Abstract
The aim of this study is to evaluate the diagnostic value of quantifying salivary gland scintigraphy in correlation to the labial biopsy findings of Sjögren’s syndrome (SS). Thirty patients suspected of having SS referred to our clinic for salivary gland scintigraphy were included to this study. All patients underwent salivary gland biopsy as well. The severity of histopathologic changes was graded according to the Chisholm and Mason scoring system. Dynamic scintigraphy was performed and region of interests (ROI) were drawn. Time activity curves for salivary glands were generated. Count rates of maximum, minimum activity after lemon juice stimuli, and last minute activities of parotid and submandibular glands were obtained. On the basis of this ROI counts, excretion fraction (EF%) was calculated for all salivary glands. The mean EF% for normal parotid gland and pathologic parotid gland was 54.5 ± 13.9 and 45.8 ± 18.42, respectively, while it was 46.7 ± 11.7 for the normal submandibular gland and 29.3 ± 18.8 for the pathologic submandibular gland. With progression in histopathologic grades from 0 to 4, the EF decreased in all salivary glands. Decreased EF in the salivary glands is correlated with the SS, and salivary gland scintigraphy is a sensitive and valid method for evaluation of the function of the salivary glands.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sjögren’s syndrome (SS) is an inflammatory autoimmune disease of unknown etiology characterised by lymphocytic infiltration and destruction of exocrine glands [1]. The target organs are the salivary and lacrimal glands, resulting particularly as a reduction of salivary function.
The diagnosis of SS depends not only on demonstrating an exocrinopathy, but also it is autoimmune in nature. The American/European consensus criteria are widely accepted [2] which are based on a positive response to a short questionnaire of ocular and oral symptoms, objective measurement of impaired lacrimal and salivary flow rates, and demonstration of autoimmunity by significant focal infiltrates on salivary gland biopsy and the presence of antibodies.
Reliable detection of impaired salivary function is crucial for the diagnosis of xerostomia in patients with early SS. The most specific diagnostic method for SS is still labial salivary gland biopsy, but it is invasive and causes some degree of patient discomfort.
Salivary gland scintigraphy (SGS) provides a detailed functional evaluation of salivation as it measures the amount and speed of radioisotope uptake and excretion for all four major salivary glands separately [3]. Salivary gland scintigraphy using qualitative or quantitative methods has been suggested as a sensitive, safe, easy to perform and objective alternative approach to functional and morphological evaluation of salivary/involvement in SS [4–9]. The aim of this study is to evaluate the diagnostic value of quantitative salivary gland uptake and secretion in patients with sicca symptoms in correlation to the labial biopsy of Sjögren’s syndrome.
Material and methods
Files of 60 patients referred to the nuclear medicine department for salivary gland scintigraphy for the differential diagnosis of dry mouth were retrospectively reviewed. Thirty patients who had both salivary gland biopsy and scintigraphy were enrolled to this study. There were 25 female and 5 male patients with a mean age of 46 years (range, 25–74) (Table 1). All had complaints of dry mouth and dry eye for a period of 12 to 36 months.
Salivary gland scintigraphy
All patients were well hydrated before salivary gland scintigraphy. Dynamic imaging was performed in modified Water’s projection after the i.v. administration of 555 mBq 99mTc pertechnetate. Imaging was started immediately after injection in order not to miss the initial portion of the curve. A single-headed gamma camera (Mediso, X-Ring/R Hungary) was used with a symmetrical 20 % window around a 140-keV photopeak. Images were acquired on 128 × 128 pixel matrix and archived for later retrieval. After i.v. injection of 99mTc pertechnetate dynamic scintigraphy was performed, with lemon juice stimulation at 20 min of the 40-min study. Regions of interests were drawn on the compressed dynamic images of the thyroid (T), parotid, and submandibular glands for both right and left sides and also to the right temporal bone as background.
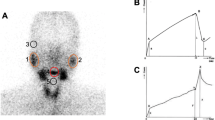
A time–activity curve for each salivary gland was generated using background subtraction. Count rates for the first minute (FM), 15 min, maximum (Max), and minimum (Min) activity after lemon juice stimuli (L) and the last minute activities for each parotid and submandibular glands were obtained (Figs. 1 and 2). FM/Max, Max/Min, T/Max, T/15, Max/L, and 15/L were calculated as well. The difference between the maximum count and the minimum count after stimulation was calculated [(maximum activity − minimum activity/maximum activity)] and the percentage of this value was defined as the excretion fraction (EF%) (Figs. 3 and 4) [3, 10, 11].
