Abstract
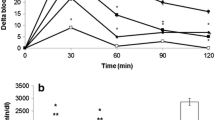
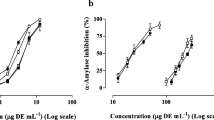
This study was designed to investigate whether the brown alga gamtae (Ecklonia cava) may inhibit α-glucosidase and α-amylase activities, and alleviate postprandial hyperglycemia in streptozotocin-induced diabetic mice. For that purpose, we prepared an enzymatic hydrolysate from gamtae (EHG) by using the carbohydrase, Celluclast. EHG evidenced prominent inhibitory effect against α-glucosidase and α-amylase. The IC50 values of EHG against α-glucosidase and α-amylase were 0.62 and 0.59 mg/mL, respectively, which evidenced the higher activities than that of acarbose. EHG did not exert any cytotoxic effect in human umbilical vein endothelial cells (HUVECs) at various concentrations (from 0.25 to 2 mg/mL). The increase of postprandial blood glucose levels were significantly suppressed in the EHG administered group than those in the streptozotocin-induced diabetic or normal mice. Moreover, the area under curve (AUC) was significantly reduced via EHG administration (6,102 vs. 10,425 mg·min/dL) in the diabetic mice as well as it delays absorption of dietary carbohydrates. These result indicated that EHG might be a potent inhibitor for α-glucosidase and α-amylase.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zimmet P, Alberti K, Shaw J. Global and societal implications of the diabetes epidemic. Nature 414: 782–787 (2001)
Baron AD. Postprandial hyperglycemia and α-glucosidase inhibitors. Diabetes Res. Clin. Pr. 40: S51–S55 (1998)
Bhandari MR, Jong-Anurakkun N, Hong G, Kawabata J. α-Glucosidase and α-amylase inhibitory activities of Nepalese medicinal herb pakhanbhed (Bergenia ciliata, Haw.). Food Chem. 106: 247–252 (2008)
Saito N, Sakai H, Sekihara H, Yajima Y. Effect of an α-glucosidase inhibitor (volibose), in combination with sulphonilureas, on glycaemic control in type 2 diabetes patients. J. Int. Med. Res. 26: 219–232 (1998)
Sels JP, Huijberts MS, Wolffenbuttel BH. Miglitol, a new α-glucosidase inhibitor. Expert Opin. Pharmaco. 1: 149–156 (1999)
Stand E, Baumgartl HJ, Fuchtenbusch M, Stemplinger J. Effect of acarbose on additional insulin therapy in type 2 diabetic patients with late failure of sulphonylurea therapy. Diabetes Obes. Metab. 1: 215–220 (1999)
Hanefeld M. The role of acarbose in the treatment of non-insulindependent diabetes mellitus. J. Diabetes Complicat. 12: 228–237 (1998)
Diaz-Gutierrez FL, Ladero JM, Diaz-Rubio M. Acarbose-induced acute hepatitis. Am. J. Gastroenterol. 93: 481–482 (1998)
Charpentier G, Riveline JP, Varroud-Vial M. Management of drugs affecting blood glucose in diabetic patients with renal failure. Diabetes Metab. 26: 73–85 (2000)
Matsui T, Tanaka T, Tamura S, Toshima A, Miyata Y, Tanaka K, Matsumoto K. α-Glucosidase inhibitory profile of catechins and theaflavins. J. Agr. Food. Chem. 55: 99–105 (2007)
Heo SJ, Hwang JY, Choi JI, Han JS, Kim HJ, Jeon YJ. Diphlorethohydroxycarmalol isolated from Ishige okamurae, a brown algae, a potent α-glucosidase and α-amylase inhibitor, alleviates postprandial hyperglycemia in diabetic mice. Eur. J. Pharmacol. 615: 252–256 (2009)
Lee SH, Park MH, Heo SJ, Kang SM, KO SC, Han JS, Jeon YJ. Dieckol isolated from Ecklonia cava inhibits α-glucosidase and α-amylase in vitro, and alleviates postprandial hyperglycemia in streptozotocin-induced diabetic mice. Food Chem. Toxicol. 48: 2633–2637 (2010)
Halliwell B, Gutteridge JMC. Free Radicals in Biology and Medicine. Halliwell B, Gutteridge JMC (eds). Oxford Science Publications, Oxford, UK. pp. 105–199 (1999)
Heo SJ, Park EJ, Lee KW, Jeon YJ. Antioxidant activities of enzymatic extracts from brown seaweeds. Bioresource Technol. 96: 1613–1623 (2005)
