Abstract
Purpose
A variety of alternative grafts to autologous iliac crest bone (ICBG) have been developed for lumbar spondylodesis, due to frequent complications following ICBG harvest. The optimal alternative graft to ICBG, however, remains elusive till now. The purpose of this study was to compare the efficacy and safety of fusion materials in lumbar degeneration diseases and to provide a ranking spectrum of the grafts.
Methods
Randomized controlled trials (RCTs) comparing different bone grafts in lumbar arthrodesis were eligible for inclusion. A network meta-analysis was performed for endpoints including fusion rate and incidence of adverse events.
Results
Twenty-seven RCTs involving 2488 patients and 13 available interventions were included. rhBMP-2 provided the highest fusion rate, being significantly superior to that of ICBG (OR = 0.21, p < 0.001), autograft local bone (ALB) (OR = 0.18, p = 0.022), rhBMP-7 (OR = 0.15, p < 0.001), allograft (OR = 0.13, p = 0.009), and DBM + ALB (OR = 0.07, p = 0.048). The treatment efficacy of allograft could be significantly enhanced by bone marrow concentrate (BMC) supplying (OR = 0.16, p = 0.010). ICBG ranks second on the frequency of complications, which is significantly higher than that of allograft (OR = 0.14, p = 0.041) and ALB (OR = 0.14, p = 0.030). All of the other comparisons showed similar efficacy and safety profiles between groups.
Conclusion
Ranking spectrums of the efficacy and safety for various bone grafts were provided graphically. Though rhBMP-2 was of the highest success rate, the application should be taken with proper caution because of the widely proposed life-threatening adverse events. ALB, ALB plus synthetic ceramic materials and allograft mixed with BMC were also proved to be potentially effective alternative graft to ICBG.
Graphic abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lumbar spinal fusion is a widespread technique for the surgical management of degenerative lumbar pathology, which might be indicated where conservative care fails to adequately control the symptoms [1, 2]. A major clinical challenge in the procedures of fusion surgery has centered around the issue of pseudarthrosis. In general, solid bony fusion depends on multiple factors: (1) patients’ age; (2) smoking status; (3) patients’ metabolism status; (4) quality of graft-bed preparation; (5) a stable and loaded construct; (6) comorbidities (e.g., osteoporosis); (7) number of fused levels; and (8) bone grafts selected [3, 4]. Among these, there is no doubt that the selection of grafts is a key determinant for the success rate of spinal fusion.
Autologous iliac crest bone graft (ICBG) can be filled into the posterolateral gutters and intervertebral to promote fusion in lumbar fusion, which was considered as the “gold standard” as it contains three inherent properties: osteoconductive, osteoinductive, and osteogenetic [5, 6]. However, the procedure of ICBG harvesting is inevitably associated with multiple donor-site-related complications including persistent iliac pain, iliac fractures, vascular and nerve injuries, hematomas and deep infections [7, 8]. In addition, the amount of available ICBG is limited, especially in multi-segment fusion, revision surgery, and patients with osteoporosis [9]. For the numerous disadvantages of ICBG, a variety of alternative bone substitutes, such as recombinant human bone morphogenetic proteins (rhBMP-2 and rhBMP-7), hydroxyapatite (HA), β-tricalcium phosphate (β-TCP), demineralized bone matrix (DBM), autograft local bone (ALB), bone marrow aspirate (BMA), silicate calcium phosphate (Si-CaP), platelet-rich plasma (PRP), and allograft, have been researched and applied separately or with various combinations to promote the process of lumbar fusion. The ideal bone substitutes should possess osteoconductive and osteoinductive properties and, when possible, osteogenetic cells to achieve a comparable fusion rate to ICBG. The grafts primarily developed to provide a conductive scaffold are ceramic products, such as HA, TCP, and Si-CaP, and DBM, while rhBMP and DBM are products equipped with osteoinductive character to facilitate osteogenesis. Other biological agents including PRP and BMA are rich in platelets (and their growth factors) and mesenchymal stem cells (MSCs) that could enhance the osteogenic potential of the scaffold materials.
In current, most of the RCTs comparing efficacy and safety of different bone substitutes are based on relatively small sample size, lacking data comparing multiple grafts to each other [3, 4, 9,10,11]. Previous head-to-head meta-analyses also could not rank these bone substitutes because some of them had not been compared one by one [12,13,14,15,16]. Therefore, this network meta-analysis (NMA) was carried out with the purpose of comparing the effectiveness and safety of all available bone grafts for the management of lumbar degenerative disease with lumbar spinal fusion and to provide a ranking spectrum of the grafts.
Methods
This review was conducted according to the guidelines outlined in Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) statement (See “Appendix 1”) [17]. A prospective protocol was created in advance and uploaded to the PROSPERO online platform.
Data sources and search strategy
Two independent researchers systematically retrieved the platforms of PubMed, EMBASE, and CENTRAL from the inception dates to Jun. 2019, using keywords including: “lumbar degenerative disease,” “lumbar spine,” “spinal fusion,” “bone graft,” “bone substitutes,” etc.
