Abstract
Magnetic resonance imaging (MRI) with maximum intensity projection (MIP) is used to evaluate the hand in rheumatoid arthritis (RA). MIP yields clear visualization of synovitis over the entirety of the bilateral hands with a single image. In this study, we assessed synovitis with MIP images, clinical findings, and power Doppler (PD) findings to examine the clinical usefulness of MIP images for RA in the hand. Thirty RA patients were assessed for swelling and tenderness in the joints included in the DAS28, and both contrast-enhanced MRI for bilateral hands and ultrasonography for bilateral wrist and metacarpophalangeal (MCP) joints were performed. Articular synovitis was scored in MIP images, and the scores were compared with those for PD. The agreement on synovitis between MIP and conventional MR images was excellent. Palpation showed low sensitivity and high specificity compared with both MIP and PD images. There were joints that were positive in MIP images only, but there were no joints that were positive in PD images only. A statistically significant correlation between the scores of MIP and PD images was found. Furthermore, the agreement between grade 2 on MIP images and positive on PD images was 0.87 (κ = 0.73) for the wrist and 0.92 (κ = 0.57) for MCP joints. Using MIP images together with palpation makes detailed evaluation of synovitis of the hand in RA easy. MIP images may predict further joint damage, since they allow semiquantitative estimation of the degree of thickening of the synovial membrane.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The hand is the most common site of rheumatoid arthritis (RA), therefore evaluation of arthritis at this site is important for diagnosis and estimation of disease activity. Physical findings and conventional radiographs have been used to evaluate the hands in RA; however, more precise methods of evaluation are now needed because of remarkable advances in RA treatment in recent years [1, 2]. As new imaging methods, magnetic resonance imaging (MRI) and joint ultrasonography (US) are currently used in RA [3]. Synovial inflammation and bone edema can be confirmed with contrast-enhanced MRI, which is not possible with plain radiographs, and it can confirm bone erosion at an earlier stage than plain radiographs [3, 4]. The correlation between synovial membrane thickness and joint destruction has been reported, and several methods to quantify thickness with contrast-enhanced MR images have been reported [5–8]. However, these methods take too long to use during outpatient treatment because quantification involves interpretation of numerous images.
We have used contrast-enhanced MR images and the images processed with maximum intensity projection (MIP), together with physical findings and plain radiographs, to evaluate the hands in RA since 2002 [9]. MIP is a method of image processing in which the brightest regions of different slices are superimposed on each other to create a single three-dimensional (3D) image [10–12]. MIP images of the hand in RA provide clear visualization of synovitis and also enable differentiation of articular synovitis and tenosynovitis anatomically [13–15]. A single MIP image of the hands allows observation of the whole of both hands, like plain radiographs. For these reasons, synovitis can be diagnosed easily in MIP images; additionally, the distribution of synovitis is very important for diagnosing whether the synovitis is due to RA, infection or other arthritis, because RA is a disease with polyarthritis [9, 13]. The time required for interpretation is less than 1/4 of that with regular contrast-enhanced MR images, and the diagnostic accuracy is equivalent to that with regular images [14]. Clinically, to diagnose precisely and efficiently, we first assess MIP images and then we check conventional MR images with reference to the MIP images. Incorporating MIP images in picture archive and communication systems (PACS) or other image systems permits easy review of the synovitis and makes it easier for both physicians and patients to understand the state of disease.
As well as MR images, we have recently been using US to evaluate synovitis. In general, gray scale (GS) and power Doppler (PD) images are recorded with US. PD-positive synovitis, in particular, is important because the findings are considered to indicate progressive joint destruction, even if the patient is in clinical remission [8, 16–19]. In this study, we compared MIP images, palpation findings, and PD findings of synovitis to examine the clinical usefulness of MIP images for evaluating the hands in RA. We also used a simple method to score MIP images of synovitis in the hands in RA and compared these scores with those from PD images to examine the clinical significance of synovitis on MIP images.
Materials and methods
Patients
Thirty RA patients were included in the study. All patients fulfilled the American Rheumatism Association 1987 criteria for RA [20]. For future prospective studies, we selected patients who were not receiving biological agents or oral doses of steroids. Tenderness and swelling in the 28 joints in the disease activity score (DAS)28 [21] were recorded, and C-reactive protein (CRP) levels were obtained; the DAS28-CRP was then calculated from these data to evaluate disease activity in each patient. All patients were examined using both MRI and US.
