Abstract
The production of free radicals can cause renal injury and play a role in the pathogenesis of acute renal failure (ARF). The indirect markers of reactive oxygen species (ROS) were evaluated in children with ARF and controls. Forty patients with ARF aged 0–10 years were selected. Twenty age- and gender-matched healthy children were included as controls. Plasma malondialdehyde, protein carbonyl, nitrite, copper, ascorbic acid, zinc, and ceruloplasmin levels were estimated in patients with ARF and controls. The plasma malondialdehyde (p < 0.01), copper (p < 0.001), ascorbic acid (p < 0.05), and ceruloplasmin (p < 0.001) levels were significantly raised in ARF patients in comparison with controls. Significantly higher levels of plasma malondialdehyde (p < 0.01), nitrite (p < 0.001), copper (p < 0.001), and ceruloplasmin (p < 0.001) and lower plasma zinc (p < 0.01) were found in ARF nonsurvivors in comparison with survivors. The cutoff levels of plasma nitrite and ceruloplasmin were found to be most accurate in predicting mortality in ARF patients and had maximum sensitivity (100%) and specificity (60.7%) among the parameters studied. In conclusion, the increased levels of oxidants and antioxidants suggest the production of ROS and their possible role in ARF pathogenesis. Plasma nitrite and ceruloplasmin concentrations demonstrated predictive ability in relation to mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute renal failure (ARF) is a clinical syndrome in which there is a rapid decrease in the glomerular filtration rate (GFR) and change in the homeostasis of the body. About 5% of patients admitted to hospitals and about 30% of admitted to intensive care units develop this complication [1]. Common etiologic factors are ischemic renal failure, acute gastroenteritis, sepsis, and hemolytic uremic syndrome (HUS) following enterohemorrhagic Escherichia coli or Shigella infection [2]. Evidence has accumulated incriminating free oxygen radicals as the causative agent of renal damage in ARF, whatever the etiology may be [3, 4]. Cell injury occurs during reperfusion of ischemic tissues when molecular oxygen is introduced into the tissues [5].
Free radicals cause lipid peroxidation of cell and organelle membranes, leading to disruption of structural integrity and capacity for cell transport and energy production, especially in proximal tubules. These free radicals are short-lived and cannot be measured directly, but their activity can be measured by estimating the byproducts and substances involved in defense against the oxidant injury. Markers of oxidant stress can be malondialdehyde (MDA), a byproduct of lipid peroxidation; protein carbonyl; nitrite; and trace metals such as copper. Defenses against free-radical injury can be substances such as ascorbic acid, ceruloplasmin, and zinc [5]. Certain enzymes, such as superoxide dismutase, lipid peroxidase, and glutathione peroxidase, are raised in ARF patients, and lipid peroxidase has been found to have predictive ability in determining the outcome of these patients [6].
The role of free radicals in causing renal injury in the pathogenesis of ARF has been mainly shown in experimental and animal studies [7, 8], but studies in humans, especially in children, are lacking. Therefore, our study was undertaken with the objective of assessing the antioxidant status of children with ARF by estimating the levels of plasma MDA, protein carbonyl, nitrite, copper, ascorbic acid, zinc, and ceruloplasmin, and also to determine their prognostic significance in these patients.
Patients and methods
Study design
This was a prospective, hospital-based study that included 40 patients with ARF of both genders in the age group 0–10 years admitted to pediatric ward of a tertiary care hospital between June 2006 and May 2007. Patients who presented with a rapid decline in renal function, i.e. oliguria (urine output <400 ml/m2 per 24 h)/anuria or normal urine output with raised serum creatinine [>2 standard deviations (SD) above the mean for age], and blood urea nitrogen (BUN) (>12 mg/dl in newborns and >18 mg/dl in older infants/children) levels with or without acidosis were included. The children who developed ARF during their hospital stay were not enrolled. Another 20 age- and gender-matched normal children attending the outpatient department of general pediatrics for routine check-up were included to serve as controls. They had normal nutritional status and no abnormality on general physical and systemic examinations. Informed consent regarding inclusion in the study was given by the parents in each case, and the study protocol was approved by the Institute Ethics Committee.
Patients were treated with hospital management protocol, which broadly included fluid restriction, insensible loss replacement with intravenous 10% dextrose solution (400 ml/m2 per 24 h), frusemide (2–4 mg/kg IV, single dose), antibiotics in modified doses as per creatinine clearance, and intermittent peritoneal dialysis wherever required. Accurate recording of weight, blood pressure, intake, and urine output was done daily.
