Abstract
We investigated whether postoperative hyperperfusion in moyamoya disease can be predicted using intraoperative laser Doppler flowmetry and/or thermography. A prospective study was conducted on 27 patients (39 hemispheres) with moyamoya disease who underwent superficial temporal artery–middle cerebral artery (STA–MCA) bypass. During surgery, regional cerebral blood flow (rCBF) was measured with a laser Doppler flowmeter and the temperature of the cortical surface was measured with an infrared thermograph. Postoperative hyperperfusion was assessed immediately after surgery based on CBF study under sedation (propofol) as >100% increase in corrected rCBF compared to preoperative values. Postoperative hyperperfusion on CBF was observed in two patients (7.4%). A significant correlation was observed between intraoperative rCBF changes and postoperative rCBF increase (Pearson’s method: r = 0.555, p = 0.0003; simple regression: Y = 1.22X + 3.289, r 2 = 0.308, p = 0.0004). Furthermore, the rCBF changes measured by laser Doppler flowmetry were significantly greater in patients with postoperative hyperperfusion (p = 0.0193) and CHS (p = 0.0193). The present study suggests that intraoperative rCBF measurement using laser Doppler flowmetry may predict a risk of post-EC–IC bypass cerebral hyperperfusion in moyamoya disease.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Postoperative cerebral hyperperfusion syndrome (CHS), which is characterized by ipsilateral throbbing headache, ocular and facial pain, seizure and focal neurological deficits, is well recognized following cerebral revascularization, especially after carotid endarterectomy (CEA) [26, 28]. While post-CEA hyperperfusion has been extensively studied, hyperperfusion after revascularization in moyamoya disease is not well documented, even though hyperperfusion as shown by regional cerebral blood flow (rCBF) is sometimes encountered after extracranial–intracranial (EC–IC) bypass in moyamoya disease [3, 4, 17, 19, 30]. It is desirable to be able to predict postoperative hyperperfusion from pre- or intra-operative rCBF data for postoperative management.
Thermographic techniques have been developed in many fields, and have also been applied in the field of neurosurgery [12, 14, 20, 23]. In particular, thermographic investigations in neurosurgery have focused on cortical circulation. Okudera et al. [23] used intraoperative thermographic imaging during surgical resection of a cerebral arteriovenous malformation and demonstrated changes in surface temperature of the cortical draining vein and the surrounding cortical surface before and after occlusion of the main feeder. We also demonstrated that thermography is useful in superficial temporal artery–middle cerebral artery (STA–MCA) bypass not only to demonstrate the distribution of blood flow, but also to evaluate quantitatively the rCBF changes at the operative field [20].
Although EC–IC bypass was reported to fail in preventing recurrence of stroke [29], some reports have demonstrated the potential effectiveness of STA–MCA anastomosis to improve compromised hemodynamic cerebral ischemia [16, 21]. Intraoperative data of anastomotic blood flow and its distribution are important to confirm an effective EC–IC bypass. Currently, anastomotic flow can be measured intraoperatively by several methods such as electromagnetic flowmeter, Doppler flowmeter, visual light spectroscopy, and videoangiography [1, 6, 33].
In the present study, we investigated whether it is possible to predict postoperative hyperperfusion following EC–IC bypass using intraoperative laser Doppler flowmetry and/or thermography in patients with moyamoya disease.
Patients and methods
Patients
Twenty-seven patients with moyamoya disease who underwent STA–MCA bypass on 39 hemispheres were included in the present study (Table 1). The patient population consisted of ten men and 17 women ranging in age from 7 to 62 years [9 children (younger than 16 years of age) and 18 adults]. Moyamoya disease was diagnosed by angiography in all patients. Surgical indications for STA–MCA bypass were transient ischemic attack in 18 patients and minor complete stroke in nine. Preoperative (within 1 week before surgery) and postoperative rCBF (immediately after surgery) were measured by cold Xe-computed tomography (CT). Xe-CT can measure rCBF quantitatively and noninvasively [5, 15, 35] and provide precise anatomic and pathologic data by correlating with the corresponding conventional CT image [35]. Decreased rCBF was defined as greater than 20% reduction compared to the normal range at the affected middle cerebral artery (MCA) territory. Regional cerebral vasoreactivity (rCVR) was measured by the acetazolamide loading test (17 mg/kg i.v.) compared to resting CBF, and diminished rCVR was classified as steal (less than 0%), disturbed (0–10%), and borderline (10–20%).