All patients gave informed consent, and the study was approved by the local Research Ethics Committee of our hospital.
MIP images
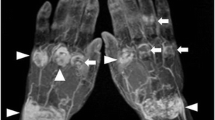
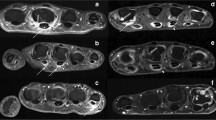
All MRI examinations of hands were performed in the supine position using a 1.5-T clinical scanner (EXCELART Vantage powered by Atlas; Toshiba, Japan), with the Atlas SPEEDER 16-channel phased array coil. Patients placed both hands on the front of their thighs. Before obtaining the contrast-enhanced fat-suppressed 3D T1-weighted images (T1WI), we obtained non-contrast-enhanced STIR, T1WI, and T2WI with coronal orientation. In all patients, an intravenous bolus of 0.1 mmol gadoteridol/kg body weight (Prohance, Eisai, Japan, Tokyo) was injected, and then, fat-suppressed contrast-enhanced 3D gradient echo T1WI were started acquisition within 3 min of injection of the contrast medium. A fat-suppressed 3D gradient echo T1WI sequence was conducted with the following parameters: repetition time/echo time (TR/TE) = 5.5/2.5 ms, number of acquisitions = 1, slice thickness = 2 mm, field-of-view = 350 × 350 mm, acquisition matrix = 256 × 256, and acquisition time = approximately 3 min 40 s. The MIP images were post-processed from fat-suppressed 3D gradient echo T1WI. In MIP images, each joint (wrist, metacarpophalangeal [MCP] joint and proximal interphalangeal [PIP] joint) was scored separately for the presence of articular synovitis on a semiquantitative scale. Since there are no previous reports of a scoring method, we used an original scale from 0 to 2 (grade 0 = no enhancement, grade 1 = partial enhancement of the joint, grade 2 = complete enhancement of the joint) (Fig. 1). We considered grade 1 or 2 to be positive for articular synovitis. Furthermore, we scored articular synovitis with fat-suppressed contrast-enhanced 3D gradient echo T1WI with reference to a previous report [14], in order to compare with the MIP scores. Two orthopedic surgeons specializing in RA conducted scoring independently, without reference to any other clinical information.
Power doppler images
US was performed on the wrist and MCP joints using a HI VISION Avius (Hitachi Medical Corporation, Tokyo, Japan) with a linear type (14–6 MHz) probe. Patients were examined while seated with the hand placed on a cushion and pronated. The dorsal side of the hand was scanned in the longitudinal plane. Radial images of the wrist were taken with Lister’s tubercle and the second metacarpal bone as landmarks, medial images of the wrist were taken around the center of the wrist, and ulnar images of the wrist were taken with the head of the ulna as the landmark. Images of the MCP joint were taken around the center of each joint. We searched for the most active area of inflammation near these landmarks. GS images with low echo regions within joints were considered to indicate thickening of the synovial membrane. In PD images, the presence of vascular signals was examined. When performing PD evaluation, the receiver gain settings were controlled to eliminate the appearance of artifacts on each joint. The PD frequency was set at 7.5 MHz, and the pulse repetition frequency was set between 800 Hz and 1,000 Hz, optimized for US of rheumatoid hands by the manufacturer. For PD images, each joint was scored on a semiquantitative scale with a score of grade 1 or higher taken as positive (grade 0 = no flow in the synovium, grade 1 = single vessel signals, grade 2 = confluent vessel signals in less than half of the area of the synovium, grade 3 = vessel signals in more than half of the area of the synovium) [22]. Intensive training is required for adequate assessment of rheumatoid hands [23, 24]. Therefore, all US examinations were performed by the same orthopedic surgeon who was trained in the examination of the small joints of rheumatoid hands. Two other orthopedic surgeons specializing in RA scored the joints independently, with no reference to any other clinical information.
Statistical analysis
The baseline characteristics of our study group are described as mean values for continuous variables including standard deviations. The agreement was estimated using the weighted kappa statistic. A κ value of 0–0.40 denotes poor agreement, 0.41–0.60 moderate, 0.61–0.80 substantial, and 0.81 or higher excellent agreement [20]. The correlation between two imaging scores was estimated using Spearman’s rank correlation coefficient test. To estimate the agreement between two imaging methods, final score decision was made after discussion between the two examiners. Statistical analysis was performed using the Ekuseru-Toukei 2012 software for Windows (Social Survey Research Information Co., Ltd.,Tokyo, Japan).
