Abstract
A retrospective study was performed to determine the sensitivities of the pyramidal signs in patients with cervical myelopathy, focusing on those with increased signal intensity (ISI) in T2-weighted magnetic resonance imaging (MRI). The relationship between prevalence of the pyramidal signs and the severity of myelopathy was investigated. We reviewed the records of 275 patients with cervical myelopathy who underwent surgery. Of these, 143 patients were excluded from this study due to comorbidities that might complicate neurological findings. The MR images of the remaining 132 patients were evaluated in a blinded fashion. The neurological findings of 120 patients with ISI (90 men and 30 women; mean age 61 years) were reviewed for hyperreflexia (patellar tendon reflex), ankle clonus, Hoffmann reflex, and Babinski sign. To assess the severity of myelopathy, the motor function scores of the upper and lower extremities for cervical myelopathy set by the Japanese Orthopaedic Association (m-JOA score) were used. The most prevalent signs were hyperreflexia (94%), Hoffmann reflex (81%), Babinski sign (53%), and ankle clonus (35%). Babinski sign (P < 0.001), ankle clonus, and Hoffmann reflex showed significant association with the lower m-JOA score. Conversely, no association was found with the upper m-JOA score. In patients with cervical myelopathy, hyperreflexia showed the highest sensitivity followed by Hoffmann reflex, Babinski sign, and ankle clonus. The prevalence of the pyramidal signs correlated with increasing severity of myelopathy. Considering their low sensitivity in patients with mild disability, the pyramidal signs may have limited utility in early diagnosis of cervical myelopathy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Making the diagnosis of cervical compressive myelopathy can be challenging, especially in the early stages, due to subtle symptoms and insidious onset [3]. The presence of pyramidal signs such as hyperreflexia, Hoffmann reflex, clonus, and Babinski sign, is a hallmark of myelopathy, which is considered a key component in the diagnostic process. However, patients with suspected myelopathy often lack these pathognomonic signs despite apparent symptoms and correlative cord compression on imaging studies. Such “atypical” neurological findings might be attributable to insufficient sensitivity of the pyramidal signs or to comorbidities that might affect neurological findings.
Although these neurological signs have been commonly used in clinical practice, few studies provided information on their prevalence and clinical utility [13, 14]. Moreover, there was significant discordance in reported prevalence of the pyramidal signs, presumably due to heterogeneity of study subjects. In addition, the relationship between prevalence of pyramidal signs and the severity of myelopathy remains to be elucidated.
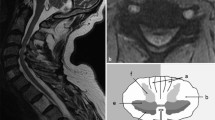
Increased signal intensity (ISI) in T2-weighted magnetic resonance imaging (MRI) is frequently associated with cervical compressive myelopathy. ISI is considered to reflect various intramedullary pathologies such as edema, gliosis, demyelination, and myelomalacia. Several studies have shown that patients with ISI were significantly more likely to show positive pyramidal signs [11, 17]. Although a majority of patients who underwent surgery for cervical myelopathy seem to have ISI, its incidence varied greatly in the literature [10, 11, 16, 17, 20, 24]. This varying incidence of ISI may be one of the factors that cause inconsistency in reported prevalence of the pyramidal signs.
We undertook this retrospective study to determine the sensitivities of the pyramidal signs in patients with cervical compressive myelopathy, focusing on those with ISI in preoperative T2-weighted MRI. We also compared the prevalence of the myelopathic signs between etiologies of cord compression. We further examined whether the presence of the pyramidal signs correlated with the severity of cervical myelopathy.
Materials and methods
A total of 275 patients with cervical compressive myelopathy underwent laminoplasty between July 1998 and April 2007. The patients were admitted to the hospital 3–7 days before surgery for preoperative evaluation including complete neurological examination by a board-certified orthopaedic surgeon and imaging studies. All patients, except those with a pacemaker, routinely had preoperative MRI.