Surgical procedures
All patients underwent STA–MCA double anastomoses. Fronto-parieto-temporal craniotomy was performed on the affected side under general anesthesia. The donor artery was harvested by dissecting the STA. Recipient arteries were selected from the cortical branches of MCA. In general, we prepared two donor arteries (the frontal and parietal branches of the STA) and two recipient arteries (the supra- and infra-Sylvian cortical branches of MCA). A silicone stent (400 μm in diameter, 3 to 4 mm in length) that we designed was inserted into the recipient artery through an arteriotomy to facilitate anastomotic procedures [22].
Intraoperative examinations
Rectal temperature was maintained between 35 and 36°C using a heating pad, and room temperature was maintained at 23°C to stabilize the background cortical surface temperature. A portable infrared camera (TVS-100ME model; Nippon Avionics Inc., Tokyo, Japan) connected to a TV monitor was used for intraoperative thermographic studies [20]. The range of thermographic measurement was 0–50°C, temperature resolution was 0.1°C, and scan speed was 10 frames/s. The cortical surface temperatures at three regions of interests (ROIs) were simultaneously displayed in real time on the monitor. The data were recorded on a 3.5-in floppy disk and analyzed with a thermal measurement computer software (Nippon Avionics Inc., Tokyo, Japan). At the completion of the anastomotic surgical procedures, we determined the change in cortical surface temperature as a result of the STA–MCA bypass by performing thermographic studies before and after temporary occlusion (20–30 s) of the bypass without using an operating microscope to avoid the influence of heat from the microscope light. We used this method instead of measuring the temperatures before and after the surgical procedures because the pre-surgical cortical temperature would have been recorded a long time before completion of the bypass procedures and may not be appropriate as the baseline value due to temperature change during surgical procedures.
Intraoperative rCBF was measured using a laser Doppler flowmeter (ALF 21; Advance Co., Tokyo, Japan) at sites corresponding to the ROIs of thermographic measurement of cortical surface temperature, including at least two sites of the supra- and infra-Sylvian areas. The effect of STA–MCA bypass on rCBF was investigated by temporary occlusion of the bypass as performed in thermography. rCBF change from the baseline was expressed as a ratio (percentage). During the operation, arterial blood gases were monitored to ensure normocapnia and normoxia.
Sensory evoked potential (SEP) recording and motor evoked potential (MEP) recording were performed as a part of intraoperative monitoring.
Postoperative management
After surgery, patients were transferred directly to the CT room and cold Xe-CT was conducted to measure rCBF without discontinuing sedation with propofol, because systemic blood pressure is unstable and sometimes difficult to control strictly when patients come out of anesthesia. The rationale for continuing propofol sedation after STA–MCA bypass is that systemic blood pressure is more easily controlled and hyperperfusion more easily prevented under sedation compared to an awake state.
All patients were managed routinely under continuous sedation with propofol for several up to 24 h after STA–MCA bypass, the length of which depended on the postoperative rCBF measured on cold Xe-CT. Even if cold Xe-CT immediately after surgery demonstrated no hyperperfusion, mild sedation was continued for less than 12 h to prevent postoperative hypertension and hyperperfusion.
Postoperative management under continuous sedation facilitates strict blood pressure control (systolic blood pressure, 110–140 mmHg), using intravenous administration of antihypertensives if necessary. When postoperative hyperperfusion was detected, postoperative systemic arterial blood pressure was more stringently controlled to lower than 120 mmHg within the autoregulatory range.
When postoperative hyperperfusion was detected on cold Xe-CT immediately after STA–MCA bypass, rCBF was re-assessed on the next day and thereafter. CHS was defined as (1) clinical symptoms such as throbbing headache, ocular and facial pain, seizure, deterioration of consciousness level, and/or focal neurological deficits, and (2) hyperperfusion as defined below, without additional postoperative ischemic lesions on CT scans or MR images [3, 4, 17, 30, 34].
CT images were obtained on the first and second postoperative days, and MR images were acquired on the seventh postoperative day in all patients to detect additional lesions. Patency of the bypasses was evaluated by MR angiography or conventional angiography.
