Abstract
Objectives
Renovascular disease (RVD) is found in about 10 % of secondary childhood hypertension. Digital subtraction angiography (DSA) is the gold standard to diagnose RVD. Non-invasive imaging methods like Doppler ultrasound (US), magnetic resonance angiography (MRA), and computed tomography angiography (CTA) are increasingly used. Our aim was to evaluate the role and accuracy of US, MRA, and CTA compared to DSA in diagnosing RVD in children.
Patients and methods
A retrospective review of 127 children with suspected renovascular hypertension was performed in children referred to Great Ormond Street Hospital between 2006 and 2014 due to clinical suspicion of renovascular hypertension and/or findings on US and/or MRA or CTA.
Results
Ninety-nine of 127 children (78 %) were diagnosed with renovascular disease and 80 were treated with percutaneous transluminal angioplasty during the same procedure. The median age at presentation was 5.6 (range, 2.5–10.6) years. Thirty-six children had unilateral renal artery stenosis in major extraparenchymal vessels, 47 bilateral stenosis and 16 intrarenal small vessel disease. US had a sensitivity of 63 % and specificity of 95 %. MRA and CTA were performed in 39 and 34 children, respectively. CTA sensitivity was slightly higher than that of MRA, 88 vs. 80 %, and specificity 81 vs. 63 %.
Conclusions
The sensitivity of MRA and CTA is still too low to reliably rule out renovascular disease in children with a strong suspicion of this diagnosis. DSA remains the gold standard to diagnose renovascular hypertension and is recommended when clinical and laboratory criteria are highly suggestive of renovascular disease even with normal radiological investigations from non-invasive methods.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
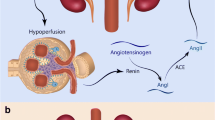
Renovascular disease is found in 10 % of secondary childhood hypertension [1–3]. Renal artery stenosis is important to accurately diagnose, as it is potentially amenable to curative treatment with several endovascular and surgical techniques.
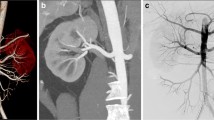
Digital subtraction angiography (DSA) is the gold standard in establishing the diagnosis of renovascular disease in children. It provides the best spatial and temporal resolution, producing excellent images of the renal arterial lumens and branches [4]. One main advantage of angiography is that therapeutic intervention with endovascular treatment can be undertaken by the interventional radiologist during the same procedure. On the other hand, DSA is an invasive diagnostic method that often needs general anesthesia in children exposed to a significant ionizing radiation exposure.
Several non-invasive imaging techniques are increasingly used to diagnose renovascular hypertension. This is based on clinical experience and studies in adult patients. The role of non-invasive imaging in children with suspected renovascular hypertension remains unclear due to the lack of studies in children. In this study, we aim to evaluate the accuracy of Doppler ultrasound (US), magnetic resonance angiography (MRA), and computed tomography angiography (CTA) compared to the gold standard DSA.
Patients and methods
A retrospective review of the medical notes, radiology reports, and diagnostic images of all children with suspected renovascular hypertension referred to Great Ormond Street Hospital for Children NHS Foundation Trust, London (GOSH) between 2006 and 2014 was performed. Children were transferred due to hypertensive emergencies or have been referred either based on positive US, MRA, or CTA scan results, or on clinical suspicion of renovascular hypertension evaluated with our established clinical and laboratory diagnostic algorithm [5].
All children underwent US and DSA at GOSH. We rarely perform MRA and CTA for this indication in our institution and we used the reports and diagnostic images of the MRA and CTA performed in the referring hospital for this study. Subsequent investigations in patients with known RVD or imaging of renal transplant vessels were excluded.
Clinical information on age at presentation, clinical symptoms, any underlying disease or diagnosed syndrome, blood pressure, number of anti-hypertensive medications, and interventional radiology procedures were recorded.
Definitions
Hypertension was classified into pre-hypertension (systolic blood pressure (SBP) between 90th and 95th centile), stage 1 hypertension (SBP >95th centile but ≤99th percentile plus 5 mmHg) and stage 2 hypertension (BP is >99th centile plus 5 mmHg).
