Abstract
The work describes a simple cotton swab-based colorimetric immunoassay as a rapid screening tool for pathogenic bacteria on poultry processing plants. This immunosensing platform can be used for the detection of pathogens present on surfaces such as glass, stainless steel and chicken meat. Unlike the reported assays, here, cotton swab plays dual function: as a sample collector from the solid surfaces and as detection platform. The immunoassay was tested for the detection of 4 different bacteria; Salmonella typhimurium, Salmonella enteritidis, Staphylococcus aureus and Campylobacter jejuni. The immunoassays were fabricated by immobilizing specific antibody for each bacterium on a cotton swab that is used to recover the cells from contaminated surfaces. Then, a sandwich immunoassay was developed by immersing the cotton swab in different colored nanobead-conjugated antibody solutions which corresponds to different bacteria. The immunoassays response was detected colorimetrically by following the change in the color intensity produced by the nanobeads due to the specific binding on the cotton swab. This simple colorimetric assay is very sensitive with a detection limit of 10 cfu.mL−1. Furthermore, no significant cross reactivity of the immunoassays with non specific bacteria was observed indicating good selectivity of the immunoassays. This simple, disposable and easy-to- use colorimetric platform shows great promise as rapid qualitative and semi quantitative detection tool for microorganisms on food processing plants and other surfaces.

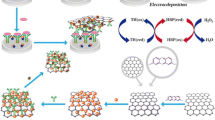
Schematic of the sandwich colorimetric immunosensor for the detection of pathogenic bacteria on poultry processing plants using cotton swabs and nanobeads.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Foodborne diseases (FBD) are referred to as the infectious and toxic diseases caused by pathogens that enter the body through the ingestion of contaminated food with bacteria, viruses, parasites, toxins or metals. These pathogens can be also transmitted to human through the food chain [1,2,3]. Bacteria such as Salmonella, Staphylococcus aureus and Campylobacter are common pathogens that cause human bacterial gastroenteritis which can be transmitted to human via consumption of poultry products [4]. Results from a recent study shows that most of the chicken infections in Saudi Arabia are caused by Campylobacter jejuni pathogen [5].
In the food-processing industry, several types of surfaces are used such as plastic, stainless steel and glass which can be also subjected to contamination by pathogens. The contamination of surfaces can be a serious public health issue. Several outbreaks were found to be due surface contamination by different bacteria. Therefore, early detection of pathogens in both contaminated food and solid surfaces is necessary for protecting consumer health and preserving food safety.
Different methods using various sensing principles have been used for the detection of pathogenic bacteria which cause foodborne diseases [6]. Conventional methods [6] such as culture-depending methods, microscopy, polymerase chain reaction (PCR), serological and biochemical methods are widely used. These methods are accurate, relatively sensitive and reliable. However, they suffer from disadvantages such as being time-consuming (culturing takes up to 3 days, PCR and biomedical assays take at least few hours to complete), unsuitable for field applications (require sophisticated laboratory equipments) and are not user friendly. Thus, significant research work is currently devoted to the development of new portable, cost effective and user-friendly tools for the rapid and on-site screening of pathogens. Immunoassays are also widely used for the detection of pathogens [7,8,9]. The most commonly used immunoassay technique is ELISA. Lateral flow assays [10], immunomagnetic [8], immunofluorescence [11] and radioimmunoassay have been also employed for pathogens detection. Some of the developed immunoassay methods provide advantages over the other conventional assays such as rapidity, high specificity and portability. A number of tests can be achieved by utilizing kits or small apparatuses which facilitates onsite and point of care analysis. Optical immunosensors have been also reported for pathogens detection based on different techniques such as fluorescence, chemiluminescence, electrochemiluminescence [12,13,14,15,16], surface plasmon resonance (SPR) [17], optical waveguide [18] and surface enhanced resonance scattering (SERS) [19].
Because of the unique electronic, structural and optical properties of nanoparticles such as magnetic [8], fluorescent and colored beads [20] and metal (gold or silver) nanoparticles [19, 21], they have been widely used to improve the sensitivity and performance of immunosensors. However, optical sensors are still expensive and techniques like SPR cannot be easily miniaturized. Moreover, most of the developed immunosensor takes more than an hour to complete.
