Abstract
The pathophysiology of elevated intracranial pressure in idiopathic intracranial hypertension (IIH) is unclear. Cerebral venous outflow obstruction and elevated intracranial venous pressure may play an etiological role. We examined jugular valve insufficiency as a potential factor contributing to intracranial hypertension. Jugular venous valve function was assessed bilaterally by duplex sonography in 20 consecutive patients with diagnosis of IIH and in 20 healthy controls matched for age, gender and body mass index. Diagnosis of valvular insufficiency was based on reflux duration during a controlled Valsalva maneuver. Intracranial venous outflow was evaluated in 11 patients (MR venography in 10, digital subtraction angiography (DSA) in two cases). As a principle result, valvular insufficiency was significantly more frequent in patients with IIH (70 vs. 30%; p < 0.05). This finding was associated with irregular leaflet structures on B-mode imaging (p < 0.01). Bilateral insufficiency was more frequent in the patient group which, however, was not significant (p = 0.08). In addition, sinovenous outflow obstruction was found in five of six patients that had undergone contrast-enhanced MR venography and DSA. The detection rate was inferior in phase-contrast MR imaging (one of five patients). In conclusion, this study gives evidence that valvular insufficiency may play a causal role in IIH. Obesity is a major risk factor for the disease and weight reduction leads to improvement of symptoms. Possibly, increased intra-abdominal pressure is transmitted into the intracranial venous system, causing intracranial hypertension. Jugular valve insufficiency may facilitate pressure transmission. As transverse sinus stenosis was a concomitant finding, these factors may be complementary.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Idiopathic intracranial hypertension (IIH) is a condition of increased intracranial pressure without evidence of mass lesion. It typically afflicts obese but otherwise healthy women of childbearing age. Symptoms of increased intracranial pressure are headache, nausea, vomiting and transient or permanent visual obscuration. Concomitant findings are papilledema and increased cerebrospinal fluid (CSF) pressure [3, 5].
Although the exact pathogenesis of intracranial pressure elevation still remains unknown, there are multiple indicators of an association with cerebral venous outflow obstruction. Bilateral transverse sinus stenosis is a frequent finding in patients with IIH [16, 17, 22]. However, it is unclear whether these stenoses are the cause or a consequence of elevated intracranial pressure [11].
There is also evidence that intracranial hypertension may be found in states of disturbed extracranial venous drainage. Different case reports describe symptomatic intracranial pressure elevation after bilateral neck dissection [28, 43], after unilateral neck dissection when the dominant internal jugular vein is resected [13] and as a consequence of increased venous pressure due to an arteriovenous fistula involving the internal jugular vein [26].
In this context, the functional status of the internal jugular valves is of interest. The intact valve efficiently prevents retrograde flow into the internal jugular vein [31, 33]. As insufficient valves may promote venous congestion, valvular insufficiency has been postulated as a causal factor in different neurological diseases [1]. In transient global amnesia, a disorder that commonly presents with precipitating activities including an involuntary Valsalva maneuver, internal jugular valve insufficiency is a frequent finding [2, 32, 37, 38]. An increased rate of valvular insufficiency has also been found in patients with exertional headache [14] and in patients with cough headache associated with uremia and deep vein thrombosis [10].
In IIH, venous pressure transmission is of particular interest, as obesity is a prominent risk factor and increased intra-abdominal pressure [39] may be more easily transmitted through insufficient valves. The primary aim of this study was therefore to evaluate the functional status of the internal jugular valves in patients with IIH. In a subset of patients, intracranial imaging studies were performed to detect disturbances of intracranial venous outflow.
