Abstract
Idiopathic intracranial hypertension is consistently associated with venous outflow disturbances. Sinus venous stenosis are found at magnetic resonance venography in the large majority of IIH patients and may have various conformations, ranging from functional smooth narrowings of sinus segments associated or not with definite flow gaps, to segmental hypoplasia or aplasia of one or more central venous collectors. Stenosis are currently believed to be a consequence of a primary altered cerebrospinal fluid (CSF) pressure since it may normalize after CSF subtraction with lumbar puncture or shunting procedures. In this paper a “self-sustained venous collapse” is proposed as a crucial causative mechanism in predisposed subjects, leading to a self-sustained intracranial hypertension in presence of a wide range of triggering factors. The proposed mechanisms predict the long-term remission of IIH syndromes frequently observed after a single or few serial CSF subtractions by lumbar puncture.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Idiopathic intracranial hypertension
Idiopathic intracranial hypertension (IIH) [1] is an infrequent and enigmatic condition almost always encountered in overweight or obese women of childbearing age. IIH is characterized by headache, often on a daily basis, papilledema, transient visual obscurations, diplopia, vertigo and tinnitus. Symptoms arise from a hypertensive intracranial status which is not associated with any detectable cause. The prognosis is generally good but up to 25% of patients may develop permanent visual deficit secondary to optical nerve atrophy [2].
IIH diagnosis requires the demonstration of an intracranial hypertensive status not associated with cerebrospinal fluid (CSF) abnormalities or with any other detectable intracranial pathology [1]. Differential diagnosis with cerebral venous thrombosis (CVT) may be challenging since an isolated intracranial hypertension may be the only presenting sign in about 1/3 of CVT cases [3] and a CVT has been found in 11.4% of patients who were presumed to have IIH [4]. A secondary form of IIH may also be sustained by exposition to a number of drugs (minocycline and tetracycline, growth hormone, steroids and vitamin A, among others) [5]. IIH has been associated with menstrual irregularities [6, 7], oral contraceptives use [7], hyperthyroidism [8], acquired or congenital prothrombotic states [6], systemic lupus erythematosus [9, 10], uraemia [11], hypothyroidism [12], sleep disturbances including obstructive sleep apnoea syndrome (OSAS) [13], extracranial venous hypertension secondary to cardiac septal defect [14], and iron deficit anaemia [15]. Although IIH may present at any age and is not infrequent in children [16], female sex and obesity are major risk factors for IIH development [6, 17, 18]. Incidence of IIH is about 1 per 100,000 in the general population. In female sex the incidence increase with body mass index (BMI) [19, 20] and rise to about 14/100,000 among childbearing aged women with at least a 10% overweight and to about 19/100,000 in those with at least a 20% increase of ideal weight. The F/M ratio in this series was 8:1 [19]. Men with idiopathic intracranial hypertension (IIH) are twice as likely as women to develop severe visual loss [21]. IIH may run with a chronic or with a periodic recurrent course [6, 22]. Recurrences may follow weight gain and have been described in pregnancy [23].
Idiopathic intracranial hypertension without papilledema
IIH may run without papilledema (IIHWOP) in a part of the patients [24, 25]. IIHWOP may mimic chronic tension type headache [26] or may be indistinguishable from transformed migraine on clinical basis [27, 28]. Consequently, it is found in 10% [29] to 14% [27] of chronic headache sufferers series. Available literature evidences suggest that IIHWOP could represent a possible, largely underestimated, risk factor for migraine progression [30].
IIHWOP prevalence in general population is not known. Reported opening pressure (OP) values in IIHWOP are lower than in IIH [31] indicating that papilledema may lack in cases with milder or, possibly, with fluctuating [32] CSF pressure increases. IIH may run without headache in non-migrainous individuals or in course of a strong migraine protective factor as pregnancy [33] suggesting that a chronic migraine-like presentation of IIHWOP may require a migrainous background. Taken together the above considerations suggest that in non-migrainous individuals IIHWOP may run without any symptom or sign suggestive of raised intracranial pressure. This gives rise to the hypothesis that IIHWOP may be much more prevalent than believed in general population and that cases presenting with papilledema or other intracranial hypertensive symptoms or signs may represent only the top of the iceberg.
