Abstract
Background
Taste and smell abnormalities (TSA) commonly occur in cancer and are associated with anorexia, early satiety, malnutrition, weight loss and reduced quality of life. A recent study found a high TSA prevalence in newly diagnosed cancer patients before treatment. This suggests that TSA may originate from the tumour itself. No previous study has examined TSA, both subjectively and objectively, in newly diagnosed, treatment-naïve cancer patients. This study aimed to address this gap.
Methods
This prospective observational study recruited consecutive, newly diagnosed, treatment-naïve patients with solid tumours at Radiation Oncology Out-patients. Self-reported taste and smell changes since becoming ill were evaluated using modified Taste and Smell Survey, and objective taste and smell tests were conducted using ‘Sniffin’ Sticks Olfactory Test® and Burghart Taste Strips®. Nutritional status was assessed with abridged Patient-Generated Subjective Global Assessment.
Results
Thirty completed the study. Seventy-four per cent had at least one TSA. Taste changes and/or abnormalities were more prevalent than smell, and subjective taste changes more common than objective abnormalities. Although less common, smell abnormalities impacted quality of life more. TSA characteristics were heterogeneous. Forty-seven per cent were at malnutrition risk. No association was found between TSA and nutritional status.
Conclusions
Over two thirds had at least one TSA and almost half were at malnutrition risk. Self-reported TSA included changes in taste and smell perception, and most commonly persistent bad taste. This study demonstrated the complexity of TSA assessment and the prevalence, severity and impact of these and related symptoms in treatment-naïve cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Taste and smell play an important role in food enjoyment, oral nutrition and quality of life. Taste results from chemical stimulation of mouth receptors for bitter, salty, sour, sweet and umami. Smell perception occurs from nasal olfactory receptor neuron activation. When food enters the mouth, a combination of taste, smell and trigeminal signals occur and sensations like texture and temperature are realised [1]. The close physiological relationship between smell and taste has led to suggestions they should be assessed concurrently [2]. A more recent study concluded that smell had little influence on taste [3]. Distinct characteristics of taste and smell exist. Smell induces appetite and flavour perception whereas taste is primarily a nutrient sensing system [4]. Local and systemic physiological changes in chronic illness affect taste and smell [5]. Age and gender affect both; females have higher acuity. Acuity deteriorates with age, especially over 75 years [6].
Taste and smell abnormalities (TSA) commonly occur in all cancer stages and are among the most distressing symptoms in advanced disease [7]. They can be categorised as changed or distorted perception [5]. Changes in taste perception include ageusia (no taste), hypogeusia (reduced) and hypergeusia (enhanced). Taste distortions include dysgeusia (bad taste) and hallucinations. Anosmia (no ability to smell), hyposmia (reduced) and hypersomnia (enhanced) are the most common smell disturbances. Smell distortions include parosmia (incorrect perception) and phantosmia (perception without odour). TSA influence food enjoyment and social interactions. Loss of perception is associated with anorexia which can be complicated by early satiety, malnutrition and weight loss and worse quality of life [8,9,10].
The pathophysiology and characteristics of cancer TSA are poorly understood. They may occur from the actual cancer, from treatment (chemotherapy, medications, radiotherapy) or from cancer-related complications (dry mouth, oral infection, anaemia) or syndromes (para-neoplastic) [11]. Stage of disease and cancer type may also be important.
The presence, magnitude and impact of TSA are under-recognised [12,13,14]. Clinical assessment is not routine. Taste and smell can be measured subjectively and objectively. Because no gold standard exists, various methods are used. Few cancer studies have combined subjective and objective evaluations of both taste and smell [15,16,17]. Objective measures focus on detection and recognition thresholds and can identify reduced perception. They may more accurately reflect impaired chemical stimulation than do the subjective measures [18]. Subjective self-reports are favoured by some, because they capture patient experience [8]. They may more reliably predict dietary changes [19]. Limitations in subjective measures are evident. Taste descriptors are poorly understood and study participants have difficulty describing TSA [20, 21]. This may explain why they appear under-reported [19].
