Abstract
The voltammetry of imidacloprid was investigated by using three kinds of carbon paste electrodes (CPEs) based on tricresyl phosphate, silicone oil, and n-tetradecane. The tricresyl phosphate-based carbon paste electrode exhibited the best analytical performance with respect to peak shape and signal intensity. The method, operated in the differential pulse voltammetric mode, was applied to the determination of imidacloprid in a river water sample and two commercial formulations and works in the concentration range from 1.7 to 30 μg mL−1, with a relative standard deviation not exceeding 2.2%. This appears to be the first application of a CPE to the voltammetric determination of neonicotinoid insecticides.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Imidacloprid ((EZ)-1-(6-chloro-3-pyridylmethyl)-N-nitroimidazolidin-2-ylideneamine) belongs to the fastest growing class of insecticides, called neonicotinoids, which are accounting for 11–15% of the total insecticide market [1–4]. Since its launch in 1991, products containing imidacloprid have gained registrations in about 120 countries and are marketed for use in agriculture (for over 140 agricultural crops), on turf, on pets, and for household pests [4, 5]. Imidacloprid is marketed under variety of names including Gaucho, Merit, Admire, Confidor, Macho and Winner. Although imidacloprid has been in use for a relatively short period compared to other common pesticides, it is considered to being used in the largest volume globally of all insecticides [4, 6, 7]. The mechanism of imidacloprid action has been studied extensively, and is relatively well known. It acts as agonist by binding to nicotinic acetylcholine receptor in the insect’s nervous system. This leads to the accumulation of acetylcholine, resulting in the paralysis and death of insects [8].
Because of wide and constantly growing areas of application, high water solubility (0.58 g L−1) and water stability of >30 days (at pH 5–7) [2], imidacloprid is increasingly present in the environment. All above facts indicate the need for new analytical methods for the determination of this neonicotinoid, both in commercial formulations and real samples from the environment, using possibly simple and low cost instrumental techniques.
Widely used analytical techniques for imidacloprid determination are high-performance liquid chromatography (HPLC) with diode-array [9–11], mass-spectrometric [12, 13], thermal lens spectrometric [14] or amperometric detection [15]. Some alternative techniques like an enzyme-linked immunosorbent assay [16, 17], fluorimetry [18] and Fourier transform infrared spectroscopy [19] have also been employed for rapid, selective analysis of different samples containing imidacloprid. Several electroanalytical methods such as differential-pulse polarography (DPP) [20] or square-wave adsorptive stripping voltammetry on a hanging mercury drop electrode (HMDE) [21]) have also been described for the determination of imidacloprid in commercial formulations and spiked river water sample. The applicability of bismuth-film modified glassy carbon electrode for the monitoring of photocatalytic degradation of imidacloprid on TiO2 catalyst was also described [22], widening thus the field of use of environment friendly bismuth-modified carbon-based electrodes [23, 24]. On the other hand, a recent publication [25] described the photochemical transformation of imidacloprid, allowing highly sensitive and selective, indirect determination of both parent compounds and their stable degradation products using HPLC with an electrochemical detector.
While voltammetric techniques have often been used to analyze pesticides in general, to our knowledge, there are no publications dealing with the electroanalytical determination of imidacloprid using carbon paste electrodes (CPEs). These electrodes have been widely used in very different types of chemical analysis, including also voltammetric determination of pesticides [26]. In the literature, the determination of some well known agricultural pollutants like paraquat, diquat, paraoxon, methyl parathion, fenitrothion, metamitron, diazinon are described [27–34]. On the other hand, a few publications only are dealing with tricresyl phosphate-based carbon paste electrode (TCP-CPE) and its applicability in voltammetric analysis. To date, this type of CPE, containing chemically active organic ester instead of common binder [35], has been employed for the determination of gold and bismuth [35], silver [36], and iodide [37]; the latter in the stripping potentiometric mode. As far as we know, there are no publications yet that would deal with the application of the TCP-CPE to the determination of organic compounds.