Normal scintigraphic pattern of a 37-year-old female patient, who had a dry mouth complaint for 18 months, and labial biopsy result was grade 0. Good radiopharmaceutical uptake and sharp and rapid fall of time–activity curve (TAC) after lemon juice stimuli. Red line: right parotid. Green line: left parotid. Blue line: right submandibular. Yellow line: left submandibular glands
Abnormal scintigraphic pattern as diminished radiopharmaceutical uptake and no response to lemon juice stimuli on TAC in a 45-year-old female patient, who had a history of dry mouth for 2 years, and labial biopsy was graded as 4. Red line: right parotid. Green line: left parotid. Blue line: right submandibular. Yellow line: left submandibular glands
Histopathological analysis
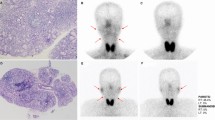
All patients had salivary gland biopsy, which were performed by an experienced physician. The severity of histopathological changes was graded according to the Chisholm and Mason scoring system [12]. According to this scoring system, the presence of lymphocytes per 4 mm2 of salivary tissue makes the basis of this scoring system where:
-
Grade 0
Absence of any lymphocytes
-
Grade 1
Slight infiltrate
-
Grade 2
Moderate infiltrate or less than one focus, where a focus is an aggregate of 50 or more lymphocytes and histiocytes
-
Grade 3
Infiltration in one focus
-
Grade 4
Presence of infiltration in more than one focus
According to histopathological grading, the patients were divided into two subgroups: grade 0 and grade 1 biopsy findings were accepted as normal or with minimal changes (group I) and grades 2 to 4 were accepted as advanced stages of SS (group II).
Statistical analysis
The difference between the calculated semiquantitative parameters was tested using ANOVA test.
Results
Out of 30 patients, 15 were accepted in group I and 15 patients were accepted in group II. The EF values are given as mean, median (in percent), and range in Table 2 for left–right parotid and submandibular glands for both normal and pathologic sides. Excretion fraction was significantly reduced in group II patients (54.5 to 45.8 % in the parotid glands (p < 0.05) and 46.7 to 29.3 % in the submandibular glands; p < 0.000). With the progression in histopathologic grades, the velocity of secretion decreased in both submandibular glands.
No significant correlation was found between tracer uptake concentration and time to peak activity for parotid and submandibular glands among group I and group II. No other quantitative scintigraphic parameters were able to differentiate between group I and group II.
Discussion
Salivary gland scintigraphy has been useful for the investigation of multiple diseases affecting salivary glands [3, 6]. In Sjögren’s syndrome, pertechnetate imaging can gauge the severity of salivary gland involvement, which may not be accurately reflected by clinical symptoms (i.e., xerostomia and other features of the sicca syndrome) [4, 5]. In other clinical situations, such as iatrogenic irradiation of the salivary glands during treatment of head and neck tumours or radioiodine treatment of thyroid cancer, salivary scintigraphy helps to assess functional damage and monitor recovery [13].
Recently, salivary gland scintigraphy has been refined toward providing quantitative information on changes in gland function after parenchymal insult, whether inflammatory or radiation induced. Most attempts have used data derived from the kinetics of uptake and clearance of radioactivity in the major salivary glands [14–17]. In addition, grading and comparison of uptake in the salivary glands in successive scans have been used objectively and reproducibly to follow changes, thus indicating progression or improvement of the pathologic condition [13, 18].
SGS is a well-standardised procedure, and results are reported in a qualitative way. The use of computer-assisted time–activity curves allows quantification of salivary gland radioactivity turnover in a defined time period during SGS [11, 12]. In this study, the decrease in EF values in both parotid and submandibular glands help to evaluate SS patients according to histopathological groups. Although semiquantitative SGS data have been studied before, this is one of the few studies using absolute measures for isotope uptake and excretion. The degree of reduction in secretion in our study agrees with the data presented in other studies of paired time–activity curves for submandibular and parotid glands in primary SS patients [3, 13, 14].
Reduced uptake in the submandibular glands more than parotids in SS patients was noted in other studies [9]; however, in this study, we were unable to show this. This discrepancy could be because of the methodological aspects. The present SS cohort had shorter duration (12 to 36 months) of dry mouth symptoms in our study compared to long-standing disease symptoms (mean 11 years) in the mentioned study. Variable sensitivities of the parotid and submandibular glands to the insult over time could be another explanation [15–17]. Besides, as shown by Gerli et al., the onset of the disease is more important than the duration [18], which might be the reason for the disconcordance between the studies.
Delayed tracer uptake and secretion in SS patients are thought to be due to a progressive reduction in exocrine function of the major salivary glands resulting from morphological changes induced by chronic immune-mediated inflammation [1, 3, 4, 19]. This thought is mainly based on indirect evidence from minor labial gland biopsy findings in SS. However, normal acinar cells are commonly observed in biopsy materials indicating that dryness cannot be solely explained by glandular destruction. Autoantibodies against muscarinic M3 receptors that inhibit parasympathetic neurotransmission by acetylcholine may also contribute to impaired glandular function through the disruption of autonomic function without mediating inflammation [6, 7]. High concentrations of cholinesterase, produced by local T lymphocytes, in the resting and stimulated salivas of SS patients has been found [20]. Besides anticholinergic receptor antibodies have also been tentatively described [20]. Although we lack the necessary expertise to measure muscarinic receptor autoantibodies, studying the influence of this new class of autoantibodies on salivation may also profit from quantitative SGS testing.