Eligibility criteria and study selection
The inclusion criteria were as following: (1) patients diagnosed with a lumbar degenerative disease undergone spinal fusion with bone graft materials; (2) definitive outcomes were reported in studies, such as fusion rate and the number of adverse events; (3) head-to-head RCT study; (4) the judgment of fusion was contingent on computed tomography (CT) or X-ray plain results. Exclusion criteria: (1) studies with single-arm design; (2) pathology other than degenerative diseases, such as infectious or inflammatory diseases, spinal tumors, and trauma; (3) studies with less than 10 subjects in any treatment arm.
There were two steps in study selection process: screening the titles and abstracts, and reviewing the full texts. Throughout the screening process, the two independent authors strictly followed the inclusion and exclusion criteria. Finally, references cited in eligible studies that were considered to be potentially relevant were also retrieved and assessed in full. In case of a disagreement between the two authors, a third investigator resolved the disagreement through discussion.
Data extraction
Two authors independently extracted the following information from each included studies: (1) Study characters: lead author, publication year, study design, the country of lead author, study period, and follow-up; (2) Patients information: number of involved subjects, number of patients dropped, percentage of male patients, and age at operation; (3) operation information (Intervention and Comparison): the types and dosages of bone grafts, and surgical methods; (4) Outcome information: success rate of fusion (based on plain/extension -flexion radiographs or thin-layer CT scan) and frequency of adverse events at final follow-up. The differences between the two authors were resolved by a third author after discussed.
Risk of bias assessment
The risk of bias was assessed using the Cochrane Collaboration’s risk of bias tool [18]. Each study was assessed on seven items: (1) random sequence generation; (2) allocation concealment; (3) performance bias; (4) detection bias; (5) incomplete outcome data; (6) reporting bias; (7) other bias. Each parameter is judged as low risk of bias, high risk of bias or unclear.
Data synthesis and statistical analysis
The primary and second outcomes analyzed were the fusion rate and the number of each specific treatment-related adverse events. We recorded all adverse events that were occurring during the course of treatment without distinguishing between their specific classifications. We used odds ratio (OR) and 95% credibility interval (95% CrI) as summary statistics to quantify the effect of treatment. A classic half-integer continuity correction was used so that studies with no events would still be included for analyses [19].
To illustrate which interventions were directly compared in the primary RCTs, we generated network plots using “network” suite of commands for Stata version 14.0 (StataCorp LLC, College Station, Texas, USA). R 3.5.3 software (R Core Team, Vienna, Austria) was used to invoke the program of WinBUGS 1.4.3 software (MRC Biostatistics Unit, Cambridge, UK) for Bayesian NMA. A random-effect model was used to compare treatments using Markov chain Monte Carlo (MCMC) methods with Gibbs sampling from 40,000 iterations obtained after a 10,000 burn-in phase. Following the processes of NMA, interventions were ranked according to their estimated effect sizes to display which treatment ranked highest, second highest, and so on, using the surface under the cumulative ranking curves (SUCRA) [20]. Statistical significance was defined as a two-sided P value of less than 0.05.
Standard pairwise meta-analysis was also performed for all direct head-to-head comparisons, using random-effect model for considering the anticipated variety in study populations. Both of the pooled effect estimates in NMA and pairwise meta-analysis were presented as the estimated summary effects (OR) combining with the 95% CrI as well as the 95% prediction intervals (95%PrI). Inconsistency is estimated as the difference between direct and indirect comparisons for each closed loop, with the method of node-splitting analysis (p < 0.05 indicated significant inconsistency).
Novel presentational approach (i.e., summary forest plot matrix) was used to display the results, including the forest plots and estimated effects both for NMA and pairwise meta-analysis, SUCRA value for each intervention, and the between-study heterogeneity, as described by Tan et al. [21]. Comparison-adjusted funnel plot was used to identify possible small-sample effect for each network using Stata software [22]. Subgroup NMA were performed for the subgroups of posterolateral lumbar fusion (PLF) and lumbar interbody fusion (LIF) on the success rate and incidence of adverse events, to assess the stability of NMA results.
Results
Study inclusion and baseline characteristics
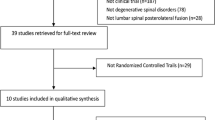
Figure 1 shows the flowchart illustrating the process of study retrieval and selection. Databases searching initially identified a total of 5185 records, and another two records were manually searched for potential eligibility. Following exclusion of the duplicates, 3604 titles/abstracts were left for screening. Finally, 47 full-text articles were assessed for final eligibility, and 27 RCTs [3, 4, 9, 23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46] were included for qualitative and quantitative syntheses
Table 1 shows a summary of the trials included in this NMA. These studies included 2488 patients with an overall female percentage of 58.2% (range 36.8–72.5%). The mean follow-up period was 19.8 ± 8.5 months with an overall dropout rate of 10.5%. Several fusion techniques were performed, including PLF in 18 studies [4, 23,24,25,26,27,28,29,30,31,32,33,34,35,36,37, 39, 40], posterior LIF (PLIF) procedures in four studies [38, 43, 44, 46], anterior LIF (ALIF) in one study [45], transforaminal LIF (TILF) in three studies [9, 41, 42], and extremely lateral LIF (XLIF) in one study [3].