In this work, a voltammetric investigation of imidacloprid was performed at different CPEs and an electroanalytical method has been developed for its determination by DPV using a TCP-CPE in aqueous Britton–Robinson buffer solution (pH 7.0) as supporting electrolyte. The developed voltammetric procedure was tested by determining imidacloprid in river water sample (Danube) and two commercial formulations of imidacloprid (Macho 200 SL and Confidor 200 SL). The results of the electroanalytical method developed were compared with those obtained by HPLC/DAD procedure.
Experimental
Chemicals and solutions
All chemicals used were of analytical reagent grade and solutions were prepared in doubly distilled water. Analytical standard of imidacloprid (Riedel-de Haën, Germany, http://www.sigmaaldrich.com/) was of 99.9% purity. The concentration of the imidacloprid stock solution was 166.7 μg mL−1 and it was further diluted as required. Britton–Robinson buffer solutions for voltammetric characterization and determination were prepared from a stock solution containing 0.04 M phosphoric (Merck, Germany, http://www.merck.de/), boric (Merck) and acetic (Merck) acids, respectively by adding 0.2 M sodium hydroxide (Merck) to obtain the required pH value. To prepare the mobile phase in HPLC experiments, acetonitrile (J. T. Baker, Netherlands, purity 99.8%, http://www.jtbaker.com/) and 0.2% phosphoric acid (made by diluting phosphoric acid (Centrohem, Serbia, http://www.centrohem.co.rs/)) were used. Commercial formulations of imidacloprid were Macho 200 SL (Hemovet, Serbia, http://www.hemovet.com/) and Confidor 200 SL (Bayer CropScience, Germany, http://www.bayercropscience.com/).
Apparatus
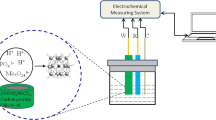
Voltammetric experiments were performed on an AUTOLAB (PGSTAT12, Ecochemie, Utrecht, The Netherlands, http://www.ecochemie.nl/) electrochemical analyzer operated via GPES 4.9 software (Ecochemie). The cell stand included a three-electrode system with CPE or glassy carbon electrode, GCE (Amel, Italy, http://www.amelchem.com/) as working, an SCE (Amel) as reference, and a platinum (Amel) auxiliary electrode. All potentials are quoted vs. SCE reference electrode.
The surface morphology of TCP-CPE was studied on a JEOL JSM-6460LV (Japan Electron Optics Laboratory, Japan, http://www.jeol.com/) scanning electron microscope (SEM).
Comparative HPLC measurements were performed using an Agilent 1100 liquid chromatograph (Agilent Technologies Inc., USA, http://www.agilent.com/), Zorbax Eclipse XDB-C18 (4.6 mm × 250 mm, 3.5 μm) column, and DA-detector.
Procedures
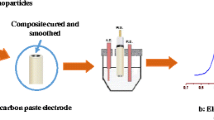
Preparation of the CPEs. Carbon pastes were made by intimate hand-mixing of “CR 5” graphite powder (Maziva Týn, Týn nad Vltavou, Czech Republic, http://www.grafit.cz/) with the corresponding liquid binder. Three different carbon pastes were prepared: “SO-CPE” type, containing Lukooil silicone oil (MV 8,000, Lučební Závody, Kolín, Czech Republic, http://www.lucebni.cz/), “C14-CPE” with n-tetradecane (Polyscience Corporation, Evanston, USA, http://www.polyscience.com/), and “TCP-CPE” prepared from tricresyl phosphate (mixture of isomers, Fluka, http://www.sigmaaldrich.com/). All three pastes, homogenized at the same ratio of 0.25 g graphite + 0.1 mL pasting liquid, were packed into a piston-driven Teflon® holder [38]. The electrode surface of CPEs (2 mm in diameter) was renewed mechanically by smoothing some paste off with a wet piece of filter paper before starting a new set of experiments.