For the detection of pathogens on solid surfaces, a recovery step of the pathogen is required prior to analysis. The swabbing method is the conventional way for recovering pathogens from plastic, stainless steel and wood that has been widely applied in food safety management protocols. Cotton swab is the conventional swabbing tool that has been widely employed for recovering pathogens form contaminated surfaces. The pathogens are then usually released from the cotton by vortexing in extraction buffer followed by plating or applying other analysis techniques [22,23,24,25].
The increased interest in food safety necessitates the development of new rapid detection methods for food contaminants [26]. The new method has to be simple, low cost and portable and can give precise results in short time. In this work, we report a novel, simple and versatile cotton swab-based sandwich immunoassay for bacteria detection. The proposed immunoassay platform is fabricated on a cotton swab functionalized with specific antibody for each bacterium. The cotton swab is used to collect the pathogens such as Salmonella Typhimurium (St), Salmonella Enteritidis (Se), Staphylococcus aureus (Sa) and Campylobacter jejuni (Cj) from poultry processing plants surfaces. Then, the cotton is immersed in colored nanobeads conjugated with specific antibody for the corresponding bacteria. This sandwich immunoassay enables the collection as well as the detection of the pathogens from solid surfaces using a single device. Different from the conventional methods which rely on using the cotton swabs only for pathogen recovery from the surface followed by releasing the pathogens and then analysis or plating, the proposed immunoassay requires only a single cotton swab to perform the entire analysis process. This platform is easy to use, rapid and low cost and can be applied for the detection of other pathogens.
Experimental section
Materials and reagents
St (ATCC14028), Se (ATCC13076), Sa (ATCC6538) and Cj (ATCC 29428) were purchased from American Type Culture Collection (Manassas, VA). Murine Anti- Salmonella Typhimurium monoclonal antibodies of St, Se, anti-Cj monoclonal antibody of Cj and rabbit polyclonal antibody of Sa were purchased from Biospacific (Emeryville, CA, USA). Nanobeads around 50 nm size with three different colors, blue, orange and green were purchased from Polyesciences Inc. (Warrington, PA, USA). Carboxy-functionalized cobalt-based magnetic nanoparticles with 50 nm diameter were purchased from Turbobeads (Zurich, Switzerland). The DNA extraction kit and real-time PCR reagents were obtained from QIAGEN (www.qiagen.com) (Manchester, UK). The HPLC grade chloroform was purchased from scharlau, (Barcelona, Spain). 1-ethyl-(3-dimethylaminopropy) carbodimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS) were purchased from Fisher Scientific (Ontario, Canada). Dipotassium hydrogen orthophosphate, potassium dihydrogen orthophosphate, sodium chloride and bovine serum albumin (BSA) were purchased from Sigma (Ontario, Canada),
Procedure
Bacteria culturing
Stock cultures of all strains except Cj were stored at -80 °C in 20% glycerol solution. Prior to use, the frozen culture is activated in trypticase soy broth (TSB, Oxoid, Hampshire, UK) at 37 °C with two consecutive transfers after 18- 20 h incubation periods. The culture is centrifuged at 10000 Xg for 10 min at 4 °C and washed twice with trypticase soy broth. Cell suspensions were prepared and the OD is adjusted at 600 nm to 0.5, which is equivalent to 108 cfu.mL−1. Then serially diluted from 1 to 108 cfu.mL−1 in trypticase soy broth. Stock cultures of Cj were grown for 4 h at 37 °C and then for 24–48 h at 42 °C under microaerophilic conditions in Boloton broth media (Oxoid LTD, UK) in an anaerobe jar with an active catalyst and a microaerophilic gas generator pack. 10 fold serial dilutions were made in Maximum Recovery Diluent (Oxoid LTD, UK) and the viable cell numbers of Cj were determined by surface plating on Columbia Blood agars (Oxoid LTD, UK).
Immobilization of antibodies on cotton swabs
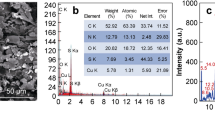
The cotton swabs were activated by immersing them in a mixture of 100 mL of 2 mM sodium periodate (NaIO4), and 1 mL of sulfuric acid overnight at room temperature. The cotton swabs were then washed with cold distilled water extensively to remove the excess oxidizing agent. Periodate oxidation of cellulose cotton activation into aldehyde is characterized by the FTIR spectroscopy (Fig. S1). FTIR spectra were taken in transmission mode by KBr pellets for the treated and untreated cotton samples.