Reported prevalence and severity depend on assessment method and disease stage. TSA have been reported during radiotherapy (and persist afterwards) but there is insufficient evidence that chemotherapy alone affects taste. Chemotherapy can influence food preference independent of taste [22]. TSA during chemotherapy may be influenced by other symptoms like anorexia, early satiety, nausea and mouth problems [21].
There is limited evidence that TSA may be tumour related [23, 24]. In the treatment-naïve, studies are limited and results contradictory. Most studies used subjective measures only [23, 25,26,27]. In many, only taste abnormalities were evaluated and not smell. A recent study found a high prevalence of subjective TSA in newly diagnosed cancer patients before treatment, which suggests that TSA may originate from the tumour itself rather than the treatment [23]. In a three-arm study, taste disorder was examined in cancer patients (n = 42), acutely hospitalised inflammatory disease patients (n = 57) and healthy controls (n = 39). Cancer patients suffered taste disorders irrespective of chemotherapeutic treatments and cancer-related inflammation appeared to have a greater impact on taste perception than an acute inflammatory process [24].
No study has examined TSA, both subjectively and objectively, in newly diagnosed, treatment-naïve cancer patients. This study aimed to address this gap. The primary objectives were to assess the prevalence, characteristics and severity of TSA and to compare subjective with objective TSA results. Secondary objectives were to examine the relationship between TSA and concurrent symptoms and nutritional status.
Methods
This prospective observational study examined the characteristics, prevalence and severity of TSA in consecutive newly diagnosed, treatment-naïve patients with solid tumours. It was approved by the local research ethics committees at St James Hospital and St Luke’s Hospital in Dublin. Consecutive patients who attended a radiation oncology outpatient clinic in a large tertiary care teaching hospital were recruited at their first referral appointment over a 5-month period (June–October 2016). Those considered too unwell to participate, with < 7 days to live or with an Eastern Co-operative Oncology Group (ECOG) performance score of 4, were excluded [28]. Radiation oncologists identified eligible participants and invited them to participate. Research nurses (PUíD, MB) met eligible participants and provided detailed verbal and written study information. Those willing to participate provided written informed consent. Data collection was conducted by the two research nurses (PUíD, MB) in a private consultation room during the same visit. All measures were administered and completed in 30–40 min. Participants refrained from eating, drinking and smoking for 1 hour beforehand.
Subjective measures
Taste and smell survey
A modified version of the Taste and Smell Survey (TSS) assessed self-reported changes in taste and smell since the start of illness [29]. This self-administered survey, originally developed for HIV studies and used in several oncology studies [23, 26, 30], has not been validated for either population. The original 16-item questionnaire was reduced to 12 (four questions about HIV drugs were omitted) for this study. Seven of the 12 items related to taste and 5 to smell changes. Persistent bad taste and changes in taste and smell perception were assessed. One point was awarded for each taste or smell change; another point if rated “rarely”/“sometimes” or “mild”/“moderate” and two points if rated “often”/“always” or “severe”/“incapacitating”. Total taste score range was 0 to 10 (0 = no change; 10 = multiple severe changes) and smell scores ranged from 0 to 5 (0 = no change; 5 = many severe changes). Higher scores indicated greater prevalence and/or severity of taste or smell change. A combined subjective chemosensory global complaint score (0–15) was then calculated. Two un-scored free text questions addressed TSA impact on quality of life.
The Abridged Patient-Generated Subjective Global Assessment (abPG-SGA) assessed nutritional status [31]. This validated nutrition screening tool has four components: (1) weight history, (2) dietary intake, (3) nutritional impact symptoms (NIS) and (4) functional status.