Findings of a well conducted recent study strongly support the hypothesis of a largely underestimated IIHWOP prevalence in general population. A strict association between bilateral transverse sinus stenosis and OP >200 mmH2O has been found in a series of 217 patients presenting neurological symptoms not reminiscent of intracranial hypertension (papilledema, visual symptoms, double vision, tinnitus and “persistent daily headache” were among the exclusion criteria). A bilateral transverse sinus stenosis (BTSS) at MRV was found in 50 subjects (23.0%). This subgroup showed a significantly higher mean OP (191.8 mmH2O, ±52.3, range 91–286) than unilateral stenosis subgroup (n = 47; mean OP = 147.1 mmH2O ± 34.6, range 78–195) or normal MRV subgroup (n = 120; mean OP = 132.3 mmH2O ± 38.0, range 65–195). An opening pressure >200 mmH2O (up to 286 mmH2O) was recorded in 24 of the 50 BTSS carriers (11.1% of the sample). In contrast, all patients showing unilateral stenosis or normal MRV had a CSF opening pressure <200 mmH2O [34]. Thus, in this large series of neurological patients without intracranial hypertensive symptoms or signs the IIHWOP prevalence was 11%.
Treatment options
Acetazolamide and topiramate share a carbonic anhydrase isoenzyme inhibition [35]. According with a recent trial, both drugs are effective in IIH management probably because of the reduction of CSF production rate [36]. Corticosteroids are highly effective in relieving IIH symptoms but the condition is sometimes precipitated by their withdrawal [37]. Intravenous administration of indomethacin has determined an intracranial pressure lowering effect both in IIH [38] and in intracranial hypertension following head trauma [39] but the efficacy of long-term IIH treatment with indomethacin is not known. According to a recent metanalysis, bariatric surgery (mainly Roux-en-Y gastric bypass) may be an effective treatment for IIH in obese patients, both in terms of symptoms resolution and visual outcome [40]. Shunting procedures [41] or optical nerve fenestration [42] are required in the minority of cases at risk for permanent visual defects. Still, repeated or even single CSF subtractions with lumbar puncture are not infrequently followed by longstanding remissions of IIH symptoms and signs [43–45]. Although not generally accepted [46], we believe that the long-term efficacy of a single or a few serial 20 ml CSF removal by LP should always be tested in IIH patients not responding to medical treatments, before planning any more invasive procedure.
Physiopathology
Despite the number of pathogenetic theories that have been proposed, the cause of IIH is still unknown. Basic mechanisms that could explain a sustained intracranial hypertensive state in otherwise healthy subjects include: (1) increased CSF production rate, (2) cerebral oedema and (3) reduced CSF absorption.
-
1.
Increase of CSF production rate A dramatic increase of CSF production rate has been documented in presence of choroidal plexus papilloma but this condition is associated with hydrocephalus rather than IIH [47]. An increased CSF production has been suspected in IIH cases associated with high oestrone levels [48] hypervitaminosis [49] but evidences that such a mechanism is of relevance in IIH are lacking [50].
-
2.
Cerebral oedema The hypothesis of a cryptogenetic cerebral swelling [51] has been convincingly ruled out by a more recent, well-designed study [52].
-
3.
Reduced CSF absorption A CSF absorption impairment is currently considered the main pathogenetic mechanism of IIH. It may be the consequence of a structural impairment of arachnoids villi [48] but a reduced CSF absorption is the mechanism leading to CSF pressure rising in the course of intracranial venous hypertension, whatever its cause [53]. CSF outflow rate into the venous compartment, in fact, largely depends on transmural pressure gradient [54]. Thus, it will be reduced by any condition leading to an increase of intracranial venous pressure. Most of neurologists have direct experience of the strict relation existing between CSF and intracranial venous pressures. A short bilateral jugular compression while monitoring the CSF pressure during a lumbar puncture, the so called Queckenstedt’s manoeuvre [55], is expected to immediately increase CSF pressure. If no changes are observed, the occlusion of spinal subarachnoid spaces communications is to be suspected.
Thus, a central venous hypertension is expected in every condition leading to a significant intracranial venous outflow resistance, including functional, malformative or thrombotic narrowing or occlusion of large cerebral venous collectors.