The study protocol (including Xe-CT) was approved by the institutional review board of our university.
Definition of cerebral hyperperfusion
Although hyperperfusion was most commonly defined as greater than 100% increase over baseline (preoperative values) in previous reports on CEA, we were not able to directly compare the postoperative rCBF with preoperative values because postoperative rCBF was measured under sedation. Therefore, we evaluated and defined postoperative hyperperfusion by a method as used in our previous report [10]: greater than 100% increase in corrected postoperative ipsilateral rCBF over preoperative value. The corrected postoperative ipsilateral rCBF was calculated based on the percent reduction in postoperative (under sedation with propofol) rCBF relative to preoperative (awake state) value on the contralateral (non-operative) side (corrected postoperative ipsilateral rCBF = postoperative ipsilateral rCBF × preoperative contralateral rCBF/postoperative contralateral rCBF). This method, using the ratio relative to the contralateral side, should be appropriate because administration of propofol decreases CBF [24, 27, 32] and rCBF [9] with no significant difference in reduction in various cerebral locations [11, 25], and propofol reduces human CBF regardless of the differences in patient background [24].
Statistical analysis
Data are expressed as mean ± standard deviation. The relationship among increased postoperative rCBF, intraoperative thermographic changes, and intraoperative rCBF changes measured by laser Doppler flowmetry were analyzed by Pearson’s correlation coefficient and simple regression. Unpaired Student’s t-test was used to determine the statistical significance of the differences in intraoperative thermographic changes or intraoperative rCBF changes measured by laser Doppler flowmetry between patients with and without postoperative hyperperfusion or CHS. A p value less than 0.05 was considered statistically significant.
Results
Table 1 demonstrates the data of individual patients.
Postoperative outcomes
All patients in this series demonstrated patency of the bypasses after surgery. Postoperative hyperperfusion on Xe-CT immediately after surgery defined as greater than 100% increase over preoperative rCBF using corrected values was observed in two patients (two surgeries). In the two patients, preoperative rCVR was normal in one and disturbed in another.
Postoperative CHS was observed in the subacute stage between 1 and 2 weeks after surgery in two patients (7.4%), who developed intracerebral hemorrhage. These two patients had transient motor aphasia, and they were the same two patients who showed greater than 100% increase immediately after surgery.
No other patients developed additional neurological deficits related to postoperative cerebrovascular complications. Furthermore, we never encountered complications related to postoperative propofol sedation, which could include systemic complications such as cardiovascular events and renal failure.
Relationship between intraoperative laser Doppler flowmetry results and postoperative rCBF
A significant correlation was obtained between intraoperative rCBF changes as a ratio measured by a laser Doppler flowmeter following temporary occlusion of the anastomosed STA and postoperative rCBF increase (Pearson’s method: r = 0.555, p = 0.0003; simple regression: Y = 1.22X + 3.289, r 2 = 0.308, p = 0.0004) (Fig. 1). According to the data of the two patients who developed postoperative hyperperfusion, approximately 45% changes in rCBF measured by laser Doppler flowmetry may predict a risk of postoperative hyperperfusion.
A significant correlation is demonstrated between intraoperative rCBF changes measured by a laser Doppler flowmeter following temporary occlusion of the anastomosed STA and postoperative rCBF increase (Pearson’s method: r = 0.555, p = 0.0003; simple regression: Y = 1.22X + 3.289, r 2 = 0.308, p = 0.0004)
Comparative analysis of laser Doppler flowmetry results and postoperative hyperperfusion
Intraoperative rCBF changes measured by laser Doppler flowmetry were significantly greater in patients with postoperative hyperperfusion (>100%: p = 0.0193) and with CHS (p = 0.0193), compared to the patients without these findings.
Relationship between intraoperative thermography and postoperative rCBF
No significant correlation was observed between intraoperative thermographic changes following temporary occlusion of the anastomosed STA and postoperative rCBF increase.
