Abstract
Key message
This study showed the negative effects of insect defoliation on the reproduction of canopy trees, where defoliation was not artificially manipulated but rather observed in a natural setting.
Abstract
The gypsy moth (Lymantria diapar) is a serious defoliator with the ability to completely defoliate forest canopies. Although the negative effects of defoliation on tree reproduction have been revealed in studies with artificially manipulated defoliation, few studies have examined the effect of insect defoliation on the reproduction of canopy trees under natural condition. In this study, visual surveys were used to clarify the effect of gypsy moth defoliation on the production of acorns by oaks (Quercus spp.). Surveys were conducted in an outbreak year at 22 sites in central Japan (13 sites with Quercus crispula and nine sites with Q. serrata). Five of the Q. crispula sites were severely defoliated (mean defoliation ranged from 67 to 88 %), while the remainder of the Q. crispula sites and all of the Q. serrata sites were lightly defoliated (0–20 %). A negative effect of gypsy moth defoliation on acorn production was detected for Q. crispula. However, there was a synchronous decrease in acorn production from the previous year and crop levels were low at all sites regardless of the defoliation severity. The consistent low crop levels were likely the result of weather-related factors. Defoliation also negatively affected the production of acorns for Q. serrata; however, severe defoliation was not present at any Q. serrata sites. This study suggests that insect defoliation can affect forest ecosystem processes, such as the regeneration of host trees and the behavior of wildlife that depend on seed production, by reducing the reproductive potential of host trees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Defoliation has negative effects on tree reproduction and growth (Kulman 1971; Schowalter et al. 1986; Obeso 1993; Morris et al. 2007; Massad 2013). Among the numerous insect defoliators, the gypsy moth (Lymantria diapar), which is widely distributed across the northern hemisphere (Alalouni et al. 2013), is well known as a serious defoliator whose larvae can defoliate multiple tree species (Liebhold et al. 1995; Onodera and Hara 2011). Severe outbreaks of gypsy moths can cause complete defoliation of forest canopies (Lovett et al. 2006).
Gypsy moth defoliation induces various responses in trees: refoliation in the same season (Collins 1961; Eschtruth and Battles 2014), reduction of seed production (Gottschalk 1990; Kasbohm 1994), reduction of radial growth (Baker 1941; Muzika and Liebhold 1999; Kosola et al. 2001; Naidoo and Lechowicz 2001; Fajvan et al. 2008), chemical changes in the leaves (Schultz and Baldwin 1982), and even mortality in extreme cases (Campbell and Sloan 1977; Gottschalk et al. 1998; Eisenbies et al. 2007). These tree responses affect forest ecosystem processes (Lovett et al. 2006; Gandhi and Herms 2010). For example, ephemeral foliage loss or mortality of canopy trees can facilitate the growth of understory tree species and plant invasions, thus, altering forest species composition (Collins 1961; Fajvan and Wood 1996; Jedlicka et al. 2004; Eschtruth and Battles 2014). Such vegetation changes and diminished seed production caused by defoliation have cascading impacts on wildlife that depend on specific vegetation or seeds (Kasbohm et al. 1995, 1998; Bell and Whitmore 1997). In addition, defoliation can also affect nutrient cycles by altering the nutritional composition and seasonal distribution of litter fall (Grace 1986). Therefore, it is important to monitor the spatial and temporal dynamics of gypsy moth defoliation that occur at a regional scale and to quantify their effect on tree performance to more effectively manage forest ecosystems.
The majority of studies revealing the negative effects of defoliation on tree reproduction to date have involved one of two methods: artificial defoliation, mainly at the branch level and chiefly for small trees or shrubs (Rockwood 1973; Janzen 1976; Stephenson 1980; Marquis 1984; Obeso 1998; Kaitaniemi et al. 1999; Hoch 2005); or the use of insecticide to prevent defoliation in some trees and then compare impacts with naturally defoliated trees (Crawley 1985; May and Killingbeck 1995; Salleo et al. 2003). Consequently, little is known about the effect of insect defoliation on the reproduction of canopy trees under natural condition (Wesołowski et al. 2015), even for the gypsy moth defoliation (Gottschalk 1990; Kasbohm 1994). This is because, first, insect outbreaks that cause severe defoliation are relatively rare events. Second, severe defoliation is short-lived because it is often followed by refoliation (Kulman 1971). Accordingly, surveys of defoliation must be conducted after defoliation reaches a peak but before refoliation progresses: a period of less than 1 month (Collins 1961; de Beurs and Townsend 2008; Eschtruth and Battles 2014). Third, measuring the reproduction of canopy trees is problematic, as the commonly used seed trap method is labor-intensive to place, maintain, and empty. This problem, however, can be overcome by using visual surveys with binoculars (Koenig et al. 1994; Nakajima 2015).
