Abstract
Background
Anti-complement factor H (CFH) autoantibody (Ab)-associated atypical hemolytic uremic syndrome (aHUS) has a poor prognosis, but no consensus exists on its treatment.
Methods
We report the follow-up of four children with anti-CFH Ab (8,000 to >32,000 arbitrary units)-associated aHUS after plasma exchanges (PEs), prednisone, and cyclophosphamide pulse therapy with the evolution of anti-CFH Ab titers and kidney function.
Results
Patient 1 received PEs + prednisone + cyclophosphamide pulses after two relapses following PEs and then PEs + rituximab. The other three patients were treated with PEs + prednisone + cyclophosphamide pulses as a first-line therapy. In our four patients, the induction protocol combining PEs + prednisone + cyclophosphamide pulses led to a rapid and sustained remission up to 6 years, 4 years and 4 months without any maintenance therapy. Kidney function was normal and anti-CFH Ab titer decreased, but remained detectable during remission without any clinical or biological signs of relapse.
Conclusions
We demonstrate the long-term efficiency and safety of cyclophosphamide pulses combined with PEs and prednisone in anti-CFH Ab-associated aHUS leading to a prolonged decrease in anti-CFH Ab titers and prevention of relapses without the need for maintenance therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atypical hemolytic uremic syndrome (aHUS) is a rare thrombotic microangiopathy mostly associated with defective regulation of the alternative complement pathway leading to inappropriate chronic complement activation. In 6–10 % of cases, aHUS is associated with the presence of autoantibodies (Ab) directed against complement factor H (CFH) resulting in a functional deficiency of CFH [1–5].
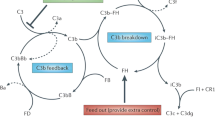
CFH plays a key role in regulating activation of the alternative complement pathway at cell surfaces. CFH prevents the formation of the C3bBb convertase and accelerates its decay, in addition to mediating proteolytic cleavage of C3b by factor I. CFH is composed of 20 short consensus repeats (SCRs) and contains at least two C3b-binding sites. The first binding site for C3b is located within the N-terminal domains SCR1-4 and regulates fluid-phase alternative pathway amplification. The second C3b-binding site is located in the C-terminal domains SCR19-20 and is involved in the regulation of alternative complement pathway activation at the endothelial cell surface. Anti-CFH Ab block the C-terminal domains SCR19-20 and impair CFH binding to C3b and to cellular surfaces, leading to uncontrolled complement activation at the cell surface [1, 3, 4]. Moreover, it has been recently demonstrated that anti-CFH Ab form immune complexes with CFH at disease onset, leading to its complete neutralization [5]. This ultimately results in endothelial cell injury and development of aHUS.
The mechanism implicated in the autoimmunization is not completely understood but anti-CFH Ab-associated aHUS seems to be secondary to a combination of acquired factors and genetic predisposition. Indeed, more than 90 % of patients with anti-CFH Ab-associated aHUS present a complete deficiency of CFHR1 and CFHR3 associated with a homozygous deletion of CFHR1 and CFHR3 [6, 7]. These patients define a novel group of aHUS, termed DEAP HUS (deficiency of CFHR proteins and CFH Ab-positive). The lack of circulating CFHR1 and CFHR3 seems to be necessary but not sufficient to develop the disease, since homozygous CFHR1 and CFHR3 deletions are also observed in the control population [6–8].
Anti-CFH Ab-associated aHUS presents a high relapsing course and carries a poor prognosis with a progression to end-stage renal disease (ESRD) in 20–35 % of cases, and a mortality rate of 10 % [9]. Plasma exchanges (PEs) for antibody clearance remain the first-line therapy at disease onset. However, PE cessation is often followed by a rebound of antibody titers and relapses frequently occur [1, 9, 10]. Therefore the association of immunosuppressive treatment is recommended, such as steroids, azathioprine, mycophenolate mofetil, cyclophosphamide, or anti-CD20 monoclonal antibody (rituximab). No consensus exists on the duration of plasmatherapy and the choice of induction and maintenance immunosuppressive regimen in this disease. Our group previously reported the remission of anti-CFH Ab-associated aHUS after PEs and cyclophosphamide pulses in three children with a median follow-up of 15 months [11]. We present herein the long-term follow-up of these children with the evolution of anti-CFH Ab titers and kidney function 4–6 years after onset. A fourth patient recently received PEs with prednisone and cyclophosphamide pulses at disease onset—his ongoing follow-up is also presented.
