Abstract
Background
Chronic subdural hematoma (CSDH) is a common disease in neurosurgical practice with substantial recurrence rate. We aimed to estimate recurrence rate of CSDH and to identify risk factors for CSDH recurrence.
Methods
We retrospectively studied consecutive cases with CSDH and performed surgical therapy in our hospital. Univariate and multivariate logistic regression analyses were performed to identify factors associated with recurrence of CSDH.
Results
A total of 226 patients with CSDH were included; 34 patients recurred after surgery with a recurrence rate of 15.0%. Univariate analysis showed that the recurrence group had more patients with homogenous hyper-dense hematoma (20.6 vs 6.3%, p = 0.035) and shorter duration of subdural drainage post-surgery (1.2 ± 1.4 vs 1.5 ± 0.9, p = 0.022) than the non-recurrence group. Logistic regression analysis revealed that duration of subdural drainage (OR = 0.66, p = 0.05) and hyper-dense of hematoma (OR = 4.94, p = 0.012) were independent predictors for CSDH recurrence.
Conclusions
Homogenous hyper-dense of hematoma and duration of subdural drainage post-surgery were independent predictors for CSDH recurrence; longer duration of postoperative subdural drainage was associated with lower risk of recurrence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chronic subdural hematoma (CSDH) is a common neurological disorder which often occurs in the elderly [9]. The incidence of CSDH in general population is estimated to range from 5 to 14 per 100,000 person-years [9, 16]. With the aging population, a significant increase in the incidence of CSDH is expected. Surgical evacuation, especially burr-hole craniotomy and drainage, is the most widely used technique for treating CSDH [12]. Most of the patients obtained great improvements in neurological function after surgery. However, some patients experienced a recurrence of hematoma requiring a reoperation. The prevalence of recurrence was reported to range from 2.5 to 33% in literature [1, 18, 24, 25]. Recurrence of hematoma often resulted in a complex clinical situation, increased psychological burden, and poor outcomes of patients, which poses a major challenge for neurosurgeons. Furthermore, the mechanism that contributes to CSDH recurrence is elusive. Therefore, the identification of factors associated with recurrence of CSDH is important for the treatment and postoperative management of these patients. Although various potential risk factors have been proposed including age, body mass index (BMI), hematoma density, separated hematoma, and use of antiplatelet or anticoagulant agents, the results remain inconclusive [2, 4, 5, 15, 20].
Therefore, the aims of this study were to estimate the recurrence rate and to identify risk factors for CSDH recurrence by retrospectively reviewing the cases of CSDH treated in our institution.
Methods
Patient population
We retrospectively reviewed patients with CSDH admitted to the Department of Neurosurgery, First Affiliated Hospital, College of Medicine, Zhejiang University, between January 2013 and September 2016. The study was approved by the ethics committee of the hospital and was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Patients with CSDH were diagnosed by head computed tomography (CT) or magnetic resonance imaging (MRI). Inclusion criteria were (1) age of 18 years or older and (2) underwent surgical treatment. Cases with missing data or co-existed with severe multi-organ dysfunction were excluded from the study. The recruitment of patients is shown in Fig. 2.
Treatment of CSDH
All patients underwent burr-hole craniotomy and drainage after admission, which is the standard of treatment for CSDH in our department. Briefly, a single burr-hole or two burr-holes over the maximum width of hematoma were drilled under local or general anesthesia. Hematoma was evacuated followed by irrigation of the subdural cavity using normal saline. A silicone catheter was inserted into the subdural space after irrigation, and a closed drainage system was connected to the catheter. The drainage system was properly fixed and maintained after the surgery. Catheters were usually removed 1 to 2 days later unless prolonged drainage was indicated. Postoperative head CT scanning was routinely performed 24 h after surgery, before discharge and 1-month post operation. Patients were routinely followed up by telephone or out-patient department visits for 1 year after discharge from hospital.
Data collection
Demographic and clinical data were extracted from case records: age, sex, height, weight, history of head trauma, Glasgow Coma Scale (GCS), head CT imaging, pre-existing diseases including hypertension, diabetes mellitus, and cardiac vascular disease, use of antiplatelet or anticoagulant agents, duration of postoperative subdural drainage, and the interval between recurrence and the first operation.