The activated aldehyde group in the cotton swab is used to couple the amino groups of the antibodies. 40 μl (50 ng.mL) of antibody was mixed well with 1 mL of phosphate buffered saline (PB saline) pH 7.4 for 3 min. Then, the activated cotton was immersed in the antibody buffer solution overnight at 4 °C. The antibody linked cotton swab was washed with PBS buffer to remove the unbound antibodies. The free aldehyde groups were blocked with BSA by incubating the cotton in 1 mL of BSA (1 mg.mL) for 30 min at room temperature followed by washing with PBS buffer for three times. The antibody-linked cotton was stored at 4 °C in PBS for further use.
The control samples were prepared by immersing the activated cotton swabs in 1 mg.mL solution of bovine serum albumin (BSA) in PB saline overnight at room temperature. BSA-linked cotton swabs were then extensively washed with PB saline to remove the unbounded BSA and stored in PBS buffer at 4 °C.
Conjugation of the antibodies to the nanobeads
300 μl of each (blue, orange and green) polymer beads or magnetic beads were washed 3 times with water. EDC/NHS solution was prepared by mixing, 100 mg of EDC and 100 mg of NHS in 10 mL of water. 300 μl of EDC/NHS solution was added to the nanobeads and mixed for 20 min at room temperature. EDC/NHS activated beads were washed three times using PB saline followed by the addition of 20 μl (50 ng.mL) of antibody and 300 μl of buffer. Each specific antibody for a particular bacterial strain was mixed with a particular colored bead. For example, Se specific antibody was incubated with the blue nanobeads, St specific antibody was incubated with the black magnetic nanobeads, Sa specific antibody was incubated with the orange nanobeads and Cj specific antibody was incubated with green nanobeads overnight at 4 °C. Antibody-linked nanobeads were then washed with buffer to remove the unreacted antibodies. Finally, the remained active sites were blocked by mixing the nanobeads with 1 mg.mL of BSA in buffer for 30 min. Unbound BSA were washed with PB saline and the antibodies-bound nanobeads were stored at 4 °C in buffer.
Screening assay
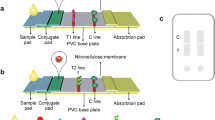
The screening procedure involved two steps. The assay steps are shown in Scheme 1. The specific capture primary antibodies (C-Abs) were conjugated to cotton swabs and developing color solution of magnetic beads or colored polymeric nanobeads were conjugated to secondary detection antibody (D-Ab). The first step, the wet cotton-immobilized specific antibody is swabbed over surfaces to pre-concentrate the bacteria from artificially contaminated surfaces with a serial dilution of target bacterial cells (a certain sampling area is chosen such as (20 cm × 20 cm) and the swabs is rubbed and rolled firmly several times across the sampling area). St, Se, Sa and Cj were used for contaminating the surfaces of the chicken meat, glass slide and stainless steel surfaces. The number of bacteria on each surface is confirmed by swabbing, releasing and culturing the cells followed by counting. After 10 min, the cotton immobilized antibody-bacteria complexes were washed with PB saline to remove the free cells from cotton.
Schematic diagram of cotton swab bioassay for screening pathogenic bacteria from contaminated surfaces. Aldehyde functionalized cotton swab was linked with bacteria-specific antibody which then binds to the target. The target complex then treated with different colored beads (in which each colored bead is linked with specific antibody strain). The pathogenic bacteria of interest present in the contaminated surfaces will be identified from the color of the cotton surface by sandwich immunocomplex formation. The unbound magnetic beads are separated by magnet and the other beads were separated by washing
The second step is the detection step in-which the bacteria is sandwiched between the cotton immobilized antibody and secondary detection antibody conjugated to colored nanobeads. The cotton swab antibody-bacteria (cotton-C-Ab-cell) complexes were immersed in developing color solution which contains colored nanopolymeric beads or magnetic nanobeads linked with the D-Ab in buffer solution for 2 min. The cotton is then washed with buffer to remove the unbound beads. The color of the beads bound to the cotton swab indicates the specific bacterial stain present in the contaminated surface as shown in Fig. 1. Control samples were done as above except, we swabbed the chicken, glass and stainless steel surfaces without bacteria contamination. For testing the selectivity of the assay, different antibody –bound cotton swabs were used to collect different types of bacterial cells from contaminated surfaces. After washing with PB saline, the cotton swabs were immersed in the four different colored nanobeads conjugated with the secondary antibodies. Finally, the cotton swabs were washed with buffer to remove the unbound beads. In the case of magnetic beads, the unbound particles can be separated either by normal washing or by passing the cotton swab over a small magnetic sheet.