Methods
Twenty consecutive patients with diagnosis of IIH were included in this prospective study. For diagnosis of IIH, the modified Dandy criteria were applied [5]. All patients underwent both a clinical and a neurological evaluation. Spinal fluid opening pressures were recorded. Blood samples were analysed for routine parameters. Underlying structural cerebral pathologies as, e.g., brain tumor, were excluded by magnetic resonance tomography. Twenty healthy volunteers served as controls. The control group was recruited throughout the same time period as the patients and was matched for age, gender and body mass index (BMI). Informed consent was obtained from all patients and controls before entering the study. The study was approved by the local ethics committee and was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
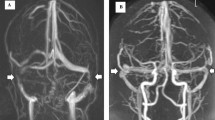
Internal jugular veins and jugular venous valve closure were assessed by both color-coded duplex sonography (HDI 5000, Philips Ultrasound Systems, Bothell WA; linear array 4–7 MHz transducer) and Doppler sonography (5 MHz). All studies were recorded on VCR for later offline analysis. The assessment of valvular insufficiency was based on previously published criteria [31]. In brief, jugular valve closure is monitored during a pressure-controlled Valsalva maneuver. The closure of competent jugular valves without any signs of insufficiency is indicated by a brief episode of venous reflux. This reflux corresponds to a short period of backward flow of blood during valve closure, followed by complete cessation of blood flow. Insufficient valves are characterized by sustained retrograde flow, visualized on color-coded duplex sonography as a circumscribed retrograde flow jet, which persists markedly after valve closure. Valvular insufficiency is diagnosed on the basis of reflux duration, with insufficient blood flow over a period longer than 0.88 s (Fig. 1). This cut-off value represents three times the standard deviation of the mean duration of backward flow in competent valves and discriminates conclusively between competent and incompetent valves. Anteroposterior venous diameter (directly above the valvular level) was determined in a transverse plane during expiration within a normal respiratory cycle (B-mode imaging).
Examples of valvular functional testing with Doppler ultrasound. The beginning of the Valsalva maneuver is indicated by arrows. a Brief episode of physiological reflux during closure of a competent valve, followed by cessation of flow. b Prolonged retrograde flow jet in a patient with diagnosis of idiopathic intracranial hypertension, indicating valvular insufficiency
Imaging of the intracranial venous system was performed in 11 patients. In five patients phase-contrast angiography (PC-MRA) was done, while in five patients contrast enhanced MR-angiography (CE-MRA) was performed. One of these underwent an additional digital subtraction angiography (DSA), which was the only imaging modality in one other patient. CE-MRA was performed with an eight-channel head coil array (i-PAT coil) allowing for parallel acquisition of independently reconstructed images (GRAPPA mode) [42].
The Chi square test was used for the comparison of the incidence of valvular insufficiency and for comparison of valvular morphology in patients with IIH and controls. After confirmation of normal distribution (Kolmogorov–Smirnov test), the unpaired two-sided Student’s t test was used for two-group comparison of baseline characteristics (age and BMI) and the diameter of the internal jugular vein. Significance was defined as p < 0.05. All analyses were performed using SPSS version 12.0 for Windows. Values are mean ± SD.
Results
As the incidence of jugular valve insufficiency depends on age, BMI and gender of individuals, the patients and the control group were carefully matched for these factors. Slight differences with respect to the individuals’ ages were not significant (p = 0.33). See Table 1 for details on clinical characteristics. Analysis of CSF revealed no signs of meningitis in any of the patients (the cell count was within normal limits). In four patients, a slightly raised protein level was detected (none above 0.7 g/l; normal range:<0.45 g/l).
Ultrasound
Fourteen out of 20 patients with IIH had either left- or right-sided, or bilaterally insufficient jugular valves. The incidence of jugular valve insufficiency was significantly lower in the matched control group (6 out of 20 individuals; p < 0.05; Table 2). The total number of insufficient valves was 19 out of 40 valves in patients with IIH, as compared to 7 out of 40 valves in controls (p < 0.01). The finding of bilaterally insufficient valves was more frequent in the patient group (n = 5 vs. n = 1 in the controls). This difference, however, was not significant (p = 0.08). Insufficiency was equally distributed between the left and the right internal jugular vein. 73% of the valves in the control group were morphologically (B-mode imaging) inconspicuous, with slender and regularly configured leaflets and a typical whip-like motion during the cardiac cycle. This finding was significantly less frequent in the patients’ group (38%; p < 0.01). The remaining valves displayed irregularly structured leaflets and limited or absent motion, or were not detectable on B-mode imaging.