The role of central venous stenosis in IIH pathogenesis
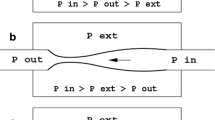
Stenosis of large intracranial venous sinuses can be found in magnetic resonance venography (MRV) in almost all affected subjects and are considered a reliable radiologic marker of intracranial hypertension, with a high specificity (93%) and sensitivity (93%) [56]. Stenosis may have various conformations. Smooth narrowing of a sinus segment associated or not with definite flow gaps are the most frequent, but segmental hypoplasia or aplasia of one or more central venous collectors, sometimes extended to a whole transverse sinus, can be not infrequently found. Significant TS calibre asymmetries can be sometimes followed along the jugular veins of the same side.
Whatever their appearance, there is evidence [57] of a venous pressure gradient across the stenosis with a potential effect in raising the cerebrospinal fluid (CSF) pressure [53]. Conversely, the venous pressure gradient may normalize soon after suboccipital CSF subtraction [57] and the stenosis may reduce or resolve after a single 20 ml CSF subtraction with lumbar puncture [43–45] or after continuous CSF diversion procedures such as lumbo-peritoneal shunt [58, 59]. These findings suggest that venous flow disturbances in IIH are most probably the effect of CSF hypertension, not its cause [60–62]. However, mounting evidences [46, 63–66] indicate that the placing of a self-expanding stent at the venous stenosis level is consistently followed by the immediate and longstanding resolution of IIH symptoms.
Thus, on one hand, an acute CSF volume reduction by LP or continuous CSF diversion may reduce the stenosis degree and the related pressure gradient. On the other hand, the stenting of segmental venous narrowing at transverse sinus level may revert the CSF hypertensive status. Taken together, these findings indicate that in IIH patients sinus venous stenosis and CSF hypertension may influence each other in a circular way whose starting point may only arbitrarily be indicated.
Sinus venous stenosis as a causative factor in IIH pathogenesis
Since the CSF pressure is higher than central venous pressure (at least in clinostatic position), any central vein not sufficiently rigid would be compressed as a consequence of transmural pressure gradient. Therefore, in subjects harbouring one ore more collapsible segments of central veins and exposed to an unknown CSF rising factor, the subsequent segmental venous narrowing and the related further CSF pressure rising may engage a positive feedback loop. This loop is presumably self-limiting: once the maximum stretching of the venous wall is reached, in fact, no further venous narrowing could parallel CSF pressure rising, leading to a relatively stable new balance at higher values of CSF and venous blood pressures [43]. This means that the new high pressures balance may be self-sustained, i.e. not susceptible to spontaneous reversing even after the cessation of the primary triggering factor, leading to a longstanding clinical syndrome. Cases presenting with a periodic pattern may reflect a spontaneous shift between the two balance states in individuals with less compliant venous walls, associated or not to other venous outflow obstacles.
The hypothesis of a causative “self-limiting venous collapse” predicts the longstanding normalization of venous and CSF outflow dynamics observed during CSF draining [67] or, at least in a part of the cases, even after a single (or a few serial) LP [43]. In fact, according with the Monro–Kellie hypothesis [68] the removal of CSF by LP is compensated by the enlargement of cerebral venous calibre that, in turn, reduces sinus venous hypertension enhancing CSF outflow and promoting a further venous enlarging. The findings of an interesting manometric/angiographic combined study on nine IIH patients confirm that venous and CSF pressures are strictly coupled in IIH patients and that during CSF drainage both pressures decrease, probably until normalization of venous pressure gradient at the stenosis level. Then only CSF pressure further decreases, while venous pressure remains constant [67].
The hypothesis of a causative role of a “self-limiting venous collapse” that we proposed on the basis of clinical observations [4] is supported by a series of independent and almost simultaneous mathematic modelling studies [69, 70]. In subjects carrying one or more collapsible segment of large central venous collectors, the proposed mechanism may account at least for the maintenance of a hypertensive intracranial status induced by a different cause. Moreover, it can explain the long-term efficacy not infrequently observed after a single or serial LP. The self-limiting property of the mechanism could also explain prognostic differences between “benign” intracranial hypertension and CVT, a disease with up to 8.3% of mortality and about 35% of permanent residual neurological symptoms [71], in which the venous hypertension is sustained by an intrinsic obstruction of venous flow, a mechanism lacking any self-limiting propriety.