Patients and methods
The main characteristics of the four children at disease onset and/or at relapses and their treatments are reported in Tables 1 and 2.
Patients
Four patients were diagnosed at our center with anti-CFH Ab-associated aHUS between 2006 and 2012. The four patients were white boys aged between 16 months and 11 years and previously healthy.
The diagnoses of anti-CFH Ab-associated aHUS were based on the presence of anti-CFH Ab titer > 100 AU [1], an acute anemia (<10 g/dl) with schistocytes, thrombocytopenia (<150 × 109/l) and acute kidney injury (serum creatinine level > normal values for age and/or estimated glomerular filtration rate (eGFR) < 90 ml/min/1.73 m2 and/or proteinuria > 0.5 g/day). GFR was estimated by the modified Schwartz formula [12].
Stool culture, detection of Shiga toxins in stool, and serum antibodies against lipopolysaccharides were negative in three of the four patients. Patient 2 presented an episode of non-hemorrhagic diarrhea preceding disease onset. Stool culture showed a non-enteropathogenic Escherichia coli O145, and anti-O145 immunoglobulin M was detected in the serum.
A scarlet fever-like infection a few days before disease onset was reported for patient 4.
In each patient, CH50, C4, CFH, complement factor I (CFI), complement factor B (CFB) antigens, membrane cofactor protein (MCP) expression, and von Willebrand factor protease activity (ADAMTS-13 protease) values were normal, while plasma concentration of C3 was reduced in two of the four patients. This study was performed according to the instutional giudelines of our local ethics committee.
Treatment protocols
The treatment protocols of patients 1, 2, and 3 have been reported previously [11]. Briefly, patient 1 was initially treated with PEs. After the first relapse, he received PEs + rituximab. The second relapse was treated with PEs + prednisone + cyclophosphamide pulses 5× 1 g/1.73 m2 (number of CD19- and CD20-positive cells was 0/μl).
The three following patients were treated with PEs + prednisone + cyclophosphamide pulses 2× 0.5 g/1.73 m2 as a first-line therapy, 11–16 days after the onset, in association with intravenous immunoglobulin (IVIg) infusions in patient 4.
Complement assays
Measurement of CH50 activity in EDTA plasma samples was performed as previously described [13]. Plasma concentrations of the complement components C4, C3, CFB antigens were measured by nephelometry (Dade Behring, www.dadebehring.com). CFH and CFI antigen concentrations were measured by sensitive ELISA methods [13]. MCP (CD46) membrane expression was determined by flow cytometry [13].
Anti-complement factor H antibody assessment
Anti-CFH Ab were detected using the ELISA method as previously described [9]. Titers of positive samples were expressed as arbitrary units per milliliter (AU/ml) and calculated using a calibration curve obtained with serial dilutions of a reference positive plasma given an arbitrary titer from 100 to 2,000 AU/ml. The positive threshold was calculated by the mean + 5 SD of those obtained in the plasma of 100 individual healthy donors. This titer was determined to be 100 AU/ml, and titers above this value were considered as positive.
Genomic CFH, CFI, MCP, and C3 DNA sequencing
For genetic analysis, genomic DNA was extracted from peripheral blood cells. Uncloned genomic DNA was amplified by means of a PCR using oligonucleotides flanking each exon of each of CFH, CFI, MCP and C3 genes. Primer sequences, length of the PCR amplified fragments, and temperatures of hybridization used for each reaction, in addition to the direct DNA sequencing procedure, have been previously described [13].
Multiplex ligation-dependent probe amplification
The multiplex ligation-dependent probe amplification (MLPA) reaction was performed as described previously [6]. Sequences of probes were designed to determine dosage for exon 23 of CFH, exon 5 of CFHR1, and exon 3 of CFHR3 along with the control probe C1INH exon 8. Reagents of the MLPA reaction were purchased from MRC Holland (Amsterdam, The Netherlands), and the reaction was carried out according to the manufacturer’s recommended protocol.