The anteroposterior venous diameter at the valvular level was significantly larger in the patients’ group, with 1.11 ±0.27 versus 0.94 ±0.28 cm in controls (p < 0.05; Table 2). Within the patients’ group, there was no difference of diameter between competent and insufficient valves (1.12 ±0.28 vs. 1.09 ±0.27 cm).
MRI
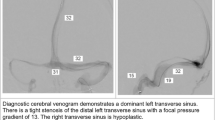
The typical finding of intracranial venous imaging was bilateral obstruction of sinovenous outflow at the level of the transverse sinuses, comprised of either bilateral stenoses or unilateral stenosis in combination with contralateral hypoplasia. However, imaging results were largely dependent on the employed technique. Only one of five patients examined by PC-MRA exhibited stenoses of the transverse sinuses, as compared with five of six patients examined by CE-MRA and DSA. In all affected patients, the stenoses were found at the transverse sinuses; one patient had an additional stenosis of the distal sagittal sinus (Fig. 2).
Contrast-enhanced MR venography findings in one patient with idiopathic intracranial hypertension. Bilateral venous outflow obstruction is revealed with a hypoplastic left transverse sinus and stenoses of the distal portion of the superior sagittal sinus (black arrow) and the right transverse sinus (white arrows)
Discussion
This study shows that insufficiency of the internal jugular valve is a frequent finding in patients with IIH. The prevalence of valvular insufficiency is more than double compared to a control group that was matched for age, gender and BMI. These results support the hypothesis that impeded venous return from the brain may play a causal role in the etiology of IIH.
Despite increasing research efforts, the underlying pathophysiology of IIH is still unclear. Different theories have been developed to explain the etiology of intracranial hypertension [3, 11, 15, 34]. Among these, increased CSF pressure as a consequence of venous outflow obstruction is of particular interest. It is recognized that dural sinus thrombosis can cause a clinical picture very similar to IIH [6]. Therefore, imaging of the venous outflow system is routinely employed in the diagnostic workup of patients with IIH. Recent studies have identified non-thrombotic dural sinus stenoses as a common finding in most patients. These abnormalities are mostly located at the level of the transverse sinuses and are often found bilaterally [16, 17, 22]. In the study by Farb and co-workers [16], transverse sinus stenoses mostly consisted of a long smooth tapered narrowing of the venous conduit. A minority had intraluminal filling defects, which were attributed to a possible swelling of arachnoid granulation.
Our findings confirm the presence of transverse sinus stenosis. However, detection of stenoses strongly depended on the imaging modality used. In all but one of the cases that were studied by CE-MRA or DSA, bilateral transverse sinus outflow obstruction was found; in one case the sagittal sinus was additionally affected. The detection rate was much lower in patients that were studied by PC-MRA, due to an inferior quality of delineation of the intracranial venous structures compared to CE studies [21]. In addition, imaging of intracranial venous outflow had been investigated in only 11 of 20 of our patients. Therefore, a more comprehensive picture of the overall—extra- and intracranial—venous outflow situation cannot be provided by this study.
It remains unclear whether venous sinus stenoses are the cause or a consequence of intracranial pressure elevation. Possibly, a stenosis may be caused by external compression of the sinus due to elevated intracranial pressure. This would explain observations of an improvement of the stenoses’ diameter after therapeutic lumbar puncture [12, 36] and lumboperitoneal shunting [4, 21, 30]. However, another study has shown persistence of transverse sinus stenoses after normalisation of CSF pressure at long term follow-up [9]. On the other hand, a venous pressure gradient across the stenotic sections has been repeatedly described [20, 23–25]. Also, case reports suggest an improvement of symptoms after stenting, hinting towards a causal link [19, 20, 35]. Based on these findings, it has been suggested that elevated intracranial pressure may be due to two interrelated components [16, 25, 29]. An elevated intracranial pressure, possibly present over a variable period of time, may lead to transverse sinus compression. In a vicious cycle, the narrowed lumen may then cause venous flow obstruction with consecutive pressure retention resulting in decompensation of intracranial pressure and in clinical presentation.
