Abstract
The aim of this study was to evaluate the process of bone repair in rats submitted to low-level laser therapy using optical densitometry. A total of 45 rats which underwent femoral osteotomy were randomly distributed into three groups: control (group I) and laser-treated groups using wavelengths in the red (λ, 660–690 nm) and in the infrared (λ, 790–830 nm) spectra (group II and group III, respectively). The animals (five per group) were killed after 7, 14, and 21 days and the femurs were removed for optical densitometry analysis. Optical density showed a significant increase in the degree of mineralization (gray level) in both groups treated with the laser after 7 days. After 14 days, only the group treated with laser therapy in the infrared spectrum showed higher bone density. No differences were observed between groups after 21 days. Such results suggest the positive effect of low-level laser therapy in bone repair is time- and wavelength-dependent. In addition, our results have confirmed that optical densitometry technique can measure bone mineralization status.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Therapeutic strategies to promote bone repair represent a major challenge to many health professionals. In order to reduce the functional incapacity and the high socioeconomic costs associated with the bone fractures, several interventions have been investigated on bone healing process, and these include the use of low-level laser therapy (LLLT) [1–5].
The LLLT involves the application of monochromatic and coherent light with low energy density that promotes nonthermal photochemistry effects on cellular level [6, 7]. It has been described that LLLT may accelerate the healing of bone defects in vivo and in vitro investigations [1]. It was showed that osteoblastic activity increased [3, 8–10], vascularization [11, 12], organization of collagen fibers [1, 13], and mitochondrial and intracellular adenosine triphosphate level changes [14]. However, others studies did not find any effect of LLLT on the repair of mineralized tissues [15, 16].
The conflicting results of LLLT on biomodulation are mainly related to diversity of the radiation parameters: treatment dose, power density, number of applications, method of applying, and wavelength [17]. For the last parameter, it has been postulated that tissue response is altered in function of wavelength used. The radiation emitted in the red region shows activation of mitochondrial respiratory chain components resulting in the initiation of a cascade of cellular reactions, whereas in the region of the infrared (invisible light), there is activation of chromophores located in cellular membrane [18]. Currently, studies involving the analysis of bone repair after LLLT have used the following methodologies: optic microscopy [1, 19], morphometry [20], immunohistochemistry [21], and biomechanic tests [22].
However, limited attention has been attributed to X-rays, which are an important tool in clinical analysis for mineralized tissues because it is noninvasive and can be easily performed. Optical densitometry has been demonstrated by some authors as a tool for the study of bone mineralization and emerges as an important complement to traditional methods for quantification and imaging of local variations of mineral bone [2, 8, 23].
The aim of this study was to evaluate, by optical densitometry, the process of bone repair of surgical defects (osteotomy) in the femur of rats submitted to LLLT, using wavelengths in the red (λ, 660–690 nm) and infrared (λ, 790–830 nm) spectra at 7, 14, and 21 days.
Material and methods
Animals and experimental design
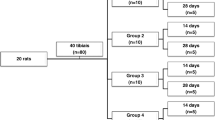
All experiments were carried out in accordance with Research Ethic Committee at the Universidade do Vale do Paraíba, Brazil (UNIVAP) for animal care (L213/2005). The experiments were performed using males Wistar rats (240–260 g), supplied with food and water ad libitum provided by the Central Animal House of the Research and Development Department (IP&D, UNIVAP). The rats were placed in appropriate cages (22 ± 3 °C, relative humidity 60 ± 10 %, 12 h:12 h light/darkness) and randomly divided into experimental groups with five animals per group.
All animals were subjected to a surgical procedure to create a bone defect (osteotomy). The 45 animals selected for the study were separated into three groups constituted by nine animals in each according to the experimental time: 7, 14, and 21 days. In group I, control group simulations of LLLT were performed after osteotomy; the animals of group II were treated with red laser (λ, 660–690 nm); and the laser therapy was performed with infrared laser (λ, 790–830 nm) in the animals of group III.
Surgical procedure
The animals received intramuscular administration of preanesthetic acepromazine (0.02 ml/kg) and butorphanol (0.01 ml/kg) followed by the anesthetic Zoletil (1.0 ml/kg). Following which, the right femoral region of the animals was shaved and a topical solution povidone-iodine was applied. Using a scalpel, an incision of approximately 3 cm was made on the skin surface and on the muscle. The osteotomy was performed by a trephine drill of 2.8 mm in diameter, connected to a low-speed electric motor (DENTEC 405 N®), with a speed of 1,100 rpm and frequency of 0.25 s, under constant irrigation of saline throughout surgery. The muscle and subcutaneous tissue were sutured using catgut 4.0 (Cirumédica, Cotia, SP, Brazil), and the skin was sutured with silk 4.0 (Ethicon, Johnson & Johnson, São José dos Campos, SP, Brazil).