Summary of the risk of bias and the risk of bias graph is presented in Fig. 2. The blinding of participants and personnel was presented to be with high risk of bias in most of the studies, while the other items were all shown to be with low or unclear risk of bias predominately.
NMA for spinal fusion rate and all recorded complications
Figure 3 displays the network plot illustrating interventions directly compared in the primary RCTs. In total, 13 individual or combined intervention regimens, including ICBG (n = 962), rhBMP-2 (n = 746), rhBMP-7 (n = 329), Si-CaP (n = 92), PRP + ICBG (n = 20), HA + BMA + ALB (n = 20), HA + ALB (n = 25), DBM + ALB (n = 28), allograft (n = 102), ALB + β-TCP + HA (n = 10), ALB (n = 82), allograft + BMC (bone marrow concentrate) (n = 40), and ALB + β-TCP (n = 32), were available for analyses
Network plots illustrating interventions directly compared in the total network meta-analysis (a), and subgroup analyses of posterior lumbar fusion (b) and lumbar interbody fusion (c). Each node represents a type of bone graft, while each line represents a direct comparison between two grafts. The nodes and lines are weighted by the numbers of related patients and trials
The results of NMA for success rate of fusion are available in the summary forest plot matrix in Fig. 4. A ranking spectrum was provided in the diagonal line depicting the efficacy order of the intervention regimens. In general, rhBMP-2 provided the highest fusion rate, which was significantly superior to that of ICBG (OR = 0.21, 95% CrI 0.11–0.36, p < 0.001), ALB (OR = 0.18, 95% CrI 0.04–0.78, p = 0.022), rhBMP-7 (OR = 0.15, 95% CrI 0.06–0.38, p < 0.001), allograft (OR = 0.13, 95% CrI 0.03–0.60, p = 0.009), and DBM + ALB (OR = 0.07, 95% CrI 0.00–0.98, p = 0.048). The treatment efficacy of allograft could be significantly enhanced by BMC supplying (OR = 0.16, 95% CrI 0.04–0.64, p = 0.010). No significant difference was demonstrated for any other comparison according to the NMA results. The DBM + ALB was associated with the least success rate of fusion. The summary forest plot matrix for NMA of the recorded complications is available in Fig. 5. Among the available interventions, the DBM + ALB is associated with the highest incidence of complications, while the β-TCP + ALB is of the most favorable safety. ICBG ranks second in the frequency of complications, which is significantly higher than that of allograft (OR = 0.14, 95% CrI 0.02–0.92, p = 0.041) and ALB (OR = 0.14, 95% CrI 0.02–0.83, p = 0.030). All of the other comparisons were shown to be similar between groups.
Summary forest plot matrix for NMA of fusion rate. The matrix consisted of the forest plots (below the diagonal) as well as the estimated effect sizes (above the diagonal) for pairwise meta-analyses and NMA, the SUCRA curves (along the diagonal ordering by SUCRA values), and the between-study variance (τ2). NMA network meta-analysis, CrI credible interval, PI prediction interval
Summary forest plot matrix for NMA of complications. The matrix consisted of the forest plots (below the diagonal) as well as the estimated effect sizes (above the diagonal) for pairwise meta-analyses and NMA, the SUCRA curves (along the diagonal ordering by SUCRA values), and the between-study variance (τ2). NMA network meta-analysis, CrI credible interval, PI prediction interval
The cluster ranking plot is shown in Fig. 6, in which the bone grafts are divided into four groups using the median SUCRA values of the two networks. In general, the allograft + BMA, ALB + β-TCP + HA, HA + BMA + ALB, and β-TCP + ALB were demonstrated to provide both increased fusion rate and decreased frequency of complications. In contrast, though rhBMP-2, Si-CaP and ICBG could provide favorable fusion rate, they were also associated with increased risk of complications, especially for ICBG. For grafts including ALB, allograft, and rhBMP-7, they provided below-median treatment efficacy, but increased safety. DBM + ALB, HA + ALB, and PRP + ICBG were divided into the most unfavorable group of grafts, which were associated with both below-median efficacy and safety.