Transverse sinus stenosis may therefore not be sufficient to explain the formation of intracranial pressure elevation. Another venous hypothesis focuses on a more general venous hypertension with intracranial venous pressure elevation being a consequence of extracranially impeded venous return [3, 7, 23]. Marked obesity, which is one of the most prominent clinical features of patients with IIH, is frequently quoted in this context. Different studies have shown obesity related increased intra-abdominal and right heart filling pressures in patients with IIH [23, 39]. An animal study found an association between increased intra-abdominal pressure and an intracranial pressure elevation [8]. Intracranial hypertension as a consequence of raised intra-abdominal pressure may explain why weight reduction is associated with clinical improvement, improvement of papilledema and lowering of CSF pressure [27, 40, 44]. Sugerman and co-workers [41] found a high rate of treatment success after surgical (bariatric) treatment of obesity. Also, it has been shown that recent weight gain is associated with clinical manifestation and relapses of IIH [18, 44].
The finding of internal jugular valve abnormalities is to be seen in this context, as valvular insufficiency may have facilitated pressure transmission from the right heart into the intracranial venous system. Our patients significantly more often displayed functionally and structurally deviant internal jugular valves as compared to the control group. In this study, the patient and the control group were carefully matched for gender, age and BMI, as these factors are known to influence the frequency of pathological findings at the internal jugular valves [31]. A slightly higher age of the controls was not significantly different from the patient group and would, if at all, have influenced the results towards a higher prevalence of valvular insufficiency in the control group.
In summary, our data contribute to an understanding of the etiology of IIH. The results of this study support the hypothesis that increased intracranial pressure in IIH may be a consequence of a more general state of venous hypertension, possibly related to obesity.
In the context of the discussed literature, there is strong evidence that venous outflow abnormalities and obstructions are a unifying mechanism in the development of intracranial hypertension. Intra- and extracranial obstruction and venous pressure elevation may be complementary factors. However, it is stressed that the venous hypothesis has its limitations, as it does not conclusively explain all distinctive characteristics of IIH. It does not explain the preponderance of women of childbearing age with this disorder. Men and children with the disease are less likely to be obese. Also, conceiving elevated right heart filling pressures as a major contributing factor, one would expect congestive heart failure as a potential cause of symptomatic intracranial hypertension, which is not the case. Possibly, venous hypertension is only one contributing factor to the disease and IIH may have to be viewed as a disease entity with a multifactorial origin and a chain of events leading to increased CSF pressure [11].