Representative case (patient no. 12)
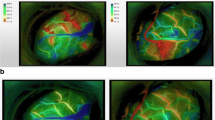
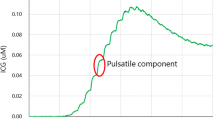
The clinical findings of a representative case of a 40-year-old woman with frequent transient motor weakness in upper and lower extremities mainly on the right side are summarized in Fig. 2. Carotid angiograms demonstrated typical findings of moyamoya disease. Preoperative cold Xe-CT studies showed bilateral reduction of resting rCBF (30% reduction compared to normal values on the right side and 42.5% reduction on the left side) (Fig. 2a) and rCVR (−11.5% on the right side and 5.8% on the left side) (Fig. 2b). First, the patient underwent left STA–MCA double bypass. Temporary occlusion of the STA–MCA bypass during surgery produced heterogeneous changes in cortical surface temperature, with a maximum change of 1.0°C (Fig. 2 d, e). Intraoperative measurements of rCBF by a laser Doppler flowmeter following temporary occlusion of the bypass also demonstrated definitive changes in rCBF (45.5% from the baseline) at the sites where the brain surface temperature changed drastically (Fig. 2f). Cold Xe-CT conducted under sedation immediately after surgery demonstrated 149.3% increase in resting rCBF (Fig. 2c). The patient developed intracerebral hemorrhage on postoperative day 12 (Fig. 2 g). She had transient motor aphasia.
A 40-year-old woman with moyamoya disease underwent left STA–MCA double bypass. Preoperative cold Xe-CT images before (a) and after (b) acetazolamide loading show reduced resting rCBF and vascular reactivity, respectively. Postoperative cold Xe-CT image (c) shows an increase in rCBF in the left MCA territory by 149% (white arrow), indicating hyperperfusion. Thermographic monitoring of the operative field shows brain surface temperature before occlusion of the anastomosed STA (d) and during occlusion of the anastomosed STA (e). Thermograms demonstrate heterogeneous changes (1.0°C at the maximum) in cortical surface temperatures by temporary occlusion of the anastomosed STA. A laser Doppler flowmeter record demonstrates change of rCBF (45.5% from baseline) during (between the two arrows) occlusion of the anastomosed STA (f). A postoperative CT scan 12 days after surgery demonstrates intracerebral hemorrhage (g)
Discussion
Hyperperfusion following cerebral revascularization is well recognized, particularly in the context of CEA [2, 7, 8, 10, 13, 18, 26, 28, 31]. Although the occurrence of hyperperfusion after EC–IC bypass is not surprising, there are few detailed studies of CHS or hyperperfusion based on CBF measurement after EC–IC bypass [34]. Some investigators have reported hyperperfusion based on rCBF after EC–IC bypass, especially in adults with moyamoya disease [3, 4, 17, 19, 30]. Transient postoperative neurological deficits caused by focal cerebral hyperperfusion are not rare in adult-onset moyamoya disease [3, 4, 17, 19, 30]. On the contrary, hyperperfusion after revascularization in children with moyamoya disease was rarely documented. Although clinical symptoms are transient with good prognosis [3, 19], hyperperfusion following EC–IC bypass occasionally results in cerebral hemorrhage [4]. Postoperative CHS and/or intracerebral hemorrhages, which may have devastating consequence, can complicate EC–IC bypasses for carotid occlusive disease, but evidence for hyperperfusion as the underlying cause of these hemorrhages is not well established. For moyamoya diseases, although most of the reports are case presentations, a few investigations on postoperative hyperperfusion have been conducted on small series of patients [3, 19]. Understanding the pathogenesis of postoperative hyperperfusion is important to establish safe and effective EC–IC bypass. Prediction of postoperative hyperperfusion, if feasible, will be a very effective method to minimize postoperative complications [34].
The present study suggests that laser Doppler flowmetry is useful to monitor changes in cortical circulation attributed to the EC–IC bypass. Consequently, we have shown that intraoperative rCBF measurement using laser Doppler flowmetry may predict a risk of postoperative cerebral hyperperfusion in moyamoya disease. In order to prevent postoperative hyperperfusion, an important postoperative management is to control the patient’s blood pressure appropriately even just after the bypass surgery, based on the intraoperative laser Doppler flowmetry findings. Furthermore, we would consider changing the surgery plan to single bypass instead of double bypass in case hyperperfusion is detected by intraoperative laser Doppler study. The intraoperative laser Doppler findings may predict quantitative circulatory changes following bypass surgery.