Results
The follow-up after cyclophosphamide pulse therapy was respectively; 6 years (for patient 1), 4 years (for patients 2 and 3) and 4 months (for patient 4). All children fully recovered without relapse. PEs + prednisone + cyclophosphamide pulse therapy led to rapid remission with normalization of blood cell count, complement C3 level and urinary sediment, improvement in kidney function, and a prolonged decrease in CFH Ab titer (Table 3). The time from the first cyclophosphamide infusion to remission was between 1 and 4 weeks. All maintenance treatments could be discontinued in the first three patients. In patient 1, the dose of prednisone was reduced to 0.5 mg/kg/day within 2 months and then slowly tapered and stopped after 2 years. In patients 2 and 3, prednisone was tapered off within 6 months. Concerning patient 4, the dose of prednisone was gradually reduced to an alternate-day administration of 0.6 mg/kg/day after 4 months of follow-up.
The main characteristics of the four children at the last follow-up are reported in Table 3.
Renal outcome
At the last follow-up, eGFR was respectively 98, 95, 113, 102 ml/min/1.73 m2. During the follow-up, serum creatinine concentration remained stable with a mean ± SD of 81.5 ± 10.7, 48.8 ± 11.5, 32.2 ± 5.2, 55.6 ± 10.7 μmol/l. None of our patients developed any extra-renal complication of aHUS.
Treatment safety
No patient presented any side effect of cyclophosphamide such as an increased susceptibility to infection, hemorrhagic cystitis, or alopecia.
Evolution of anti-CFH Ab titers
In all patients, anti-CFH Ab decreased but remained detectable during remission (Fig. 1). As expected, the anti-CFH Ab titers were higher at the acute phase than in remission. After 6 and 4 years of follow-up for the first two patients, anti-CFH Ab titer was measured at 760 and 1,620 AU. Fluctuant titers were observed over the long-term follow-up with a minimal and a maximal anti-CFH Ab titer of 630 AU and 2,200 AU, respectively, measured at 47 months and 12 months after the disease onset for patient 1 and 380 AU and 2,350 AU measured after 17 months and 48 months of follow-up for patient 2. No clinical or biological sign of relapse was noted at the time of the maximal anti-CFH Ab titer. Concerning patient 3, the last anti-CFH Ab titer was <100 AU 4 years after disease onset. Patient 4 presented a progressive decrease of anti-CFH Ab titers to 200 AU measured after 1 month under PEs. At last follow-up, he presented an anti-CFH Ab titer of 450 AU.
Evolution of anti-complement factor H autoantibody (CFH Ab) titers during the follow-up in the four patients. The two relapses of patient 1 are represented by black vertical arrows. a Results are expressed as arbitrary units over time. b Results are expressed as percents of the titer at disease onset
Complement component profile
The plasma C3 antigen levels were lower at the acute phase in the two patients who presented higher titers of anti-CFH Ab. These children quickly normalized their complement C3 levels and in all patients the plasma C3 antigen levels remained normal during follow-up. The other complement component titers such as C4, CFH, Factor I, and Factor B antigens were within normal ranges.
Genetic analysis
None of the four patients presented any mutations in the CFH, CFI, MCP, or C3 genes.
All four patients showed a homozygous deletion mutation of the CFHR1 and CFHR3 genes.
Discussion
In this study, we demonstrate the long-term efficacy and safety of cyclophosphamide pulses combined with PEs and prednisone in four patients with anti-CFH Ab-associated aHUS, leading to a sustained remission without the need for any maintenance therapy.
Anti-CFH Ab-associated aHUS represents 60 % of aHUS cases occurring between 9 and 12 years old in the French cohort [1]. In the largest cohort of 45 patients with anti-CFH Ab-mediated aHUS reported by Dragon-Durey et al. [9], this form of aHUS occurs mainly in late childhood. However, as confirmed by our report, it can also affect younger children and the presence of anti-CFH Ab should be investigated in all patients with signs evocative of aHUS.
An infectious episode may precede the onset of anti-CFH Ab-associated aHUS, such as upper respiratory tract infection or transient urticaria with facial edema [9, 15, 16]. Kwon et al. [17] also reported two cases triggered by chickenpox. In our series, we noted a scarlet fever-like infection and one episode of non-hemorrhagic diarrhea preceding the disease onset. Several studies have also described some diarrhea during the prodromal phase of anti-CFH Ab-associated aHUS [1, 10, 15, 16] in up to 53 % of cases. This indicates that post-diarrheal onset does not exclude the possibility of aHUS.