References
Akkawi NM, Agosti C, Borroni B, Rozzini L, Magoni M, Vignolo LA, Padovani A (2002) Jugular valve incompetence: a study using air contrast ultrasonography on a general population. J Ultrasound Med 21:747–751
Akkawi NM, Agosti C, Rozzini L, Anzola GP, Padovani A (2001) Transient global amnesia and venous flow patterns. Lancet 357:639
Ball AK, Clarke CE (2006) Idiopathic intracranial hypertension. Lancet Neurol 5:433–442
Baryshnik DB, Farb RI (2004) Changes in the appearance of venous sinuses after treatment of disordered intracranial pressure. Neurology 62:1445–1446
Binder DK, Horton JC, Lawton MT, McDermott MW (2004) Idiopathic intracranial hypertension. Neurosurgery 54:538–551 discussion 551-532
Biousse V, Ameri A, Bousser MG (1999) Isolated intracranial hypertension as the only sign of cerebral venous thrombosis. Neurology 53:1537–1542
Biousse V, Bousser MG (2001) Benign intracranial hypertension. Rev Neurol (Paris) 157:21–34
Bloomfield GL, Ridings PC, Blocher CR, Marmarou A, Sugerman HJ (1997) A proposed relationship between increased intra-abdominal, intrathoracic, and intracranial pressure. Crit Care Med 25:496–503
Bono F, Giliberto C, Mastrandrea C, Cristiano D, Lavano A, Fera F, Quattrone A (2005) Transverse sinus stenoses persist after normalization of the CSF pressure in IIH. Neurology 65:1090–1093
Chuang YM, Hu HH (2005) Cough headache and thoracic inlet valvular competence in uremia. Eur Neurol 53:78–80
Corbett JJ, Digre K (2002) Idiopathic intracranial hypertension: an answer to, “the chicken or the egg?”. Neurology 58:5–6
De Simone R, Marano E, Fiorillo C, Briganti F, Di Salle F, Volpe A, Bonavita V (2005) Sudden re-opening of collapsed transverse sinuses and longstanding clinical remission after a single lumbar puncture in a case of idiopathic intracranial hypertension: pathogenetic implications. Neurol Sci 25:342–344
Doepp F, Schreiber SJ, Benndorf G, Radtke A, Gallinat J, Valdueza JM (2003) Venous drainage patterns in a case of pseudotumor cerebri following unilateral radical neck dissection. Acta Otolaryngol 123:994–997
Doepp F, Valdueza JM, Schreiber SJ (2008) Incompetence of internal jugular valve in patients with primary exertional headache: a risk factor? Cephalalgia 28:182–185
Donaldson JO (1981) Pathogenesis of pseudotumor cerebri syndromes. Neurology 31:877–880
Farb RI, Vanek I, Scott JN, Mikulis DJ, Willinsky RA, Tomlinson G, terBrugge KG (2003) Idiopathic intracranial hypertension: the prevalence and morphology of sinovenous stenosis. Neurology 60:1418–1424
Fera F, Bono F, Messina D, Gallo O, Lanza PL, Auteri W, Nicoletti G, Santoro G, Quattrone A (2005) Comparison of different MR venography techniques for detecting transverse sinus stenosis in idiopathic intracranial hypertension. J Neurol 252:1021–1025
Giuseffi V, Wall M, Siegel PZ, Rojas PB (1991) Symptoms and disease associations in idiopathic intracranial hypertension (pseudotumor cerebri): a case–control study. Neurology 41:239–244
Higgins JN, Cousins C, Owler BK, Sarkies N, Pickard JD (2003) Idiopathic intracranial hypertension: 12 cases treated by venous sinus stenting. J Neurol Neurosurg Psychiatry 74:1662–1666
Higgins JN, Owler BK, Cousins C, Pickard JD (2002) Venous sinus stenting for refractory benign intracranial hypertension. Lancet 359:228–230
Higgins JN, Pickard JD (2004) Lateral sinus stenoses in idiopathic intracranial hypertension resolving after CSF diversion. Neurology 62:1907–1908
Higgins JN, Tipper G, Varley M, Pickard JD (2005) Transverse sinus stenoses in benign intracranial hypertension demonstrated on CT venography. Br J Neurosurg 19:137–140
Karahalios DG, Rekate HL, Khayata MH, Apostolides PJ (1996) Elevated intracranial venous pressure as a universal mechanism in pseudotumor cerebri of varying etiologies. Neurology 46:198–202