RAS was defined as significant reduction of intraluminal diameter (e.g. ≥ 60 %) of the renal artery and presence of collateral vessels in DSA. The size of renal arteries is affected by the age and the size of the child and by the degree of branching. DSA results were interpreted as an involvement of major renal vessels when amenable for angioplastic interventions and as intrarenal small vessel disease when not amenable for interventions including segmental, lobar, and interlobar renal arteries. Major renal arteries were represented by the main renal artery, but also by one of its major branches (anterior/posterior division), accessory or duplicated renal arteries. Involvement of the abdominal aorta, superior mesenteric artery (SMA), inferior mesenteric artery (IMA), and celiac trunk were documented.
MRA and CTA suggested renovascular disease when one or more stenoses were directly visualized, evaluated by a relevant reduction of the intraluminal diameter and presence of collateral vessels.
Renal Doppler US was defined as positive, suggesting renovascular disease when US could directly visualize a stenosis or when a parvus et tardus waveform pattern or pathologic age-dependent flow parameters (peak systolic flow > 2 m/s [6, 7], acceleration time > 80 ms, renal artery to aortic flow velocity ratio >3, and difference in resistive index) were seen. A significant difference in kidney length (≥1 cm) was regarded as a possible indirect sign of RAS.
The accuracy of the non-invasive methods was compared to the gold standard considering both the results of each patient and each kidney separately.
Statistics
Percentages are given relative to all patients with available information regarding a particular topic. Sensitivities and specifities were determined for each method using DSA as the reference standard. Age is given as median with interquartile range (IQR).
Results
Patient characteristics
Between 2006 and 2014, 127 patients were newly referred to GOSH for evaluation of renovascular hypertension and investigated with Doppler US and DSA following a diagnostic algorithm. Among these, 12 patients (9.4 %) had a single functioning kidney either due to congenital absence or involution of a multi-cystic dysplastic kidney (six patients) or surgical removal (six patients). Consequently, 242 kidneys from 127 patients were included in the per kidney analysis.
Before they were referred, 50.3 % of children (64/127) were evaluated by MRA, CTA, or both. Among these, 53 patients had positive results suggestive for renovascular disease whereas 11 patients had negative investigations. The other half of the study cohort (N = 63) underwent DSA based on clinical and laboratory criteria and Doppler US results (26 with positive and 37 negative Doppler US results).
The age at diagnosis and at performing DSA are given in Fig. 1. More than half of the children (53 %) presented before the age of 6 years, 9 % during the first 6 months of life. Median age at first presentation was 5.6 (IQR, 2.5–10.6) years, median age at time of angiography 7.5 (3.6–11.5) years. Age at time of DSA differs from that at first disease manifestation, especially in the younger (<3 years) and older age groups (≥12 years) (Fig. 1). Overall, only 38 % of patients received DSA in the first 3 months after disease manifestation independent of age at clinical presentation: In the age group less than 1 year, 38 % (5/13) had the DSA shortly after onset of their disease; 32 % of patients in the age group 1–6 years underwent angiograms within 3 months after clinical manifestation, 43 % of patients in the age group ≥ 6 years.
Information about symptoms at presentation is reported in 72 % (91/127) of children. Among these, 41 % were clinically asymptomatic on screening blood pressure measurements, whereas 59 % (54/127) had neurological, cardiological, or unspecific clinical symptoms, mainly headaches.
More than three-quarters of patients (77 %; 98/127) presented with stage 2 hypertension and systolic blood pressure levels ≥ 140 mmHg, whereas 20 % of patients (25/127) presented with stage 1 hypertension and 3 % (4/127) with pre-hypertension. Blood pressure was difficult to control medically in 89 % of patients, requiring treatment with two or more anti-hypertensive agents.
An underlying condition causing the RAS was present in 25 % of the children - with neurofibromatosis type 1 (19/127, 15 %) and Williams syndrome (7/127; 5.5 %) as the most common.
DSA findings
Ninety-nine of 127 children (78 %) were diagnosed with renovascular hypertension on DSA. Thirty-six percent of patients were diagnosed with unilateral, 47 % with bilateral stenosis in the major renal arteries (including main, branch and accessory arteries), and 16 % with unilateral/bilateral intrarenal small vessel disease (including segmental, lobar, and interlobar renal arteries). In 34 %, mid-aortic syndrome was diagnosed additionally, either combined with bilateral (26 %) or unilateral renovascular disease (8 %). Isolated unilateral main renal artery stenosis was only diagnosed in 28 % of patients. In 14 %, other main intra-abdominal arteries (SMA, IMA, and/or celiac trunk) were affected.