Salmonella typhimurium screening results: St specific antibody-conjugated cotton swab and the black magnetic beads captured the salmonella typhimurium target cells from chicken (a), glass plate (b) and stainless steel surface (c). More intense black sandwich complexes were formed with increasing concentration of bacterial cells. Visual detection limit for the samples from chicken, glass and stainless steel are 101,103 and 101 cfu.mL−1, respectively. The experiments were done in triplicates
PCR confirmation assay
Extraction of DNA
The DNA from the bacterial culture was extracted according to the protocol given by the QIAGEN (http://www.congen.de). Briefly, 200 μl of bacterial culture or 200 mg of food samples were suspended in 1 mL of lysis buffer containing cetyltrimethylammonium bromide, with 2.5 μl of proteinase and kept at 60 °C for 30 min then cooled and centrifuged. 700 μl of the solution mixture was mixed with 500 μl of chloroform and precipitated by centrifugation with 14,000 g. The portion of the supernatant was mixed with PB buffer containing guanidine hydrochloride and isopropanol in QIAquick spin column and centrifuged at 18000 g for 1 min. The flow through was discarded and the content was washed with AW2 buffer and dried. Finally, 150 μl of elution buffer was added and incubated for 1 min at room temperature and centrifuged to collect the DNA.
Real time PCR
Real time RCT was done according to the CONGEN protocol. Total volume of 20 μl of PCR mixture containing 5 μl of extracted DNA and FAM was used as a fluorescent probe. Amplification of DNA was monitored from the increase in the fluorescence of FAM at 520 nm in real time. Positive control reagent and master mixture were used for positive and negative control, respectively.
Quantitative measurements
Our assay was intended to be used for visual observation of the colour change of the cotton swabs from white to colour via bare eye. The intensity of the colour on the cotton swabs for the targeted bacteria was increased by increasing the bacteria concentrations from 10 cfu.mL−1 to 10 × 108 cfu.mL−1 for St, Se and Cj. However, for quantitative measurements, the intensity of the colour was determined by using the ImageJ program developed at National Institute of Health after taking images for the cotton swabs using a smart phone. The calibration curves were plotted as the colour intensity for each bacteria concentration as a function of bacteria concentration.
Results and discussion
Detection of pathogenic bacteria
Because of their low cost and ease of use, cotton swabs were chosen in this work to play dual function as sample collector from solid surfaces and substrate for the immunoassay. Cotton is 99% cellulose with polyhydroxy groups. These hydroxyl groups can be easily oxidized to active aldehyde groups for the immobilization of biomolecules such as antibodies. Appearance of new low intense peak in the FTIR at 1730 cm−1 in the activated cotton confirms the presence of aldehyde group. Pathogenic bacteria such as St, Se, Sa and Cj was screened with a sandwich assay. The pathogenic bacteria were sandwiched between the primary antibody immobilized on the cotton swab surface and a secondary antibody conjugated on colored nanobeads as shown in Scheme 1. The target bacteria cells were preconcentrated by capturing the cells from the surfaces by the cotton swabs to form antibody-cell complex due to antigen-antibody interactions. In the subsequent step, the secondary antibody conjugated with colored nanobeads binds to the cells captured by the primary antibody. As the antibodies are specific to the target cells, only a particular type of colored nanobeads will bind to the captured cell causing a change in the color of the cotton surface.