The Asian gypsy moth is widely distributed in Japan (Liebhold et al. 2008; Arimoto and Iwaizumi 2014) with outbreaks recorded intermittently in various regions (Koyama 1954; Akasofu 1973; Higashiura 1987; Liebhold et al. 1998; Kamata 2002; Jikumaru and Sano 2007; Mizutani 2014). Previous studies have quantified defoliation intensity primarily in young coniferous plantation forests (e.g., Ishihama et al. 2011) with few surveys conducted in natural forests (Jikumaru and Sano 2007; Mizutani 2014). Moreover, to date, no study has demonstrated the distribution of defoliation intensity at a regional scale.
The main aim of this study was to evaluate the impact of gypsy moth defoliation on the production of acorns by oaks. The study was conducted in Toyama Prefecture, central Japan, where one of the subspecies of Asian gypsy moth, Lymantria dispar japonica, is distributed (Higashiura et al. 2011; Arimoto and Iwaizumi 2014) with outbreaks recorded occasionally (Koyama 1954; Akasofu 1973). The impact of gypsy moth defoliation was quantified in two oak species, Quercus crispula and Q. serrata, which are temperate deciduous trees requiring about 4–5 months to mature acorns from spring-flowering. These two species were chosen as they are the dominant species in secondary forests in this region (Nakajima and Ishida 2014) and are both susceptible to gypsy moth defoliation (Jikumaru and Sano 2007; Onodera and Hara 2011).
The Toyama Prefectural Government has been monitoring the acorn production of several hundred trees of Q. crispula and Q. serrata across the prefecture. In this study, visual surveys of defoliation and acorn production of these oak trees were conducted during a gypsy moth outbreak year (2014) at 22 sites (13 sites with Q. crispula and nine sites with Q. serrata). The aim was to examine the distribution of defoliation intensity throughout the prefecture and evaluate the impact of defoliation on acorn production. As the two oak species exhibit annual fluctuations in acorn production (Imada et al. 1990; Shibata et al. 2002; Maeto and Ozaki 2003; Saitoh et al. 2008; Fukumoto and Kajimura 2011), data on the acorn production in the previous year (2013) were used to compare with the production in 2014.
Materials and methods
Study site
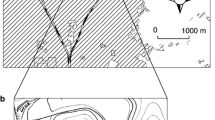
Visual surveys of defoliation and acorn production were conducted in 13 sites for Q. crispula (Fig. 1, A–M) and nine sites for Q. serrata (Fig. 1, 1–9) in Toyama Prefecture, central Japan. Elevation above sea level ranged from 350 to 1170 m for sites with Q. crispula and 90–340 m for sites with Q. serrata (Table S1). These sites had been established by the Toyama Prefectural Government to monitor acorn production and predict the mass intrusion of Asiatic black bears (Ursus thibetanus) into residential areas in years of poor acorn crops. The government monitoring protocol involves at least 20 trees per site, with the same trees being monitored every year unless the tree declined or died. This study surveyed these same trees (Table S1; n = 299 trees for Q. crispula; 184 trees for Q. serrata). These trees were canopy trees located along the roads and therefore defoliation and acorn-bearing could be visually surveyed easily.
Defoliation
Defoliation was surveyed in 2014 when an outbreak of gypsy moths occurred. In this region, incubation from overwintering egg begins around the middle of April, with pupation starting in late June. Defoliation proceeds rapidly just before pupation because most of the defoliation is caused by the last instar larvae (Furuno 1964). Refoliation induced by severe defoliation is completed within around 1 month of peak defoliation (Collins 1961; Eschtruth and Battles 2014). Therefore, the survey was conducted in July just after peak defoliation.
Using binoculars, tree-level defoliation was visually scored as the percentage loss of crown foliage, in 10 % classes (Williams et al. 1991; Gottschalk et al. 1998; Muzika and Liebhold 1999). Pictures of sample trees in 2012 with little defoliation were used as a reference. Defoliation was rated by the author alone who is skilled in rating crown foliage biomass visually in the forest monitoring program (Nakajima et al. 2011). Site-level defoliation was calculated by the mean of tree-level defoliation. No tree exhibited >20 % defoliation in the previous year, 2013.
Acorn index
In 2013 and 2014, acorn production was visually surveyed in mid- to late-August. The number of acorns was counted with binoculars for 20 branches (50 cm length each) per tree and a tree-level acorn index was calculated from these mean. The observed branches were selected at random from the sun crown in each year. This method was devised by Mizui (1991) and has been used for estimating the seed production of various tree species, including oak (Yasaka et al. 2008; Kozakai et al. 2011; Mizutani et al. 2013). A site-level acorn index was calculated by averaging the tree-level acorn indices, and categorizing this value into four crop levels: few, poor, fair, or good (Mizui 1991). The corresponding boundary values of the crop levels were 0–0.6, 0.6–1.9, 1.9–5.6, and >5.6 for Q. crispula, and 0–0.9, 0.9–2.6, 2.6–7.8, and >7.8 for Q. serrata. The values of Q. crispula were smaller than Q. serrata because the boundary values were defined to correlate negatively with the mean acorn weight of the species (Mizui 1991) and Q. crispula acorns are heavier. To examine the annual fluctuation in acorn production from 2013 to 2014, the tree-level acorn indices were compared between the 2 years in each site with a Mann–Whitney U test.