Laser therapy
The LLLT was applied immediately after osteotomy and was repeated every 48 h on the same days, three times a week, during the experimental times of 7 days (three sessions), 14 days (six sessions), and 21 days (nine sessions). The equipment used in the study was a laser, Flash Laser III (DMC Equipamentos Ltda, São Carlos, SP), which operates in two wavelengths, between 660 and 690 nm (red laser, mid-activity: InGaAlP) used for the group II and between 790 and 830 nm (infrared laser, mid-activity: GaAlAs) applied to group III. The LLLT was applied directly on the injury, with the hand piece at a 90° angle, perpendicularly positioned on the wound, using punctual technique according the protocol described in Table 1. Except for the different wavelengths of LLLT used, other parameters were identical to the groups II and III.
Optical densitometry
In order to evaluate the bone repair in the fractured areas, the bone optical density was measured by using a charge-coupled device (CCD) sensor (Dentsply International, Gendex Dental System model Visualix GX-S-HDI™ digital sensor) for collecting digital X-ray radiographs [24]. First, CCD and bone samples were stabilized in a standardized fixing device. Afterwards, each femur was radiographed with X-ray equipment (Spectro-70X™; Dabi Atlante, São Paulo, SP, Brazil) at 7 mA and 65 kVp, with 0.032 s exposure time and 40 cm focus-object distance. Radiographic images of the specimens were obtained and the digital images acquired were transferred to the computer. The bone optical density was evaluated using the VixWin™ 1.4 system (Gendex Dental System, Chicago, IL, USA). Image resolution was 635 ppi, the image size was 900 × 641 dpi, and the pixel size was 40 mm. Images were stored in TIFF format, without compression, with resolution of 600 dpi. To obtain optical density values, the images were imported into Adobe Photoshop v.6.0 (Adobe Systems, San Jose, CA, USA). Afterward, by using the histogram command, we obtained density mean values and standard deviation of areas corresponding to osteotomy-treated and control defects, which served as a parameter for calculating the difference in optical density. All the results were expressed in 8-bit gray levels (0–255). Increased bone density was indicated by higher values in gray scale [25].
Statistical analysis
The values obtained from optical densitometry analysis were analyzed using SPSS software version 11.0 (SPSS Inc, Chicago, IL, USA). The results are presented as means and standard deviations. The one-way ANOVA and the Tukey–Kramer multiple comparison pos hoc test were applied to check the significance of the mean value of gray level between the experimental groups at 7, 14, and 21 days. Differences were considered significant with the level of significance set at 5 % (p < 0.05).
Results
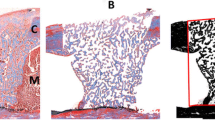
Figure 1 showed differences on the bone repair in the femoral fractured areas (white arrows) between the LLLT (II and III) and control (I) groups over the 7th, 14th, and 22nd day. After 7 days, LLLT groups (II and III) had greater (p < 0.05) mean optical density when compared with untreated group (I), with 3.4 and 5.8 %, respectively. There were no significant differences between the two treated groups in this experimental time (p > 0.05) (Fig. 2).
X-ray images of right femur osteotomies treated by laser energy. Group I (control), group II (laser therapy in the red spectrum, 660 nm), and group III (laser therapy in the infrared spectrum, 830 nm) at 7, 14, and 21 days posttreatment. Bone growth in the fractured areas is indicated by the white arrows. Note the differences in groups treated versus control group and the evolution of bone formation
Means and standard deviations of new bone formation obtained by optical density analysis in group I (control), group II (laser therapy in the red spectrum, 660 nm), and group III (laser therapy in the infrared spectrum, 830 nm) at seventh days. *p<0.01 indicates significant difference from the control group
At the 14th postoperative day, group III had the greatest mean optical density (142 ± 2.0 gray level). These values were significantly different (3 % higher; p < 0.05) only compared with group I (136 ± 2.4 gray level). In addition, the mean optical density obtained by laser therapy in the red spectrum (group II, 140 ± 1.8 gray level) was not different (p > 0.05) from other groups (Fig. 3). The mean optical density at the 21st postoperative day showed no significant difference among the experimental groups (p > 0.05) (Fig. 4).