Results
Our study included 25 women and 5 men. Their mean age was 61.5 ± 9.5 years (range 38–81 years), and their mean disease duration was 12.5 ± 11.5 years (range 4 months–45 years). Twenty-seven patients were being treated with methotrexate, at a mean dose of 7.3 ± 3.1 mg (range 2–14 mg). Steinbrocker classifications were stage 1 for 6 patients, stage 2 for 11, stage 3 for 7, and stage 4 for 6, and class 1 for 13, class 2 for 13, class 3 for 3, and class 4 for 1. The mean DAS28-CRP score for the study group was 2.23 ± 1.01 (range 0.96–5.37). There were 18 patients in remission with DAS28-CRP scores of lower than 2.3, 1 with low disease activity with a score of 2.3–2.7, 10 with moderate disease activity with scores of 2.7–4.1, and 1 with high disease activity with a score of more than 4.1.
A total of 60 wrists and 300 MCP joints were evaluated with clinical examination, MRI, and US. Three hundred PIP joints (including 60 interphalangeal [IP] joints in the thumb) were evaluated with clinical examination and MRI. Interobserver agreement was excellent for MIP image scores, conventional MRI scores, and PD image scores with a κ value of more than 0.81 at each joint area. There were statistically significant correlations between the scores for MIP images and conventional MR images for the wrist (r s = 0.904, P < 0.001), MCP joints (r s = 0.919, P < 0.001), and PIP joints (r s = 0.930, P < 0.001). Furthermore, the κ value for synovitis scores from MIP images and conventional MR images was 0.92 for the wrist, 0.88 for MCP joints, and 0.88 for PIP joints. Therefore, the agreement on synovitis scores between MIP images and conventional MR images was excellent.
Synovitis was visualized on MIP images in 48 (wrist), 84 (MCP), and 59 (PIP) joints, whereas only 25 (wrist), 26 (MCP), and 10 (PIP) joints exhibited synovitis on clinical assessment (swelling or tenderness) (Fig. 2). Using either MIP or PD images as the reference, clinical examination showed low sensitivity and high specificity in each joint area (Table 1). Using MIP images as a reference, the sensitivity of palpation was 0.52 for the wrist, 0.26 for MCP joints, and 0.12 for PIP joints; thus, the smaller the joint, the lower the sensitivity of palpation (Table 1).
The number of joints positive on MIP images and PD images is shown in Table 2. In PD images, 31 wrists and 23 MCP joints were positive for synovitis, all of which were also positive in MIP images; there were joints that were positive in MIP images only, but there were no joints that were positive in PD images only (Table 2). There were statistically significant correlations between the scores for MIP images and PD images for both wrists (r s = 0.701, P < 0.001) and MCP joints (r s = 0.518, P < 0.001). The agreement on synovitis between MIP and PD images was moderate at 0.73 (κ = 0.44) for the wrist and poor at 0.80 (κ = 0.35) for MCP joints. However, the agreement between grade 2 on MIP images and positive on PD images was substantial at 0.87 (κ = 0.73) for the wrist and moderate at 0.92 (κ = 0.57) for MCP joints.
Discussion
Clinical findings are essential for evaluating disease activity, but our results show that MIP imaging is more sensitive than palpation in detecting synovitis. This finding is consistent with previous studies regarding imaging of the joints in RA [3, 9, 15, 21, 25–28]. It is also known that most RA patients who satisfied the remission criteria with normal findings on clinical and laboratory studies had imaging-detected synovitis that predicts subsequent joint damage [3, 28]. The high sensitivity of MIP imaging in the wrist and MCP joints is not surprising because thick palmar ligaments exist in the wrist, and each joint exists side by side in MCP joints. It is striking that the smaller the joints were, the less sensitive clinical findings became, even though PIP joints could be examined from all around the joint.