Kim KN, Heo SJ, Song CB, Lee JH, Heo MS, Yeo IK, Kang KA, Hyun JW, Jeon YJ. Protective effect of Ecklonia cava enzymatic extracts on hydrogen peroxide-induced cell damage. Process Biochem. 41: 2393–2401 (2006)
Athukorala Y, Jung WK, Vasanthan T, Jeon YJ. An anticoagulative polysaccharide from an enzymatic hydrolysate of Ecklonia cava. Carbohyd. Polym. 66: 184–191 (2006)
Athukorala Y, Kim KN, Jeon YJ. Antiproliferative and antioxidant properties of an enzymatic hydrolysate from brown alga, Ecklonia cava. Food Chem. Toxicol. 44: 1065–1074 (2006)
Ahn GN, Hwang IS, Park EJ, Kim JH, Jeon YJ, Lee JH, Park JW, Jee YH. Immunomodulatory effects of an enzymatic extract from Ecklonia cava on murine splenocytes. Mar. Biotechnol. 10: 278–289 (2008)
Lee SH, Heo SJ, Hwang JY, Han JS, Jeon YJ. Protective effects of enzymatic digest from Ecklonia cava against high glucose-induced oxidative stress in human umbilical vein endothelial cells. J. Sci. Food. Agr. 90: 349–356 (2010)
Watanabe J, Kawabata J, Kurihara H, Niki R. Isolation and identification of α-glucosidase inhibitors from tochucha (Eucommia ulmoides). Biosci. Biotech. Bioch. 61: 177–178 (1997)
Fautz R, Husen B, Hechenberger C. Application of the neutral red assay (NR assay) to monolayer cultures of primary hepatocytes: Rapid colorimetric viability determination for the unscheduled DNA synthesis test (UDS). Mutat. Res. 253: 173–179 (1991)
Kim JS. Effect of Rhemanniae Radix on the hyperglycemic mice induced with streptozotocin. J. Korean Soc. Food Sci. Nutr. 33: 1133–1138 (2004)
Gray DM. Carbohydrate digestion and absorption role of small intestine. New Engl. J. Med. 29: 1225–1230 (1995)
Krentz AJ, Bailey CJ. Oral antidiabetic agents. Current role in type 2 diabetes mellitus. Drugs 65: 385–411 (2005)
Lebovitz HE. α-Glucosidase inhibitors. Endocrin. Metab. Clin. 26: 539–551 (1997)
Hara Y, Honda M. The inhibition of α-amylase by tea polyphenols. Agric. Biol. Chem. Tokyo 54: 1939–1945 (1990)
Inoue I, Takahashi K, Noji S, Awata T, Negishi K, Katayama S. Acarbose controls postprandial hyper-proinsulinemia in non-insulindependent diabetes mellitus. Diabetes Res. Clin. Pr. 36: 143–151 (1997)
Dennis JW, Laferte S, Waghorne C, Breitman ML, Kerbel RS. β1-6 Branching of Asn-linked oligosaccharides is directly associated with metastasis. Science 236: 582–585 (1987)
Gruters RA, Neefjes JJ, Tersmette M, De Goede REY, Tulp A, Huisman HG, Miedema F, Ploegh HL. Interference with HIVinduced syncytium formation and viral infectivity by inhibitors of trimming glucosidase. Nature 330: 74–77 (1987)
Winchester B, Fleet GW. Amino-sugar glycosidase inhibitors: Versatile tools for glycobiologists. Glycobiology 2: 199–210 (1992)
Li Y, Wen S, Kota BP, Peng G, Li GQ, Yamahara J. Punica granatum flower extract, a potent α-glucosidase inhibitor, improves postprandial hyperglycemia in Zucker diabetic fatty rats. J. Ethnopharmacol. 99: 239–244 (2005)
Cherng JY, Shih MF. Improving glycogenesis in Streptozotocin (STZ) diabetic mice after administration of green algae Chlorella. Life Sci. 78: 1181–1186 (2006)
Kang SI, Jin YJ, Ko HC, Choi SY, Hwang JH, Whang I, Kim MH, Shin HS, Jeong HB, Kim SJ. Petalonia improves glucose homeostasis in streptozotocin-induced diabetic mice. Biochem. Bioph. Res. Co. 373: 264–269 (2008)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, SH., Park, MH., Han, JS. et al. Bioactive compounds extracted from Gamtae (Ecklonia cava) by using enzymatic hydrolysis, a potent α-glucosidase and α-amylase inhibitor, alleviates postprandial hyperglycemia in diabetic mice. Food Sci Biotechnol 21, 1149–1155 (2012). https://doi.org/10.1007/s10068-012-0150-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-012-0150-x