Means and standard deviations of new bone formation obtained by optical density analysis in group I (control), group II (laser therapy in the red spectrum, 660 nm), and group III (laser therapy in the infrared spectrum, 830 nm) at 14th days. *p<0.01 indicates significant difference from the control group
Discussion
Several studies have compared the effect of laser irradiation on bone repair. However, there are conflicting results of LLLT on biomodulation mainly related to the diversity of the radiation parameters, also the selection of wavelength used. Thus, the purpose of this study was to evaluate by optical densitometry the repair process of bone defect (osteotomy) in rat femurs submitted to laser therapy in the red (660–690 nm) and infrared (790–830 nm) spectra, in experimental periods of 7, 14, and 21 days.
In this study, it was observed that the bone defects submitted to laser therapy in the infrared spectrum (group III) had a more advanced repair compared to group I (control) in 7 and 14 days, suggesting that the LLLT in infrared spectrum accelerates the bone repair process. These results are in accordance with those by Gerbi et al. [26] that evaluated the LLLT in femoral bone defects in rats, using laser AsGaAl (830 nm), 40 mW, continuous, spot size ~0.6 mm, and 16 J/cm2 of density energy. The histological analysis of the specimens showed an increase of collagen fibers at the initial stages of bone repair (15 days) and higher trabecular bone organization after 30 days. Khadra et al. [27] investigated the effects of LLLT (AsGaAl—830 nm in ten consecutive days) on titanium implant healing and attachment in bone. The mechanical strength, histomorphometrical analysis, and energy-dispersive X-ray microanalysis determined positive effects on bone repair process. Coombe et al. [28] evaluated the effects of LLLT (AsGaAl—830 nm, 90 mW, energy density of 1.7 to 25.1 J/cm2) daily for 10 days in human osteoblastic cells. The results showed an increase in intracellular calcium, which indicates a positive response of these cells. Thus, LLLT in the infrared spectrum determined a biomodulator effect in the bone repair process.
LLLT in red spectrum (group II) only promoted higher optical density when compared to control group at 7 days of experimental time. It has been described that the LLLT in red spectrum, when administered in initial stages of bone inflammatory response accelerate the repair process. Trelles and Mayayo [12] showed better bone repair when He–Ne laser was used in first days after bone surgery. Others authors reported that the LLLT in red spectrum increased the osteoblast number and levels of alkaline phosphatase [14], showed better results in biomechanics tests for the mature bone callus formation [27], as well as promoted a proliferation and maturation of human osteoblast, in vitro, with increase in alkaline phosphatase levels, osteopontin, and bone sialoprotein [10].
It is observed in the literature that the biomodulatory effects of LLLT depend upon the wavelength used [20]. In this study, our results demonstrate a significant increase in the quantity of mineralization status in both laser-treated groups at the 7 days of experimental time. After 14 days, only the group treated with laser therapy in the infrared spectrum showed higher bone density. Such results suggest that positive effect of LLLT in bone repair was wavelength- and time-dependent. In fact, the modulatory effects of LLLT on bone repair have been suggested as better indication for the infrared laser [20]. As the wavelength defines the depth of the laser penetration in the target tissue, the red light shows lesser penetration capacity when compared with infrared laser [29]. This phenomenon can be related to the fact that lower wavelengths are less resistant to the dispersion and do not penetrate deeply into the tissues because of their high coefficient of skin absorption. The red light (632.8 nm) penetrates 0.5–1.0 mm before losing 37 % of its intensity, while the infrared wavelengths penetrate 2 mm before losing the same percentage of energy [29]. Independently of the bone repair time, the efficiency of both wavelengths used could having a relationship to the capacity to (a) promote endothelial cells proliferation, vascular neoformation, proportioned blood irrigation, and nutrition to local cells and tissues [11, 12]; (b) stimulate the fibroblast proliferation, synthesis, and organization of collagen fibers [13]; (c) stimulate osteoprogenitor cells, osteoblasts, and osteoclasts, contributed to bone repair process [9, 10]; and (d) increase of DNA and RNA synthesis, indicated effects on cell growth and protein synthesis [14].
Although, not being statistically significant, the results of the measurements of the optical densitometry of bone repair after 21 days show higher gray level in treated groups than control group. Moreover, it is probable that 3 weeks was a sufficient time to produce the biological bone repair independently of LLLT [30], which justify the small differences between the groups.