Following approval of an institutional review board, we reviewed the clinical records of the 275 patients. Medical charts and nursing summaries were evaluated for presence of comorbidities. Patients with any of the following comorbidities were excluded from this study: diabetes mellitus (42 patients), previous or concomitant thoracic/lumbar surgery (27), cerebral palsy (24), rheumatoid arthritis (14), upper cervical lesion (11), previous cervical surgery (10), cerebrovascular disease (8), spinal trauma (7), systemic bone disease (5), peripheral neuropathy (4), hemodialysis (3), pacemaker (3), total joint replacement (1) and insufficient medical record (1). Of the 275 patients, 143 were excluded, and in these, there was an overlap in the comorbidities.
The conditions for MRI study during the period from July 1998 to September 2002 were as previously described [19]. The following four types of 1.5-T MRI systems have been used since September 2002: MAGNETOM Avanto (Siemens AG Medical Solutions, Erlangen, Germany), Signa HDx (GE Healthcare, Milwaukee, WI), Signa EXCITE HD (GE Healthcare, Milwaukee, WI) and EXCELART Vantage (Toshiba Medical Systems, Tochigi, Japan). Imaging was performed with fast spin-echo sequences.
The preoperative MR images of the remaining 132 patients were independently reviewed by two board-certified orthopaedic surgeons who were masked to any other information on the patients. There was initially disagreement between the readers in two patients as to presence of ISI in T2-weighted images. The concordance rate was 98.5% and kappa coefficient was 0.90, indicating good interobserver reliability. These two patients were then excluded. Another ten patients were excluded due to absence of ISI. All patients with ISI had substantial cord compression as evidenced by effacement of the subarachnoid space and indentation of the spinal cord.
For the remaining 120 patients, the preoperative neurological findings were reviewed: Hyperreflexia: Patellar tendon reflex (PTR) was rated as exaggerated, normal, or diminished. Exaggerated PTR was considered as positive.
-
Ankle clonus sustained clonus was considered as positive.
-
Hoffmann reflex flexion of the ipsilateral thumb and/or index finger was considered as positive.
-
Babinski sign extension of the great toe was considered as positive. Equivocal or indifferent toe was considered as negative.
The signs were judged as positive when positive sign was present unilaterally or bilaterally. To rate the preoperative motor function, the motor function scores of the upper and lower extremities for cervical myelopathy set by the Japanese Orthopaedic Association (upper and lower m-JOA score) (Table 1) were also reviewed [8].
The overall sensitivity of each pyramidal sign was calculated and then calculated separately according to the etiology of cervical myelopathy. We used the chi-square test for categorical comparisons of the data, and used the Mann–Whitney U test to analyze the relationship between the positive pyramidal signs and upper/lower m-JOA scores. All statistics were calculated using SPSS, version 13.0 (SPSS Inc., Chicago, IL). A P value of less than 0.05 was considered statistically significant.
Results
A total of 120 patients (90 men and 30 women) met the study criteria. The mean age at surgery was 61 years (range 29–85 years). The mean follow-up period was 48 months (median 41 months; range 4–117 months). Of the 120 patients, 116 have been followed up for more than 12 months. There were 81 patients with cervical spondylotic myelopathy (CSM) and 39 patients with ossification of the posterior longitudinal ligament (OPLL). The preoperative severity of the motor dysfunction was rated by the upper/lower m-JOA score (Table 2).
Sensitivities of the pyramidal signs
Hyperreflexia showed the highest sensitivity followed by Hoffmann reflex, Babinski sign, and ankle clonus (Table 3). The incidences of unilateral positive signs were as follows: exaggerated PTR, 8% (10/120 patients); Hoffmann reflex, 20% (24/118); Babinski sign, 12% (14/116); and ankle clonus, 9% (9/104). We found no significant difference in the sensitivities of these pyramidal signs between CSM and OPLL although clonus was more frequently observed in patients with OPLL than with CSM (P = 0.05).
Patients without exaggerated PTR
We found seven men who did not show exaggerated PTR. Their mean age at surgery was 55 years (range 46–66 years). Of these seven, five patients (71%) were also negative for Hoffmann reflex.
Patients with negative Hoffmann reflex
We found 23 patients (21 men and 2 women) who were negative for Hoffmann reflex. Their mean age at surgery was 59 years (range 34–75 years). Of these 23, preoperative MRI revealed the effacement of the subarachnoid space at C6/7 in 13 patients and at C7/T1 in 1 patient.