Radiological imaging evaluation
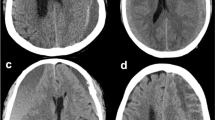
Preoperative head CT imaging of each patient was assessed by two investigators blinded to patients’ clinical conditions. Midline shift, location of hematoma, and hematoma density were reviewed. Hematomas were classified into four types according to their density on CT as previously described Nakaguchi et al. [14]: homogenous (including three subtypes, iso-dense, homogenous hypo-dense, homogenous hyper-dense), laminar, separated, and trabecular (Fig. 1). The laminar type was defined as a hematoma exhibiting a high-density layer along the inner membrane. The separated type contains two components of different densities, and a boundary lies between them. The trabecular type was defined as a hematoma with inhomogeneous contents, and the low-density to iso-dense hematoma was separated by high-density septa. Patients with bilateral CSDHs that displayed different types of hematoma were classified according to the hematoma that showed greater bleeding tendency [20].
Definition of and treatment for recurrence
Recurrence of CSDH was defined as a re-accumulation of hematoma in the ipsilateral subdural space accompanied with neurological deficits, for which a reoperation was required. Reoperation also evolves a burr-hole evacuation of hematoma and closed drainage.
Statistical analysis
Statistical analyses were performed by SPSS Statistics software (Version 17.0, Chicago, IL). Continuous variables were given as means and standard deviations or as medians and interquartile ranges. Continuous variables were compared by Student’s t test if normally distributed or by Mann-Whitney U test if non-normally distributed. Categorical variables were expressed as numbers and percentages. Categorical data were compared by chi-square test. Binary logistic regression analysis was then performed to identify risk factors for CSDH recurrence. Multiple comparisons between recurrence rates of each hematoma type were conducted by chi-square tests and Bonferroni corrections. Variables that were significant in univariate analysis were included in logistic analysis. Statistical significance was determined at a p value < 0.05.
Results
Baseline characteristics
A total of 226 patients with CSDH were included in our study. The baseline demographic characteristics and radiographic findings of the study population are summarized in Table 1. There were 184 (81.4%) males and 42 (18.6%) females, and the mean age was 65.1 ± 13.5 years. One hundred sixty-one (71.2%) patients reported history of head trauma. The hematoma was on the left side in 93 (41.2%) patients, on the right side in 67 (29.6%) patients, and bilateral in 66 (29.2%) patients. With regard to hematoma density, homogenous hypo-dense type was observed in 47 patients (20.8%), homogenous iso-dense type in 55 patients (24.3%), homogenous hyper-dense type in 19 patients (8.4%), laminar type in 9 patients (4.0%), separated type in 33 subjects (14.6%), and trabecular type in 63 subjects (27.9%). Surgery was successful in all patients, and the mean duration of drainage after the initial operation was 1.4 ± 1.0 days.
Recurrence rate of CSDH
Thirty-four (15.0%) patients experienced a recurrence of CSDH that required reoperation. The average interval between the initial operation and the recurrence was 59.9 days. All the patients with recurrence of CSDH underwent burr-hole and drainage as second operation and achieved good treatment outcome; no additional third operation was needed for the subjects with recurrence (Fig. 2).
Risk factors for recurrence of CSDH
Univariate analysis of the possible risk factors showed that patients in the recurrence group had shorter duration of postoperative subdural drainage than those in non-recurrence group (1.2 ± 1.4 vs 1.5 ± 0.9, p = 0.022). And, types of hematoma were associated with postoperative recurrence (p = 0.035) (Table 2). Multiple comparisons of recurrence rate between hematoma types showed that the homogeneous iso-dense type had the lowest recurrence rate (7.3%), while the homogeneous hyper-dense hematoma had the highest recurrence rate (36.8%), and there were no significant differences between the recurrence rate of homogenous hypo-dense (21.3%), laminar (11.1%), separated (15.2%), and trabecular (11.1%) types (Table 3). Subjects with bilateral hematoma (p = 0.192), using antiplatelet or anticoagulant drugs (p = 0.144), or pre-existing heart disease (p = 0.120) tended to have higher rate of recurrence, although they were not statistically significant. There were no differences in age, gender, BMI, and history of hypertension and diabetes mellitus between recurrence group and non-recurrence group (Table 2).