Voltammetry on CPEs. In model systems and real samples imidacloprid was measured in 4.00 mL of the solution of different concentration, to which 1.00 mL of Britton–Robinson buffer solution was added. Scan rate in linear scan voltammetry (LSV) was 100 mV s−1. Measurement parameters in DPV were as follows: pulse amplitude 50 mV, pulse width 50 ms, scan rate 25 mV s−1 [22, 39]. The solutions in the case of model systems and commercial formulations were measured without filtering. The solutions were deaerated by passing a nitrogen stream through them for 10 min, and the voltammograms were registered at ambient temperature.
Scanning Electron Microscopy. The surface morphology of “CR 5” graphite powder and TCP-CPE was studied on a JEOL scanning electron microscope at an accelerating voltage of 20 kV in low vacuum mode.
Chromatography. For the HPLC/DAD analysis, all aliquots were filtered through Millex 0.22 μm syringe filters. The mobile phase was a mixture of water (0.2% phosphoric acid) and acetonitrile (70:30, v/v) [14]. The separation was performed at isocratic regime, the flow rate was 1.0 mL min−1 and the column temperature was held at 25 °C. Imidacloprid was detected at working wavelength of 270 nm with a reference wavelength of 500 nm and retention time of 3.5 min.
Real Sample Preparation. The water sample was collected from the Danube River (Novi Sad, Republic of Serbia) and stored in the dark at 4 °C. The aliquots of river water sample were spiked with the standard solution of imidacloprid and kept in the dark at 4 °C for 1 h before analysis without any kind of sample pretreatment. Analyses of commercial formulations Confidor 200SL and Macho 200SL were performed after a stepwise dilution with doubly distilled water to the required concentration range. Filtering of the prepared solutions was done just in the case of HPLC measurements.
Results and discussion
Voltammetric investigation of imidacloprid at CPEs
The applicability of the TCP-CPE prepared was investigated with the main objective of using it for the determination of imidacloprid, and the voltammetric response was compared with those obtained at SO-CPE and C14-CPE at pH 7.0 (Fig. 1), knowing that the neutral or slightly alkaline pH is the most acceptable for the determination of the nitro-group containing neonicotinoid insecticides [20–22, 39–41]. Relying on previous polarographic investigations [20, 41], the detection of imidacloprid was based on the irreversible reduction of its nitro group. Optimal working electode was selected by comparing the intensity and shape of the LSV, and first of all DPV, signals obtained with different CPEs (Fig. 1a and b). The advantage of the TCP-CPE over SO-CPE and C14-CPE is evident. Namely, the shapes of voltammograms recorded for TCP-CPE in the both LSV and DPV experiments are clearly defined, with lowest residual current and highest peak currents in comparison with other CPEs. Such behavior of the TCP-CPE is probably a result of its polarity, which facilitates contact of polar imidacloprid molecules with the electrode surface, in contrast to CPEs with nonpolar binders. The imidacloprid peak at TCP-CPE is located a bit more negative than at SO-CPE (Fig. 1b) and GCE (not shown), but this did not affect the determination, because of the wide potential window in the cathodic range at TCP-CPE. As was shown earlier, the optimal measuring parameters in DPV determination of imidacloprid were as follows: pulse amplitude, 50 mV; pulse width, 50 ms; and scan rate, 25 mV s−1 [22, 39]. Although the previous results [42] showed that the effect of dissolved oxygen on TCP-CPE signal was not significant, purging of solutions with nitrogen (10 min) led to a lower background current and better reproducibility of determination in this fairly negative region. Therefore, all systems were purged with above mentioned inert gas. It has been shown earlier that the use of membrane plasticizers (e.g. TCP, dioctyl phthalate) in CPEs as pasting liquids, in comparison with usual binders as Nujol or SO, can significantly reduce the amount of oxygen absorbed in the graphite [35]. Hence, no special attention was paid to the oxygen in the paste. It is also well known that the electrochemical properties of carbon-based electrodes can be improved and stabilized by potential cycling. In our case, before the measurement, all electrodes were subjected to electrochemical activation by potential cycling in the range from −0.60 to −1.60 V (10 cycles). Increasing the number of potential cycles did not lead to the further widening of the potential window, lowering of the background current and signal stabilization.