Relationship between the severity of myelopathy and prevalence of the pyramidal signs
We further analyzed the relationship between prevalence of the pyramidal signs and the severity of myelopathy as measured by the upper/lower m-JOA score using the Mann–Whitney U test (Table 4). None of the pyramidal signs showed correlation with the upper m-JOA score. Statistical difference was found in Babinski sign (P < 0.001), ankle clonus (P = 0.006), and Hoffmann reflex (P = 0.031). Positive pyramidal sign was more frequently found with increasing severity of motor dysfunction in the lower extremities (Fig. 1).
Discussion
Our study had three main findings. First, we determined the sensitivities of the pyramidal signs in patients with cervical compressive myelopathy with ISI in T2-weighted MRI. Hyperreflexia showed the highest sensitivity (94%) followed by Hoffmann reflex (81%), Babinski sign (53%), and ankle clonus (35%). Second, we obtained similar prevalence of pyramidal signs in CSM and OPLL. Of note, patients with OPLL were more likely to have ankle clonus. Third, we found that prevalence of the pyramidal signs closely correlated with increasing severity of myelopathy as measured by the lower m-JOA score. In contrast, no significant correlation was found with the upper m-JOA score.
In previous studies, not much attention has been paid to heterogeneity of study subjects, which could have limited the external validity. In the current study, we focused on patients with ISI and excluded the patients with comorbidities that might complicate neurological findings. The clinical characteristics of subjects in the current study were almost equivalent to those reported in recent surgical series [12, 18, 21, 24].
The prevalence of the pyramidal signs obtained in this study was almost consistent with those reported previously [2, 11, 13, 14, 17]. We found considerable difference in sensitivity among these signs as depicted by Lunsford et al. [14]. In line with most of the preceding studies, we found that sensitivities of exaggerated PTR and Hoffmann reflex were relatively high. Thus, for screening myelopathic patients, PTR and Hoffmann reflex may be useful. In comparison with our results, Rhee et al. [17] reported much lower sensitivity values for exaggerated PTR (33%), Hoffmann reflex (59%), Babinski sign (13%), and ankle clonus (13%). However, in 20 patients who showed cord signal change, the prevalence in their study increased to 45, 80, 15 and 25%, respectively.
All patients who lack exaggerated PTR were male and slightly younger than average. The majority of them (5/7 patients) were also negative for Hoffmann reflex. There was also male predominance in patients lacking Hoffmann reflex (21/23 patients). In line with our findings, Glaser et al. [6] have reported that female patients have higher incidence of positive Hoffmann reflex than males. These findings indicate females may have higher excitability of deep tendon reflex as compared with males. Further study is needed to clarify possible gender differences in prevalence of the pyramidal signs. Interestingly, the lack of Hoffmann reflex was not necessarily associated with the cord compression at C6/7 or C7/T1 where the reflex center is presumed to exist.
Few studies compared the prevalence of pyramidal signs between CSM and OPLL. In the present study, we found similar prevalence of hyperreflexia, Hoffmann reflex, and Babinski sign in CSM and OPLL. Of note, ankle clonus was found more frequently in patients with OPLL than in those with CSM (47 vs. 28%; P = 0.05). Although concomitant thoracic OPLL or ossification of the ligamentum flavum might play a role, higher prevalence of clonus in OPLL patients remains to be elucidated.
We also found that prevalence of the pyramidal signs significantly correlated with the severity of myelopathy, especially with the severity of motor dysfunction in the lower extremities (Fig. 1). This finding underscores the validity of these signs as a clinical indicator of impaired motor tract. In particular, the prevalence of Babinski sign closely correlated with increasing severity of myelopathy as measured by the lower m-JOA score. Although varying prevalence values of Babinski sign (13–54%) [2, 11, 13, 14, 17] have been reported in the literature, this discordance may be attributable to the varying severity of myelopathy. Our findings are not in line with those recently reported by Rhee et al. [17]. They compared the mean preoperative Nurick scores of 31 patients with any myelopathic sign to that of 8 patients without any myelopathic sign. They concluded that there was no correlation between the preoperative Nurick score and the presence of myelopathic signs. A relatively small number of the subjects in their study might have resulted in different conclusion from ours.