On the contrary, in the present study, intraoperative thermographic changes did not correlate significantly with postoperative rCBF immediately after STA–MCA bypass, although there is a significant correlation between intraoperative thermal changes and rCBF changes determined by laser Doppler flowmetry. One of the reasons for these findings is a very small change in brain surface temperature, a maximum change of less than 2°C, in the present study. Technical error has to be reduced to a minimum to demonstrate a statistically significant result within the narrow range of thermal changes. A second possible reason is that room temperature was not as stable as the condition in our previous study [20]. Constant room temperature has to be maintained to keep the background cortical surface temperature as stable as possible, as mentioned in the “Patients and methods” section. It is possible that the room temperature was unexpectedly changeable and the air conditioner might have generated a soft wind in some surgeries.
We measured postoperative rCBF immediately after EC–IC bypass under sedation in the present study. Although no patients developed additional neurological complications except for the two patients with transient symptoms caused by CHS, prolonged sedation may carry the risk of missing a newly developed postoperative neurological deficit. According to the present protocol, patients without postoperative hyperperfusion were also put on mild sedation for less than 12 h. CT images were obtained on the first and the second postoperative day. However, it is not easy to detect newly developed neurological deficits promptly because of mild continuous sedation. This is a possible shortcoming of postoperative continuous sedation.
Although postoperative hyperperfusion was most commonly defined as greater than 100% increase over baseline (preoperative values) in previous reports on CEA, few reports provide a definition for EC–IC bypass [3, 19]. A large scale study is required to examine the incidence of postoperative hyperperfusion on rCBF following EC–IC bypass. We obtained identical results for postoperative rCBF in the analyses by intraoperative laser Doppler flowmetry and thermography, which may indicate that the evaluation methods used in the present study are valid and adequate.
Conclusions
Intraoperative changes of rCBF determined by laser Doppler flowmetry correlate significantly with postoperative rCBF measured on Xe-CT immediately after STA–MCA bypass in patients with moyamoya disease. Furthermore, the rCBF changes measured by laser Doppler flowmetry were significantly greater in patients with postoperative hyperperfusion. Intraoperative measurement of rCBF change using laser Doppler flowmetry may be useful in predicting a risk of post-EC–IC bypass cerebral hyperperfusion in moyamoya disease.
References
Amin-Hanjani S, Du X, Mlinarevich N, Meglio G, Zhao M, Charbel FT (2005) The cut flow index: an intraoperative predictor of the success of extracranial–intracranial bypass for occlusive cerebrovascular disease. Neurosurgery 56(1 Suppl):75–85
Fujimoto S, Toyoda K, Inoue T, Hirai Y, Uwatoko T, Kishikawa K, Yasumori K, Ibayashi S, Iida FM, Okada Y (2004) Diagnostic impact of transcranial color-coded real-time sonography with echo contrast agents for hyperperfusion syndrome after carotid endarterectomy. Stroke 35:1852–1856
Fujimura M, Mugikura S, Kaneta T, Shimizu H, Tominaga T (2009) Incidence and risk factors for symptomatic cerebral hyperperfusion after superficial temporal artery–middle cerebral artery anastomosis in patients with moyamoya disease. Surg Neurol 71:442–447
Fujimura M, Shimizu H, Mugikura S, Tominaga T (2009) Delayed intracerebral hemorrhage after superficial temporal artery–middle cerebral artery anastomosis in a patient with moyamoya disease: possible involvement of cerebral hyperperfusion and increased vascular permeability. Surg Neurol 71:223–227
Horowitz M, Yonas H, Albright AL (1995) Evaluation of cerebral blood flow and hemodynamic reserve in symptomatic moyamoya disease using stable Xenon-CT blood flow. Surg Neurol 44:251–262