Contrast-enhanced MRI is an imaging modality for visualization of the inflamed synovium in which there is increased blood flow and increased capillary permeability [13], [29]. A correlation between the thickness of the synovial membrane detected with contrast-enhanced MRI and joint destruction has been reported [5–7]. Several assessments for synovitis have been used, such as rheumatoid arthritis MRI score (RAMRIS), which provides semiquantitative assessments [30–32]; measuring the maximum enhanced thickness of the synovium [6]; and the total volume of the synovial membrane calculated by summation of each slice [7]. All of these methods take time, which makes them difficult to use in busy outpatient settings. Our scoring of synovitis in MIP images is semiquantitative for detecting the volume of the synovial membrane, since the MIP image is a superimposed single 3D image. This scoring is convenient and useful because we can assess simply, using only a single image, and the interobserver agreement is excellent. However, there may be drawbacks with MIP images. Firstly, hyperemia may imitate synovitis, and secondly, the detailed location of the affected anatomical structure is restricted in MIP images. Therefore, we must cross-check MIP images with reference to conventional MR images, although the agreement on synovitis between MIP and conventional MR images was excellent.
US examination is less expensive and more convenient for patients than MRI and can be easily used for repeated examinations. GS and PD images are recorded for the assessment of joint inflammation. Synovial hypertrophy is evaluated primarily on GS images, while PD images are utilized to demonstrate activity related to synovial hypertrophy [3, 33, 34]. The clinical significance of GS findings remains disputed [15, 35], and there is also the problem of interobserver agreement [22, 23] (interobserver agreement of GS scores was moderate in this study [data not shown]). The presence of microvascular blood flow in synovial hypertrophy is interpreted as active synovitis and predicts ongoing joint damage even in patients in clinical remission [3, 8, 16, 17, 19, 28, 36, 37]. A high agreement between PD and contrast-enhanced MRI findings of synovitis has been reported [3, 38, 39]. In this study, a statistically significant correlation between MIP and PD findings was shown, and the agreement between grade 2 on MIP images and positive on PD images was substantial for the wrist and moderate for MCP joints. That is, intensive enhancement on MIP images could be a risk factor for further joint damage.
Our results showed that all PD-positive joints were also MIP-positive. We propose two reasons for this result. Firstly, the sensitivity of MIP is so high that it includes inactive synovial membrane thickness. It is not yet clear if there is a level of MRI-detected synovitis below which patients will not show progressive joint destruction [40]. Secondly, the sensitivity of PD is too low. Several factors are known to influence the sensitivity of detecting synovitis by GS and PD. Equipment characteristics and resolution, as well as varying parameter settings, affect sensitivity [34]. Recently, global US scoring systems for synovitis in RA have been challenging to use. Examining a large number of joints takes a considerable amount of time [21, 23, 38]. The minimum number of joints required to evaluate global disease activity is currently being discussed [34]. Also, MRI provides a local, not global, estimation of synovitis; therefore, MRI imaging cannot be used to evaluate global disease activity. One advantage of MRI is that MR images can be reread remotely at a later date; in contrast, US cannot identify abnormalities that are overlooked and not imaged at the time of the original examination. However, increasing use of ultrasound video-loops and 3D ultrasound may assist in this regard in the future [41].
One of the limitations of our study is that the PD images were recorded using a machine widely used in Japan. More sensitive PD signals may be observed using a very high-end machine in the future. In addition to equipment-dependent effects, operator-dependent factors, including factors affecting both image acquisition and interpretation, have to be considered, although all US examinations in our study were performed by a specialist trained in the US examination of rheumatoid hands. Another limitation of this study is that partial enhancement of the joint was graded as grade 1, and complete enhancement of the joint was graded as grade 2, but prospective studies should be conducted to determine the clinical significance of this scoring system. Furthermore, the significance of joints in which PD images are negative and MIP images are positive must be also clarified in a prospective study.
We conclude that using MIP images together with palpation, plain radiographs and conventional MR images allows detailed and comprehensive evaluation of the hand in RA. MIP images may predict ongoing joint damage, since they permit easy semiquantitative evaluation of the degree of thickening of the synovial membrane.