Extra-renal complications are not uncommon in anti-CFH Ab-associated aHUS and include hepatitis (50 %), pancreatitis (23 %), and seizures (10–23 %) [1, 9, 10, 16, 18]. In addition, severe myocarditis has been reported in four patients [9, 19]. In our study, no patient presented any extra-renal manifestation.
Anti-CFH Ab-mediated aHUS is related to a poor prognosis. Dragon-Durey et al. [9] reported 58 % of relapse, among which 68 % occurred in the first 6 months. After a mean follow-up of 39 months, 39 % of patients had chronic kidney disease, 27 % had reached ESRD, and 9.5 % had died. Only 25 % of patients had a favorable outcome without sequelae. Accordingly, Noris et al. [16] reported on eight patients with anti-CFH Ab-mediated aHUS of whom 62 % progressed to ESRD or died, 12 % presented a complete remission, and 25 % had a partial remission within the 3 years from disease onset. In this present study, all our patients exhibited a favorable outcome following cyclophosphamide pulses, with no relapse and normal renal function. Two of them presented persistent hypertension as frequently reported in the literature [1, 20].
Dragon-Durey et al. [9] identified low C3 titers, low Factor B levels, and high plasma creatinine values at disease onset as major pejorative prognosis factors for relapse and renal survival. Indeed, anti-CFH Abs are associated with a systemic alternative complement pathway activation leading to the consumption of C3 and Factor B at disease onset. Patients may present with a low C3 titer sometimes associated with a low Factor B level while C4 titers are within the normal range [4, 9, 10, 21]. Among our series of four patients, two children presented isolated low C3 titers at time of diagnosis, which then returned to normal values during remission. These two patients with low C3 levels also exhibited the highest titers of anti-CFH Ab at onset, which is consistent with the literature [9]. However, despite the above-mentioned pejorative factor of low C3 level, their long-term outcome was favorable after a follow-up of 6 and 4 years.
The demonstration of anti-CFH Ab is crucial for etiological diagnosis in the context of aHUS and has a direct impact on the treatment choice and on prognosis. Therefore, the screening of Ab should be performed in all cases of aHUS and necessarily before PE therapy that removes the autoantibodies [22]. The anti-CFH Ab titers at disease onset are higher than in remission but remain detectable in most patients during remission [9, 15]. In our report, cyclophosphamide pulse therapy led to a decrease of anti-CFH Ab levels in all patients, which remained detectable with fluctuant titers over time without any clinical or biological sign of relapse. Some studies [9, 15] have tried to determine a threshold above which the risk of relapse is increased, but to date, there are insufficient data to statistically determine such a threshold. Indeed, in the present report, patient 1 with the relapsing course presented anti-CFH Ab levels of 2,000 and 5,800 AU at the first and second relapse respectively, before introduction of cyclophosphamide. It is interesting to note that at month 12, after cyclophosphamide pulses, anti-CFH Ab titer raised to 2,200 AU without any sign of relapse. Similarly, patient 2 had an anti-CFH Ab titer of 2,350 AU after 48 months of follow-up and did not present any relapse. Currently, the threshold predicting an increased risk of relapse and indicating the need for intensified therapy is not yet clearly established. This may be explained by individual variability, intrinsic susceptibility, and the lack of prospective follow-up in a large cohort. Undoubtedly, the follow-up of anti-CFH Ab titers represents a useful marker to monitor disease evolution and treatment efficacy during the acute phase and remission.