Hoshino T, Katayama Y, Sakatani K, Kano T, Murata Y (2006) Intraoperative monitoring of cerebral blood oxygenation and hemodynamics during extracranial–intracranial bypass surgery by a newly developed visible light spectroscopy system. Surg Neurol 65:569–576
Hosoda K, Kawaguchi T, Shibata Y, Kamei M, Kidoguchi K, Koyama J, Fujita S, Tamaki N (2001) Cerebral vasoreactivity and internal carotid artery flow help to identify patients at risk for hyperperfusion after carotid endarterectomy. Stroke 32:1567–1573
Jorgensen L, Schroeder T (1993) Defective cerebrovascular autoregulation after carotid endarterectomy. Eur J Vasc Surg 7:370–379
Kaisti K, Metsahonkala L, Teras M, Oikonen V, Aalto S, Jaaskelainen S, Hinkka S, Scheinin H (2002) Effects of surgical levels of propofol and sevoflurane anesthesia on cerebral blood flow in healthy subjects studied with positron emission tomography. Anesthesiology 96:1358–1370
Kawamata T, Okada Y, Kawashima A, Yoneyama T, Yamaguchi K, Ono Y, Hori T (2009) Post-carotid endarterectomy cerebral hyperperfusion can be prevented by minimizing intraoperative cerebral ischemia and strict postoperative blood pressure control under continuous sedation. Neurosurgery 64:447–454
Kimme P, Ledin T, Sjoberg F (2007) Dose effect of sevoflurane and isoflurane anesthetics on cortical blood flow during controlled hypotension in the pig. Acta Anaesthesiol Scand 51:607–613
Koga H, Mori K, Ono H, Kuwahara M, Matsuse E (1987) Intraoperative regional thermography during surgery for brain tumor. Neurol Med Chir (Tokyo) 27:1033–1038
Komoribayashi N, Ogasawara K, Kobayashi M, Saitoh H, Terasaki K, Inoue T, Ogawa A (2006) Cerebral hyperperfusion after carotid endarterectomy is associated with preoperative hemodynamic impairment and intraoperative cerebral ischemia. J Cereb Blood Flow Metab 26:878–884
Kondo S, Okada Y, Iseki H, Hori T, Takakura K, Kobayashi A, Nagata H (2000) Thermological study of drilling bone tissue with a high-speed drill. Neurosurgery 46:1162–1168
Nambu K, Suzuki R, Hirakawa K (1995) Cerebral blood flow: measurement with xenon-enhanced dynamic helical CT. Radiology 195:53–57
Nussbaum ES, Erickson DL (2000) Extracranial–intracranial bypass for ischemic cerebrovascular disease refractory to maximal medical therapy. Neurosurgery 46:37–43
Ogasawara K, Komoribayashi N, Kobayashi M, Fukuda T, Inoue T, Yamadate K, Ogawa (2005) A neural damage caused by cerebral hyperperfusion after arterial bypass surgery in a patient with moyamoya disease:case report. Neurosurgery 56:E1380
Ogasawara K, Yukawa H, Kobayashi M, Mikami C, Konno H, Terasaki K, Inoue T, Ogawa A (2003) Prediction and monitoring of cerebral hyperperfusion after carotid endarterectomy by using single-photon emission computerized tomography scanning. J Neurosurg 99:504–510
Ohue S, Kumon Y, Kohno K, Watanabe H, Iwata S, Ohnishi T (2008) Postoperative temporary neurological deficits in adults with moyamoya disease. Surg Neurol 69:281–287
Okada Y, Kawamata T, Kawashima A, Hori T (2007) Intraoperative application of thermography in extracranial-intracranial bypass. Neurosurgery 60(4 Suppl 2):362–365
Okada Y, Shima T, Nishida M, Yamane K, Yamada T, Yamanaka C (1998) Effectiveness of superficial temporal artery–middle cerebral artery anastomosis in adult moyamoya disease: cerebral hemodynamics and clinical course in ischemic and hemorrhagic varieties. Stroke 29:625–630
Okada Y, Shima T, Yamane K, Yamanaka C, Kagawa R (1999) Cylindrical or T-shaped silicone rubber stents for microanastomosis —technical note. Neurol Med Chir (Tokyo) 39:55–58
Okudera H, Kobayashi S, Toriyama T (1994) Intraoperative regional and functional thermography during resection of cerebral arteriovenous malformation. Neurosurgery 34:1065–1067
Oshima T, Karasawa F, Satoh T (2002) Effects of propofol on cerebral blood flow and the metabolic rate of oxygen in humans. Acta Anaesthesiol Scand 46:831–835
Ouchi T, Ochiai R, Takeda J, Tsukada H, Kakiuchi T (2006) Combined effects of propofol and mild hypothermia on cerebral metabolism and blood flow in rhesus monkey: a positron emission tomography study. J Anesth 20:208–214