It is noteworthy that in some IIH patients the venous flow abnormalities may reflect a congenital hypoplasia of central veins segments [72] or the presence of endoluminal obstructions caused by giant Pacchioni granulations [73, 74]. A minor central venous calibre variation, either compression or dilatation [75], may be physiologic. It has been proposed that the dural venous sinuses could act as a volume–pressure reservoir to accommodate the physiologic and spontaneous changes of CSF volume and pressure [59]. A low-range collapsibility of sinus venous walls is therefore expected to be present in normal conditions. Thus, in both congenital venous hypoplasia and intrinsic venous filling defects, the physiological redundancy of total venous outflow cross section may result reduced under a critical value, therefore enhancing the susceptibility of the venous and CSF outflow systems to a self-sustained high-pressure CSF and venous pressure balance shift, even in the absence of a pathologically elevated venous sinus compliance.
Triggering factors
The self-limited venous collapse might be regarded as a crucial IIH predisposing mechanism. Still, a primary event triggering the CSF and sinus venous higher-pressure balances shift is probably always required. There is evidence that a central venous hypertension not associated with anatomical or functional stenosis may result from derangements of cerebral blood flow autoregulation, leading to a transient or persistent hyperperfusion of the brain [76]. In subjects carrying one ore more collapsible segments of large central venous sinuses, the increased venous outflow resulting from brain hyperaemia could represent an adequate trigger of the self-limiting venous collapse mechanism [77, 78].
It is of interest that an instability of the brain perfusion can be observed in migraine with aura [79] and in migraine without aura [80] possibly as a consequence of the cortical spread depression phenomena. This could account for the high frequency of IIHWOP observed in headache sufferers [81].
Another recent interesting study [82] has found a high prevalence of jugular valve incompetence in IIH patients carrying central venous stenosis. Possibly, an increased abdominal pressure transmitted into the intracranial venous system through a jugular valve incompetence may have a role in triggering the self-limiting venous collapse in affected individuals.
Possibly, other primary factors, either promoting a CSF hypertension or an intracranial venous hypertension, could reveal implied in the pressures balance shift. This could account for the wide range of comorbidity conditions observed in IIH, including endocrinological dysfunctions, drug exposures, and any cause of raised extracranial venous pressure such as OSAS, weight gain or pregnancy.
Conclusions
Available literature evidences strongly suggest that an increased resistance of sinus venous outflow caused by functional, malformative or obstructive narrowing or occlusion of large cerebral venous collectors are almost always required to develop a IIH syndrome. Accordingly, central sinus venous stenosis should be view as a crucial pathogenetic predisposing factor for the development of a chronic or recurrent IIH with or without papilledema, rather than a secondary phenomenon lacking any pathogenetic role. The hypothesized mechanism relays on the transition from a normal CSF and venous intracranial pressures balance to a relatively stable, self-sustained, higher-pressure balance promoted by a self-limiting collapse of one or more segments of central veins. A trigger factor is probably almost always required. Recent evidences suggest that, in many cases, an instability of brain perfusion autoregulation and/or a jugular valve incompetence may trigger a self-sustained intracranial hypertension in presence of sinus venous stenosis. Still, other factors leading to transient or persistent central venous hypertension or to an increase of CSF production rate may be required as complementary factors. The pathologic high CSF and venous pressures balance is potentially reversible by an adequate perturbation at either side of the loop, such as CSF removal/diversion or endovascular venous stenting.