Relation between defoliation and acorn index
To clarify the effect of defoliation on acorn production, Pearson’s correlations between defoliation and acorn indices were calculated at both the site- and tree-level. In the tree-level analysis, pooled data across the sites were used. In addition, to control for variation among sites, a generalized linear mixed model with a negative binomial distribution and a log link function was used. The total acorn count for each tree was used as the dependent variable. Tree-level defoliation was included as a fixed effect, site was included as a random effect, and the log of the number of observed branches per tree (log 20) was included as an offset variable. The model was developed using R 3.1.2 (R Core Team 2014) with the aod package (Lesnoff and Lancelot 2012).
Results
Defoliation
The sites could be divided into two categories according to the intensity of defoliation (Fig. 2; Table S1). Five sites of Q. crispula (A–E) were categorized as severely defoliated sites, containing completely defoliated trees and an average (site-level) defoliation of between 67 and 88 %. These sites were located in the south-west of Toyama Prefecture, between 500 and 1000 m elevation (Fig. 1). The remaining eight sites of Q. crispula (F–M) and all nine Q. serrata sites were categorized as lightly defoliated sites. In these sites, the maximum tree-level defoliation was not greater than 50 % and the site-level defoliation ranged from 0 to 20 %.
Distribution of tree-level defoliation throughout the study area in 2014. The box represents the median (central thick lines) and quartiles (box width). Letters A–M and 1–9 correspond to site names listed in Fig. 1. The number of trees in each site is given at the top
In the severely defoliated sites, many cadavers of gypsy moth larvae were observed on the tree trunks, which occur as a result of pathogenic microbes, and characteristic of severe outbreak years (Aoki 1974; Jikumaru and Sano 2007). The larvae, pupae, or imago of gypsy moths were also observed in most of the lightly defoliated sites. Therefore, it was speculated that most of the defoliation was caused by gypsy moth larvae. When acorn production was surveyed in August, about one and a half months after peak defoliation, severely defoliated trees had refoliated and no tree had died.
Acorn production
The site-level acorn indices with Q. crispula exhibited a synchronous decrease among sites from 2013 to 2014 (Fig. 3; Table S1). The tree-level acorn index of 2014 was significantly lower than that of 2013 in 12 of the 13 sites (P < 0.05). Crop levels in 2013 were classified as ‘fair’ in five sites and ‘poor’ in eight sites, while in 2014 crop levels at all sites were classified as ‘few’, regardless of whether the site was severely or lightly defoliated. In contrast, the acorn indices with Q. serrata exhibited a synchronous increase from 2013 to 2014, significantly so in eight of the nine sites (P < 0.05). Crop levels in 2013 were mainly classed as ‘few’ (seven of the nine sites), while in 2014 most sites (five) were classed as ‘fair’.
Fluctuations in site-level acorn index (the mean number of acorns on a 50 cm branch) from 2013 to 2014. Letters A–M and 1–9 correspond to site names listed in Fig. 1
Effect of defoliation on acorn production
There were negative correlations between defoliation and acorn indices at both the site- (Fig. 4) and tree-levels (Fig. 5) for both species. Just 6 % (11/184) of Q. serrata trees did not contain any acorns (acorn index equal to zero), whereas 63 % (187/299) of Q. crispula trees had no acorns. For Q. crispula, 45 % (80/178) of lightly defoliated trees (defoliation ≤20 %) had no acorns, while a notably higher proportion (99 %, 67/68) of severely defoliated trees (defoliation ≥80 %) had no acorns. The results of the generalized linear mixed model revealed the negative effects of defoliation on acorn index at the tree level (for Q. crispula, defoliation slope estimate = −0.033, SE = 0.011, P = 0.0038; for Q. serrata, estimate = −0.029, SE = 0.011, P = 0.011).
Discussion
This study has revealed the negative effects of insect defoliation on the reproduction of canopy trees, where defoliation has not been artificially manipulated but rather observed in a natural setting.
Carbohydrate is traditionally considered a limiting resource for seed production in species with mast seeding behavior (intermittent synchronous seed production at regional scale). Meanwhile, recent studies have revealed that temperate deciduous canopy tree species, including oak, primarily use the current year’s photosynthate for seed production regardless of their reproductive interval (Ichie et al. 2013; Hoch et al. 2013). Therefore, seed production of these species is independent from old carbohydrate reserves. Their findings suggest that seed production may be limited by the accumulation of other nutrients, such as nitrogen and phosphorus, rather than carbohydrates (Ichie et al. 2013; Hoch et al. 2013). However, defoliation causes a reduction in the crown leaf area of a tree, decreasing its current year’s carbohydrate gain (Palacio et al. 2012). Defoliation also causes a reduction in the stem and root carbohydrate content, especially for severely defoliated trees that subsequently undergo refoliation (Wargo 1972; Wargo et al. 1972; Kosola et al. 2001; Rieske and Dillaway 2008; Palacio et al. 2012; Piper and Fajardo 2014). In addition, defoliation depresses the nitrogen uptake capacity of roots (Kosola et al. 2001). Accordingly, the negative effects of gypsy moth defoliation on the production of acorns by oak revealed in this study (Figs. 4, 5) are likely caused by deficiencies in the availability of carbohydrates or other nutrients that result in the abortion of immature acorns. Another possible explanation for the impact of defoliation on acorn production is that gypsy moth larvae directly consume oak flowers, which bloom just after the leaf flush in spring. However, Gottschalk (1990) suggested that the abortion of immature acorns has more significant effect than the consumption of flowers.