Comparison of LSV (a) and DPV (b) signals obtained at different working electrodes: SO-CPE (1), C14-CPE (2) and TCP-CPE (3) in the same imidacloprid solution; and the influence of pH (the pHs of the solutions were indicated at the curves) on LSV signals (c) and peak current (d) at TCP-CPE. Measurement parameters: v = 100 mV s−1 (a, c) or 25 mV s−1 (b), pulse amplitude 50 mV (b), pulse width 50 ms (b), c = 33.34 μg mL−1 (a–d), pH = 7.0 (a, b)
Before application of TCP-CPE for a quantitative determination of imidacloprid, additional characterization of the working electrode was performed. LSV curves were recorded in the pH range 2.0 to 8.0, to study the pH dependence of peak potentials, whereby they shifted to more negative values with the increase of pH. The sharpest, most symmetrical and intense peaks (Fig. 1c and d) were obtained for neutral and slightly alkaline solutions, which is in good agreement with previous voltammetric investigations of neonicotinoids with nitro-group [20–22, 39–41]. On the basis of these statements, and with the aim of avoiding the possible hydrolysis of the TCP binder in alkaline media, the pH 7.0 was chosen for imidacloprid determination. Reproducibility studies at TCP-CPE were also performed, first of all to check the signal stability and possible changes in the signal shape because of potential absorption or adsorption processes. Figure 2, on which records are presented for repeating measurements of the 15.11 μg mL−1 imidacloprid solution, shows good reproducibility of the analytical signal in the time interval of approximately 30 min, with no significant changes in the electrode properties during measurements.
The reproducibility of the analytical signal of imidacloprid at TCP-CPE at the concentration level of 15.11 μg mL−1 in 30 min time interval. Other experimental parameters are the same as in Fig. 1b
Surface characteristics of TCP-CPE were examined by SEM. Characteristic particles of CR5 graphite powder with average size of 5 µm are recognizable in the structure of the TCP-CPE, rather connected to each other, forming relatively compact mass (Fig. 3).
Determination of imidacloprid in model systems and selected real samples
The quantitative DPV determination of imidacloprid at TCP-CPE in model systems is based on the linear relationship between the peak current intensity and imidacloprid concentration. The analytical parameters of the developed DPV method and the comparative HPLC-DAD measurements are shown in the Table 1. As can be seen, imidacloprid could be determined by DPV (Fig. 4) in the concentration range of 1.73–30.0 μg mL−1, while the RSD (assessed by six replicate measurements at 3.26 μg mL−1) did not exceed 1.4%.
Differential pulse voltammograms recorded at TCP-CPE for different imidacloprid concentrations in Britton–Robinson buffer solution. Other experimental parameters are the same as in Fig. 1b
The applicability of the voltammetric procedure was tested by determining imidacloprid in several real samples. As can be seen from Fig. 5, matrix from the water sample (Danube) and commercial formulations (e.g. Confidor 200 SL) did not block the electrode surface and did not show voltammetric interferences, which are favorable facts for the determination. Standard addition method in the concentration range as in the case of model systems was applied for the determination of the active compound in real samples (Fig. 5). Good correlation between amounts determined and declared or added, as well as low RSDs, reflect the high accuracy and precision of the proposed method (Table 2). On the other hand, a somewhat lower precision observed for imidacloprid in commercial formulations in comparison with water samples arises probably from the sampling process of viscous and sticky liquid samples. In addition, the results obtained by DPV method compared well with HPLC/DAD analysis, both in the mean insecticide content and precision (Table 2). Though the chromatographic technique provides significantly more information about the examined system, voltammetry using TCP-CPEs can represent an alternative, rapid, inexpensive and, compared to polarographic/voltammetric techniques working with mercury electrodes, also an environmentally acceptable tool for giving information about the insecticide concentrations in commercial formulations and river water samples.