First current weight and height were measured for body mass index (BMI) according to the World Health Organisation BMI classification [32]. Percentage weight loss was calculated from weight history at 1 or 6 months prior. Second, dietary intake during the previous month was assessed. The amount (“normal” or “> normal” = 0; “< normal” = 1) and type (“normal food, but < usual” = 1; “little solid food” = 2; “liquid” or “nutritional supplements only” = 3; “very little of anything” = 4; “enteral/parenteral nutrition” = 0) of food consumed were scored. Thirdly, participants were asked if the following NIS interfered with food intake in the previous 2 weeks: anorexia, constipation, diarrhoea, dry mouth, dysphagia, early satiety, fatigue, mouth sores, nausea, pain, taste and smell problems and vomiting. Symptoms were weighted (per abPG-SGA instructions) and scores per symptom ranged from 1 to 3. Total possible scores for this section were 0–24. Last, functional status was categorised by a numerical scale of 0–4 (0 = normal activity with no limitations; 4 = completely disabled, rarely out of bed). Scores from each component were combined for a total score. The minimum total abPG-SGA score was 0 and the maximum 36. A total ≥ 6 indicated malnutrition risk.
Objective measures
Taste assessment
Validated taste strips (Burghart Messtechnik GmbH, Wedel, Germany) tested the four basic taste modalities (bitter, salty, sour and sweet) [33]. Umami was not tested as this descriptor is not familiar or widely used in Ireland. Four strips, each impregnated with one of the four, were applied individually, to the centre of the tongue for whole-mouth testing. The participant closed their mouth for 10 seconds (timed with a stopwatch). The taste strip was then removed and the participant identified the taste as bitter, salty, sour or sweet in a forced response. One point was awarded for each correct answer for a total score range of 0–4 (0 = none correct; 4 = all correct). Incorrect answers indicated an abnormality for that particular modality. After each strip, the participant cleansed the palate with 10–20 ml of still cold bottled water (Tesco, Perthshire, UK). Thirty seconds then elapsed before the next strip was presented. The strips were offered in random order except for bitter. This was always last (as per user manual) because of the risk of persistent unpleasant taste.
Smell assessment
Olfactory function was measured by Sniffin’ Sticks® (Burghart Messtechnik GmbH, Wedel, Germany) [34]. This odour identification test, validated for healthy volunteers and patients with olfactory disorders, consisted of 12 felt tip pens each filled with a unique liquid odour [35]. The pens were presented in the following order (pens 1–12): orange, leather, cinnamon, peppermint, banana, lemon, liquorice, coffee, clove, pineapple, rose and fish. The cap was removed and the pen tip positioned 2 cm below the nostrils for 4 seconds (timed by stopwatch). Simultaneously, a card (uniquely paired with each pen) displayed four possible smells (only one of which was correct). In a forced choice procedure, they chose the item that best matched the presented odour. The objective tests detected normal and reduced taste and smell perception, but could not detect increased perception.
Results
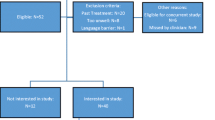
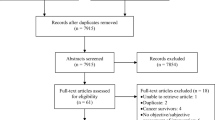
Forty consecutive newly diagnosed treatment-naïve cancer patients with solid tumours were screened. Of those eligible (n = 37), 31 participated and 30 full data sets completed. Demographic characteristics are in Table 1. The median combined global chemosensory complaint score was 1 (range 0–11). Twenty-two (74%) participants had at least one self-reported or objective TSA and ten (33%) had both (Fig. 1). Taste abnormalities were more prevalent than smell and self-reported taste changes more common than objective. Four had both subjective and objective taste complaints; the remainder (n = 15) had either one or the other. Three had objective taste abnormalities without subjective changes.