Although a negative effect of defoliation on acorn production was detected for Q. crispula (Figs. 4, 5), acorn production decreased from 2013 to 2014 and crop levels were low (‘few’) in 2014 at both severely and lightly defoliated Q. crispula sites (Fig. 3). Synchronous fluctuations in oak acorn production at geographical scales are thought to be driven by weather-related factors (e.g., Koenig and Knops 2013, 2014), and these may also explain the consistent low crop levels recorded for this species in 2014. If a gypsy moth outbreak occurred in a year with high crop potential, driven by weather-related factors, it is possible that only severely defoliated sites exhibit low crop levels (Kasbohm 1994).
For Q. serrata, defoliation also negatively impacted acorn production (Figs. 4, 5), even though no site was severely defoliated (Fig. 2). This result suggests that relatively low level defoliation can negatively impact acorn production.
In Japan, there are years when many Asiatic black bears intrude into residential areas in autumn. These are thought to coincide with years of poor acorn crops (Oka et al. 2004; Mizutani et al. 2013; Nakajima 2013) because acorns are a key food for bears during autumn, and because bears will alter their home ranges and foraging behaviors depending on annual fluctuations in acorn production (Kozakai et al. 2011). Therefore, increased human–bear conflicts are predicted to occur as a result of the reduced production of acorns by oaks following gypsy moth defoliation. However, the results of this study suggest that at a regional scale, the impact of gypsy moth defoliation in 2014 on acorn production was not great, because severe defoliation was observed only in Q. crispula sites (Fig. 2) where crop levels were low regardless of the defoliation severity (Fig. 3).
In North America, it is common for gypsy moth defoliation to cause tree mortality, with mortality rates higher in less vigorous trees, suppressed trees, and in trees that suffer severe defoliation for two consecutive years (Campbell and Sloan 1977; Gottschalk et al. 1998; Eisenbies et al. 2007). No tree deaths was observed in this study, at least between peak defoliation and the acorn production survey approximately one and a half months later. The resilience of trees in this study to defoliation might be because they were not suppressed, and did not experience consecutive years of severe defoliation, or because Japan’s moist climate creates an environment of generally low tree mortality from insect defoliation (Kamata 2002). Nevertheless, studies have shown that severe defoliation does negatively affect tree growth and vigor, with effects persisting for the following years (Campbell and Sloan 1977; Muzika and Liebhold 1999; Fajvan et al. 2008). Hence, the defoliation observed in this study (in 2014) will likely affect acorn production for the coming years.
Interestingly, the gypsy moth infestation did not cause severe defoliation at any Q. serrata site (Fig. 2). This is not because Q. serrata are less susceptible to gypsy moth defoliation, but rather because the gypsy moth outbreak in 2014 was elevation-dependent. The most severely defoliated area during the outbreak occurred between 500 and 1000 m elevation (A–E in Fig. 1), where Q. crispula dominate. Quercus serrata oaks occur mainly at lower elevations (Nakajima and Ishida 2014). In support of this theory, there were completely defoliated Q. serrata in site A at 570 m a.s.l. (pers. obs.), where Q. crispula trees were surveyed and mixed with Q. serrata trees. The cause of this elevation-dependent outbreak is likely to be related to factors such as the density of host trees and natural enemies (Kamata 2002; Haynes et al. 2012).
Studies from North America have shown interesting relationships among gypsy moth populations, acorn production, and small mammal populations: failed acorn production lead to reductions in the population sizes of small mammals that depend on the acorns, which subsequently lowers predation rates on gypsy moths leading to outbreaks (Liebhold et al. 2000). If outbreaks diminish acorn production as revealed in this study, positive feedback loops could be present. It is possible that relationships such as those observed in North America also operate in Japan (Liebhold et al. 1998) because acorn masting also affects rodent population dynamics in Japan (Saitoh et al. 2008) and because the predation of gypsy moths by rodent has been observed (Fukuyama et al. 1990; Liebhold et al. 1998). However, the main predators of gypsy moths in Japan are birds (Higashiura 1980; Furuta 1982). Long-term monitoring studies are needed to clarify the existence of such interactions among gypsy moths, small mammal populations, and oak trees.