Conclusion
This study demonstrates the applicability of CPEs for the cathodic voltammetric determination of imidacloprid insecticide. LSV and DPV signals of imidacloprid were investigated at tricresyl phosphate-, silicone oil- and n-tetradecane-based CPEs. The sensitivity of the applied voltammetric procedures depends, among others, on the composition of the electrode and on the electrochemical pretreatment of the electrode surface. Also, the TCP-CPE with the most hydrophilic pasting liquid exhibited the most favorable analytical performance in respect of the peak shape and signal intensity.
The DPV with TCP-CPE in Britton–Robinson buffer solution of pH 7.0 was found to be applicable for imidacloprid determination with the linearity of response in the concentration range of 1.73 to 30.0 μg mL−1 and the respective LOD and LOQ values of 0.52 and 1.73 μg mL−1. The developed procedure might be applied as a convenient screening method for the determination of imidacloprid in selected real samples, even without complicated sample clean-up, which saves time, money and the environment. The voltammetric results were justified by the comparative HPLC/DAD measurements. This contribution is apparently the first application of a CPE for voltammetric investigation of neonicotinoid insecticides.
References
TDC Environmental (2003) Insecticide market trends and potential water quality implications. TDC Environmental, LLC, San Mateo, CA, USA
Jeschke P, Nauen R (2005) Neonicotinoid insecticides. In: Gilbert L, Iatrou K, Gill SS (eds) Comprehensive molecular insect science, vol 5. Elsevier, Amsterdam, pp 53–105
McCaffery A, Slater R (2005) IRAC Symposium on insecticide sustainability: neonicotinoids ESA annual meeting
Drobne D, Blažič M, Van Gestel CAM, Lešer V, Zidar P, Jemec A, Trebše P (2008) Toxicity of imidacloprid to the terrestrial isopod Porcellio scaber (Isopoda, Crustacea). Chemosphere 71:1326–1334
Liu HX, Song JM, Zhang SS, Qu LB, Zhao YF, Wu YJ, Liu HM (2005) Analysis of residues of imidacloprid in tobacco by high-performance liquid chromatography with liquid-liquid partition cleanup. Pest Manag Sci 61:511–514
Cox C (2001) Insecticide factsheet/Imidacloprid. J Pest Ref 21:15–21
Ware GW, Whitacre DM (2004) An introduction to insecticides. MeisterPro information resources, a division of Meister media worldwide, 4th edn. Willoughby, Ohio, USA
Iwasa T, Motoyama N, Ambrose JT, Roe MR (2004) Mechanism for the differential toxicity of neonicotinoid insecticides in the honey bee, Apis mellifera. Crop Prot 23:371–378
Obana H, Okihashi M, Akutsu K, Kitagawa Y, Hori S (2002) Determination of acetamiprid, imidacloprid and nitenpyram residues in vegetables and fruits by high-performance liquid chromatography with diode-array detection. J Agric Food Chem 50:4464–4467
Mandić A, Lazić S, Ökrész Sz, Gaál F (2005) Determination of the insecticide imidacloprid in potato (Solanum tuberosum L.) and onion (Allium cepa) by high-performance liquid chromatography with diode-array detection. J Anal Chem 12:1134–1138
Seccia S, Fidente P, Montesano D, Morrica P (2008) Determination of neonicotinoid insecticides residues in bovine milk samples by solid-phase extraction clean-up and liquid chromatography with diode-array detection. J Chromatogr A 1214:115–120
Obana H, Okihashi M, Akutsu K, Kitagawa Y, Hori S (2003) Determination of neonicotinoid pesticide residues in vegetables and fruits with solid phase extraction and liquid chromatography mass spectrometry. J Agric Food Chem 51:2501–2505