Different methods have been used to evaluate the bone repair [1, 19–22]. Digital radiographic examination is an important tool in clinical analysis for mineralized tissues and some studies indicated that this technique could offer quantification and imaging of local variations of bone repair by optical densitometry. Taba et al. [23] studied a radiographic method to assess the bone density based on gray level of digitalized radiographic image (optical densitometry) and conclude that this method shows similar results when compared with the histological findings with the characterization of bone mineral compound being related. Using this methodology, Liu et al. [2] assessed the effects of LLLT (λ, 830 nm) on tibial fractures in rabbits and showed higher bone density after light therapy. In addition, Nascimento et al. [8] evaluated the bone repair in osteopenic rats after LLLT associated with calcitonin administration and observed an increase of optical density with this therapeutic regimen.
In summary, the results suggest that LLLT accelerates bone repair in the initial phase independent of the wavelength used, and this effect remains for 14 days when using infrared laser. Therefore, it is concluded that LLLT induced a biomodulatory positive effect on the healing process of bone defects, which was time- and wavelength-dependent. In addition, our results have confirmed that optical densitometry technique can measure bone mineralization status.
References
Pinheiro ALB, Gerbi MEM, Limeira Júnior FA, Ponzi EAC, Marques AMC, Carvalho CM, Santos RC, Oliveira PC, Nóia M, Ramalho LMP (2009) Bone repair following bone grafting hydroxyapatite guided bone regeneration and infra-red laser photobiomodulation: a histological study in a rodent model. Lasers Med Sci 24:234–240. doi:10.1007/s10103-008-0556-0
Liu X, Lyon R, Meier HT, Thometz J, Haworth ST (2007) Effect of lower-level laser therapy on rabbit tibial fracture. Photomed Laser Surg 25(6):487–494. doi:10.1089/pho.2006.2075
Lopes CB, Pinheiro AL, Sathaiah S, Da Silva NS, Salgado MA (2007) Infrared laser photobiomodulation (lambda 830 nm) on bone tissue around dental implants: a Raman spectroscopy and scanning electronic microscopy study in rabbits. Photomed Laser Surg 25(2):96–101. doi:10.1089/pho.2006.2030
Lirani-Galvão AP, Jorgetti V, Da Silva OP (2006) Comparative study of how low-level laser therapy and low-intensity pulsed ultrasound affect bone repair in rats. Photomed Laser Surg 24(6):735–740. doi:10.1089/pho.2006.24.735
Nicolau RA, Jorgetti V, Rigau J, Pacheco MT, Dos Reis LM, Zângaro RA (2003) Effect of low-power GaAlAs laser (660 nm) on bone structure and cell activity: an experimental animal study. Lasers Med Sci 18:89–94. doi:10.1007/s10103-003-0260-z
Karu T (1987) Phobobiological fundamental of low power laser therapy. IEEE J Quantum Electron 23:1703
Schindl A, Schindl M, Pernerstorfer-Schön H, Schindl L (2000) Low-intensity laser therapy: a review. J Investig Med 48(5):312–326
Nascimento SB, Cardoso CA, Ribeiro TP, Almeida JD, Albertini R, Munin E, Arisawa EA (2010) Effect of low-level laser therapy and calcitonin on bone repair in castrated rats: a densitometric study. Photomed Laser Surg 28(1):45–49. doi:10.1089/pho.2008.2396
Da Silva RV, Camilli JA (2006) Repair of bone defects treated with autogenous bone graft and low-power laser. J Craniofac Surg 17:297–301
Freitas IGF, Baranauskas V, Cruz-Höfling MA (2000) Laser effects on osteogenesis. Appl Surf Sci 154–155:548–554
Schlager A, Kronberger P, Petschke F, Ulmer H (2000) Low-power laser light in the healing of burns: a comparison between two different wavelengths (635 nm and 690 nm) and a placebo group. Lasers Surg Med 27:39–42. doi:10.1002/1096-9101(2000) 27:1<39::AID-LSM5>3.0.CO;2-4
Trelles MA, Mayayo E (1987) Bone fracture consolidates faster with low-power laser. Lasers Surg Med 7(1):36–45
Garavello-Freitas I, Baranauskas V, Joazeiro P, Padovani CR, Dal Pai-Silva M, Da Cruz-Hofling MA (2003) Low-power laser irradiation improves histomorphometrical parameters and bone matrix organization during tibia wound healing in rats. J Photochem B Photobiol 70:81–89. doi:10.1016/S1011-1344(03)00058-7