References
Smolen JS, Landewe R, Breedveld FC, Dougados M, Emery P, Gaujoux-Viala C, Gorter S, Knevel R, Nam J, Schoels M, Aletaha D, Buch M, Gossec L, Huizinga T, Bijlsma JW, Burmester G, Combe B, Cutolo M, Gabay C, Gomez-Reino J, Kouloumas M, Kvien TK, Martin-Mola E, McInnes I, Pavelka K, van Riel P, Scholte M, Scott DL, Sokka T, Valesini G, van Vollenhoven R, Winthrop KL, Wong J, Zink A, van der Heijde D (2010) EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Ann Rheum Dis 69:964–975
Smolen JS, Aletaha D, Bijlsma JW, Breedveld FC, Boumpas D, Burmester G, Combe B, Cutolo M, de Wit M, Dougados M, Emery P, Gibofsky A, Gomez-Reino JJ, Haraoui B, Kalden J, Keystone EC, Kvien TK, McInnes I, Martin-Mola E, Montecucco C, Schoels M, van der Heijde D (2010) Treating rheumatoid arthritis to target: recommendations of an international task force. Ann Rheum Dis 69:631–637
Colebatch AN, Edwards CJ, Ostergaard M, van der Heijde D, Balint PV, D’Agostino MA, Forslind K, Grassi W, Haavardsholm EA, Haugeberg G, Jurik AG, Landewe RB, Naredo E, O’Connor PJ, Ostendorf B, Potocki K, Schmidt WA, Smolen JS, Sokolovic S, Watt I, Conaghan PG (2013) EULAR recommendations for the use of imaging of the joints in the clinical management of rheumatoid arthritis. Annals of the rheumatic diseases
Suter LG, Fraenkel L, Braithwaite RS (2011) Role of magnetic resonance imaging in the diagnosis and prognosis of rheumatoid arthritis. Arthritis Care Res 63:675–688
Boyesen P, Haavardsholm EA, Ostergaard M, van der Heijde D, Sesseng S, Kvien TK (2011) MRI in early rheumatoid arthritis: synovitis and bone marrow oedema are independent predictors of subsequent radiographic progression. Ann Rheum Dis 70:428–433
Conaghan PG, O’Connor P, McGonagle D, Astin P, Wakefield RJ, Gibbon WW, Quinn M, Karim Z, Green MJ, Proudman S, Isaacs J, Emery P (2003) Elucidation of the relationship between synovitis and bone damage: a randomized magnetic resonance imaging study of individual joints in patients with early rheumatoid arthritis. Arthritis Rheum 48:64–71
Ostergaard M, Hansen M, Stoltenberg M, Gideon P, Klarlund M, Jensen KE, Lorenzen I (1999) Magnetic resonance imaging-determined synovial membrane volume as a marker of disease activity and a predictor of progressive joint destruction in the wrists of patients with rheumatoid arthritis. Arthritis Rheum 42:918–929
Brown AK, Conaghan PG, Karim Z, Quinn MA, Ikeda K, Peterfy CG, Hensor E, Wakefield RJ, O’Connor PJ, Emery P (2008) An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum 58:2958–2967
Mori G, Tokunaga D, Takahashi KA, Hojo T, Fujiwara H, Arai Y, Taniguchi D, Takatori R, Imai K, Otakara E, Ito H, Nishimura T, Kubo T (2008) Maximum intensity projection as a tool to diagnose early rheumatoid arthritis. Mod Rheumatol Jpn RheumAssoc 18:247–251
Okumura A, Watanabe Y, Dohke M, Ishimori T, Amoh Y, Oda K, Dodo Y (1999) Contrast-enhanced three-dimensional MR portography. Radiographics: Rev Publ Radiol Soc North Am Inc 19:973–987
Aufort S, Charra L, Lesnik A, Bruel JM, Taourel P (2005) Multidetector CT of bowel obstruction: value of post-processing. Eur Radiol 15:2323–2329
Ferencik M, Ropers D, Abbara S, Cury RC, Hoffmann U, Nieman K, Brady TJ, Moselewski F, Daniel WG, Achenbach S (2007) Diagnostic accuracy of image postprocessing methods for the detection of coronary artery stenoses by using multidetector CT. Radiology 243:696–702
Navalho M, Resende C, Rodrigues AM, Ramos F, Gaspar A, Pereira da Silva JA, Fonseca JE, Campos J, Canhao H (2012) Bilateral MR imaging of the hand and wrist in early and very early inflammatory arthritis: tenosynovitis is associated with progression to rheumatoid arthritis. Radiology 264:823–833
Karlo C, Zanetti M, Stolzmann P, Steurer-Dober I, Brunner F, Hodler J, Pfirrmann CW (2011) Synovitis maps for the assessment of inflammatory diseases of the hand. Eur Radiol 21:1499–1508
