Abstract
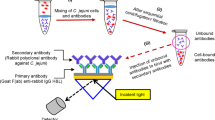
Campylobacter jejuni is a worldwide foodborne pathogen recognized as a leading cause of human gastrointestinal enteritis. A rapid, sensitive, and specific method is required to monitor food and water in cases of contamination by this pathogen. This report presents a novel immunochromatographic test (ICT) using fluorescent microspheres labeled with polyclonal antibodies of C. jejuni as the capture reagent dispensed onto the conjugate pad. Polyclonal antibodies against the outer membrane protein PEB1 of C. jejuni were used as the detective reagent at the test line, whereas the goat anti-rabbit IgG was used on the control line. PEB1 was obtained by gene cloning and expression to prepare its antibody. In this study, a simple and rapid ICT is reported for detecting C. jejuni for the first time with a detection limit of 106 CFU/ mL.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Tauxe RV. Incidence, trends, and sources of campylobacteriosis in developed countries: An overview. pp. 42–43. In: The Increasing Incidence of Human Campylobacteriosis. Report and proceedings of a WHO consultation of experts. (WHO/CDS/CSR/APH 2001.7) World Health Organization, Cophenhagen, Denmark (2001)
EFSA (European Food Safety Agency). The community summary report on trends and sources of zoonoses, zoonotic agents in the European Union in 2007. J. EFSA 223: 223–226 (2009)
Bardon J, Kolar M, Cekanova L, Hejnar P, Koukalova D. Prevalence of Campylobacter jejuni and its resistance to antibiotics in poultry in the Czech Republic. Zoonoses Public Hlth. 56: 111–116 (2009)
Tauxe RV. Epidemiology of Campylobacter jejuni infections in the United States and other industrialized nations. pp. 9–19. In: Campylobacter jejuni Current Status and Future Trends. Nachamkin I, Blaser M, Tompkins L. (eds). American Society for Microbiology, Washington, DC, USA (1992)
Habib I, Sampers I, Uyttendaele M, Berkvens D, Zutter L. Performance characteristics and estimation of measurement uncertainty of three plating procedures for Campylobacter enumeration in chicken meat. Int. J. Food Microbiol. 25: 65–74 (2008)
LaGier MJ, Joseph LA, Passaretti TV, Musser KA, Cirino NM. A real-time multiplexed PCR assay for rapid detection and differentiation of Campylobacter jejuni and Campylobacter coli. Mol. Cell. Probe. 18: 275–282 (2004)
Wang G, Clark CG, Taylor TM, Pucknell C, Barton C, Price L, Woodward DL, Rodgers FG. Colony multiplex PCR assay for identification and differentiation of Campylobacter jejuni, C. coli, C. lari, C. upsaliensis, and C. fetus subsp. fetus. J. Clin. Microbiol. 40: 4744–4747 (2002)
Burnett TA, Hornitzky MA, Kuhnert P, Djordjevic SP. Speciating Campylobacter jejuni and Campylobacter coli isolates from poultry and humans using six PCR-based assays. FEMS Microbiol. Lett. 216: 201–209 (2002)
Wei D, Oyarzabal OA, Huang TS, Balasubramanian S, Sista S, Simonian AL. Development of a surface plasmon resonance biosensor for the identification of Campylobacter jejuni. J. Microbiol. Meth. 69: 78–85 (2007)
Kanki M, Sakata J, Taguchi M, Kumeda Y, Ishibashi M, Kawai T, Kawatsu K, Yamasaki W, Inoue K, Miyahara M. Effect of sample preparation and bacterial concentration on Salmonella enterica detection in poultry meat using culture methods and PCR assaying of preenrichment broths. Int. J. Food Microbiol. 26: 1–3 (2009)
Fauchere JL, Kervella M, Rosenau A, Mohanna K, Veron M. Adhesion to HeLa cells of Campylobacter jejuni and C. coli outer membrane components. Res. Microbiol. 140: 379–392 (1989)
Pei Z, Blaser MJ. PEB1, the major cell-binding factor of Campylobacter jejuni, is a homolog of the binding component in Gram-negative nutrient transport systems. J. Biol. Chem. 268: 18717–18725 (1993)
Du LF, Li ZJ, Tang XY, Huang JQ, Sun WB. Immunogenicity and immunoprotection of recombinant PEB1 in Campylobacter jejuni-infected mice. World J. Gastroenterol. 14: 6244–6248 (2008)
Kawatsu K, Kumeda Y, Taguchi M, Yamazaki-Matsune W, Kanki M, Inoue K. Development and evaluation of immunochromatographic assay for simple and rapid detection of Campylobacter jejuni and Campylobacter coli in human stool specimens. J. Clinteral. Microbiol. 46: 1226–1231 (2008)
Sharp JM, Doran PM. Strategies for enhancing monoclonal antibody accumulation in plant cell and organ cultures. Biotechnol. Progr. 17: 979–992 (2001)
Bibila TA, Robinson DK. In pursuit of the optimal fed-batch process for monoclonal antibody production. Biotechnol. Progr. 11: 1–13 (1995)
Farboud B, Aotaki KA, Miyata T, Hjelmeland LM, Handa JT. Development of a polyclonal antibody with broad epitope specificity for advanced glycation endproducts and localization of these epitopes in Bruch’s membrane of the aging eye. Mol. Vis. 5: 11–15 (1999)
Bradley M, Ashokkumar M, Grieser F. Sonochemical production of fluorescent and phosphorescent latex particles. J. Am. Chem. Soc. 125: 525–529 (2003)
Endtz H, Ang C, Van Den Braak N, Luijendijk A, Jacobs B, De Man P, Van Duin J, Van Belkum A, Verbrugh H. Evaluation of a new commercial immunoassay for rapid detection of Campylobacter jejuni in stool samples. Eur. J. Clin. Microbiol. 19: 794–797 (2000)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, D., Wu, X., Li, B. et al. Rapid detection of Campylobacter jejuni using fluorescent microspheres as label for immunochromatographic strip test. Food Sci Biotechnol 22, 585–591 (2013). https://doi.org/10.1007/s10068-013-0118-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-013-0118-5