Figure 1 shows the detection results for St using the proposed approach. The cotton swabs linked to St C-Ab was swabbed over surfaces of chicken meat, glass and stainless steel which were artificially contaminated with St. After that, the complex was treated with a solution of St D-Ab linked to magnetic nanoparticles and a black color was detected. Figure 1a, b, c show the immunoassay color response for St bacteria concentrations (10 to 108 cfu.mL−1) spiked on chicken meat, glass and stainless steel surfaces, respectively. The unbounded magnetic beads were collected either by washing or by passing permanent magnet sheet close to the cotton swab. Thus, the magnetic beads offer an advantage over the polymer nanobeads by the capability of eliminating the last washing step. As shown in Fig. 1, the intensity of the black color on the cotton surface increases proportionally with increasing the concentration of the cell counts for all the samples. When the cotton swab is treated with high concentration of St bacteria, more cells was captured by the capture antibodies and therefore, higher number of magnetic beads were attached to the beads. The detection limit (LOD) of this simple assay can be determined visually by human bare eye. For the St results, it is clear that the visual LOD of St from the chicken meat and the stainless steel plates were 101 cfu.mL−1, whereas, the LOD from the glass slide was 102 cfu.mL−1. The strong physical adsorption of St bacterial cells on glass is one possible reason for the higher LOD for St on glass plates. The LOD obtained using our method (10 cfu.mL−1) for the chicken and stainless steel surfaces is in comparable with the previous results done by using immunomagnetic nanosphere and immunofluorescent nanosphere- based assays [26].
We have then applied the assay for the detection of the other bacteria strains; Se, Sa and Cj and the results are shown in the supporting information file (Fig. S2,3,4). Similar trend was observed in all cases where the color intensity increased with increasing the bacteria cell count. The LODs for Se were 102,101 and 101 cfu.mL−1 for the glass, chicken and stainless steel surfaces, respectively. This result indicates better sensitivity of our assay compared with the previously reported results by FRET- based detection of Se in milk and water (102 to103 cfu.mL−1) [27].
For the Sa assay, the LOD was 101 cfu.mL for all the three surfaces. Zelada-Guillén et al. have developed a biosensor for the detection of Sa on the skin using anti Sa DNA aptamer as a recognition receptor and single-walled carbon nanotubes (SWCNTs) as an ion-to-electron potentiometric transducer. This sensor has achieved higher LOD (103 cfu.mL−1) [28] compared to our method.
For Cj, the LOD was also 10 cfu.mL−1 for the samples from stainless steel surface. However, the LODs on chicken and glass plates were 102 cfu.mL−1 presumably due to the strong interaction between those surfaces and campylobacter. Recent report on quartz crystal microbalance (QCM)-based sensor for the detection of Cj has shown a LOD of 150 cfu.mL-1 [29] that is comparable to our LOD. A comparison of the detection range and LOD of the four bacteria stains on different surfaces is shown in Table 1. This method obviously has comparable and - in some cases - even better sensitivity than the reported methods. Moreover, our colorimetric assay offers several advantages over reported assays in terms of simplicity, low cost and capability of using it for on-site detection as it is instrument-free. On the other hand, as shown in Fig. 2, by using ImagJ software, we can determine the color intensity and plot a relationship between the cell count of each bacteria and the intensity of colors on the cotton swabs indicating that this method can be used not only for semi-quantitative analysis but also for accurate quantitative detection provided that the photos are taken from the same angle.
PCR confirmation assays
The confirmation of binding of each bacteria with its corresponding specific antibody were further confirmed by real-time PCR. After performing the immunoassay on the cotton swabs, the bacteria were eluted in buffer and the DNA was extracted following the protocol described in the experimental part. Figure 3 shows the real-time PCR results for various concentrations of the three targeted bacteria Se, Sa and Cj DNA collected from contaminated stainless plates. Figure 3 shows that there is an increase in the DNA amplification with increasing the cell counts of the three bacteria from 10 to 108 cfu.mL−1 which is in agreement with the color intensity response obtained by our method indicating the accuracy of our assay.
Selectivity tests
Selectivity is a major indicator of the success of the assay performance. Figure 4 summarizes the cross reactivity results for the four targeted bacteria. Four cotton swabs conjugated with antibody specific for each bacteria strain were used individually for swabbing four surfaces which were artificially contaminated with St, Se, Sa and Cj. Each cotton swab was further incubated with a developing solution, which contains a cocktail of nanobeads with different colors. Each color conjugated with specific antibodies for the various targeted bacteria. After extensive washing, the colored beads on cotton swabs treated with St, Se, Sa and Cj were washed away. Only the cotton swabs treated with specific bacteria for a particular assay exhibited a color which corresponds to the specific bacteria. No significant color was observed on the non specific assays indicating good selectivity of the immunoassays.