Conclusion
Semiquantitative analysis of salivary gland scintigraphy provides further information on the decreased secretion velocity in the submandibular and parotid glands. EF value seems to be a sensitive and valuable parameter for the differantiation of patients with pathological secretion function in the salivary glands.
References
Manoussakis MN, Moutsopoulos HM (2000) Sjögren’s syndrome: autoimmune epithelitis. Baillière’s Best Practice & Research Clin Rheumatol 14:73–95
Vitali C, Bombardieri S, Moutsopoulos HM (2002) Classification criteria for Sjögren’s syndrome: a revised version of the European criteria proposed by the Americane European consensus group. Ann Rheum Dis 61:554–558
Umehara I, Yamada I, Murata Y, Takahashi Y, Okada N, Shibuya H (1999) Quantitative evaluation of salivary gland scintigraphy in Sjögren’s syndrome. J Nucl Med 40:64–69
Schall GL, Anderson LG, Wolf RO et al (1971) Xerostomia in Sjögren’s syndrome: evaluation by sequential salivary scintigraphy. JAMA 216:2109–2116
Arrago JP, Rain JD, Brocheriou C, Rocher F (1987) Scintigraphy of the salivary glandsin Sjögren’s syndrome. J Clin Pathol 40:1463–1467
Daniels TE, Powell MR, Sylvester RA, Talal N (1979) An evaluation of salivaryscintigraphy in Sjögren’s syndrome. Arthritis Rheum 22:809–814
Sugihara T, Yoshimura Y (1988) Scintigraphic evaluation of the salivary glands in patients with Sjögren’s syndrome. Int J Oral Maxillofac Surg 17:71–75
Bohuslavizki KH, Brenner W, Wolf H et al (1995) Value of quantitative salivary gland scintigraphy in the early stage of Sjögren’s syndrome. Nucl Med Commun 16:917–922
Aung W, Yamada I, Umehara I, Ohbayashi N, Yoshino N, Shibuya H (2000) Sjögren’s syndrome: comparison of assessments with quantitative salivary gland scintigraphy and contrast sialography. J Nucl Med 41:257–262
Klutmann S, Bohuslavizki KH, Kröger S, Bleckmann C, Brenner W, Mester J et al (1999) Quantitative salivary gland scintigraphy. J Nucl Med Technol 27:20–26
Kosuda S, Suzuki K, Kawakami R, Akita S, Mezaki T, Inokuma S et al (1993) Reassessment of usefulness of salivary scintigraphy in diagnosis of Sjögren’s syndrome. Kaku Igaku (Jpn J Nucl Med) 30:161–170
Chisholm DM, Mason DK (1968) Labial salivary gland biopsy in Sjögren’s syndrome. J Clin Pathol 21:656–660
Liem IH, Olmos RA, Balm AJ et al (1996) Evidence for early and persistent impairment of salivary gland excretion after irradiation of head and neck tumours. Eur J Nucl Med 23:1485–1490
Kohn WG, Ship JA, Atkinson JC, Patton LL, Fox PC (1992) Salivary gland 99mTc-scintigraphy: a grading scale and correlation with major salivary gland flow rates. J Oral Pathol Med 21:70–74
Vigh L, Carlsen O, Hartling OJ (1997) Uptake index and stimulated salivary gland response in 99Tcm-pertechnetate salivary gland scintigraphy in normal subjects. Nucl Med Commun 18:363–366
Demangeat R, Didon-Poncelet A, Cherfan J, Demangeat JL (2000) Stimulated salivary pertechnetate clearance revisited: correlation with dynamic scintigraphic indices in sicca syndrome. Clin Nucl Med 25:888–894
Bohuslavizki KH, Brenner W, Tinnemeyer S et al (1995) Quantitative salivary gland scintigraphy derived from 166 normals. Radiol Oncol 29:297–305
Gerli R, Muscat C, Giasanti M et al (1997) Quantitative assessment of salivary gland inflamatory inflamation in primary Sjögren’s syndrome: its relationship to different demographic clinical and serological features of the disorder. Br J Rheumatol 36:969–975
Vinagre F, Santos MJ, Prata A, Canas da Silva J, Santos AI (2009) Assessment of salivary gland function in Sjögren’s syndrome: the role of salivary gland scintigraphy. Autoimmun Rev 8:672–676
Konttinen YT, Porola P, Konttinen LLA, Ne M, Poduval P (2006) Immunohistopathology of Sjögren’s syndrome. Autoimmun Rev 6:6–20
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aksoy, T., Kiratli, P.O. & Erbas, B. Correlations between histopathologic and scintigraphic parameters of salivary glands in patients with Sjögren’s syndrome. Clin Rheumatol 31, 1365–1370 (2012). https://doi.org/10.1007/s10067-012-2024-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-012-2024-2