In conclusion, this study has shown that gypsy moth defoliation reduced the production of acorns by two oak species. This finding lends further support to the suggestion that insect defoliation can dramatically affect forest ecosystem processes, such as the regeneration of host trees and the behavior of wildlife that depend on seed production, by reducing the reproductive potential of host trees.
Author contribution statement
HN conceived, designed, and executed this study and wrote the manuscript.
References
Akasofu Y (1973) Extraordinary outbreak of gypsy moth in Takaoka City. For Pests 22:214–217 (in Japanese)
Alalouni U, Schädler M, Brandl R (2013) Natural enemies and environmental factors affecting the population dynamics of the gypsy moth. J Appl Entomol 137:721–738. doi:10.1111/jen.12072
Aoki J (1974) Mixed infection of the gypsy moth, Lymantria dispar japonica MOTSCHULSKY (Lepidoptera: Lymantriidae), in a larch forest by Entomophthora aulicae (REICH.) SOROK. and Paecilomyces canadensis (VUILL.) BROWN et SMITH. Appl Entomol Zool 9:185–190. doi:10.1303/aez.9.185
Arimoto M, Iwaizumi R (2014) Identification of Japanese Lymantria species (Lepidoptera: Lymantriidae) based on PCR–RFLP analysis of mitochondrial DNA. Appl Entomol Zool 49:159–169. doi:10.1007/s13355-013-0235-x
Baker WL (1941) Effect of gypsy moth defoliation on certain forest trees. J Forestry 39:1017–1022
Bell JL, Whitmore RC (1997) Eastern towhee numbers increase following defoliation by gypsy moths. Auk 114:708–716. doi:10.2307/4089290
Campbell RW, Sloan RJ (1977) Forest stand responses to defoliation by the gypsy moth. For Sci Monogr 19:1–34
Collins S (1961) Benefits to understory from canopy defoliation by gypsy moth larvae. Ecology 42:836–838. doi:10.2307/1933521
Crawley MJ (1985) Reduction of oak fecundity by low-density herbivore populations. Nature 314:163–164. doi:10.1038/314163a0
de Beurs KM, Townsend PA (2008) Estimating the effect of gypsy moth defoliation using MODIS. Remote Sens Environ 112:3983–3990. doi:10.1016/j.rse.2008.07.008
Eisenbies MH, Davidson C, Johnson J, Amateis R, Gottschalk K (2007) Tree mortality in mixed pine–hardwood stands defoliated by the European gypsy moth (Lymantria dispar L.). For Sci 53:683–691
Eschtruth AK, Battles JJ (2014) Ephemeral disturbances have long-lasting impacts on forest invasion dynamics. Ecology 95:1770–1779. doi:10.1890/13-1980.1
Fajvan MA, Wood JM (1996) Stand structure and development after gypsy moth defoliation in the Appalachian Plateau. For Ecol Manage 89:79–88. doi:10.1016/S0378-1127(96)03865-0
Fajvan MA, Rentch J, Gottschalk K (2008) The effects of thinning and gypsy moth defoliation on wood volume growth in oaks. Trees 22:257–268. doi:10.1007/s00468-007-0183-6
Fukumoto H, Kajimura H (2011) Effects of asynchronous acorn production by co-occurring Quercus trees on resource utilization by acorn-feeding insects. J For Res 16:62–67. doi:10.1007/s10310-010-0208-7
Fukuyama K, Yamaguchi H, Koizumi C (1990) Comparative analysis of mortality factors of artificial larval populations of the gypsy moth, Lymantria dispar (Lepidotera: Lymantriidae) in Hokkaido: I. Mortality factors in five different types of forests. Appl Entomol Zool 25:205–213. doi:10.1303/aez.25.205
Furuno T (1964) On the feeding quantity of the gypsy moth (Lymantria dispar LINNE) and the camphor silk moth (Dictyoploca japonica BUTLER). J Jpn For Soc 46:14–19 (in Japanese with English summary)
Furuta K (1982) Natural control of Lymantria dispar L. (Lep., Lymantriidae) population at low density levels in Hokkaido (Japan). Zeitschrift für Angewandte Entomologie 93:513–522. doi:10.1111/j.1439-0418.1982.tb03629.x
Gandhi KJK, Herms DA (2010) Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biol Invasions 12:389–405. doi:10.1007/s10530-009-9627-9
Gottschalk KW (1990) Gypsy moth effects on mast production. In: McGee CE (ed) Proceedings of the workshop: Southern Appalachian mast management. Knoxville, Tennessee, pp 42–50
Gottschalk KW, Colbert JJ, Feicht DL (1998) Tree mortality risk of oak due to gypsy moth. Eur J For Path 28:121–132. doi:10.1111/j.1439-0329.1998.tb01173.x
Grace JR (1986) The influence of gypsy moth on the composition and nutrient content of litter fall in a Pennsylvania oak forest. For Sci 32:855–870