References
Friedman DI, Jacobson DM (2002) Diagnostic criteria for idiopathic intracranial hypertension. Neurology 59:1492–1495
Corbett JJ, Savino PJ, Thompson HS, Kansu T, Schatz NJ, Orr LS, Hopson D (1982) Visual loss in pseudotumor cerebri: follow-up of 57 patients from 5 to 41 years and a profile of 14 patients with permanent severe visual loss. Arch Neurol 39:461–474
Biousse V, Ameri A, Bousser MG (1999) Isolated intracranial hypertension as the only sign of cerebral venous thrombosis. Neurology 53:1537–1542
Agarwal P, Kumar M, Arora V (2010) Clinical profile of cerebral venous sinus thrombosis and the role of imaging in its diagnosis in patients with presumed idiopathic intracranial hypertension. Indian J Ophthalmol 58(2):153–155
Dhungana S, Sharrack B, Woodroofe N (2010) Idiopathic intracranial hypertension. Acta Neurol Scand 121:71–82
Glueck CJ, Aregawi D, Goldenberg N, Golnik KC, Sieve L, Wang P (2005) Idiopathic intracranial hypertension, polycystic-ovary syndrome, and thrombophilia. J Lab Clin Med 145:72–82
Finsterer J, Kuntscher D, Brunner S, Krugluger W (2007) Pseudotumor cerebri from sinus venous thrombosis, associated with polycystic ovary syndrome and hereditary hypercoagulability. Gynecol Endocrinol 23:179–182
Merkenschlager A, Ehrt O, Müller-Felber W, Schmidt H, Bernhard MK (2008) Reversible benign intracranial hypertension in a child with hyperthyroidism. J Pediatr Endocrinol Metab 21:1099–1101
Green L, Vinker S, Amital H, Amir T, Bar-Dayan Y, Levi Y, Schoenfeld Y (1995) Pseudotumor cerebri in systemic lupus erythematosus. Semin Arthritis Rheum 25:103–108
Barahona-Hernando R, Ríos-Blanco JJ, Méndez-Mesón I, Pérez-Valero I, Soto-Abánades CI, Noval S, Carceller F, Robles-Marhuenda A, Gil-Aguado A (2009) Idiopathic intracranial hypertension and systemic lupus erythematosus: a case report and review of the literature. Lupus 18:1121–1123
Chang D, Nagamoto G, Smith WE (1992) Benign intracranial hypertension and chronic renal failure. Clevel Clin J Med 59:419–422
Huseman CA, Torkelson RD (1984) Pseudotumor cerebri following treatment of hypothalamic and primary hypothyroidism. Am J Dis Child 138:927–931
Wall M, Purvin V (2009) Idiopathic intracranial hypertension in men and the relationship to sleep apnea. Neurology 72:300–301 Epub Oct 15 2008
Jicha GA, Suarez GA (2003) Pseudotumor cerebri reversed by cardiac septal defect repair. Neurology 60:2016–2017
Mollan SP, Ball AK, Sinclair AJ, Madill SA, Clarke CE, Jacks AS, Burdon MA, Matthews TD (2009) Idiopathic intracranial hypertension associated with iron deficiency anaemia: a lesson for management. Eur Neurol 62:105–108
Tibussek D, Schneider DT, Vandemeulebroecke N, Turowski B, Messing-Juenger M, Willems PH, Mayatepek E, Distelmaier F (2010) Clinical spectrum of the pseudotumor cerebri complex in children. Childs Nerv Syst 26:313–321
Rowe FJ, Sarkies NJ (1999) The relationship between obesity and idiopathic intracranial hypertension. Int J Obes Relat Metab Disord 23:54–59
Hannerz J, Ericson K (2009) The relationship between idiopathic intracranial hypertension and obesity. Headache 49:178–184
Durcan FJ, Corbett JJ, Wall M (1988) The incidence of pseudotumour cerebri population studies in Iowa and Louisiana. Arch Neurol 45:875–877
Radhakrishnan K, Ahlskog JE, Cross SA, Kurland LT, O’Fallon WM (1993) Idiopathic intracranial hypertension (pseudotumor cerebri). Descriptive epidemiology in Rochester, Minn., 1976 to 1990. Arch Neurol 50:78–80
Bruce BB, Kedar S, Van Stavern GP, Monaghan D, Acierno MD, Braswell RA, Preechawat P, Corbett JJ, Newman NJ, Biousse V (2009) Idiopathic intracranial hypertension in men. Neurology 72(4):304–309