Multivariate logistic regression analysis demonstrated that preoperative homogenous hyper-dense hematoma (p = 0.012, OR = 4.94, trabecular as reference) and duration of postoperative subdural drainage (p = 0.050, OR = 0.66) were independent risk factors for recurrence of CSDH, and longer duration of drainage was associated with lower risk of recurrence (Table 4).
Discussion
In the present study, we identified preoperative homogenous hyper-dense hematoma and duration of postoperative subdural drainage was associated with recurrence of hematoma, and the risk of recurrence decreased with longer duration of drainage.
As a common neurosurgical disease usually occurs in older adults, CSDH showed favorable treatment outcomes in most patients. Nevertheless, some patients experienced recurrence after surgical treatment. Although various risk factors for recurrence of CSDH have been proposed in literature [4], a definitive conclusion is still difficult to be drawn. In the present study, we included 226 cases with CSDH. Thirty-four patients experienced recurrence of hematoma with a recurrence rate of 15.0%, which was comparable to that of previous studies reporting the prevalence of CSDH recurrence from 2.5 to 33%.
By univariate analysis, we found that patients with shorter duration of subdural drainage and homogeneous hyper-dense hematoma were more likely to experience recurrence. Logistic regression analysis further demonstrated that these two factors were independent predictors for CSDH recurrence.
Postoperative subdural drainage has long been a research focus for CSDH recurrence. Several studies had shown that patients with subdural drainage had significantly lower recurrence rate than those without drainage after surgery [17, 22, 23]. However, few studies have investigated the relationships between duration of subdural drainage and recurrence. Although some researchers pointed out that longer duration of drainage might lead to lower rate of recurrence, their reliability was largely affected due to their small sample sizes and lack of rigorous statistic assessment [7, 10]. In our study, both the univariate analysis and multivariate logistic analysis demonstrated that longer duration of drainage was associated with lower recurrence rate of CSDH. Therefore, our study indicated that longer duration of drainage should be used in order to reduce recurrence rate. However, risk of infectious complication may increase with prolonged duration of drainage. Thus, further prospective studies are needed to examine the optimal duration of drainage, which both reduced recurrence of hematoma and do not increase the risk of infection.
A large amount of studies have investigated the relationships between preoperative hematoma density and recurrence of hematoma, yet results were varied [3, 6, 15]. Some research found no associations between them [3], while others reported that trabecular, hyper-dense, and mixed density hematomas had higher rate of recurrence [6, 15]. In the present study, both univariate and multivariate logistic regression analyses revealed that homogenous hyper-dense hematoma was more likely to recur after surgery. This was consistent with previous studies reporting higher recurrence rate in patients with high-density hematoma [6, 8, 11]. High density of hematoma indicated that the CSDH was in the acute phase and was not fully developed, thus a higher recurrence rate was usually observed after surgery in this type of CSDH [8, 19]. Therefore, surgery should be delayed in patients displaying high-density hematoma unless severe symptoms were present. Although a lot of studies found separated hematoma was associated with higher recurrence rate, no correlation was observed between them in our study.
The relationships between antiplatelet and anticoagulant administration and CSDH recurrence have been controversial. Some studies concluded that there was no association between them [13, 21], while others revealed that use of antiplatelet or anticoagulant was a risk factor for recurrence [3]. This discrepancy might be explained by the different anticoagulation levels of patients in these studies; high level of anticoagulation was associated with recurrence, while low level of anticoagulation was not correlated with recurrence. In this study, the recurrence group (11.8%) tended to have more patients administrating antiplatelet and anticoagulant drugs than the non-recurrence group (5.2%), even though it was not statistically significant, possibly because only a few patients (14 patients, 6.2%) used these drugs in our study population. In addition, we also investigated patient age, BMI, hypertension, diabetes mellitus, and recurrence of hematoma; no associations were observed.
