Summary.
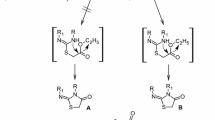
A series of peptide and dipeptide derivatives conjugated with a 1,2,3-triazolo[4,5-d]pyrimidinedione residue were synthesized. The new compounds were evaluated in vitro for cytotoxicity against HAV-27 and HSV-1 and showed moderate activity. The prepared compounds were tested for antimicrobial activity against four different bacterial species, and they displayed different degrees of antibacterial activities or inhibitory actions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Part V: Ali OM, Abdel-Rahman AA-H (2007) Monatsh Chem (in press)
AA Deshmukh MK Mody T Ramalingam PB Sattur (1984) Indian J Chem 23B 793 Occurrence Handle1:CAS:528:DyaL2cXmtFWjt7s%3D
A Agrody M El-Hakim M Abdellatif A Fakery E El-Sayed K El-Ghareab (2000) Acta Pharm 50 111
AH Bedair NA Elhady M Abdellatif A Fakery A Agrody (2000) Il Farmaco 55 708 Occurrence Handle10.1016/S0014-827X(00)00097-5 Occurrence Handle1:CAS:528:DC%2BD3MXis1Ojug%3D%3D
AH Bedair HA Emam NA Elhady KAR Ahmed AM Elagroudy (2001) Il Farmaco 56 965 Occurrence Handle10.1016/S0014-827X(01)01168-5 Occurrence Handle1:CAS:528:DC%2BD3MXpt1artb8%3D
FA Eid AH Abdelwaheb GA Elhagali M Khafagy (2004) Acta Pharm 54 13 Occurrence Handle1:CAS:528:DC%2BD2cXjtVamtbg%3D
SP Hiremath A Ullagaddi K Shivaramayya MG Purohit (1999) Indian J Heterocycl Chem 3 145
CJ Shishoo MB Devani GV Ullas S Anathan VS Bhadti (1981) J Heterocycl Chem 18 43 Occurrence Handle1:CAS:528:DyaL3MXkt1Cqtb4%3D
S Nega J Ainso A Diazj F Junquere (1990) J Heterocycl Chem 27 269
R Gupta AK Gupta S Paul PL Kachroo (1998) Indian J Chem 37B 1211 Occurrence Handle1:CAS:528:DyaK1MXhtVyms7o%3D
AE Rashad OA Heikal AOH Elnezhawy FME Abdelmegeid (2005) Heteroatom Chem 16 226 Occurrence Handle10.1002/hc.20114 Occurrence Handle1:CAS:528:DC%2BD2MXktVCltLg%3D
B Loye JH Musser RE Brown H Jones R Kahenan FC Huang A Khandwala P Sonnio-Goldman MJ Leibowitz (1985) J Med Chem 28 363 Occurrence Handle10.1021/jm00381a016
M Pemmsin C Lnu-Due F Hoguet C Gaultier J Narcisse (1988) Eur J Chem 23 543
LM Hall MM Devine (1990) Plant Physiol 93 962 Occurrence Handle1:CAS:528:DyaK3cXlsVOiurY%3D
Z Liu G Yang XJ Qin (2001) Chem Technol Biotechnol 76 1154 Occurrence Handle10.1002/jctb.500 Occurrence Handle1:CAS:528:DC%2BD3MXotVSgtbY%3D
W Chen G Jin (2003) Heteroatom Chem 14 607 Occurrence Handle10.1002/hc.10198 Occurrence Handle1:CAS:528:DC%2BD3sXptFWqsbw%3D
TD Bradshaw MC Bibby JA Double I Fichtner PA Cooper MC Alley S Donohue SF Stinson JE Tomaszewjski EA Sausville MFG Stevens (2002) Mol Cancer Ther 1 239 Occurrence Handle1:CAS:528:DC%2BD38XitlSrtrc%3D
MD Wittman JF Kadow (1996) US Patent 5,489,589, 6 Feb; Chem Abstr 124 289947a
A Casini A Scozzafava CT Supuran (2002) Environ Health Perspect 110 801 Occurrence Handle1:CAS:528:DC%2BD38XovFKhsbY%3D Occurrence Handle10.1289/ehp.02110801
K-L Yu WE Harte P Spinazze JC Martin MM Mansuri (1993) Bioorg Med Chem Lett 3 535 Occurrence Handle10.1016/S0960-894X(01)81222-1 Occurrence Handle1:CAS:528:DyaK3sXmtVers78%3D
IAI Ali IA Al-Masoudi B Saeed NA Al-Masoudi P La Colla (2005) Heteroatom Chem 16 148 Occurrence Handle10.1002/hc.20082 Occurrence Handle1:CAS:528:DC%2BD2MXjtVyjsLc%3D
NA Al-Masoudi IA Al-Masoudi IAI Ali YA Al-Soud B Saeed P La Colla (2005) Heteroatom Chem 16 576 Occurrence Handle10.1002/hc.20149 Occurrence Handle1:CAS:528:DC%2BD2MXht1KgurnF
NA Al-Masoudi IA Al-Masoudi IAI Ali YA Al-Soud B Saeed P La Colla (2006) Acta Pharm 56 175 Occurrence Handle1:CAS:528:DC%2BD28XltValsL4%3D
Ali IAI, Ali OM, Abdel-Rahman AA-H (2007) Monatsh Chem (in press)
El-Essawy FA, Khattab AF, Abdel-Rahman AA-H (2007) Monatsh Chem (in press)
K Senga M Ichiba S Nishigaki (1977) Heterocycles 6 1915 Occurrence Handle1:CAS:528:DyaE1cXltVOktg%3D%3D Occurrence Handle10.3987/R-1977-11-1915
R Schwyzer H Kappeler (1961) Helv Chim Acta 44 1991 Occurrence Handle10.1002/hlca.19610440723 Occurrence Handle1:CAS:528:DyaF38XktlKls7k%3D
RS Farag AS Shalaby GA El-Baroty NA Ibrahim (2004) Phytother Res 18 30 Occurrence Handle10.1002/ptr.1348 Occurrence Handle1:CAS:528:DC%2BD2cXhvFehsr8%3D
Janssen AM, Scheffer JJ, Svendsen AB (1986) Planta Medica: 395
MT Shaaban ME El-Sharif (2001) African J Mycol Biotechnol 9 IssueID2 15
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Rahman, AH. Amino Acid Derivatives, VI [1]: Synthesis, Antiviral, and Antimicrobial Evaluation of α-Amino Acid Esters Bearing a 1,2,3-Triazolo[4,5-d]pyrimidinedione Side Chain. Monatsh. Chem. 139, 61–68 (2008). https://doi.org/10.1007/s00706-007-0745-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-007-0745-8