Both short-term and long-term outcome vary in the literature according to the therapy initiated, but standardized approaches to initial and maintenance therapy are lacking for anti-CFH Ab-associated aHUS. In the most recent guidelines [23, 24], PE therapy is considered the first-line therapy for patients during the acute phase of aHUS and should be started as early as possible within the first 24 h of presentation. However, fresh-frozen plasma (FFP) infusion, PE, or IVIg alone have failed to demonstrate significant long-term efficacy in patients with anti-CFH Ab-associated aHUS and discontinuing plasmatherapy is associated with high relapse rates and progression to ESRD [1, 4, 9, 10]. These findings have encouraged the early use of immunosuppressive treatment such as prednisone, azathioprine, rituximab, mycophenolate mofetil, and cyclophosphamide in anti-CFH Ab-mediated aHUS. To date, little data is available on the long-term efficiency of such therapies and concern only 14 patients (Table 4). Three of these received a combination of PEs and prednisone leading to remission during 14, 24, and 31 months, respectively, under persistent dose of prednisone for two of them [1, 10, 19]. In three patients with relapses under PEs, azathioprine induced a sustained remission with a normal kidney function at 5, 24, and 28 months of follow-up [1, 10, 19]. However, this treatment could not be withdrawn, and in one patient, azathioprine did not allow discontinuation of plasmatherapy. Rituximab has been used in anti-CFH Ab-mediated aHUS with variable outcome. Patient 1 in the present report relapsed despite rituximab infusions. Four other patients received rituximab. In one adult with PE dependence, the addition of prednisone and rituximab allowed improvement of renal function and discontinuation of PEs [9]. The three other patients received rituximab in preparation for kidney transplantation, among whom two were transplanted without relapse up to 8 months and 4 years, respectively [9, 25]. Mycophenolate mofetil was used on native kidneys in two patients who fully recovered up to 12 and 21 months [4, 9] and in one kidney transplant recipient in association with PEs, prednisone, basiliximab, and tacrolimus [25]. The latter patient remained free of relapse 2 years post-transplantation. One patient received PEs successively associated with mycophenolate mofetil, rituximab, and lastly, cyclophosphamide, 11 months after onset because of multidrug resistance. Despite these treatments, the child was in ESRD 42 months after the acute phase [4]. Eculizumab, an anti-C5 humanized monoclonal antibody, has demonstrated excellent efficiency on native or transplanted kidneys in patients with aHUS due to genetic abnormalities of the alternative complement pathway. Its use alone or in combination with PEs can induce prolonged remission, allow withdrawal of plasmatherapy, control plasma-refractory forms and prevent post-transplant relapses [18, 26, 27]. Therefore, eculizumab appears to be becoming the treatment of choice for aHUS without initial plasmatherapy. However, whereas maintenance eculizumab therapy has proven effective in aHUS, a single dose or treatment discontinuation has been associated with severe relapses [18, 28]. To our knowledge, eculizumab has not yet been used in patients with anti-CFH Ab-mediated aHUS. Contrary to cyclophosphamide pulses that induce a sustained decrease in CFH autoantibody production, eculizumab inhibits the activation of the terminal complement pathway without any effect on CFH autoantibody production. Therefore, it is unclear how long eculizumab therapy should be maintained to prevent relapses in patients with anti-CFH Ab-mediated aHUS. Finally, the use of eculizumab should be in balance with its very high cost and the increased risk of Neisseria meningitidis infection requiring vaccination and permanent prophylaxis.
In our report, following a short cyclophosphamide pulse therapy in association with steroids, all of our patients presented an excellent outcome with normal kidney function and no relapse throughout the follow-up of 6 years, 4 years, and 4 months without any maintenance therapy. No side effect such as infection, hemorrhagic cystitis, or alopecia have been observed in any of the four patients.
In conclusion, the identification of anti-CFH Ab is currently an indication for PEs associated with immunosuppressive therapy. Immunosuppressive drugs should be started early after the confirmation of the diagnosis. We recommend the use of cyclophosphamide as a first-line therapy or rapidly as a second-line therapy in case of failure of other treatments, before renal damage becomes irreversible. We suggest following the CFH Ab titer every 2 months for 6 months and then 3 times per year. Repeated measurements are recommended in case of pallor, hematuria, or proteinuria. If the CFH Ab titer increases, frequent blood and urine tests should be performed in order to detect signs of relapse. We would only propose an additional cyclophosphamide pulse in case of HUS recurrence. Prospective follow-up of larger cohorts of patients would be helpful to better determine the relapse risk factors and define therapeutic strategies.