However, some limitations of our study need to be mentioned. First, this is a retrospective study and thus is potentially subject to selection bias. Second, this study was conducted in a single center, which may affect the generalizability of our study results. Despite these limitations, our relatively large sample size and rigorous statistical analysis supported our conclusions.
Conclusions
In conclusion, homogeneous hyper-dense hematoma and duration of postoperative subdural drainage were independent predictors for CSDH recurrence, and the risk of recurrence decreased with longer duration of drainage. However, further prospective studies are needed to define the optimal duration of postoperative subdural drainage.
References
Almenawer SA, Farrokhyar F, Hong C, Alhazzani W, Manoranjan B, Yarascavitch B, Arjmand P, Baronia B, Reddy K, Murty N, Singh S (2014) Chronic subdural hematoma management: a systematic review and meta-analysis of 34,829 patients. Ann Surg 259:449–457. https://doi.org/10.1097/SLA.0000000000000255
Amirjamshidi A, Abouzari M, Eftekhar B, Rashidi A, Rezaii J, Esfandiari K, Shirani A, Asadollahi M, Aleali H (2007) Outcomes and recurrence rates in chronic subdural haematoma. Br J Neurosurg 21:272–275
Chon KH, Lee JM, Koh EJ, Choi HY (2012) Independent predictors for recurrence of chronic subdural hematoma. Acta Neurochir 154:1541–1548. https://doi.org/10.1007/s00701-012-1399-9
Han MH, Ryu JI, Kim CH, Kim JM, Cheong JH, Yi HJ (2017) Predictive factors for recurrence and clinical outcomes in patients with chronic subdural hematoma. J Neurosurg 127:1117–1125. https://doi.org/10.3171/2016.8.JNS16867
Jack A, O’Kelly C, McDougall C, Findlay JM (2015) Predicting recurrence after chronic subdural haematoma drainage. Canadian J Neurological Sci Le J Canadien Des Sci Neurologiques 42:34–39. https://doi.org/10.1017/cjn.2014.122
Jeong SI, Kim SO, Won YS, Kwon YJ, Choi CS (2014) Clinical analysis of risk factors for recurrence in patients with chronic subdural hematoma undergoing burr hole trephination. Korean J Neurotrauma 10:15–21. https://doi.org/10.13004/kjnt.2014.10.1.15
Kiymaz N, Yilmaz N, Mumcu C (2007) Controversies in chronic subdural hematoma: continuous drainage versus one-time drainage. Medical Sci Monitor: Int Medical J experimental Clinical Res 13:Cr240–Cr243
Ko BS, Lee JK, Seo BR, Moon SJ, Kim JH, Kim SH (2008) Clinical analysis of risk factors related to recurrent chronic subdural hematoma. J Korean Neurosurgical Soc 43:11–15. https://doi.org/10.3340/jkns.2008.43.1.11
Kolias AG, Chari A, Santarius T, Hutchinson PJ (2014) Chronic subdural haematoma: modern management and emerging therapies. Nat Rev Neurol 10:570–578. https://doi.org/10.1038/nrneurol.2014.163
Kotwica Z, Brzezinski J (1991) Chronic subdural haematoma treated by burr holes and closed system drainage: personal experience in 131 patients. Br J Neurosurg 5:461–465
Lin CC, Lu YM, Chen TH, Wang SP, Hsiao SH, Lin MS (2014) Quantitative assessment of post-operative recurrence of chronic subdural haematoma using mean haematoma density. Brain Inj 28:1082–1086. https://doi.org/10.3109/02699052.2014.901559
Lind CR, Lind CJ, Mee EW (2003) Reduction in the number of repeated operations for the treatment of subacute and chronic subdural hematomas by placement of subdural drains. J Neurosurg 99:44–46. https://doi.org/10.3171/jns.2003.99.1.0044
Lindvall P, Koskinen LO (2009) Anticoagulants and antiplatelet agents and the risk of development and recurrence of chronic subdural haematomas. J Clinical Neuroscience: Official J Neurosurgical Soc Australasia 16:1287–1290. https://doi.org/10.1016/j.jocn.2009.01.001