Cluster ranking plot which divided the bone grafts into four groups (colored as red, blue, green and purple) using the median SUCRA values of network meta-analyses for efficacy and safety. Values close to 100% indicate increased spinal fusion rate or increased incidence of adverse events. rhBMP recombinant human bone morphogenetic protein, BMA bone marrow aspirate, ALB autograft local bone, β-TCP β-tricalcium phosphate, HA hydroxyapatite, Si-CaP silicate calcium phosphate, ICBG autologous iliac crest bone graft, PRP platelet-rich plasma, DBM demineralized bone matrix, BMC bone marrow concentrate
Subgroup analyses
Supplementary Figures S1–4 show the forest plot matrices for the efficacy and safety of the available bone grafts based on the subgroups of PLF and LIF, and the corresponding ranking spectrums are available in Supplementary Table S1. RhBMP-2 was shown to be the most effective bone graft in both subgroups, providing significantly superior fusion rate than ICBG (OR = 0.24, 95% CrI 0.13–0.44, p < 0.001) and rhBMP-7 (OR = 0.17, 95% CrI 0.07–0.45, p < 0.001) in PLF subgroup (Supplementary Figure S1), and ICBG (OR = 0.06, 95% CrI 0.00–0.61, p = 0.017) in LIF subgroup (Supplementary Figure S2), respectively. None of the other head-to-head comparisons showed significant difference on the fusion rate. Similar incidence of complications was presented among the available grafts in the subgroups of PLF and LIF (Supplementary Figure S3–4). When compared with the total NMA, the subgroups provided similar ranking of the available bone grafts, indicating that no obvious unstability of the NMA results exists.
Inconsistency assumption and small-sample effect test
The results of inconsistency test are provided in Supplementary Figure S5. Only a single closed triangle loop (ICBG–allograft–ALB) was available in the integrated networks for spondylodesis efficacy and safety (Fig. 3a). No significant inconsistency was found between the direct and indirect comparisons in the closed loops, according to results of the node-split analysis (p > 0.05).
Comparison-adjusted funnel plot is presented in Supplementary Figure S6a–f, giving no obvious asymmetry, but some small-sample trials in each network located in the bottom of the funnels. Thus, no obviously detected publication bias exists, but irreducible small-sample effect may lead to the risk of bias.
Discussion
The main finding of our study was that rhBMP-2, allograft + BMA, ALB + β-TCP + HA, Si-CaP, β-TCP + ALB, and HA + BMA + ALB were associated with a tendency of increased success rate of lumbar fusion than that of ICBG, but of these Si-CaP and rhBMP-2 were found to lead to above-median incidence of complications.
To achieve solid spinal fusion in the situation of lumbar degenerative diseases, many alternative biological and synthetic bone substitutes have been identified or currently under development [47]. The optimal alternative to the ICBG, nevertheless, remains elusive till now. As a low molecular weight glycoprotein which belongs to the transforming growth factor-β superfamily, rhBMP-2 possesses strong osteoinductive property and has been widely accepted as the most effective osteobiologic agent to induce arthrodesis since the introduction in spinal fusion [48, 49]. There are several pieces of high-level evidence from meta-analyses that have compared the efficacy of rhBMP-2 and ICBG, which consistently reported superior spinal fusion rate for the rhBMP-2-treated group [12, 13, 50]. In the meta-analysis of individual participant data performed by Simmonds et al. [12], RCTs of rhBMP-2 versus ICBG in spinal fusion surgery for degenerative disk disease and related conditions were included for analysis, and a 12% higher radiographic fusion rate was provided with rhBMP-2 than with ICBG. Chen et al. [50] conducted a meta-analysis basing on 10 high-quality RCTs to compare the efficacy of rhBMP-2 and ICBG for lumbar fusion, showing significantly decreased risk of fusion failure at all time intervals (6, 12, and 24 months) for rhBMP-2 group than ICBG group. Similar result was demonstrated in our study, which showed that rhBMP-2 is the most effective bone graft substitute among all available grafts, providing significantly increased fusion rate than ICBG, ALB, rhBMP-7, allograft, and DBM + ALB.
Despite these encouraging results following the application of rhBMP-2, the utilization of rhBMP-2 in lumbar spondylodesis is still an off-label procedure which has not been approved by the Food and Drug Administration of USA [51]. Recent articles have presented several adverse events associated with rhBMP-2 application, including heterotopic bone growth, increased risk of malignancy, bony resorption or osteolysis, retrograde ejaculation (RE), radiculitis, and direct neural toxicity [50, 52,53,54,55]. Fu et al. [14] reported a significantly increased overall cancer risk at 24 months following treating with rhBMP-2. Poorman1 et al. [56] also reported increased odds of developing radiculitis or neurological complications attributed to BMP use, when compared with non-BMP group. Even so, the small number of adverse events has limited the power to detect the difference between groups, precluding definite conclusions. In our results, rhBMP-2 is associated with an above-median but lower-than-ICBG incidence of overall adverse events. Mostly, the adverse events associate with ICBG may be caused by graft harvesting, which should be less life-threatening than the former mention adverse events caused by rhBMP-2 application. Thus, to weight the benefit and damage that rhBMP-2 may bring to patients is quite essential, and application procedure should be taken with proper caution to ensure the graft to be contained within the cage or area where bone should grow.