Conclusion
In this work, a simple, rapid, low-cost detection assay for pathogenic bacteria on the surfaces of poultry processing plant is described. A specific antibody immobilized on cotton swab is used for preconcentrating the pathogens. The captured bacteria are then detected colorimetrically with a secondary antibody conjugated to specific colored nanobeads. We have demonstrated the detection of Salmonella Typhimurium, Salmonella Enteritidis, Staphylococcus aureus and Campylobacter jejuni by the formation of various colors on cotton surfaces. Our method offers advantages over other assays in terms of simplicity and low cost since the cotton swab was used for both collecting the sample as well as for detection. Moreover, the assay showed good sensitivity and selectivity. It can be used by non-skilled personnel as it is instrument-free. However, this assay is semi- quantitative and can be integrated with a smart phone in order to obtain quantitative results. We believe that this immunoassay can have a potential application for the rapid screening of bacteria in food and contaminated surfaces.
References
Canny GO, McCormick BA (2008) Bacteria in the intestine, helpful residents or enemies from within? Infect Immun 76:3360–3373
Zhang Y-J, Li S, Gan R-Y, Zhou T, Xu D-P, Li H-B (2015) Impacts of gut bacteria on human health and diseases. Int J Mol Sci 16:7493–7519
Dhama K, Rajagunalan S, Chakraborty S, Verma AK, Kumar A, Tiwari R, Kapoor S (2013) Food-borne pathogens of animal origin-diagnosis, prevention, control and their zoonotic significance: a review. Pak J Biol Sci 16:1076–1085
Cantas L, Suer K (2014) Review: the important bacterial Zoonoses in “one health” concept. Front Public Health 2:144
Yehia HM, Al-Dagal MM (2014) Prevalence of campylobacter jejuni in chicken produced by major poultry companies in Saudi Arabia. Int J Food Contam 1:1–5
Mohammed Zourob SE, Anthony Turner (2008) Principles of bacterial detection: biosensors, recognition receptors and microsystems. https://doi.org/10.1007/978-0-387-75113-9
Zhao X, Lin CW, Wang J, Oh DH (2014) Advances in rapid detection methods for foodborne pathogens. J Microbiol Biotechnol 24(3):297–312
Wang Z, Wang D, Chen J, Sela DA, Nugen SR (2016) Development of a novel bacteriophage based biomagnetic separation method as an aid for sensitive detection of viable Escherichia coli. Analyst 141:1009–1016
Casale WL, Pestka JJ, Hart LP (1988) Enzyme-linked immunosorbent assay employing monoclonal antibody specific for deoxynivalenol (vomitoxin) and several analogs. J Agric Food Chem 36:663–668
Morales-Narváez E, Naghdi T, Zor E, Merkoçi A (2015) Photoluminescent lateral-flow immunoassay revealed by graphene oxide: highly sensitive paper-based pathogen detection. Anal Chem 87:8573–8577
Song E, Yu M, Wang Y, Hu W, Cheng D, Swihart MT, Song Y (2015) Multi-color quantum dot-based fluorescence immunoassay array for simultaneous visual detection of multiple antibiotic residues in milk. Biosens Bioelectron 72:320–325
Poltronieri P, Mezzolla V, Primiceri E, Maruccio G (2014) Biosensors for the detection of food pathogens. Foods 3:511–526
Elshafey R, Siaj M, Zourob M (2015) DNA aptamers selection and characterization for development of label-free impedimetric aptasensor for neurotoxin anatoxin-a. Biosens Bioelectron 68:295–302
Mungkarndee R, Tumcharern G, Thiramanus R, Techakriengkrai I, Sukwattanasinitt M (2015) Fluorescence sensor arrays for identification of foodborne pathogens. Anal Methods 7:7431–7435
Mairal T, Nadal P, Svobodova M, O'Sullivan CK (2014) FRET-based dimeric aptamer probe for selective and sensitive Lup an 1 allergen detection. Biosens Bioelectron 54:207–210