Eighty children could be treated with angioplasty during the same intervention. Among these, 52 patients underwent unilateral and 28 patients bilateral angioplasty. Additionally, six patients required angioplasty of mid-aorta.
MRA versus DSA
MR angiography had been performed in 30 % (39/127) of the children prior to referral to us. Their median age was 8.0 (3.6–11.6) years. A total of 77 % of these MRA investigations had shown a positive result. On the patient level, we found a sensitivity of 82 % for MRA and specificity of 36 % (Table 1). Sensitivity was similar (80 %) when considering each kidney separately while specificity improved to 62 % (Table 1). MRA had a sensitivity and specificity of 88 and 94 % in detecting mid-aortic syndrome.
MRA missed the diagnosis of renal artery stenosis in ten kidneys: The location of RAS was the main renal artery in six of these cases, anterior or posterior branch artery in two and a segmental artery in two cases.
Median age of children with false-negative MRA results was 5.5 (3.8–8.2) years, 11 (9.6–15.4) years in children with false-positive MRA results. A pattern of reasons of false-negative MRA results like younger age or smaller vessel size could not be identified.
CTA versus DSA
CT angiography had been performed in 27 % (34/127) of children before they were referred to us. The median age of performing CTA in children was 6.2 (4.2–9.7) years. Renovascular disease had been diagnosed in 29 of 34 CTA scans (85 %). Sensitivity was 93 % and specifity 50 % on analyses per patient (Table 2). Assessing accuracy of CTA based on each kidney separately, sensitivity was 88 % and specifity 81 % (Table 2). Interestingly, CTA failed to diagnose mid-aortic syndrome (sensitivity only 45 %) whereas specifity in diagnosing mid-aortic syndrome was 96 %. Five diagnoses of renovascular disease were missed by CTA. All stenoses were identified in the main renal artery by DSA, combined with an additional stenosis in one of the branch renal arteries in one case. The median age of children with false-negative CTA results was 11 (7.5–15.3) years and 8.3 (6.3–10.2) years in children with incorrectly identified RAS (false-positive results) by CTA.
MRA and CTA versus DSA
Both MR and CT angiography had been performed in nine children (7 %) and 16 kidneys. In combination, CTA and MRA showed a sensitivity of 100 % in detection of renal artery stenosis (analyzed on kidney level) but only 50 % in diagnosing mid-aortic syndrome. Specificity of the combined investigations was 75 % in included children and 86 % on the kidney level. CTA and MRA showed consistent results in seven of nine patients. MRA missed bilateral renal artery stenosis in one child and overdiagnosed unilateral renal artery stenosis in another patient whereas CTA was true positive/true negative in these patients.
Renal Doppler US versus DSA
All children had been investigated with renal Doppler US in our pediatric radiology unit. There was a suspicion of renal artery stenosis in 50 % of these investigations. The sensitivity of Doppler US based on Doppler US criteria and number of patients, compared with DSA, was 63 %, whereas specifity was 96 % (Table 3).
If the criteria with difference in kidney length ≥1 cm were included, 12 more patients were identified. Among these 12 patients, ten were diagnosed with renal artery stenosis on DSA (true-positive results). Sensitivity of Doppler US increased to 73 %, specificity dropped to 89 % if the length criterion was also included (Table 4).
Analyzed on a kidney level (n kidneys = 242), sensitivity was 65 % and specifity 95 % (Table 5). Separately analyzing children with single kidneys, sensitivity was 40 % and specificity 100 % but number of included children were small (n single kidneys = 12).
Seventy-one renal artery stenoses were not identified by Doppler US, located in 39 main, 11 branch, and 14 segmental and smaller arteries. Seven main renal artery stenoses appeared combined with intrarenal changes of small renal arteries.
Summarized, 65 % (46/71) of missed renal artery stenoses were located in the main renal artery. Median age of children with false-negative results was 6 (3.4–10.2) years compared to 7.5 (3.6–11.5) years in all 127 patients.
Discussion
In this retrospective study of 127 children with suspected RAS, we found the sensitivity to diagnose renal artery stenosis to be 88 % for CTA, 80 % for MRA, and 73 % for Doppler US. These results are worse than those seen in prospective studies in adult patients with sensitivities of 92–98 % for MRA and 64–94 % for CTA [8–14]. The sensitivity is the most important parameter for these investigations, as it is crucial not to miss any treatable RAS. Of importance is that our study is biased towards better performance of MRA and CTA, as findings on those modalities were one main reason for referral to us. It is thus impossible to know how many children had false-negative CTA or MRA that never had formal DSA.