Apart from the rhBMP-2, another molecule belonging to BMP family, which is called rhBMP-7 or osteogenic protein-1 (OP-1), has been shown to be able to initiate the cascade of bone formation in a variety of clinical situations including lumbar spondylodesis [57]. Up to now, the effectiveness and safety of rhBMP-7 relative to ICBG remain controversial [13, 15, 26]. The current NMA showed that OP-1 was associated with nonsignificantly inferior efficacy than ICBG and located on the median level of safety among all available graft materials that was nonsignificantly superior than ICBG. Similarly, Ye et al. [15] also found that there was no significant difference between the rhBMP-7 and ICBG groups, but rhBMP-7 appeared to yield a lower fusion rate in the instrumented PLF subgroup. Additionally, though rhBMP-7 group recorded lower rate of adverse events, no significant difference was found between the two groups. Thus, the current review does not recommend the rhBMP-7 as an effective alternative to ICBG due to no additional benefit would be produced, while it tended to yield a decreased fusion rate.
The ALB is often used as an alternative graft to ICBG, which provides almost same characteristics as bone graft from ICBG, including three-dimensional osteoconductive scaffold, osteoinductive potential provided by inherent BMPs, and osteogenetic activity derived from the osteoblasts [58]. The bone chips obtained during laminectomy are of predominantly cortical composition, with only a small percentage of trabecular or unmineralized bone which consists of the main components of marrow cavity. The ICBG is a graft rich in cancellous trabecular, which would be theoretically superior to ALB on the fusion rate due to increased osteoinductive activity. Our NMA found that ALB provided lower fusion rate than ICBG, but the subtle difference did not reach a statistical significance basing on the available patient samples. Concerning the safety of the grafts, ALB was identified to be with the least incidence of complications, which was found to be significantly less frequent than that of ICBG. Thus, ALB still could be used as an alternative graft to ICBG in lumbar arthrodesis, to provide nonsignificantly inferior fusion rate but obviously decreased risk of postoperative complications.
Calcium phosphate (CaP) ceramics, such as HA, β-TCP, and Si-CaP, are another set of bone graft substitutes which mainly exhibits osteoconductivity through their intrinsic three-dimensional scaffold [51, 59]. In general, these ceramic-based grafts are biocompatible with an appropriate safety profile and are able to mimic physiological bone [60, 61]. When augmented with osteoinductive growth factors or autologous mesenchymal stem cells or local bone, the ceramics could be equipped with ability to induce bone regeneration and osteogenic ability. What is more, ceramic materials application could also prevent the complications associated with autograft harvesting, and large-scale production is allowed. This study analyzed a total of five CaP ceramics-based intervention regimens, including TCP + HA + ALB, Si-CaP, TCP + ALB, HA + ALB + BMA, and HA + ALB, in which augmenting with osteoinductive materials were provided to enhance the osteogenesis process. Apart from the HA + ALB, a tendency of increased fusion rate than that of ICBG was provided by the other four grafts combinations. Thus, CaP ceramics are recommended to be used in combination with autogeneous bone as alternatives to ICBG to obtain solid fusion. We failed to assess the effectiveness of purely osteoconductive scaffolds in spinal fusion, but unsatisfactory results of stand-alone CaP bone graft substitutes have been previously reported [62,63,64].
Allogenic bone graft is another conventional alternative to ICBG used for spondylodesis, which biologically appears to be inferior due to the lack of osteoinductivity and osteogenic potential [65]. Nevertheless, in the current study, we found that when mixed with BMA, the fusion rate of bone allograft was significantly elevated to be ranked only second to rhBMP-2. DBM is a class of commercially available grafts derived from allograft, which theoretically has all types of BMPs involved in osteoinduction, albeit with lower concentrations. This may apply another potential alternative to ICBG for spine fusion. However, few data about DBM application were available for analysis.
Limitations
There were some limitations that should be noted. First, the small samples enrolled in primary trials might not provide sufficient power to detect small differences between groups (type II error). Therefore, some larger controlled trials of higher quality should be conducted to draw more definite conclusions. Second, the assessment of solid spinal fusion mainly depended on radiological evaluations. It must be taken into consideration that a predictive value of no more than 70% has been reported for the evaluation procedures with radiological methods [66, 67]. Some novel assessment methods, therefore, are required to provide more precise assessment on fusion rate. Finally, some potential clinical heterogeneity, such as the different fusion techniques selected, numbers of segments fused, the utilization of internal fixation instrumentation, and the amounts of the grafts provided, may confused the reliability of results. Hence, subgroup analyses were carried out for some of these confounding factors to decrease potential heterogeneity, giving stable ranking orders similar to the total NMA.