References
Dragon-Durey MA, Loirat C, Cloarec S, Macher MA, Blouin J, Nivet H, Weiss L, Fridman WH, Frémeaux-Bacchi V (2005) Anti-Factor H autoantibodies associated with atypical hemolytic uremic syndrome. J Am Soc Nephrol 16:555–563
Skerba C, Józsi M, Zipfel PF, Dragon-Durey MA, Frémeaux-Bacchi V (2009) Autoantibodies in haemolytic uraemic syndrome. Thromb Haemost 101:227–232
Józsi M, Strobel S, Dahse HM, Liu WS, Hoyer PF, Oppermann M, Skerka C, Zipfel PF (2007) Anti-factor H autoantibodies block C-terminal recognition function of factor H in hemolytic uremic syndrome. Blood 110:1516–1518
Strobel S, Hoyer PF, Mache CJ, Sulyok E, Liu WS, Richter H, Oppermann M, Zipfel PF, Józsi M (2010) Functional analyses indicate a pathogenic role of factor H autoantibodies in atypical haemolytic uraemic syndrome. Nephrol Dial Transplant 25:136–144
Blanc C, Roumenina LT, Ashraf Y, Hyvärinen S, Sethi SK, Ranchin B, Niaudet P, Loirat C, Gulati A, Bagga A, Fridman WH, Sautès-Fridman C, Jokiranta TS, Frémeaux-Bacchi V, Dragon-Durey MA (2012) Overall neutralization of complement factor H by autoantibodies in the acute phase of the autoimmune form of atypical hemolytic uremic syndrome. J Immunol 185:3528–3537
Dragon-Durey MA, Blanc C, Marliot F, Loirat C, Blouin J, Sautes-Fridman C, Fridman WH, Frémeaux-Bacchi V (2009) The high frequency of complement factor H related CFHR1 gene deletion is restricted to specific subgroups of patients with atypical haemolitic uraemic syndrome. J Med Genet 46:447–450
Jozsi M, Licht C, Strobel S, Zipfel SL, Richter H, Heinen S, Zipfel PF, Skerka C (2008) Factor H autoantibodies in atypical hemolytic uremic syndrome correlate with CFHR1/CFHR3 deficiency. Blood 111:1512–1514
Hughes AE, Orr N, Esfandiary H, Diaz-Torres M, Goodship T, Chakravarthy U (2006) A common CFH haplotype, with deletion of CFHR1 and CFHR3, is associated with lower risk of age-related macular degeneration. Nat Genet 38:1173–1177
Dragon-Durey MA, Sethi SK, Bagga A, Blanc C, Blouin J, Ranchin B, André JL, Takagi N, Cheong HI, Hari P, Le Quintrec M, Niaudet P, Loirat C, Fridman WH, Frémeaux-Bacchi V (2010) Clinical features of anti-factor H autoantibody-associated hemolytic uremic syndrome. J Am Soc Nephrol 21:2180–2187
Lee BH, Kwak SH, Shin JI, Lee SH, Choi HJ, Kang HG, Ha IS, Lee JS, Dragon-Durey MA, Choi Y, Cheong HI (2009) Atypical hemolytic uremic syndrome associated with complement factor H autoantibodies and CFHR1/CFHR3 deficiency. Pediatr Res 66:336–340
Boyer O, Balzamo E, Charbit M, Biebuyck-Gougé N, Salomon R, Dragon-Durey MA, Frémeaux-Bacchi V, Niaudet P (2010) Pulse of cyclophosphamide therapy and clinical remission in atypical hemolytic uremic syndrome with anti-complement factor H autoantibodies. Am J Kidney Dis 55:923–927
Schwartz GJ, Muñoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20:629–637
Sellier-Leclerc AL, Frémeaux-Bacchi V, Dragon-Durey MA, Macher MA, Niaudet P, Guest G, Boudailliez B, Bouissou F, Deschenes G, Gie S, Tsimaratos M, Fischbach M, Morin D, Nivet H, Alberti C, Loirat C, French Society of Pediatric Nephrology (2007) Differential impact of complement mutations on clinical characteristics in atypical hemolytic uremic syndrome. J Am Soc Nephrol 18:2392–2400
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents (2004) The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 114:555–576
Hofer J, Janecke AR, Zimmerhackl LB, Riedl M, Rosales A, Giner T, Cortina G, Haindl CJ, Petzelberger B, Pawlik M, Jeller V, Vester U, Gadner B, Van Husen M, Moritz ML, Jungraithmayr T (2013) Complement factor H-related protein 1 deficiency and factor H antibodies in pediatric patients with atypical hemolytic uremic syndrome. Clin J Am Soc Nephrol 8:407–415