Seccia S, Fidente P, Barbini Attard D, Morrica P (2005) Multiresidue determination of neonicotinoid insecticide residues in drinking water by liquid chromatography with electrospray ionization mass spectrometry. Anal Chim Acta 553:21–26
Guzsvány V, Madžgalj A, Trebše P, Gaál F, Franko M (2007) Determination of selected neonicotinoid insecticides by liquid chromatography with thermal lens spectrometric detection. Environ Chem Lett 5:203–208
de Erenchun NR, de Balugera ZG, Goicolea MA, Barrio RJ (1997) Determination of imidacloprid and its major metabolite in soils by liquid chromatography with pulsed reductive amperometric detection. Anal Chim Acta 349:199–206
Watanabe E, Eun H, Baba K, Arao T, Ishii Y, Endo S, Ueji M (2004) Enzyme-linked immunosorbent assay for the neonicotinoid insecticide imidacloprid in agricultural samples. J Agric Food Chem 52:2756–2762
Watanabe E, Eun H, Baba K, Arao T, Ishii Y, Endo S, Ueji M (2004) Rapid and simple screening analysis for residual imidacloprid in agricultural products with commercially available ELISA. Anal Chim Acta 521:45–51
Vilchez JL, Valencia MC, Navalón A, Molinero-Morales B, Capitán-Vallvey LF (2001) Flow injection analysis of the insecticide imidacloprid in water samples with photochemically induced fluorescence detection. Anal Chim Acta 439:299–305
Quintás G, Armenta S, Garrigues S, de la Guardia M (2004) Fourier transform infrared determination of imidacloprid in pesticide formulations. J Braz Chem Soc 15:307–312
Navalón A, El-Khattabi R, González-Casado A, Vilchez JL (1999) Differential-pulse polarographic determination of the insecticide imidacloprid in commercial formulations. Mikrochim Acta 130:261–265
Guiberteau A, Galeano T, Mora N, Parrilla P, Salinas F (2001) Study and determination of the pesticide imidacloprid by square wave adsorptive stripping voltammetry. Talanta 53:943–949
Guzsvány V, Kádár M, Papp Z, Bjelica L, Gaál F, Tóth K (2008) Monitoring of photocatalytic degradation of selected neonicotinoid insecticides by cathodic voltammetry with a bismuth film electrode. Electroanalysis 20:291–300
Economou A (2005) Bismuth-film electrodes: recent developments and potentialities for electroanalysis. Trends Anal Chem 24:334–340
Claux B, Vittori O (2007) Bismuth film electrode as an alternative for mercury electrodes: Determination of azo dyes and application for detection in food stuff. Electroanalysis 19:2243–2246
Rancan M, Sabatini GA, Achilli G, Galletti CG (2006) Determination of imidacloprid and metabolites by liquid chromatography with an electrochemical detector and post column photochemical reactor. Anal Chim Acta 555:20–24
Švancara I, Vytřas K, Kalcher K, Walcarius A, Wang J (2009) Carbon paste electrodes in facts, numbers, and notes: a review on the occasion of the 50-years jubilee of carbon paste in electrochemistry and electroanalysis. Electroanalysis 21:7–28
El Mhammedi MA, Bakasse M, Bachirat R, Chtaini A (2008) Square wave voltammetry for analytical determination of paraquat at carbon paste electrode modified with fluoroapatite. Food Chem 110:1001–1006
El Mhammedi MA, Bakasse M, Najih R, Chtaini A (2009) A carbon paste electrode modified with kaolin for the detection of diquat. Appl Clay Sci 43:130–134
Chough SH, Mulchandani A, Mulchandani P, Chen W, Wang J, Rogers KR (2002) Organophosphorus hydrolase-based amperometric sensor: modulation of sensitivity and substrate selectivity. Electroanalysis 14:273–276