Kesler A, Hadayer A, Goldhammer Y, Almog Y, Korczyn AD (2004) Idiopathic intracranial hypertension: risk of recurrences. Neurology 63:1737–1739
Huna-Baron R, Kupersmith MJ (2002) Idiopathic intracranial hypertension in pregnancy. J Neurol 249:1078–1081
Lipton HL, Michelson PE (1972) Pseudotumor cerebri syndrome without papilledema. J Am Med Assoc 220:1591–1592
Marcelis J, Silberstein SD (1992) Idiopathic intracranial hypertension without papilledema. Arch Neurol 48:392–399
Bono F, Messina D, Giliberto C, Cristiano D, Broussard G, D’Asero S, Condino F, Mangone L, Mastrandrea C, Fera F, Quattrone A (2008) Bilateral transverse sinus stenosis and idiopathic intracranial hypertension without papilledema in chronic tension-type headache. J Neurol 255(6):807–812
Mathew NT, Ravishankar K, Sanin LC (1996) Coexistence of migraine and idiopathic intracranial hypertension without papilledema. Neurology 46:1226–1230
Wang SJ, Silberstein SD, Patterson S, Young WB (1998) Idiopathic intracranial hypertension without papilledema: a case–control study in a headache center. Neurology 51:245–249
Vieira DS, Masruha MR, Gonçalves AL, Zukerman E, Senne Soares CA, Naffah-Mazzacoratti Mda G, Peres MF (2008) Idiopathic intracranial hypertension with and without papilloedema in a consecutive series of patients with chronic migraine. Cephalalgia 28:609–613
De Simone R, Ranieri A, Fiorillo C, Bilo L, Bonavita V (2010) Is idiopathic intracranial hypertension without papilledema a risk factor for migraine progression? Neurol Sci. doi:10.1007/s10072-010-0229-1
Kathleen Digre B, Beau Nakamoto K, Judith Warner EA, Wendy Langeberg J, Susan Baggaley K, Bradley Katz J (2009) A comparison of idiopathic intracranial hypertension with and without papilledema. Headache 49:185–193
Torbey MT, Geocadin RG, Razumovsky AY, Rigamonti D, Williams MA (2003) Utility of CSF pressure monitoring to identify idiopathic intracranial hypertension without papilledema in patients with chronic daily headache. Cephalalgia 24:495–502
De Simone R, Marano E, Bilo L, Briganti F, Esposito M, Ripa P et al (2006) Idiopathic intracranial hypertension without headache. Cephalalgia 26:1020–1021
Bono F, Cristiano D, Mastrandrea C et al (2010) The upper limit of normal CSF opening pressure is related to bilateral transverse sinus stenosis in headache sufferers. Cephalalgia 30(2):145–151
Dodgson SJ, Shank RP, Maryanoff BE (2000) Topiramate as an inhibitor of carbonic anhydrase isozymes. Epilepsia 41(Suppl 1):S35–S39
Celebisoy N, Gökçay F, Sirin H, Akyürekli O (2007) Treatment of idiopathic intracranial hypertension: topiramate vs acetazolamide, an open-label study. Acta Neurol Scand 116:322–327
Liu GT, Kay MD, Bienfang DC, Schatz NJ (1994) Pseudotumour cerebri associated with corticosteroid withdrawal in inflammatory bowel disease. Am J Ophthalmol 117:352–357
Förderreuther S, Straube A (2000) Indomethacin reduces CSF pressure in intracranial hypertension. Neurology 55:1043–1045
Jensen K, Cold GE, Astrup J, Ohrström J (1990) Indomethacin in severe head injury. Lancet 336(8709):246
Fridley J, Foroozan R, Sherman V, Brandt ML, Yoshor D (2010) Bariatric surgery for the treatment of idiopathic intracranial hypertension. J Neurosurg. doi:10.3171/2009.12.JNS09953
Garton HJ (2004) Cerebrospinal fluid diversion procedures. J Neuroophthalmol 24:146–155
Nithyanandam S, Manayath GJ, Battu RR (2008) Optic nerve sheath decompression for visual loss in intracranial hypertension: report from a tertiary care center in South India. Indian J Ophthalmol 56:115–120
De Simone R, Marano E, Fiorillo C, Briganti F, Di Salle F, Volpe A, Bonavita V (2005) Sudden re-opening of collapsed transverse sinuses and longstanding clinical remission after a single lumbar puncture in a case of idiopathic intracranial hypertension: pathogenetic implications. Neurol Sci 25:342–344