Interestingly, prevalence of pyramidal signs did not correlate with the severity of motor dysfunction in the upper extremities. The lack of correlation with the upper m-JOA score can be attributable to the following reasons: first of all, the motor function of the upper extremities is affected not only by the severity of the cord compression but also by the level of the cord compression. Second, the motor function of the upper extremities might be maintained by compensatory mechanisms of the brain even with cord compromise [9, 15]. Probably for these reasons, the degree of cord compromise may not linearly correlate with the upper m-JOA score.
To assess the severity of motor dysfunction, we used the JOA scoring system, which has been validated as an accurate assessment of the severity of myelopathy [23]. Although the JOA score is widely used in the literature, the European Myelopathy Score (EMS), a European modification of the JOA score, has been recently proposed [7]. EMS might provide additional information on the severity of myelopathy by further assessing proprioception and coordination.
The association between prevalence of the pyramidal signs and the severity of myelopathy also indicates limited utility of these signs in the early diagnosis of cervical myelopathy. In the early stages, patients with cervical myelopathy may only have numbness in their hands and subtle gait disturbance [5]. Our data suggest that prevalence of pyramidal signs in such population would be considerably low. It seems reasonable to assume that only a small fraction of such patients would be positive for Babinski sign and ankle clonus. Therefore, additional diagnostic modalities seem to be needed for early detection of cervical compressive myelopathy. In this view, neurophysiologic assessment (e.g., motor and somatosensory evoked potentials) has a potentially important role, particularly in screening for subclinical cord compromise [1, 4].
There are several limitations to this study inherent to its retrospective nature. The first limitation is that the examiners were not masked to other elements of medical history and the results of imaging studies. It is possible that the lack of masking has influenced the neurological assessment, a bias that has been demonstrated previously [22]. Thus, the sensitivity obtained in this study might have been overestimated since the examiners might have had a tendency to judge an equivocal sign as positive. The second limitation is the lack of strict standardization in the execution of neurological examination, which could have influenced the validity of the present study. However, variation between the examiners would have been minimal since we had consistently tried to standardize our performance through an instructive session on neurological examination every 6 months. The third limitation is that only documented comorbidities were evaluated. Further evaluation or imaging studies might have revealed additional coexistent conditions.
Conclusions
In patients with cervical myelopathy, hyperreflexia showed the highest sensitivity followed by Hoffmann reflex, Babinski sign, and ankle clonus. For screening myelopathic patients, PTR and Hoffmann reflex may be useful because of high sensitivity. We found that the prevalence of the pyramidal signs closely correlated with increasing severity of myelopathy. Considering the low prevalence in patients with mild disability, our findings suggest that these signs have limited usefulness in early diagnosis of cervical compressive myelopathy.
References
Bednarik J, Kadanka Z, Vohanka S, Novotny O, Surelova D, Filipovicova D, Prokes B (1998) The value of somatosensory and motor evoked potentials in pre-clinical spondylotic cervical cord compression. Eur Spine J 7:493–500
Chiles BW III, Leonard MA, Choudhri HF, Cooper PR (1999) Cervical spondylotic myelopathy: patterns of neurological deficit and recovery after anterior cervical decompression. Neurosurgery 44:762–769 (discussion 769–770)
Dillin WH, Watkins RG (1992) Clinical syndromes in cervical myelopathy. In: Rothman RH, Simeone FA (eds) The spine, 3rd edn. W.B. Saunders, Philadelphia, pp 560–570