Karu TI, Pyatibrat L, Kalendo G (1995) Irradiation with He-Ne laser increases ATP level in cells cultivated in vitro. J Photochem B Photobiol 27:219–223. doi:10.1016/1011-1344(94)07078-3
Luger EJ, Rochkind S, Wollman Y, Kogan G, Dekel S (1998) Effect of low power laser irradiation on the mechanical properties of bone fracture healing in rats. Lasers Surg Med 22:97–102. doi:10.1002/(SICI)1096-9101(1998) 22:2<97::AID-LSM5>3.0.CO;2-R
Anneroth G, Hall G, Rydén H, Zetterqvist L (1988) The effect of low-energy infrared laser radiation on wound healing in rats. Br J Oral Maxillofac Surg 26:12–17
Pinheiro AL, Gerbi ME (2006) Photoengineering of bone repair processes. Photomed Laser Surg 24(2):169–178. doi:10.1089/pho.2006.24.169
Vladimirov YA, Osipov AN, Klebanov GI (2004) Photobiological principles of therapeutic applications of laser radiation. Biochemistry 69(1):81–90
Martínez MEM, Pinheiro ALB, Ramalho LMP (2008) Effect of IR laser photobiomodulation on the repair of bone defects grafted with organic bovine bone. Lasers Med Sci 23:313–317. doi:10.1007/s10103-007-0488-0
Queiroga AS, Sousa FB, Araújo JMS, Santos SD, Sousa CD’FS, Quintans TC, Almeida TP, Nonaka CFW, Batista LV, Limeira Junior FA (2008) Evaluation of bone repair in the femur of rats submitted to laser therapy in different wavelengths: an image segmentation method of analysis. Laser Phys 18(9):1087–1091. doi:10.1134/S1054660X08090156
Matsumoto MA, Ferino RV, Monteleone GF, Ribeiro DA (2009) Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med Sci 24:195–201. doi:10.1007/s10103-008-0544-4
Shakouri SK, Soleimanpour J, Salekzamani Y, Oskuie MR (2010) Effect of low-level laser therapy on the fracture healing process. Lasers Med Sci 25(1):73–77. doi:10.1007/s10103-009-0670-7
Taba M, Novaes AB, Souza SLS, Grisi MFM, Palioto DB, Pardini LC (2003) Radiographic evaluation of dental implants with different surface treatments: an experimental study in dogs. Implant Dent 12(3):252–258
Martins MV, Da Silva MA, Medici Filho E, De Moraes LC, Castilho JC, Da Rocha RF (2005) Evaluation of digital optical density of bone repair in rats medicated with ketoprofen. Braz Dent J 16:207–212
Sakakura CE, Giro G, Gonçalves D, Pereira RM, Orrico SR, Marcantonio E Jr (2006) Radiographic assessment of bone density around integrated titanium implants after ovariectomy in rats. Clin Oral Implants Res 17:134–138
Gerbi MEM, Pinheiro ALB, Marzola C, Limeira Júnior FA, Ramalho LMP, Ponzi EAC, Soares AO, Carvalho LCB, Lima HV, Gonçalves TO (2005) Assessment of bone repair associated with the use of organic bovine bone and membrane irradiated at 830 nm. Photomed Laser Surg 23(4):382–388. doi:10.1089/pho.2005.23.382
Khadra M, Ronold HJ, Lyngstadaas SP, Ellingsen JE, Haanaes HR (2004) Low-level laser therapy stimulates bone-implant interaction: an experimental study in rabbits. Clin Oral Implants Res 15:325–332. doi:10.1111/j.1600-0501.2004.00994.x
Coombe AR, Ho C-TG, Philips JR, Chapple CC, Yum LWP, Darendeliler MA, Junter N (2001) The effects of low level laser irradiation on osteoblastic cells. Clin Orthop Res 4:3–14. doi:10.1034/j.1600-0544.2001.040102.x
Basford JR (1995) Low intensity laser therapy: still not an established clinical tool. Lasers Surg Med 16(3):331–342. doi:10.1002/lsm.1900160404
Javadieh F, Bayat M, Abdi S, Mohsenifar Z, Razi S (2009) The effects of infrared low-level laser therapy on healing of partial osteotomy of tibia in streptozotocin-induced diabetic rats. Photomed Laser Surg 27(4):641–646. doi:10.1089/pho.2008.2370
Acknowledgments
R.A. Souza thanks Fundação de Amparo a Pesquisa do Estado de Minas Gerais for the research grant support (APQ-02900-10).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barbosa, D., de Souza, R.A., Xavier, M. et al. Effects of low-level laser therapy (LLLT) on bone repair in rats: optical densitometry analysis. Lasers Med Sci 28, 651–656 (2013). https://doi.org/10.1007/s10103-012-1125-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-012-1125-0