Law JW-F, Ab Mutalib N-S, Chan K-G, Lee L-H (2014) Rapid methods for the detection of foodborne bacterial pathogens: principles, applications, advantages and limitations. Front Microbiol 5:770
Bhunia AK, Nanduri V, Bae E, Hirleman ED, (2009) Biosensors, foodborne pathogen detection. Encyclopedia of Industrial Biotechnology. Wiley. https://doi.org/10.1002/9780470054581.eib158
Mukundan H, Anderson AS, Grace WK, Grace KM, Hartman N, Martinez JS, Swanson BI (2009) Waveguide-based biosensors for pathogen detection. Sensors 9:5783–5809
Chen L, Mungroo N, Daikuara L, Neethirajan S (2015) Label-free NIR-SERS discrimination and detection of foodborne bacteria by in situ synthesis of Ag colloids. J Nanobiotechnology 13:1–9
Charlermroj R, Himananto O, Seepiban C, Kumpoosiri M, Warin N, Oplatowska M, Gajanandana O, Grant IR, Karoonuthaisiri N, Elliott CT (2013) Multiplex detection of plant pathogens using a microsphere immunoassay technology. PLoS One 8:e62344
Wang Y, Alocilja EC (2015) Gold nanoparticle-labeled biosensor for rapid and sensitive detection of bacterial pathogens. J Biol Eng 9:16
Moore C, Corden S, Sinha J, Jones R (2008) Dry cotton or flocked respiratory swabs as a simple collection technique for the molecular detection of respiratory viruses using real-time NASBA. JVirolog Met 153(2):84–89
Qu L-L, Geng Y-Y, Bao Z-N, Riaz S, Li H (2016) Silver nanoparticles on cotton swabs for improved surface-enhanced Raman scattering, and its application to the detection of carbaryl. Microchim Acta 183(4):1307–1313
Park GW, Chhabra P, Vinj J (2017) Swab sampling method for the detection of human norovirus on surfaces. J Vis Exp (120):e55205. https://doi.org/10.3791/55205
DiRenzo GV, Campbell Grant EH, Longo AV, Che-Castaldo C, Zamudio KR, Lips KR (2017) Imperfect pathogen detection from non-invasive skin swabs biases disease inference. Methods Ecol Evol 1–10. https://doi.org/10.1111/2041-210X.12868
Wen C-Y, Hu J, Zhang Z-L, Tian Z-Q, Ou G-P, Liao Y-L, Li Y, Xie M, Sun Z-Y, Pang D-W (2013) One-step sensitive detection of salmonella typhimurium by coupling magnetic capture and fluorescence identification with functional nanospheres. Anal Chem 85:1223–1230
Duan N, Gong W, Wang Z, Wu S (2016) An aptasensor based on fluorescence resonance energy transfer for multiplexed pathogenic bacteria determination. Anal Methods 8:1390–1395
Zelada-Guillén GA, Sebastián-Avila JL, Blondeau P, Riu J, Rius FX (2012) Label-free detection of Staphylococcus aureus in skin using real-time potentiometric biosensors based on carbon nanotubes and aptamers. Biosens Bioelectron 31:226–232
Masdor NA, Altintas Z, Tothill IE (2016) Sensitive detection of Campylobacter jejuni using nanoparticles enhanced QCM sensor. Biosens Bioelectron 78:328–336
Salam F, Tothill IE (2009) Detection of Salmonella typhimurium using an electrochemical immunosensor. Biosens Bioelectron 24:2630–2636
Wang X, Niazi S, Yukun H, Sun W, Wu S, Duan N, Hun X, Wang Z (2017) Homogeneous time-resolved FRET assay for the detection of Salmonella typhimurium using aptamer-modified NaYF4:Ce/Tb nanoparticles and a fluorescent DNA label. Microchim Acta 184(10):4021–4027
Duan N, Chang B, Zhang H, Wang Z, Wu S (2016) Salmonella typhimurium detection using a surface-enhanced Raman scattering-based aptasensor. Int J Food Microbiol 218:38–43
Ma X, Song L, Xia Y, Jiang C, Wang Z (2017) A novel colorimetric detection of S. typhimurium based on Fe3O4 magnetic nanoparticles and gold nanoparticles. Food Anal Methods 10:2735–2742