Taste
The median subjective taste complaint score was 1 (range 0–9). Three distinct types of self-reported taste complaints were present. The first was bad taste (dysguesia), reported by 13 (43%) (Fig. 1). Descriptors of this included “bitter” (n = 4), “salty” (n = 3), “sour” (n = 2), “metallic” (n = 2), “sweet” (n = 1) and “bad taste” (n = 1). The frequency varied; “sometimes” for six, “rarely” for four and “often” for three. The second was reduced taste perception (Fig. 1). Six (20%) reported at least one modality was “weaker” since becoming ill or “could not be tasted at all”. One described all four tastes as “weaker”. The third complaint was increased perception (Fig. 1). Seven (23%) reported at least one taste modality as “stronger” and one said all four were. Changes in perception (either “weaker” or “stronger”) were most frequent for salt and sweet. “Weaker” tastes were mostly “mild” (n = 5) or “moderate” (n = 4). “Stronger” tastes were “insignificant” (n = 6), “mild” (n = 2) or “moderate” (n = 1). Only one reported the taste complaint impacted quality of life.
In objective tests, everyone correctly identified at least one of the four tastes. The median objective taste abnormality score was 4/4 (range 0–4). Seven (23%) did not recognise one or more tastes (four of whom had no subjective changes). Recognition failures occurred for all tastes: bitter (n = 5), salt (n = 4), sour (n = 4) and sweet (n = 2).
There was a lack of concordance between subjective and objective results and the pattern of abnormalities was heterogeneous. For instance, three reported “bitter” tasted stronger, but only one was able to objectively identify it. Of the four who reported “sweet” was weaker, three correctly identified “sweet” on objective tests. Similar patterns were seen with “sour” and “salt”.
Smell
The median subjective smell complaint score was 0/5 (range 0–4). In objective tests, seven were hyposmic (score 6–9) and one anosmic (score 0–5). Similar prevalence was recorded for subjective and objective abnormalities (Fig. 1). Nevertheless, of the eight with hyposmia or anosmia, none self-reported a “weaker” sense of smell. Five (17%) self-reported smells as “stronger” and rated this as “insignificant” (n = 3) and “moderate” (n = 2). “Weaker” smells were self-reported by two (neither of whom had objective abnormality), rated “insignificant” by one and “severe” by the other. Four reported changed smell perception reduced quality of life and three of these cited nausea induced by smells as the issue.
Nutritional status
The median abPG-SGA score was 5 (range 0–19). Fourteen (47%) were at risk of malnutrition (score ≥ 6). Of these, nine had taste abnormalities; seven smell abnormalities and six both. All participants had above normal (over-weight n = 14; obese n = 8) or normal (n = 8) BMI. Four had lost weight in the previous 2 weeks (3 < 5%; 1 > 10%), six in the prior month and ten in the prior 6 months. Seven of the ten (who had lost weight) had at least one TSA. Ten reported reduced oral intake, and eight of these had TSA. Three attributed reduced food intake to a taste and two to a smell abnormality. More than half (n = 17) reported ≥ 1 NIS. The most common NIS were poor appetite (n = 10), fatigue (n = 9) and pain (n = 8) (Fig. 2). Chi-squared tests investigated the relationship between TSA and nutritional status but were not statistically significant. Linear regression tests showed no association between TSA and weight loss or symptom burden.
Discussion
TSA were common and complex, and characteristics heterogeneous irrespective of whether self-reported or objective assessment was used. Self-reported changes were more common for taste. Taste abnormalities occurred more frequently than smell and inter-individual variations were common. A self-reported unspecified bad taste was the single most common abnormality found. While taste abnormalities were more prevalent, smell changes were more likely to be moderate or severe and interfere with quality of life.
This study used both self-reported and objective measures to evaluate taste and smell in consecutive, treatment-naïve cancer patients. When TSS and objective taste results were compared, 16 inconsistencies were identified. The terms “salty” and “sweet” were easily recognised, but many appeared confused about the terms “sour” and “bitter” and struggled to distinguish between them. Detailed explanations were needed before some could respond. This has been noted elsewhere and may confound results [17, 21].