Haynes KJ, Liebhold AM, Johnson DM (2012) Elevational gradient in the cyclicity of a forest-defoliating insect. Popul Ecol 54:239–250. doi:10.1007/s10144-012-0305-x
Higashiura Y (1980) Analysis of factors affecting bird predation on gypsy moth egg masses by using Holling’s disc-quation. Res Popul Ecol 22:147–162. doi:10.1007/BF02513542
Higashiura Y (1987) Larval densities and a life-table for the gypsy moth, Lymantria dispar, estimated using the head-capsule collection method. Ecol Entomol 12:25–30. doi:10.1111/j.1365-2311.1987.tb00981.x
Higashiura Y, Yamaguchi H, Ishihara M, Ono N, Tsukagoshi H, Yokobori S, Tokishita S, Yamagata H, Fukatsu T (2011) Male death resulting from hybridization between subspecies of the gypsy moth, Lymantria dispar. Heredity 106:603–613. doi:10.1038/hdy.2010.92
Hoch G (2005) Fruit-bearing branchlets are carbon autonomous in mature broad-leaved temperate forest trees. Plant Cell Environ 28:651–659. doi:10.1111/j.1365-3040.2004.01311.x
Hoch G, Siegwolf RW, Keel SG, Körner C, Han Q (2013) Fruit production in three masting tree species does not rely on stored carbon reserves. Oecologia 171:653–662. doi:10.1007/s00442-012-2579-2
Ichie T, Igarashi S, Yoshida S, Kenzo T, Masaki T, Tayasu I (2013) Are stored carbohydrates necessary for seed production in temperate deciduous trees? J Ecol 101:525–531. doi:10.1111/1365-2745.12038
Imada M, Nakai T, Nakamura T, Mabuchi T, Takahashi Y (1990) Acorn dispersal in natural stands of Mizunara (Quercus mongolica var. grosseserrata) for twenty years. J Jpn For Soc 72:426–430
Ishihama N, Yasaka M, Ohno Y, Hasui S, Nakagawa M, Takiya M (2011) Damage by outbreaks of gypsy moth in newly planted larch species plantations at Higashikagura, central Hokkaido. Trans Mtg Hokkaido Br Jpn For Soc 59:59–62 (in Japanese)
Janzen DH (1976) Effect of defoliation on fruit-bearing branches of the Kentucky coffee tree, Gymnoclaudus dioicus (Leguminosae). Am Midl Nat 95:474–478. doi:10.2307/2424414
Jedlicka J, Vandermeer J, Aviles-Vazquez K, Barros O, Perfecto I (2004) Gypsy moth defoliation of oak trees and a positive response of red maple and black cherry: An example of indirect interaction. Am Midl Nat 152:231–236. doi:10.1674/0003-0031(2004)152[0231:GMDOOT]2.0.CO;2
Jikumaru S, Sano T (2007) Distribution of late instar Lymantria dispar cadavers killed by Entomophaga maimaiga on trunks of several tree species in southwestern Japan. Can J Bot 85:25–30. doi:10.1139/b06-146
Kaitaniemi P, Neuvonen S, Nyyssönen T (1999) Effects of cumulative defoliations on growth, reproduction, and insect resistance in mountain birch. Ecology 80:524–532. doi:10.1890/0012-9658(1999)080[0524:EOCDOG]2.0.CO;2
Kamata N (2002) Outbreaks of forest defoliating insects in Japan, 1950–2000. Bull Entomol Res 92:109–117. doi:10.1079/BER2002159
Kasbohm JW (1994) Response of black bears to gypsy moth infestation in Shenandoah National Park, Virginia. PhD thesis, Virginia Polytechnic Institute and State University, Blacksburg, Virginia
Kasbohm JW, Kraus JG, Vaughan MR (1995) Food habits and nutrition of black bears during a gypsy moth infestation. Can J Zool 73:1771–1775. doi:10.1139/z95-208
Kasbohm JW, Vaughan MR, Kraus JG (1998) Black bear home range dynamics and movement patterns during a gypsy moth infestation. Ursus 10:259–267
Koenig WD, Knops JMH (2013) Large-scale spatial synchrony and cross-synchrony in acorn production by two California oaks. Ecology 94:83–93. doi:10.1890/12-0940.1
Koenig WD, Knops JMH (2014) Environmental correlates of acorn production by four species of Minnesota oaks. Popul Ecol 56:63–71. doi:10.1007/s10144-013-0408-z
Koenig WD, Knops JMH, Carmen WJ, Stanback MT, Mumme RL (1994) Estimating acorn crops using visual surveys. Can J For Res 24:2105–2112. doi:10.1139/x94-270
Kosola KR, Dickmann DI, Paul EA, Parry D (2001) Repeated insect defoliation effects on growth, nitrogen acquisition, carbohydrates, and root demography of poplars. Oecologia 129:65–74. doi:10.1007/s004420100694
Koyama R (1954) Two epizootic diseases of gypsy moth. For Pests 3:296–298 (in Japanese)
Kozakai C, Yamazaki K, Nemoto Y, Nakajima A, Koike S, Abe S, Masaki T, Kaji K (2011) Effect of mast production on home range use of Japanese black bears. J Wildl Manage 75:867–875. doi:10.1002/jwmg.122