We advocate performing DSA if the clinical findings are highly suspicious of renovascular hypertension, as all other methods miss relevant and potentially treatable cases of renal artery stenosis. We have developed a clinical diagnostic algorithm to guide in this [5]. This is especially important in the younger age group and in small vessels, where data suggest non-invasive imaging is more likely to fail [15, 16]. The algorithm includes the evaluation of the grade of hypertension (e.g., hypertension stage 2), number of anti-hypertensive medication (at least ≥ 2 drugs), secondary symptoms of hypertension (most common cardiac and cerebral), underlying syndromes with higher risk for vasculopathies, signs for vasculitis, known previous vascular insults (e.g., umbilical arterial catheter, previous trauma or radiation), existing bruit during auscultation of renal arteries, raised peripheral plasma renin and moderate hypokalemia. The included study cohort is highly prescreened, but also shows the efficacy of the screening based on clinical and laboratory criteria. The positive angiography rate was remarkably high with 78 %.
Although 78 % of included children were diagnosed with renovascular disease by DSA, only 38 % of patients with suspected renovascular hypertension underwent angiogram within 3 months after clinical presentation independent from initial age. Earlier performance of DSA in children with highly suspected renovascular hypertension is recommended. The tendency to perform DSA in younger age groups delayed with > 3 months after first manifestation can reflect the technical challenges of the examination in this age group.
In our study, 99 of 127 children were diagnosed with renovascular disease and 80 children were treated with angioplasty during the same procedure. A minority (28 %) had only isolated unilateral main renal artery stenosis. A large group showed widespread vascular disease with bilateral stenosis, mid-aortic involvement and/or involvement of the splanchnic circulation. This confirms the observation that children often have a complex renal artery disease with different locations of RAS [15]. As seen in our study, in general, RAS in children involves the extraparenchymal main, accessory, branch arteries, and/or intraparenchymal segmental renal arteries, less frequently the lobar and intralobar arteries [15, 17, 18]. The most important issue is if the size of the renal artery allows angioplasty as a curative treatment option. Apart from the degree of branching, age and size of the child influence the size of renal arteries. These aspects need to be taken into account when evaluating the accuracy of non-invasive methods in pediatric renovascular disease.
Doppler US
Renal vessel and abdominal vessel Doppler US is an important baseline investigation, as it also can detect tumors (such as neuroblastoma or pheochromocytoma) and show discrepant renal lengths or other renal pathologies. US is a simple and safe technique but requires experienced pediatric staff and a cooperative child to obtain adequate images. In general, the experience with Doppler US in pediatrics is rather limited apart from diagnosing renal artery stenosis in transplanted kidneys [19]. Adult series have shown a sensitivity of Doppler US of 60–100 % and specifity of 70–100 % in the detection of renal artery stenoses [10, 20–25]. The sensitivity in our study was in the lower end of that range, 63 %, improving to 73 % when the diagnostic criteria were extended to include kidney length difference of ≥1 cm. Our findings are consistent with other pediatric studies in smaller cohorts in which false-negative or false-positive results are not uncommon. A sensitivity for Doppler US of 65–88 % and specificity of 83–99 % has been reported in studies of up to 47 children [6, 26–28]. All pediatric studies are consistent with the observation that Doppler US has difficulties with the detection of stenoses of small renal artery branches, segmental renal arteries or accessory renal arteries [5, 6, 15, 26, 28, 29]. In our cohort, 65 % (46/71) of missed renal artery stenoses were, however, located in the main renal artery prior to any branching.
CTA
The quality of the three-dimensional images using contrast-enhanced multidetector computer tomography angiography (CTA) has improved significantly with technical advancements. Several recent studies comparing renal CTA to DSA in adults with renal artery stenosis have shown comparable accuracy [11, 30]. Renal CTA in adults has shown sensitivity of 64–94 % and specificity of 62–97 % for hemodynamically significant renal artery stenosis [8–11, 31].
There is only one small retrospective pediatric study of 24 children with clinically suspected renovascular disease, where CTA correctly predicted the DSA results of RAS in five of the six patients with a confirmed diagnosis [32].