Conclusions
In summary, ranking spectrums of the efficacy and safety for various bone grafts were graphically provided, to guide the selection of potential alternatives to ICBG in spondylodesis. RhBMP-2 was of the highest success rate, which obtained statistical significance when compared to ICBG, ALB, allograft, and DBM + ALB. However, the application of rhBMP-2 should be taken with proper caution concerning the widely proposed life-threatening adverse events though with low incidence. ALB alone, ALB plus synthetic ceramic materials and allograft mixed with BMC were also proved to be potentially effective alternative graft to ICBG.
References
Fritzell P, Hägg O, Jonsson D et al (2004) Cost-effectiveness of lumbar fusion and nonsurgical treatment for chronic low back pain in the Swedish Lumbar Spine Study: a multicenter, randomized, controlled trial from the Swedish Lumbar Spine Study Group. Spine (Phila Pa 1976) 29:421–434.
Liu X, Wang Y, Qiu G et al (2014) A systematic review with meta-analysis of posterior interbody fusion versus posterolateral fusion in lumbar spondylolisthesis. Eur Spine J 23:43–56
Pimenta L, Marchi L, Oliveira L et al (2013) A prospective, randomized, controlled trial comparing radiographic and clinical outcomes between stand-alone lateral interbody lumbar fusion with either silicate calcium phosphate or rh-BMP2. J Neurol Surg A Cent Eur Neurosurg 74:343–350
Hurlbert RJ, Alexander D, Bailey S et al (2013) rhBMP-2 for posterolateral instrumented lumbar fusion: a multicenter prospective randomized controlled trial. Spine (Phila Pa 1976) 38:2139–2148.
Shen F, Samartzis D, An H (2005) Cell technologies for spinal fusion. Spine 5:231S–S239
Marchesi DG (2000) Spinal fusions: bone and bone substitutes. Eur Spine J 9:372–378
Myeroff C, Archdeacon M (2011) Autogenous bone graft: donor sites and techniques. J Bone Joint Surg Am 93:2227–2236
Banwart JC, Asher MA, Hassanein RS (1995) Iliac crest bone graft harvest donor site morbidity a statistical evaluation. Spine (Phila Pa 1976) 20:1055–1060.
VonderHoeh NH, Voelker A, Heyde CE (2017) Results of lumbar spondylodeses using different bone grafting materials after transforaminal lumbar interbody fusion (TLIF). Eur Spine J 26:2835–2842
Boden SD, Zdeblick TA, Sandhu HS et al (2000) The use of rhBMP-2 in interbody fusion cages. Definitive evidence of osteoinduction in humans: a preliminary report. Spine (Phila Pa 1976) 25:376–381.
Johnson RG (2014) Bone marrow concentrate with allograft equivalent to autograft in lumbar fusions. Spine (Phila Pa 1976) 39:695–700.
Simmonds MC, Brown JV, Heirs MK et al (2013) Safety and effectiveness of recombinant human bone morphogenetic protein-2 for spinal fusion: a meta-analysis of individual-participant data. Ann Intern Med 158:877–889
Papakostidis C, Kontakis G, Bhandari M et al (2008) Efficacy of autologous iliac crest bone graft and bone morphogenetic proteins for posterolateral fusion of lumbar spine: a meta-analysis of the results. Spine (Phila Pa 1976) 33:E680–692.
Fu R, Selph S, McDonagh M et al (2013) Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med 158:890–902
Ye F, Zeng Z, Wang J et al (2018) Comparison of the use of rhBMP-7 versus iliac crest autograft in single-level lumbar fusion: a meta-analysis of randomized controlled trials. J Bone Miner Metab 36:119–127
Parajón A, Alimi M, Navarro-Ramirez R et al (2017) Minimally Invasive Transforaminal Lumbar Interbody Fusion: Meta-analysis of the Fusion Rates. What is the Optimal Graft Material? Neurosurgery 81:958–971
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Higgins JPT, Altman DG, Gøtzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Higgins JPT, Green S (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0. The Cochrane Collaboration, 2011. Available from www. cochrane-handbook.org.
Salanti G, Ades AE, Ioannidis JP (2011) Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64:163–171
Tan SH, Cooper NJ, Bujkiewicz S et al (2014) Novel presentational approaches were developed for reporting network meta-analysis. J Clin Epidemiol 67:672–680
Egger M, Davey Smith G, Schneider M et al (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Cho JH, Lee JH, Yeom JS et al (2017) Efficacy of Escherichia coli-derived recombinant human bone morphogenetic protein-2 in posterolateral lumbar fusion: an open, active-controlled, randomized, multicenter trial. Spine J 17:1866–1874
Delawi D, Jacobs W, van Susante JL et al (2016) OP-1 Compared with Iliac Crest Autograft in Instrumented Posterolateral Fusion: A Randomized, Multicenter Non-Inferiority Trial. J Bone Joint Surg Am 98:441–448
Kang J, An H, Hilibrand et al (2012) A Grafton and local bone have comparable outcomes to iliac crest bone in instrumented single-level lumbar fusions. Spine (Phila Pa 1976) 37:1083–1091.