Nakaguchi H, Tanishima T, Yoshimasu N (2001) Factors in the natural history of chronic subdural hematomas that influence their postoperative recurrence. J Neurosurg 95:256–262. https://doi.org/10.3171/jns.2001.95.2.0256
Ohba S, Kinoshita Y, Nakagawa T, Murakami H (2013) The risk factors for recurrence of chronic subdural hematoma. Neurosurg Rev 36:145–149. https://doi.org/10.1007/s10143-012-0396-z
Santarius T, Hutchinson PJ (2004) Chronic subdural haematoma: time to rationalize treatment? Br J Neurosurg 18:328–332
Santarius T, Kirkpatrick PJ, Ganesan D, Chia HL, Jalloh I, Smielewski P, Richards HK, Marcus H, Parker RA, Price SJ, Kirollos RW, Pickard JD, Hutchinson PJ (2009) Use of drains versus no drains after burr-hole evacuation of chronic subdural haematoma: a randomised controlled trial. Lancet (London, England) 374:1067–1073
Schwarz F, Loos F, Dunisch P, Sakr Y, Safatli DA, Kalff R, Ewald C (2015) Risk factors for reoperation after initial burr hole trephination in chronic subdural hematomas. Clin Neurol Neurosurg 138:66–71. https://doi.org/10.1016/j.clineuro.2015.08.002
Stanisic M, Hald J, Rasmussen IA, Pripp AH, Ivanovic J, Kolstad F, Sundseth J, Zuchner M, Lindegaard KF (2013) Volume and densities of chronic subdural haematoma obtained from CT imaging as predictors of postoperative recurrence: a prospective study of 107 operated patients. Acta Neurochir 155:323–333. https://doi.org/10.1007/s00701-012-1565-0
Stanisic M, Pripp AH (2017) A reliable grading system for prediction of chronic subdural hematoma recurrence requiring reoperation after initial burr-hole surgery. Neurosurgery 81:752–760. https://doi.org/10.1093/neuros/nyx090
Torihashi K, Sadamasa N, Yoshida K, Narumi O, Chin M, Yamagata S (2008) Independent predictors for recurrence of chronic subdural hematoma: a review of 343 consecutive surgical cases. Neurosurgery 63:1125–1129. https://doi.org/10.1227/01.NEU.0000335782.60059.17
Tsutsumi K, Maeda K, Iijima A, Usui M, Okada Y, Kirino T (1997) The relationship of preoperative magnetic resonance imaging findings and closed system drainage in the recurrence of chronic subdural hematoma. J Neurosurg 87:870–875. https://doi.org/10.3171/jns.1997.87.6.0870
Wakai S, Hashimoto K, Watanabe N, Inoh S, Ochiai C, Nagai M (1990) Efficacy of closed-system drainage in treating chronic subdural hematoma: a prospective comparative study. Neurosurgery 26:771–773. https://doi.org/10.1227/00006123-199005000-00006
Weigel R, Schmiedek P, Krauss JK (2003) Outcome of contemporary surgery for chronic subdural haematoma: evidence based review. J Neurol Neurosurg Psychiatry 74:937–943. https://doi.org/10.1136/jnnp.74.7.937
Yu GJ, Han CZ, Zhang M, Zhuang HT, Jiang YG (2009) Prolonged drainage reduces the recurrence of chronic subdural hematoma. Br J Neurosurg 23:606–611. https://doi.org/10.3109/02688690903386983
Acknowledgments
The authors sincerely thank all the doctors in the neurosurgery department of the First Affiliated Hospital of Zhejiang University College of Medicine for their help in conducting this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the ethics committee of the First Affiliated Hospital, College of Medicine, Zhejiang University, and was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
You, W., Zhu, Y., Wang, Y. et al. Prevalence of and risk factors for recurrence of chronic subdural hematoma. Acta Neurochir 160, 893–899 (2018). https://doi.org/10.1007/s00701-018-3513-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-018-3513-0