Noris M, Caprioli J, Bresin E, Mossali C, Pianetti G, Gamba S, Daina E, Fenili C, Castelletti F, Sorosina A, Piras R, Donadelli R, Maranta R, Van der Meer I, Conway EM, Zipfel PF, Goodship TH, Remuzzi G (2010) Relative role of genetic complement abnormalities in sporadic and familial aHUS and their impact on clinical phenotype. Clin J Am Soc Nephrol 5:1844–1859
Kwon T, Belot A, Ranchin B, Baudouin V, Frémeaux-Bacchi V, Dragon-Durey MA, Cochat P, Loirat C (2009) Varicella as a trigger of atypical haemolytic uraemic syndrome associated with complement dysfunction: two cases. Nephrol Dial Transplant 24:2752–2754
Nürnberger J, Philipp T, Witzke O, Opazo Saez A, Vester U, Baba HA, Kribben A, Zimmerhackl LB, Janecke AR, Nagel M, Kirschfink M (2009) Eculizumab for atypical hemolytic uremic syndrome. N Eng J Med 360:542–544
Abarrategui-Garrido C, Martinez-Barricarte R, Lopez-Trascasa M, de Cordoba SR, Sanchez-Corral P (2009) Characterization of complement factor H-related proteins in plasma reveals novel genetic variations of CFHR1 associated with atypical hemolytic uremic syndrome. Blood 114:4261–4271
Geerdink LM, Westra D, van Wijk JA, Dorresteijn EM, Lilien MR, Davin JC, Kömhoff M, Van Hoeck K, van der Vlugt A, van den Heuvel LP, van de Kar NC (2012) Atypical hemolytic uremic syndrome in children: complement mutations and clinical characteristics. Pediatr Nephrol 27:1283–1291
Moore I, Strain L, Pappworth I, Kavanagh D, Barlow PN, Herbert AP, Schmidt CQ, Staniforth SJ, Holmes LV, Ward R, Morgan L, Goodship TH, Marchbank KJ (2010) Association of factor H autoantibodies with deletion of CFHR1, CFHR3, CFHR4 and with mutations in CFH, CFI, CD46, and C3 in patients with atypical haemolytic uraemic syndrome. Blood 115:379–387
Roumenina LT, Loirat C, Dragon-Durey MA, Halbwachs-Mecarelli L, Sautes-Fridman C, Frémeaux-Bacchi V (2011) Alternative complement pathway assessment in patients with atypical HUS. J Immunol Methods 365:8–26
Ariceta G, Besbas N, Johnson S, Karpman D, Landau D, Licht C, Loirat C, Pecoraro C, Taylor CM, Van de Kar N, Vandewalle J, Zimmerhackl LB, European Paediatric Study Group for HUS (2009) Guidelines for the investigation and initial therapy of diarrhea negative haemolytic uraemic syndrome. Pediatr Nephrol 24:687–696
Taylor CM, Machin S, Wigmore SJ, Goodship TH (2010) Clinical practice guidelines for the management of atypical haemolytic uraemic syndrome in the United Kingdom. Br J Haematol 148:37–47
Kwon T, Dragon-Durey MA, Macher MA, Baudouin V, Maisin A, Peuchmaur M, Fremeaux-Bacchi V, Loirat C (2008) Successful pre-transplant management of a patient with anti-factor H autoantibodies-associated haemolytic uraemic syndrome. Nephrol Dial Transplant 23:2088–2090
Zimmerhackl LB, Hofer J, Cortina G, Mark W, Würzner R, Jungraithmayr TC, Khursigara G, Kliche KO, Radauer W (2010) Prophylatic eculizumab after renal transplantation in atypical hemolytic-uremic syndrome. N Eng J Med 362:1746–1748
Barbour T, Johnson S, Cohney S, Hughes P (2012) Thrombotic micorangiopathy and associated renal disorders. Nephrol Dial Transplant 27:2673–2685
Larrea CF, Cofan F, Oppenheimer F, Campistol JM, Escolar G, Lozano M (2010) Efficacy of eculizumab in the treatment of recurrent atypical hemolytic-uremic syndrome after renal transplantation. Transplantation 89:903–904
Acknowledgments
We thank the Assistance Publique - Hôpitaux de Paris and the Fondation M. Horlait-Dapsens for providing a residency grant to Gwenaëlle Sana.
Financial disclosure
The authors declare that they have no relevant financial interests.
The results presented in this paper have not been published previously in whole or part.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sana, G., Dragon-Durey, MA., Charbit, M. et al. Long-term remission of atypical HUS with anti-factor H antibodies after cyclophosphamide pulses. Pediatr Nephrol 29, 75–83 (2014). https://doi.org/10.1007/s00467-013-2558-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-013-2558-9