Piepgras D, Morgan M, Sundt TJ, Yanagihara T, Mussman L (1988) Intracerebral hemorrhage after carotid endarterectomy. J Neurosurg 68:532–536
Pinaud M, Lelausque J, Chetanneau A, Fauchoux N, Menegalli D, Souron R (1990) Effects of propofol on cerebral hemodynamics and metabolism in patients with brain trauma. Anesthesiology 73:404–409
Sundt TJ, Sharbrough F, Piepgras D, Kearns T, Messick JJ, O'Fallon W (1981) Correlation of cerebral blood flow and electroencephalographic changes during carotid endarterectomy: with results of surgery and hemodynamics of cerebral ischemia. Mayo Clin Proc 56:533–543
The EC/IC Bypass Study Group (1985) Failure of extracranial-intracranial arterial bypass to reduce the risk of ischemic stroke. Results of an international randomized trial. N Engl J Med 313:1191–1200
Uno M, Nakajima N, Nishi K, Shinno K, Nagahiro S (1998) Hyperperfusion syndrome after extracranial–intracranial bypass in a patient with moyamoya disease. Case report. Neuro Med Chir (Tokyo) 38:420–424
van Mook W, Rennenberg R, Schurink G, van Oostenbrugge R, Mess W, Hofman P, de Leeuw P (2005) Cerebral hyperperfusion syndrome. Lancet Neurol 4:877–888
Vandesteene A, Trempont V, Engelman E, Deloof T, Focroul M, Schoutens A, de Rood M (1988) Effect of propofol on cerebral blood flow and metabolism in man. Anaesthesia 43(Suppl):42–43
Woitzik J, Horn P, Vajkoczy P, Schmiedek P (2005) Intraoperative control of extracranial–intracranial bypass patency by near-infrared indocyanine green videoangiography. J Neurosurg 102:692–698
Yamaguchi K, Kawamata T, Kawashima A, Hori T, Okada Y (2010) Incidence and predictive factors of cerebral hyperperfusion after extracranial–intracranial bypass for occlusive cerebrovascular diseases. Neurosurgery 67:1548–1554
Yonas H, Wolfson SK Jr, Gur D, Latchaw RE, Good WF, Leanza R, Jackson DL, Jannetta PJ, Reinmuth OM (1984) Clinical experience with the use of xenon-enhanced CT blood flow mapping in cerebral vascular disease. Stroke 15:443–450
Acknowledgement
This study was supported by research funds of Department of Neurosurgery, Tokyo Women’s Medical University.
Disclosure
The authors have no personal financial or institutional interest in any of the drugs, materials, or devices described in this article
Conflict of Interest
All authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Yasuhiro Yonekawa, Zurich, Switzerland
Kawamata et al. have shown an elegant method to predict a risk of postoperative hyperperfusion after STA-MCA bypassfor Moyamoya disease by using laser Dopplerflowmetryintraoperatively at the time of EC-IC bypass surgery, what could be confirmed by postoperative Xe-CT scan-|rCBF measurement. This simple method would contribute to the management of postoperative hyperperfusion, although its occurrence has been reported to be rather infrequent but can be associated with considerable sequelae.
By the way, it was my little surprise that intraoperative thermography change did not correlate with postoperative rCBF change. This might be due to inherent methodological problems namelythis can be delicately influenced byroom temperature and/or heat produced by the light of operating microscope. I would be interested for the purpose to use the Peltier stack which would have less above mentioned problems and its usefulnessfulness was very impressive at the time of parent artery temporary occlusionalong with simplicity of its handling in aneurysm surgery (1).
References
1. Ogata N, Fournier JY, Imhof HG, Yonekawa Y: Thermal diffusion blood flow monitoring during aneurysm surgery. ActaNeurochir (Wien) 138: 726–731, 1996
Rights and permissions
About this article
Cite this article
Kawamata, T., Kawashima, A., Yamaguchi, K. et al. Usefulness of intraoperative laser Doppler flowmetry and thermography to predict a risk of postoperative hyperperfusion after superficial temporal artery–middle cerebral artery bypass for moyamoya disease. Neurosurg Rev 34, 355–362 (2011). https://doi.org/10.1007/s10143-011-0331-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-011-0331-8