Backhaus M, Kamradt T, Sandrock D, Loreck D, Fritz J, Wolf KJ, Raber H, Hamm B, Burmester GR, Bollow M (1999) Arthritis of the finger joints: a comprehensive approach comparing conventional radiography, scintigraphy, ultrasound, and contrast-enhanced magnetic resonance imaging. Arthritis Rheum 42:1232–1245
Naredo E, Collado P, Cruz A, Palop MJ, Cabero F, Richi P, Carmona L, Crespo M (2007) Longitudinal power Doppler ultrasonographic assessment of joint inflammatory activity in early rheumatoid arthritis: predictive value in disease activity and radiologic progression. Arthritis Rheum 57:116–124
Scire CA, Montecucco C, Codullo V, Epis O, Todoerti M, Caporali R (2009) Ultrasonographic evaluation of joint involvement in early rheumatoid arthritis in clinical remission: power Doppler signal predicts short-term relapse. Rheumatol (Oxford, England) 48:1092–1097
Foltz V, Gandjbakhch F, Etchepare F, Rosenberg C, Tanguy ML, Rozenberg S, Bourgeois P, Fautrel B (2012) Power Doppler ultrasound, but not low-field magnetic resonance imaging, predicts relapse and radiographic disease progression in rheumatoid arthritis patients with low levels of disease activity. Arthritis Rheum 64:67–76
Peluso G, Michelutti A, Bosello S, Gremese E, Tolusso B, Ferraccioli G (2011) Clinical and ultrasonographic remission determines different chances of relapse in early and long standing rheumatoid arthritis. Ann Rheum Dis 70:172–175
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Wakefield RJ, Green MJ, Marzo-Ortega H, Conaghan PG, Gibbon WW, McGonagle D, Proudman S, Emery P (2004) Should oligoarthritis be reclassified? Ultrasound reveals a high prevalence of subclinical disease. Ann Rheum Dis 63:382–385
Szkudlarek M, Court-Payen M, Jacobsen S, Klarlund M, Thomsen HS, Ostergaard M (2003) Interobserver agreement in ultrasonography of the finger and toe joints in rheumatoid arthritis. Arthritis Rheum 48:955–962
D’Agostino MA, Maillefert JF, Said-Nahal R, Breban M, Ravaud P, Dougados M (2004) Detection of small joint synovitis by ultrasonography: the learning curve of rheumatologists. Ann Rheum Dis 63:1284–1287
Naredo E, Moller I, Moragues C, de Agustin JJ, Scheel AK, Grassi W, de Miguel E, Backhaus M, Balint P, Bruyn GA, D’Agostino MA, Filippucci E, Iagnocco A, Kane D, Koski JM, Mayordomo L, Schmidt WA, Swen WA, Szkudlarek M, Terslev L, Torp-Pedersen S, Uson J, Wakefield RJ, Werner C (2006) Interobserver reliability in musculoskeletal ultrasonography: results from a “Teach the Teachers” rheumatologist course. Ann Rheum Dis 65:14–19
Lindegaard H, Vallo J, Horslev-Petersen K, Junker P, Ostergaard M (2001) Low field dedicated magnetic resonance imaging in untreated rheumatoid arthritis of recent onset. Ann Rheum Dis 60:770–776
Szkudlarek M, Narvestad E, Klarlund M, Court-Payen M, Thomsen HS, Ostergaard M (2004) Ultrasonography of the metatarsophalangeal joints in rheumatoid arthritis: comparison with magnetic resonance imaging, conventional radiography, and clinical examination. Arthritis Rheum 50:2103–2112
Ostendorf B, Scherer A, Modder U, Schneider M (2004) Diagnostic value of magnetic resonance imaging of the forefeet in early rheumatoid arthritis when findings on imaging of the metacarpophalangeal joints of the hands remain normal. Arthritis Rheum 50:2094–2102
Brown AK, Quinn MA, Karim Z, Conaghan PG, Peterfy CG, Hensor E, Wakefield RJ, O’Connor PJ, Emery P (2006) Presence of significant synovitis in rheumatoid arthritis patients with disease-modifying antirheumatic drug-induced clinical remission: evidence from an imaging study may explain structural progression. Arthritis Rheum 54:3761–3773
Ostendorf B, Peters R, Dann P, Becker A, Scherer A, Wedekind F, Friemann J, Schulitz KP, Modder U, Schneider M (2001) Magnetic resonance imaging and miniarthroscopy of metacarpophalangeal joints: sensitive detection of morphologic changes in rheumatoid arthritis. Arthritis Rheum 44:2492–2502