Delawi D, Dhert WJ, Rillardon L et al (2010) A prospective, randomized, controlled, multicenter study of osteogenic protein-1 in instrumented posterolateral fusions: report on safety and feasibility. Spine (Phila Pa 1976) 35:1185–91.
Dawson E, Bae HW, Burkus JK et al (2009) Recombinant human bone morphogenetic protein-2 on an absorbable collagen sponge with an osteoconductive bulking agent in posterolateral arthrodesis with instrumentation. A prospective randomized trial. J Bone Joint Surg Am 91:1604–1613
Dimar JR 2nd, Glassman SD, Burkus JK et al (2009) Clinical and radiographic analysis of an optimized rhBMP-2 formulation as an autograft replacement in posterolateral lumbar spine arthrodesis. J Bone Joint Surg Am 91:1377–1386
Dai LY, Jiang LS (2008) Single-level instrumented posterolateral fusion of lumbar spine with beta-tricalcium phosphate versus autograft: a prospective, randomized study with 3-year follow-up. Spine (Phila Pa 1976) 33:1299–1304.
Glassman SD, Carreon LY, Djurasovic M et al (2008) RhBMP-2 versus iliac crest bone graft for lumbar spine fusion: a randomized, controlled trial in patients over sixty years of age. Spine (Phila Pa 1976) 33:2843–2849.
Dimar JR, Glassman SD, Burkus KJ et al (2006) Clinical outcomes and fusion success at 2 years of single-level instrumented posterolateral fusions with recombinant human bone morphogenetic protein-2/compression resistant matrix versus iliac crest bone graft. Spine (Phila Pa 1976) 31:2534–2539.
Kanayama M, Hashimoto T, Shigenobu K et al (2006) A prospective randomized study of posterolateral lumbar fusion using osteogenic protein-1 (OP-1) versus local autograft with ceramic bone substitute: emphasis of surgical exploration and histologic assessment. Spine (Phila Pa 1976) 31:1067–1074.
Korovessis P, Koureas G, Zacharatos S et al (2005) Correlative radiological, self-assessment and clinical analysis of evolution in instrumented dorsal and lateral fusion for degenerative lumbar spine disease. Autograft versus coralline hydroxyapatite. Eur Spine J 14:630–638
Glassman SD, Dimar JR, Carreon LY et al (2005) Initial fusion rates with recombinant human bone morphogenetic protein-2/compression resistant matrix and a hydroxyapatite and tricalcium phosphate/collagen carrier in posterolateral spinal fusion. Spine (Phila Pa 1976) 30:1694–1698.
Johnsson R, Strömqvist B, Aspenberg P (2002) Randomized radiostereometric study comparing osteogenic protein-1 (BMP-7) and autograft bone in human noninstrumented posterolateral lumbar fusion: 2002 Volvo Award in clinical studies. Spine (Phila Pa 1976) 27:2654–2661.
Vaccaro AR, Patel T, Fischgrund J et al (2004) A pilot study evaluating the safety and efficacy of OP-1 Putty (rhBMP-7) as a replacement for iliac crest autograft in posterolateral lumbar arthrodesis for degenerative spondylolisthesis. Spine (Phila Pa 1976) 29:1885–1892.
Vaccaro AR, Whang PG, Patel T et al (2008) The safety and efficacy of OP-1 (rhBMP-7) as a replacement for iliac crest autograft for posterolateral lumbar arthrodesis: minimum 4-year follow-up of a pilot study. Spine J 8:457–465
Coughlan M, Davies M, Mostert AK et al (2018) A Prospective, Randomized, Multicenter Study Comparing Silicated Calcium Phosphate versus BMP-2 Synthetic Bone Graft in Posterolateral Instrumented Lumbar Fusion for Degenerative Spinal Disorders. Spine (Phila Pa 1976) 43:E860-E868.
Hart R, Komzák M, Okál F et al (2014) Allograft alone versus allograft with bone marrow concentrate for the healing of the instrumented posterolateral lumbar fusion. Spine J 14:1318–1324
Ohtori S, Suzuki M, Koshi T et al (2011) Single-level instrumented posterolateral fusion of the lumbar spine with a local bone graft versus an iliac crest bone graft: a prospective, randomized study with a 2-year follow-up. Eur Spine J 20:635–639
Nandyala SV, Marquez-Lara A, Fineberg SJ et al (2014) Prospective, randomized, controlled trial of silicate-substituted calcium phosphate versus rhBMP-2 in a minimally invasive transforaminal lumbar interbody fusion. Spine (Phila Pa 1976) 39:185–191.
