis and has to be addressed in the future. Further study is warranted to validate whether intraoperative micro-Doppler ultrasonography can be used clinically to predict postoperative cerebral hyperperfusion.
Conclusions
Intraoperative micro-Doppler ultrasonography monitoring was useful to characterize cortical hemodynamics and the flow alterations after direct bypass surgeries in AMMD, PMMD, and ACVD patients. In AMMD, significantly low velocity in the cortical artery was observed before anastomosis, and bypass surgery reversed the flow and significantly increased flow velocity. The data on a limited number of PMMD cases showed unique hemodynamics of the cortical artery before anastomosis, characterized by a higher frequency of retrograde flow and preserved velocity. In ACVD, hemodynamic abnormalities in the cortical artery were comparatively mild in terms of velocity and flow direction, and anastomosis produced mild changes. The V max increase rate was significantly higher in patients with postoperative cerebral hyperperfusion, and large increases might alert a risk of postoperative hyperperfusion. A larger number of patients and more reliable quantification methods should be used to validate the present clinical results.
References
Akdemir H, Oktem S, Menkü A, Tucer B, Tuğcu B, Günaldi O (2007) Image-guided microneurosurgical management of small arteriovenous malformation: role of neuronavigation and intraoperative Doppler sonography. Minim Invasive Neurosurg 50:163–169
Awano T, Sakatani K, Yokose N, Kondo Y, Igarashi T, Hoshino T, Nakamura S, Fujiwara N, Murata Y, Katayama Y, Shikayama T, Miwa M (2010) Intraoperative EC-IC bypass blood flow assessment with indocyanine green angiography in moyamoya and non-moyamoya ischemic stroke. World Neurosurg 73:668–674
Bishop CCR, Powell S, Rutt D (1986) Transcranial Doppler measurement of middle cerebral artery blood flow velocity: a validation study. Stroke 17:913–915
Czabanka M, Peña-Tapia P, Schubert GA, Heppner FL, Martus P, Horn P, Schmiedek P, Vajkoczy P (2011) Proposal for a new grading of moyamoya disease in adult patients. Cerebrovasc Dis 32:41–50
Czabanka M, Peña-Tapia P, Schubert GA, Woitzik J, Vajkoczy P, Schmiedek P (2008) Characterization of cortical microvascularization in adult Moyamoya disease. Stroke 39:1703–1709
Fujimura M, Mugikura S, Kaneta T, Shimizu H, Tominaga T (2009) Incidence and risk factors for symptomatic cerebral hyperperfusion after superficial temporal artery–middle cerebral artery anastomosis in patients with moyamoya disease. Surg Neurol 71:442–447
Gesang DZ, Zhang D, Zhao JZ, Wang S, Zhao YL, Wang R, Sun JJ, Meng Z (2009) Laser Doppler flowmeter study on regional cerebral blood flow in early stage after standard superficial temporal artery–middle cerebral artery bypass surgery for moyamoya disease. Chin Med J 122:2412–2418
Houkin K, Yoshimoto T, Kuroda S, Ishikawa T, Takahashi A, Abe H (1996) Angiographic analysis of moyamoya disease—how does moyamoya disease progress? Neurol Med Chir (Tokyo) 36:783–788
Kapsalaki EZ, Lee GP, Robinson JS 3rd, Grigorian AA, Fountas KN (2008) The role of intraoperative micro-Doppler ultrasound in verifying proper clip placement in intracranial aneurysm surgery. J Clin Neurosci 15:153–157
Kawamata T, Kawashima A, Yamaguchi K, Hori T, Okada Y (2011) Usefulness of intraoperative laser Doppler flowmetry and thermography to predict a risk of postoperative hyperperfusion after superficial temporal artery–middle cerebral artery bypass for moyamoya disease. Neurosurg Rev 34:355–362
Kawamata T, Okada Y, Kawashima A, Yoneyama T, Yamaguchi K, Ono Y, Hori T (2009) Postcarotid endarterectomy cerebral hyperperfusion can be prevented by minimizing intraoperative cerebral ischemia and strict postoperative blood pressure control under continuous sedation. Neurosurgery 64:447–454