Ostergaard M, Peterfy C, Conaghan P, McQueen F, Bird P, Ejbjerg B, Shnier R, O’Connor P, Klarlund M, Emery P, Genant H, Lassere M, Edmonds J (2003) OMERACT Rheumatoid Arthritis Magnetic Resonance Imaging Studies. Core set of MRI acquisitions, joint pathology definitions, and the OMERACT RA-MRI scoring system. J Rheumatol 30:1385–1386
Conaghan P, Bird P, Ejbjerg B, O’Connor P, Peterfy C, McQueen F, Lassere M, Emery P, Shnier R, Edmonds J, Ostergaard M (2005) The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the metacarpophalangeal joints. Ann Rheum Dis 64(Suppl 1):i11–i21
Ejbjerg B, McQueen F, Lassere M, Haavardsholm E, Conaghan P, O’Connor P, Bird P, Peterfy C, Edmonds J, Szkudlarek M, Genant H, Emery P, Ostergaard M (2005) The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the wrist joint. Ann Rheum Dis 64(Suppl 1):i23–i47
Walther M, Harms H, Krenn V, Radke S, Faehndrich TP, Gohlke F (2001) Correlation of power Doppler sonography with vascularity of the synovial tissue of the knee joint in patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum 44:331–338
Mandl P, Naredo E, Wakefield RJ, Conaghan PG, D’Agostino MA (2011) A systematic literature review analysis of ultrasound joint count and scoring systems to assess synovitis in rheumatoid arthritis according to the OMERACT filter. J Rheumatol 38:2055–2062
Szkudlarek M, Klarlund M, Narvestad E, Court-Payen M, Strandberg C, Jensen KE, Thomsen HS, Ostergaard M (2006) Ultrasonography of the metacarpophalangeal and proximal interphalangeal joints in rheumatoid arthritis: a comparison with magnetic resonance imaging, conventional radiography and clinical examination. Arthritis Res Ther 8:R52
Taylor PC, Steuer A, Gruber J, Cosgrove DO, Blomley MJ, Marsters PA, Wagner CL, McClinton C, Maini RN (2004) Comparison of ultrasonographic assessment of synovitis and joint vascularity with radiographic evaluation in a randomized, placebo-controlled study of infliximab therapy in early rheumatoid arthritis. Arthritis Rheum 50:1107–1116
Naredo E, Moller I, Cruz A, Carmona L, Garrido J (2008) Power Doppler ultrasonographic monitoring of response to anti-tumor necrosis factor therapy in patients with rheumatoid arthritis. Arthritis Rheum 58:2248–2256
Terslev L, Torp-Pedersen S, Savnik A, von der Recke P, Qvistgaard E, Danneskiold-Samsoe B, Bliddal H (2003) Doppler ultrasound and magnetic resonance imaging of synovial inflammation of the hand in rheumatoid arthritis: a comparative study. Arthritis Rheum 48:2434–2441
Horikoshi M, Suzuki T, Sugihara M, Kondo Y, Tsuboi H, Uehara T, Hama M, Takase K, Ohno S, Ishigatsubo Y, Yoshida Y, Sagawa A, Ikeda K, Ota T, Matsumoto I, Ito S, Sumida T (2010) Comparison of low-field dedicated extremity magnetic resonance imaging with articular ultrasonography in patients with rheumatoid arthritis. Mod Rheumatol Jpn Rheum Assoc 20:556–560
Conaghan PG, McQueen FM, Bird P, Peterfy CG, Haavardsholm EA, Gandjbakhch F, Boyesen P, Coates L, Ejbjerg B, Eshed I, Foltz V, Hermann KG, Freeston J, Lillegraven S, Lassere M, Wiell C, Anandarajah A, Duer-Jensen A, O’Connor P, Genant HK, Emery P, Ostergaard M (2011) Update on research and future directions of the OMERACT MRI inflammatory arthritis group. J Rheumatol 38:2031–2033
McNally EG (2008) Ultrasound of the small joints of the hands and feet: current status. Skelet Radiol 37:99–113
Disclosures
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taniguchi, D., Tokunaga, D., Oda, R. et al. Maximum intensity projection with magnetic resonance imaging for evaluating synovitis of the hand in rheumatoid arthritis: comparison with clinical and ultrasound findings. Clin Rheumatol 33, 911–917 (2014). https://doi.org/10.1007/s10067-014-2526-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-014-2526-1