Fan S, Xiao F, Liu L, Zhao F, Zeng B (2008) Sensitive voltammetric response of methylparathion on single-walled carbon nanotube paste coated electrodes using ionic liquid as binder. Sensors and Actuators B 132:34–39
Liu G, Lin Y (2008) Electrochemical stripping analysis of organophosphate pesticides and nerve agents. Electrochem Commun 7:339–343
Lei Y, Mulchandani P, Chen W, Mulchandani A (2007) Biosensor for direct determination of fenitrothion and EPN using recombinant pseudomonas putida JS444 with surface-expressed organophosphorous hydrolase. 2. Modified carbon paste electrode. Appl Biochem Biotechnol 136:243–250
Arranz A, de Betoño SF, Moreda JM, Cid A, Arranz JF (1997) Preconcentration and voltammetric determination of the herbicide metamitron with a silica-modified carbon paste electrode. Mikrochim Acta 127:273–279
Ly SY (2008) Assay of diazinon pesticides in cucumber juice and in the deep brain cells of a live carp. Microchim Acta 163:283–288
Švancara I, Vytřas K (1993) Voltammetry with carbon paste electrodes containing membrane plasticizers used for PVC-based ion-selective electrodes. Anal Chim Acta 273:195–204
Švancara I, Kalcher K, Diewald W, Vytřas K (1996) Voltammetric determination of silver at ultratrace levels using a carbon paste electrode with improved surface characteristics. Electroanalysis 8:336–342
Švancara I, Konvalina J, Schachl K, Kalcher K, Vytřas K (1998) Stripping voltammetric determination of iodide with synergistic accumulation at a carbon paste electrode. Electroanalysis 10:436–441
Švancara I, Metelka R, Vytřas K (2005) Piston-driven carbon paste electrode holders for electrochemical measurements. In: Vytřas K, Kalcher K (eds) Sensing in electroanalysis, vol 1. University of Pardubice, Pardubice, Czech Republic, pp 7–18
Guzsvány V, Papp Z, Bjelica L, Gaál F (2008) Voltammetric monitoring of photocatalytic degradation of imidacloprid, nitenpyram and acetamiprid neonicotinoid insecticides. In: Vytřas K, Kalcher K, Švancara I (eds) Sensing in electroanalysis, vol 3. University of Pardubice, Pardubice, Czech Republic, pp 105–116
Guzsvány V, Gaál F, Bjelica L, Ökrész Sz (2005) Voltammetric determination of imidacloprid and thiamethoxam. J Serb Chem Soc 5:735–743
Guzsvány V, Kádár M, Gaál F, Tóth K, Bjelica L (2006) Rapid differential pulse polarographic determination of thiamethoxam in commercial formulations and some real samples. Microchim Acta 154:321–328
Kalcher K, Švancara I, Metelka R, Vytřas K, Walcarius A (2006) Heterogeneous carbon electrochemical sensors. In: Grimes CA, Dickey EC, Pishko MV (eds) Encyclopedia of Sensors, vol 4. American Scientific, Stevenson Ranch, California, U.S.A., pp 283–429
Acknowledgements
Authors acknowledge financial support of the Ministry of Science and Technological Development of the Republic of Serbia (Project No. 142029), the Secretariat for Science and Technological Development, AP Vojvodina, Republic of Serbia (Grant No. 114-451-02011/2007-01) and Central European Exchange Programme for University Studies (Project No. CII-CZ-0212-02-0809). Czech co-authors are also thankful for a partial support of the Ministry of Education, Youth, and Sports of the Czech Republic (Project No. MSM0021627502).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Papp, Z., Švancara, I., Guzsvány, V. et al. Voltammetric determination of imidacloprid insecticide in selected samples using a carbon paste electrode. Microchim Acta 166, 169–175 (2009). https://doi.org/10.1007/s00604-009-0181-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-009-0181-z