Kawashima A, Kawamata T, Yamaguchi K, Hori T, Okada Y (2010) Successful superficial temporal artery–anterior cerebral artery direct bypass using a long graft for moyamoya disease: technical note. Neurosurgery 67 (3 Suppl Operative):ons145–149
Kim SK, Seol HJ, Cho BK, Hwang YS, Lee DS, Wang KC (2004) Moyamoya disease among young patients: its aggressive clinical course and the role of active surgical treatment. Neurosurgery 54:840–846
Lee M, Guzman R, Bell-Stephens T, Steinberg GK (2011) Intraoperative blood flow analysis of direct revascularization procedures in patients with moyamoya disease. J Cereb Blood Flow Metab 31:262–274
Lee M, Zaharchuk G, Guzman R, Achrol A, Bell-Stephens T, Steinberg GK (2009) Quantitative hemodynamic studies in moyamoya disease: a review. Neurosurg Focus 26:E5
Mücke T, Wolff KD, Wagenpfeil S, Hölzle F, Scholz M (2010) Reliability of near-infrared angiography and micro-Doppler sonography for evaluating microvascular anastomoses. Plast Reconstr Surg 126:1506–1514
Nakagawa A, Fujimura M, Arafune T, Suzuki H, Sakuma I, Tominaga T (2008) Intraoperative infrared brain surface blood flow monitoring during superficial temporal artery–middle cerebral artery anastomosis in a patient with moyamoya disease: clinical implication of the gradation value in postoperative clinical course—a case report. Acta Neurochir Suppl 102:159–163
Nishimoto A, Takeuchi S (1968) Abnormal cerebral vascular network related to the internal carotid arteries. J Neurosurg 29:255–260
Obara K, Fukuuchi Y, Kobari M, Watanabe S, Dembo T (1997) Cerebral hemodynamics in patients with moyamoya disease and in patients with atherosclerotic occlusion of the major cerebral arterial trunks. Clin Neurol Neurosurg 99(Suppl 2):S86–S89
Ogasawara K, Ogawa A (2006) JET study (Japanese EC-IC Bypass Trial). Nippon Rinsho 64(Suppl 7):524–527
Okada Y, Kawamata T, Kawashima A, Hori T (2007) Intraoperative application of thermography in extracranial-intracranial bypass surgery. Neurosurgery 60(4 Suppl 2):362–365
Okada Y, Kawamata T, Kawashima A, Yamaguchi K, Ono Y, Hori T (2012) The efficacy of superficial temporal artery–middle cerebral artery anastomosis in patients with moyamoya disease complaining of severe headache. J Neurosurg 116:672–679
Okada Y, Shima T, Nishida M, Yamane K, Yamada T, Yamanaka C (1998) Effectiveness of superficial temporal artery–middle cerebral artery anastomosis in adult moyamoya disease: cerebral hemodynamics and clinical course in ischemic and hemorrhagic varieties. Stroke 29:625–630
Okada Y, Shima T, Yamane K, Yamanaka C, Kagawa R (1999) Cylindrical or T-shaped silicone rubber stents for microanastomosis—technical note. Neurol Med Chir (Tokyo) 39:55–58
Ruan LT, Duan YY, Cao TS, Zhuang L, Huang L (2006) Color and power Doppler sonography of extracranial and intracranial arteries in Moyamoya disease. J Clin Ultrasound 34:60–69
Suzuki J, Kodama N (1983) Moyamoya disease: a review. Stroke 14:104–109
Suzuki J, Takaku A (1969) Cerebrovascular “moyamoya” disease. Disease showing abnormal net-like vessels in base of brain. Arch Neurol 20:288–299
Takanashi J (2011) Moyamoya disease in children. Brain Dev 33:229–234
Takase K, Kashihara M, Hashimoto T (1997) Transcranial Doppler ultrasonography in patients with moyamoya disease. Clin Neurol Neurosurg 99(Suppl 2):S101–S105
Yamaguchi K, Kawamata T, Kawashima A, Hori T, Okada Y (2010) Incidence and predictive factors of cerebral hyperperfusion after extracranial–intracranial bypass for occlusive cerebrovascular diseases. Neurosurgery 67:1548–1554
Acknowledgments
We thank the radiological technicians at the Departments of Neurosurgery and Radiology, Tokyo Women’s Medical University, Tokyo, Japan, for measurements of rCBF and CVR on cold Xe-CT. This study was supported by research funds of the Department of Neurosurgery, Tokyo Women’s Medical University.