The advantages of renal CTA are that it is non-invasive, can be conducted quickly without the need for sedation or general anesthesia, allows easy generation of 3D images, while sparing the risks of conventional DSA. However, CTA gives a significant radiation exposure, even with low-dose protocols [33]. CTA also has the ability to assess non-vascular structures and is sensitive for detecting non-vascular but surgically correctable causes of hypertension in children, such as occult renal or suprarenal neoplasms [32], which may be occasionally missed by laboratory tests, US and DSA.
MRA
Unlike CTA, contrast-enhanced MRA of the renal arteries and abdominal aorta in children often requires sedation or general anesthesia. MRA is otherwise an ideal non-invasive imaging technique and can evaluate renal parenchymal disease with high anatomical detail. The spatial resolution of MRA examinations has improved significantly with technological advancements, and recent studies in adults have shown excellent resolution of the main artery [12]. A meta-analysis of 25 studies including 998 adult patients reported sensitivity and specificity of non-enhanced MRA of 94 and 85 % [14]. For gadolinium-enhanced MRA, sensitivity was 97 %, specifity 93 % [8, 10, 12–14, 31].
There exists again very little pediatric data. MRA was less sensitive and specific than CTA in our cohort of 39 patients and 74 kidneys. False-negative results were mainly seen in younger children. A pattern of reasons of false-negative results could however not be identified. Not only stenoses in small intrarenal arteries—segmental, lobar, and intralobar renal arteries—or accessory renal arteries were missed by MRA, but predominantly main renal artery stenoses. This observation thus supports our recommendation to perform DSA in children with suspected renal artery stenosis. False-positive results were seen in children with unilateral duplicated renal arteries on one side, one being more gracile but not stenotic in DSA.
The largest drawback with our study is that it was based on retrospective data using results obtained in many different pediatric radiology departments around the world with variable techniques. This means that we have no full knowledge about the exact technique that was used for the investigation. We observed an improvement of equipment and techniques over time. Clinically important (i.e., treatable) causes of hypertension will often be missed, even with the best equipment and techniques currently available. We have discussed performing a prospective study with CTA and MRA acquired during the same admission to our unit. This would, however, substantially have increased the radiation burden and many children would have needed additional general anesthesia. From our clinical point of view, such an extended study was not ethically justified and practicable, although it would be the best scientific approach. The advantage with our clinical approach is that it gives real-world knowledge of how well CTA and MRA work in different units around the world and entail further diagnostic steps. Better results of MRA and CTA techniques will exist in major international centers, but we report here our experience in the diagnostic approach to a rare but important diagnosis in childhood in the largest published pediatric cohort to date.
In summary, there is no reliable non-invasive imaging study in depicting renal artery stenosis in children. Neither CTA nor MRA was shown to be sensitive enough to diagnose all renovascular disease in children. There are two important differences between pediatric and adult practice. Firstly, the disease process (and anatomical distribution) is different in children, and attention must be directed to lesions of very small (often <1 mm) arteries. Secondly, the importance of reliably diagnosing every case of RVD in adult patients might be slightly less. Non-invasive imaging can, however, be used to rule out extraparenchymal processes, diagnose parenchymal disease, or to plan surgery and to follow-up post-surgery.
It seems likely that future technical developments will improve the performance of CTA and MRA. In particular, it may be possible to identify ischemic areas of renal parenchyma using MR techniques that evaluate perfusion delay. This might be achievable using a bolus of intravenous contrast and the rapid acquisition of a series of images at high temporal and moderate spatial resolution. The decision to undertake CTA or MRA in the diagnostic process of renovascular hypertension needs to be individualized and probably adapted to the age and the clinical condition of a patient. CTA is often more suitable for imaging of the renal arteries in small and young children and has the advantage of requiring no sedation or general anesthesia, whereas MRA might be preferred in older children, sparing radiation exposure without requiring sedation.
We recommend a clinical algorithm to decide when to do a DSA before or despite negative Doppler US, MRA, or CTA results [5]. In children with clinically a high suspicion of renovascular disease, DSA remains the gold standard.