Kulman HM (1971) Effects of insect defoliation on growth and mortality of trees. Ann Rev Entomol 16:289–324. doi:10.1146/annurev.en.16.010171.001445
Lesnoff M, Lancelot R (2012) aod: Analysis of overdispersed data. R package version 1.3. http://cran.r-project.org/package=aod
Liebhold AM, Gottschalk KW, Muzika R, Montgomery ME, Young R, O’Day K, Kelley B (1995) Suitability of North American tree species to the gypsy moth: a summary of field and laboratory tests. General technical report NE-221. USDA Forest Service, Northeastern Forest Experiment Station, Radnor
Liebhold AM, Higashiura Y, Unno A (1998) Forest type affects predation on gypsy moth (Lepidoptera: Lymantriidae) pupae in Japan. Environ Entomol 27:858–862. doi:10.1093/ee/27.4.858
Liebhold A, Elkinton J, Williams D, Muzika RM (2000) What causes outbreaks of the gypsy moth in North America? Popul Ecol 42:257–266. doi:10.1007/PL00012004
Liebhold AM, Turcáni M, Kamata N (2008) Inference of adult female dispersal from the distribution of gypsy moth egg masses in a Japanese city. Agric For Entomol 10:69–73. doi:10.1111/j.1461-9563.2007.00359.x
Lovett GM, Canham CD, Arthur MA, Weathers KC, Fitzhugh RD (2006) Forest ecosystem responses to exotic pests and pathogens in eastern north America. Bioscience 56:395–405. doi:10.1641/0006-3568(2006)056[0395:FERTEP]2.0.CO;2
Maeto K, Ozaki K (2003) Prolonged diapause of specialist seed-feeders makes predator satiation unstable in masting of Quercus crispula. Oecologia 137:392–398. doi:10.1007/s00442-003-1381-6
Marquis RJ (1984) Leaf herbivores decrease fitness of a tropical plant. Science 226:537–539. doi:10.1126/science.226.4674.537
Massad TJ (2013) Ontogenetic differences of herbivory on woody and herbaceous plants: a meta-analysis demonstrating unique effects of herbivory on the young and the old, the slow and the fast. Oecologia 172:1–10. doi:10.1007/s00442-012-2470-1
May JD, Killingbeck KT (1995) Effects of herbivore-induced nutrient stress on correlates of fitness and on nutrient resorption in scrub oak (Quercus ilicifolia). Can J For Res 25:1858–1864. doi:10.1139/x95-200
Mizui N (1991) Classification of seed production based on the correlation between seed-weight and seed-number in deciduous broadleaved tree species. J Jpn For Soc 73:258–263 (in Japanese with English summary)
Mizutani M (2014) Gypsy moth outbreaks and their impacts on acorn production of Quercus crispula in Fukui Prefecture in 2013. Chubu For Res 62:63–66 (in Japanese)
Mizutani M, Nakajima H, Kodani J, Nogami T, Tada M (2013) Relationship between the acorn crops of Fagaceae trees and the mass intrusions of bears into residential areas in the Hokuriku region. J Jpn For Soc 95:76–82. doi:10.4005/jjfs.95.76 (in Japanese with English summary)
Morris WF, Hufbauer RA, Agrawal AA, Bever JD, Borowicz VA, Gilbert GS, Maron JL, Mitchell CE, Parker IM, Power AG, Torchin ME, Vázquez DP (2007) Direct and interactive effects of enemies and mutualists on plant performance: a meta-analysis. Ecology 88:1021–1029. doi:10.1890/06-0442
Muzika RM, Liebhold AM (1999) Changes in radial increment of host and nonhost tree species with gypsy moth defoliation. Can J For Res 29:1365–1373. doi:10.1139/x99-098
Naidoo R, Lechowicz MJ (2001) Effects of gypsy moth on radial growth of deciduous trees. For Sci 47:338–348
Nakajima H (2013) Estimation of beech (Fagus crenata) seed crops from female inflorescence scars: Relationship between seed crops and degree of intrusion into residential areas by Asiatic black bears in Toyama Prefecture, Japan. J Jpn For Soc 95:71–75. doi:10.4005/jjfs.95.71 (in Japanese with English summary)
Nakajima H (2015) Estimating sound seedfall density of Fagus crenata using a visual survey. J For Res 20:94–103. doi:10.1007/s10310-014-0440-7
Nakajima H, Ishida M (2014) Decline of Quercus crispula in abandoned coppice forests caused by secondary succession and Japanese oak wilt disease: Stand dynamics over twenty years. For Ecol Manage 334:18–27. doi:10.1016/j.foreco.2014.08.021
Nakajima H, Kume A, Ishida M, Ohmiya T, Mizoue N (2011) Evaluation of estimates of crown condition in forest monitoring: comparison between visual estimation and automated crown image analysis. Ann For Sci 68:1333–1340. doi:10.1007/s13595-011-0132-9
Obeso JR (1993) Does defoliation affect reproductive output in herbaceous perennials and woody plants in different ways? Func Ecol 7:150–155. doi:10.2307/2389881