King JO, Mitchell PJ, Thomson KR, Tress BM (1995) Cerebral venography and manometry in idiopathic intracranial hypertension. Neurology 45:2224–2228
King JO, Mitchell PJ, Thomson KR, Tress BM (2002) Manometry combined with cervical puncture in idiopathic intracranial hypertension. Neurology 58:26–30
Kornhuber ME, Markau S, Surov A, Herde J, Langer T (2007) Pseudotumor cerebri due to proximal haemodialysis fistula. Nephrol Dial Transplant 22:654
Kupersmith MJ, Gamell L, Turbin R, Peck V, Spiegel P, Wall M (1998) Effects of weight loss on the course of idiopathic intracranial hypertension in women. Neurology 50:1094–1098
Lam BL, Schatz NJ, Glaser JS, Bowen BC (1992) Pseudotumor cerebri from cranial venous obstruction. Ophthalmology 99:706–712
Malm J, Kristensen B, Markgren P, Ekstedt J (1992) CSF hydrodynamics in idiopathic intracranial hypertension: a long-term study. Neurology 42:851–858
McGonigal A, Bone I, Teasdale E (2004) Resolution of transverse sinus stenosis in idiopathic intracranial hypertension after L-P shunt. Neurology 62:514–515
Nedelmann M, Eicke BM, Dieterich M (2005) Functional and morphological criteria of internal jugular valve insufficiency as assessed by ultrasound. J Neuroimaging 15:70–75
Nedelmann M, Eicke BM, Dieterich M (2005) Increased incidence of jugular valve insufficiency in patients with transient global amnesia. J Neurol 252:1482–1486
Nedelmann M, Teschner D, Dieterich M (2007) Analysis of internal jugular vein insufficiency—a comparison of two ultrasound methods. Ultrasound Med Biol 33:857–862
Raichle ME, Grubb RL Jr, Phelps ME, Gado MH, Caronna JJ (1978) Cerebral hemodynamics and metabolism in pseudotumor cerebri. Ann Neurol 4:104–111
Rajpal S, Niemann DB, Turk AS (2005) Transverse venous sinus stent placement as treatment for benign intracranial hypertension in a young male: case report and review of the literature. J Neurosurg 102:342–346
Rohr A, Dorner L, Stingele R, Buhl R, Alfke K, Jansen O (2007) Reversibility of venous sinus obstruction in idiopathic intracranial hypertension. AJNR Am J Neuroradiol 28:656–659
Sander D, Winbeck K, Etgen T, Knapp R, Klingelhofer J, Conrad B (2000) Disturbance of venous flow patterns in patients with transient global amnesia. Lancet 356:1982–1984
Schreiber SJ, Doepp F, Klingebiel R, Valdueza JM (2005) Internal jugular vein valve incompetence and intracranial venous anatomy in transient global amnesia. J Neurol Neurosurg Psychiatry 76:509–513
Sugerman HJ, DeMaria EJ, Felton WL 3rd, Nakatsuka M, Sismanis A (1997) Increased intra-abdominal pressure and cardiac filling pressures in obesity-associated pseudotumor cerebri. Neurology 49:507–511
Sugerman HJ, Felton WL 3rd, Salvant JB Jr, Sismanis A, Kellum JM (1995) Effects of surgically induced weight loss on idiopathic intracranial hypertension in morbid obesity. Neurology 45:1655–1659
Sugerman HJ, Felton WL 3rd, Sismanis A, Kellum JM, DeMaria EJ, Sugerman EL (1999) Gastric surgery for pseudotumor cerebri associated with severe obesity. Ann Surg 229:634–640 discussion 640-632
Tintera J, Gawehn J, Bauermann T, Vucurevic G, Stoeter P (2004) New partially parallel acquisition technique in cerebral imaging: preliminary findings. Eur Radiol 14:2273–2281
Weiss KL, Wax MK, Haydon RC 3rd, Kaufman HH, Hurst MK (1993) Intracranial pressure changes during bilateral radical neck dissections. Head Neck 15:546–552
Wong R, Madill SA, Pandey P, Riordan-Eva P (2007) Idiopathic intracranial hypertension: the association between weight loss and the requirement for systemic treatment. BMC Ophthalmol 7:15
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nedelmann, M., Kaps, M. & Mueller-Forell, W. Venous obstruction and jugular valve insufficiency in idiopathic intracranial hypertension. J Neurol 256, 964–969 (2009). https://doi.org/10.1007/s00415-009-5056-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-009-5056-z