Abbreviations
- CTA:
-
Computed tomography angiography
- DSA:
-
Digital subtraction angiography
- GOSH:
-
Great Ormond Street Hospital London
- IQR:
-
Interquartile range
- MRA:
-
Magnetic resonance tomography
- RAS:
-
Renal artery stenosis
- RVD:
-
Renovascular disease
- US:
-
Doppler ultrasound
References
Gill DG, Mendes de Costa B, Cameron JS, Joseph MC, Ogg CS, Chantler C (1976) Analysis of 100 children with severe and persistent hypertension. Arch Dis Child 51:951–956
Wyszynska T, Cichocka E, Wieteska-Klimczak A, Jobs K, Januszewicz P (1992) A single pediatric center experience with 1025 children with hypertension. Acta Paediatr 81:244–246
Bayazit AK, Yalcinkaya F, Cakar N, Duzova A, Bircan Z, Bakkaloglu A, Canpolat N, Kara N, Sirin A, Ekim M, Oner A, Akman S, Mir S, Baskin E, Poyrazoglu HM, Noyan A, Akil I, Bakkaloglu S, Soylu A (2007) Reno-vascular hypertension in childhood: a nationwide survey. Pediatr Nephrol 22:1327–1333
Tullus K (2011) Renal artery stenosis: is angiography still the gold standard in 2011? Pediatr Nephrol 26:833–837
Tullus K, Brennan E, Hamilton G, Lord R, McLaren CA, Marks SD, Roebuck DJ (2008) Renovascular hypertension in children. Lancet 371:1453–1463
Brun P, Kchouk H, Mouchet B, Baudouin V, Raynaud A, Loirat C, Azancot-Benisty A (1997) Value of Doppler ultrasound for the diagnosis of renal artery stenosis in children. Pediatr Nephrol 11:27–30
Conkbayir I, Yucesoy C, Edguer T, Yanik B, Yasar Ayaz U, Hekimoglu B (2003) Doppler sonography in renal artery stenosis. An evaluation of intrarenal and extrarenal imaging parameters. Clin Imaging 27:256–260
Eklof H, Ahlstrom H, Magnusson A, Andersson LG, Andren B, Hagg A, Bergqvist D, Nyman R (2006) A prospective comparison of duplex ultrasonography, captopril renography, MRA, and CTA in assessing renal artery stenosis. Acta Radiol 47:764–774
Frush DP (2008) Pediatric abdominal CT angiography. Pediatr Radiol 38(Suppl 2):S259–266
Rountas C, Vlychou M, Vassiou K, Liakopoulos V, Kapsalaki E, Koukoulis G, Fezoulidis IV, Stefanidis I (2007) Imaging modalities for renal artery stenosis in suspected renovascular hypertension: prospective intraindividual comparison of color Doppler US, CT angiography, GD-enhanced MR angiography, and digital substraction angiography. Ren Fail 29:295–302
Sabharwal R, Vladica P, Coleman P (2007) Multidetector spiral CT renal angiography in the diagnosis of renal artery fibromuscular dysplasia. Eur J Radiol 61:520–527
Hacklander T, Mertens H, Stattaus J, Lurken M, Lerch H, Altenburg A, Rautenbach J, Cramer BM (2004) Evaluation of renovascular hypertension: comparison of functional MRI and contrast-enhanced MRA with a routinely performed renal scintigraphy and DSA. J Comput Assist Tomogr 28:823–831
Stacul F, Gava S, Belgrano M, Cernic S, Pagnan L, Pozzi Mucelli F, Cova MA (2008) Renal artery stenosis: comparative evaluation of gadolinium-enhanced MRA and DSA. Radiol Med 113:529–546
Tan KT, van Beek EJ, Brown PW, van Delden OM, Tijssen J, Ramsay LE (2002) Magnetic resonance angiography for the diagnosis of renal artery stenosis: a meta-analysis. Clin Radiol 57:617–624
Vo NJ, Hammelman BD, Racadio JM, Strife CF, Johnson ND, Racadio JM (2006) Anatomic distribution of renal artery stenosis in children: implications for imaging. Pediatr Radiol 36:1032–1036
Garel L, Dubois J, Robitaille P, Russo P, Filiatrault D, Grignon A, Dube J (1995) Renovascular hypertension in children: curability predicted with negative intrarenal Doppler US results. Radiology 195:401–405
Srinivasan A, Krishnamurthy G, Fontalvo-Herazo L, Nijs E, Meyers K, Kaplan B, Cahill AM (2011) Spectrum of renal findings in pediatric fibromuscular dysplasia and neurofibromatosis type 1. Pediatr Radiol 41:308–316
Deal JE, Snell MF, Barratt TM, Dillon MJ (1992) Renovascular disease in childhood. J Pediatr 121:378–384
Stringer DA, O’Halpin D, Daneman A, Liu P, Geary DF (1989) Duplex Doppler sonography for renal artery stenosis in the post-transplant pediatric patient. Pediatr Radiol 19:187–192