Huang H, Jiang C, Feng Z et al (2014) Comparing the process of creeping substitution between allograft bone and local bone grafting in lumbar interbody fusion. Eur Spine J 23:2068–2074
Sys J, Weyler J, Van Der Zijden T et al (2011) Platelet-rich plasma in mono-segmental posterior lumbar interbody fusion. Eur Spine J 20:1650–1657
Putzier M, Strube P, Funk JF et al (2009) Allogenic versus autologous cancellous bone in lumbar segmental spondylodesis: a randomized prospective study. Eur Spine J 18:687–695
Burkus JK, Sandhu HS, Gornet MF (2006) Influence of rhBMP-2 on the healing patterns associated with allograft interbody constructs in comparison with autograft. Spine (Phila Pa 1976) 31:775–781.
Haid RW Jr, Branch CL Jr, Alexander JT et al (2004) Posterior lumbar interbody fusion using recombinant human bone morphogenetic protein type 2 with cylindrical interbody cages. Spine J 4:527–538
Cha CW, Boden SD (2003) Gene therapy applications for spine fusion. Spine 28:S74–84
Agarwal R, Williams K, Umscheid CA et al (2009) Osteoinductive bone graft substitutes for lumbar fusion: a systematic review. J Neurosurg Spine 11:729–740
Carlisle E, Fischgrund JS (2005) Bone morphogenetic proteins for spinal fusion. Spine J 5:240S–249S
Chen Z, Ba G, Shen T et al (2012) Recombinant human bone morphogenetic protein-2 versus autogenous iliac crest bone graft for lumbar fusion: a meta-analysis of ten randomized controlled trials. Arch Orthop Trauma Surg 132:1725–1740
Cahill KS, Chi JH, Day A et al (2009) Prevalence, complications, and hospital charges associated with use of bone-morphogenetic proteins spinal fusion procedures. JAMA 302:58–66
Benglis D, Wang MY, Levi AD (2008) A comprehensive review of the safety profile of bone morphogenetic protein in spine surgery. Neurosurgery 62:ONS423–431.
Kanatani M, Sugimoto T, Kaji H et al (1995) Stimulatory effect of bone morphogenetic protein-2 on osteoclast-like cell formation and bone-resorbing activity. J Bone Miner Res 10:1681–1690
Carragee EJ, Hurwitz EL, Weiner BK (2011) A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: emerging safety concerns and lessons learned. Spine J 11:471–491
Rowan FE, O’Malley N, Poynton A (2012) RhBMP-2 use in lumbar fusion surgery is associated with transient immediate post-operative leg pain. Eur Spine J 21:1331–1337
Poorman GW, Jalai CM, Boniello A (2017) Bone morphogenetic protein in adult spinal deformity surgery: a meta-analysis. Eur Spine J 26:2094–2102
Wozney JM (2002) Overview of bone morphogenetic proteins. Spine (Phila Pa 1976) 27:S2–8
Eder C, Chavanne A, Meissner J et al (2011) Autografts for spinal fusion: osteogenic potential of laminectomy bone chips and bone shavings collected via high speed drill. Eur Spine J 20:1791–1795
Lee JH, Lee DH, Ryu HS et al (2003) Porous beta-calcium pyrophosphate as a bone graft substitute in a canine bone defect model. Key Eng Mater 240–2:399–402
Zimmermann G, Moghaddam A (2011) Allograft bone matrix versus sythetic bone graft substitutes. Injury 42:S16–21
Miyazaki M, Tsumura H, Wang JC et al (2009) An update on bone substitutes for spinal fusion. Eur Spine J 18:783–799
Kraiwattanapong C, Boden SD, Louis-Ugbo J et al (2005) Comparison of Healos/bone marrow to INFUSE(rhBMP-2/ACS) with a collagen-ceramic sponge bulking agent as graft substitutes for lumbar spine fusion. Spine 30:1001–1007
Tortolani PJ, Park AE, Louis-Ugbo J et al (2004) The effects of doxorubicin (adriamycin) on spinal fusion: An experimental model of posterolateral lumbar spinal arthrodesis. Spine J 4:669–674
Goodwin CB, Brighton CT, Guyer RD et al (1999) A double-blind study of capacitively coupled electrical stimulation as an adjunct to lumbar spinal fusions. Spine 24:1349–1356
Malloy KM, Hilibrand AS (2002) Autograft versus allograft in degenerative cervical disease. Clin Orthop Relat Res 394:27–38
Brodsky AE, Kovalsky ES, Khalil MA et al (1991) Correlation of radiologic assessment of lumbar spine fusions with surgical exploration. Spine 16:S261–S265
Dawson EG, Clader TJ, Bassett LW et al (1985) A comparison of different methods used to diagnose pseudarthrosis following posterior spinal fusion for scoliosis. J Bone Joint Surg Am 67:1153–1159
Acknowledgments
I would like to express special thanks to my partners for the encouragement and support they gave me during my study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
See Table
2.
Rights and permissions
About this article
Cite this article
Feng, Jt., Yang, Xg., Wang, F. et al. Efficacy and safety of bone substitutes in lumbar spinal fusion: a systematic review and network meta-analysis of randomized controlled trials. Eur Spine J 29, 1261–1276 (2020). https://doi.org/10.1007/s00586-019-06257-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-019-06257-x