Similar inconsistencies were seen in smell. Only two of 15 with smell abnormality had both self-reported and objective complaints. Many reported smells were “stronger”, but the objective tests were only able to detect reduced smell. Those who self-reported “weaker” smell did not demonstrate reduced perception on objective smell test. The number of olfactory stimuli that humans can discriminate is unknown [36] but likely to be several thousand. We examined only 12 smells and so abnormalities were only captured for those. Peppermint, orange and clove were most often recognised.
Although no one was underweight, many of the group studied were at malnutrition risk. The NIS burden was the greatest contributory factor to this, ahead of weight loss and lower dietary intake. While no association between TSA and NIS was found, this deserves further scrutiny and may improve our understanding of TSA.
TSA prevalence in this study was higher than our previous report which had a similar design but used only self-report measures [28]. This may be because the subjective and objective instruments measured different variables over varied timeframes. Objective tests assessed taste or smell at a single time point. The TSS, in contrast, captured self-reported taste and smell changes “since becoming ill”. Future studies should consider age- and gender-matched cancer-free controls to explore if these findings could be tumour related.
The variations in TSA prevalence, characteristics and severity we noted have been described in other clinical settings and cancer cohorts [16, 23, 38, 39]. Possible explanations include diverse aetiologies and variations in cancer type and stage. This suggests the need for thorough assessment and individualised interventions. No single intervention can address the complex abnormalities observed.
This study had several limitations. Cancer types included breast, gynaecological and colorectal cancers only. The gender profile was biased to women and may have influenced results; women seem more likely to report TSA than men [6]. The age profile was also young for a cancer population. Accurate details of prescribed medication were not available to the researchers, but none had commenced anti-cancer treatment. The sample size was small and participants assessed at one time-point. Future studies should consider larger sample sizes and longitudinal designs to capture TSA natural history.
The abPG SGA screened for NIS in the prior 2 weeks. Only one in five reported that TSA interfered with food intake, so impact on food intake was limited. We did not formally measure food intake, but future studies should do this.
The TSS has not been validated and this should be done as no population norms are available. Despite other studies, no firm recommendations for cancer populations can be made. We modified it to exclude questions about drugs as accurate medication information was not available. The scoring system [21] had limited severity weights and this should also be addressed.
This study demonstrated the complexity of TSA assessment in treatment-naïve patients with solid tumours and the prevalence, severity and impact of these and related symptoms even before treatment. Further research should elicit the best ways to measure TSA in early stage cancer and relationship to dietary intake, malnutrition, weight loss and other NIS. Clinically, there is a need to better recognise TSA and their impact. This has multidisciplinary implications. Strategies for individualised care need to consider the variety of abnormalities observed.
Conclusions
Study participants were all newly diagnosed, treatment-naïve, solid tumour cancer patients with mostly good performance status. Nevertheless, over two thirds had at least one TSA and almost half were at malnutrition risk in spite of normal and above normal BMI.
TSA characteristics were heterogeneous. Although less common, smell abnormalities impacted quality of life more. Reported TSA included changes in taste and smell perception, and most commonly persistent bad taste. Prevalence was high for both taste and smell abnormalities but differed by assessment method. Taste abnormalities were more prevalent than smell and subjective changes more prevalent than objective. Participants were not knowledgeable about taste descriptors. There was considerable inter-individual variation in the results. This may be related to methodological challenges or may simply reflect the need for individualised assessment and management. This study is the first to combine subjective and objective TSA assessment in treatment-naïve cancer patients.