Obeso JR (1998) Effects of defoliation and girdling on fruit production in Ilex aquifolium. Func Ecol 12:486–491. doi:10.1046/j.1365-2435.1998.00216.x
Oka T, Miura S, Masaki T, Suzuki W, Osumi K, Saitoh S (2004) Relationship between changes in beechnut production and Asiatic black bears in northern Japan. J Wildl Manage 68:979–986. doi:10.2193/0022-541X(2004)068[0979:RBCIBP]2.0.CO;2
Onodera K, Hara H (2011) Suitability of plant species as food for Asian gypsy moth larvae of the Hokkaido population. Bull Hokkaido For Res Inst 48:47–54 (in Japanese with English summary)
Palacio S, Hernández R, Maestro–Martínez M, Camarero JJ (2012) Fast replenishment of initial carbon stores after defoliation by the pine processionary moth and its relationship to the re-growth ability of trees. Trees 26:1627–1640. doi:10.1007/s00468-012-0739-y
Piper FI, Fajardo A (2014) Foliar habit, tolerance to defoliation and their link to carbon and nitrogen storage. J Ecol 102:1101–1111. doi:10.1111/1365-2745.12284
R Core Team (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rieske LK, Dillaway DN (2008) Response of two oak species to extensive defoliation: Tree growth and vigor, phytochemistry, and herbivore suitability. For Ecol Manage 256:121–128. doi:10.1016/j.foreco.2008.04.015
Rockwood LL (1973) The effect of defoliation on seed production of six Costa Rican tree species. Ecology 54:1363–1369. doi:10.2307/1934200
Saitoh T, Vik JO, Stenseth NC, Takanishi T, Hayakashi S, Ishida N, Ohmori M, Morita T, Uemura S, Kadomatsu M, Osawa J, Maekawa K (2008) Effects of acorns abundance on density dependence in a Japanese wood mice (Apodemus speciosus) population. Popul Ecol 50:159–167. doi:10.1007/s10144-008-0076-6
Salleo S, Nardini A, Raimondo F, Gullo MAL, Pace F, Giacomich P (2003) Effects of defoliation caused by the leaf miner Cameraria ohridella on wood production and efficiency in Aesculus hippocastanum growing in north-eastern Italy. Trees 17:367–375. doi:10.1007/s00468-003-0247-1
Schowalter TD, Hargrove WW, Crossley DA (1986) Herbivory in forested ecosystems. Ann Rev Entomol 31:177–196. doi:10.1146/annurev.en.31.010186.001141
Schultz JC, Baldwin IT (1982) Oak leaf quality declines in response to defoliation by gypsy moth larvae. Science 217:149–151. doi:10.1126/science.217.4555.149
Shibata M, Tanaka H, Iida S, Abe S, Masaki T, Niiyama K, Nakashizuka T (2002) Synchronized annual seed production by 16 principal tree species in a temperate deciduous forest, Japan. Ecology 83:1727–1742. doi:10.1890/0012-9658(2002)083[1727:SASPBP]2.0.CO;2
Stephenson AG (1980) Fruit set, herbivory, fruit reduction, and the fruiting strategy of Catalpa speciosa (Bignoniaceae). Ecology 61:57–64. doi:10.2307/1937155
Wargo PM (1972) Defoliation-induced chemical changes in sugar maple roots stimulate growth of Armillaria mellea. Phytopathology 62:1278–1283. doi:10.1094/Phyto-62-1278
Wargo PM, Parker J, Houston DR (1972) Starch content in roots of defoliated sugar maple. For Sci 18:203–204
Wesołowski T, Rowiński P, Maziarz M (2015) Interannual variation in tree seed production in a primeval temperate forest: does masting prevail? Eur J For Res 134:99–112. doi:10.1007/s10342-014-0836-0
Williams DW, Fuester RW, Metterhouse WW, Balaam RJ, Bullock RH, Chianese RJ (1991) Oak defoliation and population density relationships for the gypsy moth (Lepidoptera: Lymantriidae). J Econ Entomol 84:1508–1514. doi:10.1093/jee/84.5.1508
Yasaka M, Takiya M, Watanabe I, Oono Y, Mizui N (2008) Variation in seed production among years and among individuals in 11 broadleaf tree species in northern Japan. J For Res 13:83–88. doi:10.1007/s10310-007-0052-6
Acknowledgments
I am grateful to my colleagues at Toyama Prefectural Government for their helpful support. This study was funded by Toyama Prefecture, Japan.
Conflict of interest
The author declares that he have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Koike.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nakajima, H. Defoliation by gypsy moths negatively affects the production of acorns by two Japanese oak species. Trees 29, 1559–1566 (2015). https://doi.org/10.1007/s00468-015-1237-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-015-1237-9