Conflict of interest
All authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Yasuhiro Yonekawa, Zürich, Switzerland
Morisawa et al. are to be congratulated for delivering interesting information concerning the hemodynamics of cortical flow in three different cerebrovascular occlusive diseases, PMMD, AMMD, and ACVD, in comparison with a control group obtained at the time of aneurysm surgery. These examinations have been done with the aid of micro-Doppler sonography that we are using only to check vascular patency, especially after construction of an EC–IC bypass or in aneurysm surgery. Originally, the authors advocated the theory that the increase of the post-anastomotic V max could predict a hyperperfusion syndrome; however, they somewhat toned down, presumably due to an insufficient number of cases, stating that their finding may help in preventing this undesirable complication. To be honest, I miss some additional explanation or speculation about the mechanism responsible for such hemodynamic differences between patients with moyamoya disease, both pediatric and adult, or between moyamoya and atherosclerotic occlusive disease, even though some information regarding leptmeningeal anastomosis and rCVR has been given. Perhaps such an explanation could be possible with additional data obtained by measuring the intraarterial pressure or the local rCBF with the use of a thermocouple [1, 2]. Such measurements would also furnish evidence of changes in cortical flow direction after bypass surgery.
Table
V max: cortical flow | Retrograde flow | ∆V max | |
|---|---|---|---|
PMMD | ⇒ | +++ | + |
AMMD | ⇓ | ++ | +++ |
ACVD | ⇒ | + | ++ |
References
1 Yonekawa Y: Brain vascularization by extracranial–intracranial arterial bypass. In Sindou M (ed). Practical hand book of neurosurgery. Vol 1. Springer, Wien, 2009, pp 355-381
2 Ogata N, Fournier JY, Imhof HG, Yonekawa Y: Thermal diffusion blood flow monitoring during aneurysm surgery. Acta Neurochir (Wien) 138: 726-731, 1996
Kiyohiro Houkin, Sapporo, Japan
The authors clearly have demonstrated quantitative flow measurement in moyamoya disease using micro-Doppler ultrasonography. This work is praiseworthy for clinical application. They have also revealed that in pediatric moyamoya cases, the cortical flow shows retrograde flow and its V max (systolic maximum flow velocity) is higher than the adult moyamoya cases. This may mean that retrograde flow due to the pial anastomosis in pediatric cases is well established than in adult case.
As it is well-known, the collateral flow is dependent on the stages of this disease. In addition, the flow dynamics may depend on its location (area of craniotomy). We have to be careful to evaluate these quantitative data. I am confident that the intraoperative flow measurement using micro-Doppler ultrasonography is practical and convenient in intraoperative flow measurement based on my personal experience. Moreover, the hyperperfusion that is a serious issue after successful direct bypass to moyamoya disease can be predicted using this very convenient tool. More additional clinical experience with this tool is necessary and the establishment of a standard way of reliable evaluation using this weapon is expected.
Rights and permissions
About this article
Cite this article
Morisawa, H., Kawamata, T., Kawashima, A. et al. Hemodynamics and changes after STA–MCA anastomosis in moyamoya disease and atherosclerotic cerebrovascular disease measured by micro-Doppler ultrasonography. Neurosurg Rev 36, 411–419 (2013). https://doi.org/10.1007/s10143-012-0441-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-012-0441-y