Williams GJ, Macaskill P, Chan SF, Karplus TE, Yung W, Hodson EM, Craig JC (2007) Comparative accuracy of renal duplex sonographic parameters in the diagnosis of renal artery stenosis: paired and unpaired analysis. Am J Roentgenol 188:798–811
Solar M, Zizka J, Krajina A, Michl A, Raupach J, Klzo L, Ryska P, Ceral J (2011) Comparison of duplex ultrasonography and magnetic resonance imaging in the detection of significant renal artery stenosis. Acta Med (Hradec Kralove) 54:9–12
Voiculescu A, Hofer M, Hetzel GR, Malms J, Modder U, Grabensee B, Hollenbeck M (2001) Noninvasive investigation for renal artery stenosis: contrast-enhanced magnetic resonance angiography and color Doppler sonography as compared to digital subtraction angiography. Clin Exp Hypertens 23:521–531
Distler A, Spies KP (1991) Diagnostic procedure in renovascular hypertension. Clin Nephrol 36:174–180
Postma CT, van Aalen J, de Boo T, Rosenbusch G, Thien T (1992) Doppler ultrasound scanning in the detection of renal artery stenosis in hypertensive patients. Br J Radiol 65:857–860
Strandness DE Jr (1990) Duplex scanning in diagnosis of renovascular hypertension. Surg Clin North Am 70:109–117
Chhadia S, Cohn RA, Vural G, Donaldson JS (2013) Renal Doppler evaluation in the child with hypertension: a reasonable screening discriminator? Pediatr Radiol 43:1549–1556
Kchouk H, Brun P, Sentou Y, Raynaud A, Gaux JC, Loirat C (1997) Renal stenosis in hypertensive children. Doppler/arteriographic correlation. J Mal Vasc 22:86–90
Castelli PK, Dillman JR, Kershaw DB, Khalatbari S, Stanley JC, Smith EA (2014) Renal sonography with Doppler for detecting suspected pediatric renin-mediated hypertension—is it adequate? Pediatr Radiol 44:42–49
Li JC, Wang L, Jiang YX, Dai Q, Cai S, Lv K, Qi ZH (2006) Evaluation of renal artery stenosis with velocity parameters of Doppler sonography. J Ultrasound Med 25:735–742, quiz 743–734
Glockner JF, Vrtiska TJ (2007) Renal MR and CT angiography: current concepts. Abdom Imaging 32:407–420
Vasbinder GB, Nelemans PJ, Kessels AG, Kroon AA, Maki JH, Leiner T, Beek FJ, Korst MB, Flobbe K, de Haan MW, van Zwam WH, Postma CT, Hunink MG, de Leeuw PW, van Engelshoven JM (2004) Accuracy of computed tomographic angiography and magnetic resonance angiography for diagnosing renal artery stenosis. Ann Intern Med 141:674–682, discussion 682
Vade A, Agrawal R, Lim-Dunham J, Hartoin D (2002) Utility of computed tomographic renal angiogram in the management of childhood hypertension. Pediatr Nephrol 17:741–747
Kurian J, Epelman M, Darge K, Meyers K, Nijs E, Hellinger JC (2013) The role of CT angiography in the evaluation of pediatric renovascular hypertension. Pediatr Radiol 43:490–501, quiz 487–499
Contributors’ statements
Agnes Trautmann: Dr. Trautmann designed the data collection, collected the data, carried out the initial analyses, drafted the initial manuscript and approved the final manuscript as submitted.
Derek Roebuck: Dr. Roebuck reviewed and revised the manuscript, and approved the final manuscript as submitted.
Clare A. McLaren: Ms. McLaren reviewed and revised the manuscript and approved the final manuscript as submitted.
Eileen Brennan: Ms. Eileen Brennan reviewed and revised the manuscript and approved the final manuscript as submitted.
Kjell Tullus: Dr. Tullus conceptualized and designed the study, supervised data collection, critically reviewed and revised the manuscript, and approved the final manuscript as submitted.
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The retrospective study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
For this type of study formal consent is not required.
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Trautmann, A., Roebuck, D.J., McLaren, C.A. et al. Non-invasive imaging cannot replace formal angiography in the diagnosis of renovascular hypertension. Pediatr Nephrol 32, 495–502 (2017). https://doi.org/10.1007/s00467-016-3501-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-016-3501-7