References
Hummel T, Landis BN, Hüttenbrink KB (2011) Smell and taste disorders. GMS Curr Top Otorhinolaryngol Head Neck Surg 10:1–15
Wrobel BB, Leopold DA (2004) Clinical assessment of patients with taste and smell disorder. Otolaryngol Clin N Am 37(6):1127–1142
Stinton N, Ali Atif M, Barkat N et al (2010) Influence of smell loss on taste function. Behav Neurosci 124(2):256–264
Boseveldt S, de Graff K (2017) The differential role of smell and taste for eating behaviour. Perception 46(3–4):307–319
Henkin RI, Levy LM, Fordyce A (2013) Taste and smell function in chronic disease: a review of clinical and biochemical evaluations of taste and smell dysfunction in over 5000 patients at the taste and smell Clinic in Washington, DC. Am J Otolaryngol 34(5):477–489
Yoshinaka M, Ikebe K, Uota M, Ogawa T, Okada T, Inomata C, Takeshita H, Mihara Y, Gondo Y, Masui Y, Kamide K, Arai Y, Takahashi R, Maeda Y (2016) Age and sex differences in the taste sensitivity of young adult, young-old and old-old Japanese. Geriatr Gerontol Int 16(12):1281–1288
Kirkova J, Walsh D, Rybicki L, Davis MP, Aktas A, Tao Jin, Homsi J (2010) Symptom severity and distress in advanced cancer. Palliat Med 24(3):330–339
Mahmoud FA, Aktas A, Walsh D, Hullihen B (2011) A pilot study of taste changes among hospice inpatients with advanced cancer. Am J Hosp Palliat Care 28:487–492
McLoughlin L, Mahon SM (2012) Understanding taste dysfunction in patients with cancer. Clin J Oncol Nurs 16(2):171–178
Epstein JB, Barasch A (2010) Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol 46:77–81
Gamper E, Zabernigg A, Wintner LM et al (2012) Coming to your senses: detecting taste and smell alterations in chemotherapy patients. A systematic review. J Pain Symptom Manag 44:880–895
Hutton JL, Baracos VE, Wismer WV (2007) Chemosensory dysfunction is a primary factor in the evolution of declining nutritional status and quality of life in patients with advanced cancer. J Pain Symptom Manag 33:156–165
Newell S, Sanson-Fisher R, Girgis A et al (1998) How well do medical oncologists' perceptions reflect their patients’ reported physical and psychosocial problems? Data from a survey of five oncologists. Cancer J 83:1640–1651
Zabernigg A, Gamper EM, Giesinger JM, Rumpold G, Kemmler G, Gattringer K, Sperner-Unterweger B, Holzner B (2010) Taste alterations in cancer patients receiving chemotherapy: a neglected side effect? Oncologist 15:913–920
Ovesen L, Hannibal J, Sorensen M et al (1991) Food intake, eating-related complaints, and smell and taste sensations in patients with cancer of the lung, ovary and breast undergoing chemotherapy. Clin Nutr 10(6):336–334
Steinbach S, Hummel T, Bohner C et al (2009) Qualitative and quantitative assessment of taste and smell changes in patients undergoing chemotherapy for breast cancer or gynecologic malignancies. J Clin Oncol 27(11):1899–1905
Cohen J, Laing DG, Wilkes FJ, Chan A, Gabriel M, Cohn RJ (2014) Taste and smell dysfunction in childhood cancer survivors. Appetite 75:135–140
Brisbois TD, Hutton JL, Baracos VE, Wismer WV (2006) Taste and smell abnormalities as an independent cause of failure of food intake in patients with advanced cancer - an argument for the application of sensory science. J Palliat Care 22:111–114
Spotten L, Corish C, Lorton C et al (2017) Subjective and objective taste and smell changes in cancer: a narrative review. Ann Oncol 28:969–984
Huldij A, Giesbers A, Klein Poelhuis EH et al (1986) Alterations in taste appreciation in cancer patients during treatment. Cancer Nurs 9(1):38–42
Bernhardson BM, Tischlman C, Rutquist LE (2008) Self–reported taste and smell changes during cancer chemotherapy. Support Care Cancer 16(3):275–283
Boltong A, Campbell K (2013) Taste changes: a problem for patients and their dietitians. Nutr Diet 70:262–269