Dvorak J, Sutter M, Herdmann J (2003) Cervical myelopathy: clinical and neurophysiological evaluation. Eur Spine J 12(suppl 2):S181–187
Emery SE (2001) Cervical spondylotic myelopathy: diagnosis and treatment. J Am Acad Orthop Surg 9:376–388
Glaser JA, Cure JK, Bailey KL, Morrow DL (2001) Cervical spinal cord compression and the Hoffmann sign. Iowa Orthop J 21:49–52
Herdmann J, Linzbach M, Kranz M, Dvorak J, Bock WJ (1994) The European Myelopathy Score. In: Bauer BL, Brock M, Klinger M (eds) Advances in neurosurgery. Springer, Berlin, pp 266–268
Hirabayashi K, Miyakawa J, Satomi K, Maruyama T, Wakano K (1981) Operative results and postoperative progression of ossification among patients with ossification of cervical posterior longitudinal ligament. Spine (Phila Pa 1976) 6:354–364
Holly LT, Dong Y, Albistegui-DuBois R, Marehbian J, Dobkin B (2007) Cortical reorganization in patients with cervical spondylotic myelopathy. J Neurosurg Spine 6:544–551
Holly LT, Moftakhar P, Khoo LT, Shamie AN, Wang JC (2008) Surgical outcomes of elderly patients with cervical spondylotic myelopathy. Surg Neurol 69:233–240
Houten JK, Noce LA (2008) Clinical correlations of cervical myelopathy and the Hoffmann sign. J Neurosurg Spine 9:237–242
Iwasaki M, Okuda S, Miyauchi A, Sakaura H, Mukai Y, Yonenobu K, Yoshikawa H (2007) Surgical strategy for cervical myelopathy due to ossification of the posterior longitudinal ligament: part 1: clinical results and limitations of laminoplasty. Spine (Phila Pa 1976) 32:647–653
Kamata M, Satomi K (1997) Classification of cervical myelopathy (in Japanese). MB Orthop 10:1–6
Lunsford LD, Bissonette DJ, Zorub DS (1980) Anterior surgery for cervical disc disease. Part 2: treatment of cervical spondylotic myelopathy in 32 cases. J Neurosurg 53:12–19
Mikulis DJ, Jurkiewicz MT, McIlroy WE, Staines WR, Rickards L, Kalsi-Ryan S, Crawley AP, Fehlings MG, Verrier MC (2002) Adaptation in the motor cortex following cervical spinal cord injury. Neurology 58:794–801
Morio Y, Teshima R, Nagashima H, Nawata K, Yamasaki D, Nanjo Y (2001) Correlation between operative outcomes of cervical compression myelopathy and MRI of the spinal cord. Spine (Phila Pa 1976) 26:1238–1245
Rhee JM, Heflin JA, Hamasaki T, Freedman B (2009) Prevalence of physical signs in cervical myelopathy: a prospective, controlled study. Spine (Phila Pa 1976) 34:890–895
Sakaura H, Hosono N, Mukai Y, Oshima K, Iwasaki M, Yoshikawa H (2008) Preservation of the nuchal ligament plays an important role in preventing unfavorable radiologic changes after laminoplasty. J Spinal Disord Tech 21:338–343
Seichi A, Takeshita K, Kawaguchi H, Nakajima S, Akune T, Nakamura K (2004) Postoperative expansion of intramedullary high-intensity areas on t2-weighted magnetic resonance imaging after cervical laminoplasty. Spine (Phila Pa 1976) 29:1478–1482 (discussion 1482)
Suri A, Chabbra RP, Mehta VS, Gaikwad S, Pandey RM (2003) Effect of intramedullary signal changes on the surgical outcome of patients with cervical spondylotic myelopathy. Spine J 3:33–45
Tsuji T, Asazuma T, Masuoka K, Yasuoka H, Motosuneya T, Sakai T, Nemoto K (2007) Retrospective cohort study between selective and standard c3–7 laminoplasty. Minimum 2-year follow-up study. Eur Spine J 16:2072–2077
Van Gijn J, Bonke B (1977) Interpretation of plantar reflexes: biasing effect of other signs and symptoms. J Neurol Neurosurg Psychiatry 40:787–789
Yonenobu K, Abumi K, Nagata K, Taketomi E, Ueyama K (2001) Interobserver and intraobserver reliability of the Japanese Orthopaedic Association scoring system for evaluation of cervical compression myelopathy. Spine (Phila Pa 1976) 26:1890–1894 (discussion 1895)
Yukawa Y, Kato F, Ito K, Horie Y, Hida T, Machino M, Ito ZY, Matsuyama Y (2008) Postoperative changes in spinal cord signal intensity in patients with cervical compression myelopathy: comparison between preoperative and postoperative magnetic resonance images. J Neurosurg Spine 8:524–528
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chikuda, H., Seichi, A., Takeshita, K. et al. Correlation between pyramidal signs and the severity of cervical myelopathy. Eur Spine J 19, 1684–1689 (2010). https://doi.org/10.1007/s00586-010-1364-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-010-1364-3