Scoffings DJ, Pickard JD, Higgins JNP (2007) Resolution of transverse sinus stenoses immediately after CSF withdrawal in idiopathic intracranial hypertension. J Neurol Neurosurg Psychiatry 78:911–912
Lee SW, Gates P, Morris P, Whan A, Riddington L (2009) Idiopathic intracranial hypertension; immediate resolution of venous sinus ‘‘obstruction” after reducing cerebrospinal fluid pressure to <10 cmH2O. J Clin Neurosci 16:1690–1692
Brazis PW (2008) Clinical review: the surgical treatment of idiopathic pseudotumour cerebri (idiopathic intracranial hypertension). Cephalalgia 28:1361–1373
Eisenberg HM, McComb JG, Lorenzo AV (1974) Cerebrospinal fluid overproduction and hydrocephalus associated with choroid plexus papilloma. J Neurosurg 40:381–385
Donaldson J, Horak E (1982) Cerebrospinal fluid oestrone in pseudotumour cerebri. J Neurol Neurosurg Psychiatry 45:734–736
Walker RW (2001) Idiopathic intracranial hypertension: any light on the mechanism of the raised pressure? J Neurol Neurosurg Psychiatry 71:1–5
Gideon P, Sorensen PS, Thomsen C, Ståhlberg F, Gjerris F, Henriksen O (1994) Assessment of CSF dynamics and venous flow in the superior sagittal sinus by MRI in idiopathic intracranial hypertension: a preliminary study. Neuroradiology 36:350–354
Gideon P, Sorensen PS, Thomsen C, Ståhlberg F, Gjerris F, Henriksen O (1995) Increased brain water self-diffusion in patients with idiopathic intracranial hypertension. Am J Neuroradiol 16:381–387
Bastin ME, Sinha S, Farrall AJ, Wardlaw JM, Whittle IR (2003) Diffuse brain oedema in idiopathic intracranial hypertension: a quantitative magnetic study. J Neurol Neurosurg Psychiatry 74:1693–1696
Karakalios DG, Rekate HL, Khayata MH, Apostolides PJ (1996) Elevated intracranial venous pressure as a universal mechanism in pseudotumor cerebri of varying etiologies. Neurology 46:198–202
King JO, Mitchell PJ, Thomson KR, Tress BM (1995) Cerebral venography and manometry in idiopathic intracranial hypertension. Neurology 45:2224–2228
Pearce JMS (2006) Queckenstedt’s manoeuvre. J Neurol Neurosurg Psychiatry 77:728
Farb RI, Vanek I, Scott JN, Mikulis DJ, Willinsky RA, Tomlinson G, terBrugge KG (2003) Idiopathic intracranial hypertension: the prevalence and morphology of sinovenous stenosis. Neurology 60:1418–1424
King JO, Mitchell PJ, Thomson KR, Tress BM (2002) Manometry combined with cervical puncture in idiopathic intracranial hypertension. Neurology 58:26–30
Higgins JNP, Pickard JD (2004) Lateral sinus stenosis in idiopathic intracranial hypertension resolving after CSF diversion. Neurology 62:1907–1908
Baryshnik DB, Farb RI (2004) Changes in the appearance of venous sinuses after treatment of disordered intracranial pressure. Neurology 62:1445–1446
Rohr A, Dorner L, Stingele R, Buhl R, Alfke K, Jansen O (2007) Reversibility of venous sinus obstruction in idiopathic intracranial hypertension. AJNR Am J Neuroradiol 28:656
Stienen A, Weinzierl M, Ludolph A, Tibussek D, Häusler M (2008) Obstruction of cerebral venous sinus secondary to idiopathic intracranial hypertension. Eur J Neurol 15:1416–1418
Bono F, Giliberto C, Mastrandrea C, Cristiano D, Lavano A, Fera F, Quattrone A (2005) Transverse sinus stenosis persist after normalization of the CSF pressure in IIH. Neurology 65:1090–1093
Higgins JN, Cousins C, Owler BK, Sarkies N, Pickard JD (2003) Idiopathic intracranial hypertension: 12 cases treated by venous sinus stenting. J Neurol Neurosurg Psychiatry 74:1662–1666
Arac A, Lee M, Steinberg GK, Marcellus M, Marks MP (2009) Efficacy of endovascular stenting in dural venous sinus stenosis for the treatment of idiopathic intracranial hypertension. Neurosurg Focus 27(5):E14