Spotten L, Corish C, Lorton C et al (2016) Subjective taste and smell changes in treatment-naïve people with solid tumours. Support Cancer Care 24:3201–3208
Schalk P, Kohl M, Herrmann HJ, Schwappacher R, Rimmele ME, Buettner A, Siebler J, Neurath MF, Zopf Y (2018) Influence of cancer and acute inflammatory disease on taste perception: a clinical pilot study. Support Care Cancer 26:843–851
Williams LR, Cohen MH (1978) Altered taste thresholds in lung cancer. Am J Clin Nut 31(1):122–125
Kamath S, Booth P, Lad TE, Kohrs MB, McGuire WP (1983) Taste thresholds of patients with cancer of the esophagus. Cancer 52(2):386–389
Belqaid K, Orrevall Y, McGreevy J, Månsson-Brahme E, Wismer W, Tishelman C, Bernhardson BM (2014) Self-reported taste and smell alterations in patients under investigation for lung cancer. Acta Oncol 53:1405–1412
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP (1982) Toxicity and response criteria of the eastern cooperative oncology group. Am J Clin Oncol 5:649–655
Heald AE, Pieper CF, Schiffman SS (1998) Taste and smell complaints in HIV-infected patients. AIDS 12:1667–1674
Leyrer CM, Chan MD, Peiffer AM, Horne E, Harmon M, Carter AF, Hinson WH, Mirlohi S, Duncan SE, Dietrich AM, Lesser GJ (2014) Taste and smell disturbances after brain irradiation: a dose-volume histogram analysis of a prospective observational study. Pract Radiat Oncol 4(2):130–135
Gabrielson DK, Scaffidi D, Leung E, Stoyanoff L, Robinson J, Nisenbaum R, Brezden-Masley C, Darling PB (2013) Use of an abridged scored patient-generated subjective global assessment (abPG-SGA) as a nutritional screening tool for cancer patients in an outpatient setting. Nutr and Cancer 65(2):234–239
World Health Organisation: BMI Classification. (Internet: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi - accessed 31 October 2017)
Landis BN, Welge-Luessen A, Brämerson A, Bende M, Mueller CA, Nordin S, Hummel T (2009) "taste strips" - a rapid, lateralized, gustatory bedside identification test based on impregnated filter papers. J Neurol 256(2):242–248
Kobal G, Hummel TH, Sekinger B et al (1996) Sniffin’sticks’: screening of olfactory performance. Rhinology 34:222–226
Hummel T, Sekinger B, Wolf SR, Pauli E, Kobal G (1997) Sniffin’ sticks – olfactory performance assessed by the combined testing of odour identification, odour discrimination and olfactory threshold. Chem Senses 22:39–52
Gerkin RC, Castro JB (2015) The number of olfactory stimuli that humans can discriminate is still unknown. eLife 4 https://elifesciences.org/articles/08127
Bernhardson BM, Tishelman C, Rytqvist LE (2007) Chemosensory changes experienced by patients undergoing cancer chemotherapy: a qualitative interview study. J Pain Symptom Manag 34(4):403–412
Steinbach S, Hundt W, Schmalfeldt B, Böhner C, Berktold S, Wolf P, Harbeck N (2012) Effect of platinum-containing chemotherapy on olfactory, gustatory, and hearing function in ovarian cancer patients. Arch Gynecol Obstet 286(2):473–480
Sánchez-Lara K, Sosa-Sánchez R, Green-Renner D, Rodríguez C, Laviano A, Motola-Kuba D, Arrieta O (2010) Influence of taste disorders on dietary behaviors in cancer patients under chemotherapy. Nutr J 9:15
Acknowledgements
We would like to acknowledge the nursing and administration staff at the Radiation Oncology Out-patient at St James Hospital for their assistance with data collection processes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
This study has not been previously published.
We have full control of all primary data and can allow the journal to review the data if requested.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uí Dhuibhir, P., Barrett, M., O’Donoghue, N. et al. Self-reported and objective taste and smell evaluation in treatment-naive solid tumour patients. Support Care Cancer 28, 2389–2396 (2020). https://doi.org/10.1007/s00520-019-05017-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-019-05017-3