Chai Y, Li S, Horikawa S, Park MK, Vodyanoy V, Chin BA (2012) Rapid and sensitive detection of salmonella typhimurium on eggshells by using wireless biosensors. J Food Prot 75:631–636
Lee SH, Jung BY, Rayamahji N, Lee HS, Jeon WJ, Choi KS, Kweon CH, Yoo HS (2009) A multiplex real-time PCR for differential detection and quantification of Salmonella spp., Salmonella enterica serovar Typhimurium and Enteritidis in meats. J Vet Med Sci 10:43–51
Si S-H, Li X, Fung Y-S, Zhu D-R (2001) Rapid detection of Salmonella enteritidis by piezoelectric immunosensor. Microchem J 68:21–27
Song Y, Li W, Duan Y, Li Z, Deng L (2014) Nicking enzyme-assisted biosensor for Salmonella enteritidis detection based on fluorescence resonance energy transfer. Biosens Bioelectron 55:400–404
Botaro BG, Cortinhas CS, Março LV, Moreno JFG, Silva LFP, Benites NR, Santos MV (2013) Detection and enumeration of Staphylococcus aureus from bovine milk samples by real-time polymerase chain reaction. J Dairy Sci 96:6955–6964
Chen Z, Zhang K, Yin H, Li Q, Wang L, Liu Z (2015) Detection of Salmonella and several common Salmonella serotypes in food by loop-mediated isothermal amplification method. Food Sci Human Wellness 4:75–79
Banada PP, Chakravorty S, Shah D, Burday M, Mazzella FM, Alland D (2012) Highly sensitive detection of Staphylococcus aureus directly from patient blood. PLoS One 7:e31126
Brakstad OG, Aasbakk K, Maeland JA (1992) Detection of Staphylococcus aureus by polymerase chain reaction amplification of the nuc gene. J Clin Microbiol 30:1654–1660
Wang X, Du Y, Li Y, Li D, Sun R (2011) Fluorescent identification and detection of Staphylococcus aureus with carboxymethyl chitosan/CdS quantum dots bioconjugates. J Biomater Sci Polym Ed 22:1881–1893
Esteban-Fernández de Ávila B, Pedrero M, Campuzano S, Escamilla-Gómez V, Pingarrón JM (2012) Sensitive and rapid amperometric magnetoimmunosensor for the determination of Staphylococcus aureus. Anal Bioanal Chem 403:917–925
Miao T, Wang Z, Li S, Wang X (2011) Sensitive fluorescent detection of Staphylococcus aureus using nanogold linked CdTe nanocrystals as signal amplification labels. Microchim Acta 172:431–437
Xiong J, Wang W, Fu Z (2017) Fluorimetric sandwich affinity assay for Staphylococcus aureus based on dual-peptide recognition on magnetic nanoparticles. Microchim Acta 184(10):4197–4202
LLT T, Cao C, Høgberg J, Wolff A, Bang DD (2012) Isolation and detection of campylobacter jejuni from chicken fecal samples by immunomagnetic separation–PCR. Food Control 24:23–28
Saiyudthong S, Phusri K, Buates S (2015) Rapid detection of campylobacter jejuni, campylobacter coli, and campylobacter lari in fresh chicken meat and by-products in Bangkok, Thailand, using modified multiplex PCR. J Food Prot 78:1363–1369
Xu D, Wu X, Li B, Li P, Ming X, Chen T, Wei H, Xu F (2013) Rapid detection of Campylobacter jejuni using fluorescent microspheres as label for immunochromatographic strip test. Food Sci Biotechnol 22:585–591
Acknowledgements
M. Z. would like to acknowledge the fund from the ORG office at Alfaisal University under grant Number: 413130609151. S. A. would like to acknowledge the financial support from the government of Saudi Arabia for the scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 540 kb)
Rights and permissions
About this article
Cite this article
Alamer, S., Eissa, S., Chinnappan, R. et al. A rapid colorimetric immunoassay for the detection of pathogenic bacteria on poultry processing plants using cotton swabs and nanobeads. Microchim Acta 185, 164 (2018). https://doi.org/10.1007/s00604-018-2696-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2696-7