Bussière M, Falero R, Nicolle D, Proulx A, Patel V, Pelz D (2010) Unilateral transverse sinus stenting of patients with idiopathic intracranial hypertension. Am J Neuroradiol. doi:10.3174/ajnr.A1890
Donnet A, Metellus P, Levrier O, Mekkaoui C, Fuentes S, Dufour H, Conrath J, Grisoli F (2008) Endovascular treatment of idiopathic intracranial hypertension. Clinical and radiologic outcome of 10 consecutive patients. Neurology 70:641–647
Pickard JD, Czosnyka Z, Czosnyka M, Owler B, Higgins JN (2008) Coupling of sagittal sinus pressure and cerebrospinal fluid pressure in idiopathic intracranial hypertension—a preliminary report. Acta Neurochir Suppl 102:283–285
Mokri B (2001) The Monro–Kellie hypothesis: applications in CSF volume depletion. Neurology 56:1746–1748
Stevens SA, Lakin W, Thakore N, Penar PL, Tranmer B (2005) A modeling study of idiopathic intracranial hypertension. In: Ursino M (ed) Modelling in medicine and biology VI. WIT Press, Southampton
Stevens SA, Stimpson J, Lakin WD, Thakore NJ, Penar PL (2008) A model for idiopathic intracranial hypertension and associated pathological ICP wave-forms. IEEE Trans Biomed Eng 55(2 Pt 1):388–398
Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F, ISCVT Investigators (2004) Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke 35:664–670
Connor SEJ, Siddiqui MA, Stewart VR, O’Flynn EAM (2008) The relationship of transverse sinus stenosis to bony groove dimensions provides an insight into the aetiology of idiopathic intracranial hypertension. Neuroradiology 50:999–1004
Arjona A, Delgado F, Fernandez-Romero E (2003) Intracranial hypertension secondary to giant arachnoid granulations. J Neurol Neurosurg Psychiatry 74:418
Kiroglu Y, Yaqci B, Cirak B, Karabulut N (2008) Giant arachnoid granulation in a patient with benign intracranial hypertension. Eur Radiol 18:2329–2332
Farb RI, Forghani R, Lee SK, Mikulis DJ, Agid R (2007) The venous distension sign: a diagnostic sign of intracranial hypotension at MR imaging of the brain. AJNR Am J Neuroradiol 28:1489–1493
Bateman GA (2002) Vascular hydraulics associated with idiopathic and secondary intracranial hypertension. Am J Neuroradiol 23:1180–1186
Bateman GA (2004) Idiopathic intracranial hypertension: priapism of the brain? Med Hypotheses 63:549–552
Bateman GA, Stevens SA, Stimpson J (2009) A mathematical model of idiopathic intracranial hypertension incorporating increased arterial inflow and variable venous outflow collapsibility. J Neurosurg 110:446–456
Wolf ME, Jäger T, Bäzner H, Hennerici M (2009) Changes in functional vasomotor reactivity in migraine with aura. Cephalalgia 29:1156–1164
Bono F, Messina D, Giliberto C, Cristiano D, Broussard G, Fera F, Condino F, Lavano A, Quattrone A (2006) Bilateral transverse sinus stenosis predicts IIH without papilledema in patients with migraine. Neurology 67:419–423
Denuelle M, Fabre N, Payoux P, Chollet F, Geraud G (2008) Posterior cerebral hypoperfusion in migraine without aura. Cephalalgia 28:856–862
Nedelmann M, Kaps M, Mueller-Forell W (2009) Venous obstruction and jugular valve insufficiency in idiopathic intracranial hypertension. J Neurol 256:964–969
Conflict of interest
The authors declare that they have no conflict of interest related to the publication of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Simone, R., Ranieri, A. & Bonavita, V. Advancement in idiopathic intracranial hypertension pathogenesis: focus on sinus venous stenosis. Neurol Sci 31 (Suppl 1), 33–39 (2010). https://doi.org/10.1007/s10072-010-0271-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-010-0271-z