Blood sample collection and estimation
Taking all aseptic precautions, blood was collected from patients with ARF at the time of admission to hospital, through venipuncture from peripheral vein in a heparinized disposable syringe and transferred to sterile, deionized polyethylene vials. Plasma was separated immediately by centrifugation at 2,000 rpm for 5 min and was stored in separate deionized vials at 2–4°C. Plasma MDA was estimated by thiobarbituric acid test [9]. Protein carbonyl, nitrite, ascorbic acid, and ceruloplasmin were analyzed by the methods of Reznick and Packer [10], Moshage et al. [11], Roe [12], and Ravin [13], respectively. Plasma copper and zinc concentrations were estimated by atomic absorption spectrophotometry in the Department of Botany, Faculty of Science, Banaras Hindu University.
Statistical analysis
Data were analyzed using SPSS 12.0 software. Student’s t test was applied to the parameters with normal (Gaussian) distribution, and Mann–Whitney U test was used to the parameters with distribution different from normal. Comparing controls and patients, MDA, protein carbonyl, copper, ascorbic acid, and zinc showed normal distributions, whereas nitrite and ceruloplasmin did not. When comparison was made between survivors and nonsurvivors, protein carbonyl, nitrite and ceruloplasmin did not show normal distribution. The receiver operator characteristic (ROC) curve was obtained by plotting the sensitivity on the y-axis and 1-specificity on the x-axis for different parameters to determine the cutoff levels. The level depicting the maximum area under the curve (AUC) was taken as the cutoff value.
Results
Mean ages of the children with ARF and those in the control group was 39.2 (±39.2) months and 41.6 (±40.5) months, respectively. There were 13 patients (32.5%) younger than 1 year, 17 (42.5%) between 1–5 years of age, and 10 (25%) in the >5–10 years of age group. The overall male to female ratio was 2.3:1 both in ARF and control subjects. Etiologies of ARF patients are presented in Table 1. The majority of patients (77.5%) belonged to the renal group, followed by prerenal (15%) and postrenal (7.5%) etiologies. Septicemia (22.5%), HUS (17.5%), and perinatal asphyxia (10%) constituted the predominant renal causes of ARF. The mean duration of oliguria in oliguric ARF was 88.5 ± 66.5 h. The mean serum creatinine was 3.7 ± 2.25 mg/dl, with maximum level of 11.5 mg/dl. Peritoneal dialysis was required in nine (22.5%) patients.
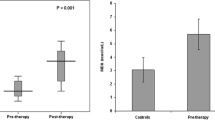
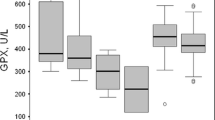
Mean plasma MDA (p < 0.01), copper (p < 0.001), ascorbic acid (p < 0.05), and ceruloplasmin (p < 0.001) levels were significantly raised in patients with ARF in comparison with controls, whereas mean plasma protein carbonyl, nitrite, and zinc concentrations were comparable with each other (Table 2). Of 40 patients with ARF, 12 (30%) died during the hospital stay. Of those 12, five had septicemia, two had HUS, and one patient each died of congenital nephrotic syndrome, steroid resistant nephrotic syndrome, severe malaria, perinatal asphyxia, and hepatorenal syndrome.
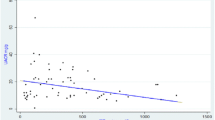
Observation of mean plasma MDA, protein carbonyl, nitrite, copper, antioxidants, and serum creatinine levels between ARF survivors and nonsurvivors are presented in Table 3. The mean plasma MDA (p < 0.01), nitrite (p < 0.001), copper (p < 0.001), and ceruloplasmin (p < 0.001) values were significantly raised and zinc was reduced (p < 0.01) in ARF nonsurvivors in comparison with survivors. The mean serum creatinine level was also significantly (p < 0.01) higher in nonsurvivors compared with survivors. An analysis was also done to determine the cutoff levels of plasma MDA, nitrite, copper, zinc, and ceruloplasmin by ROC curve in predicting mortality, and the observations are shown in Table 4. Plasma nitrite and ceruloplasmin levels had maximum sensitivity (100%), followed by MDA (91.7%) levels. However, the tests had lower specificity (60.7–64.3%). Although copper and zinc levels also had high sensitivity (91.7–100%), the specificity was very low (10.7–46.4%).
Discussion
The healthy kidney generates small amounts of reactive oxygen species (ROS) in the course of renal oxidative metabolism. The relatively low amount of ROS generated by the kidneys is tolerated without any apparent adverse effects. The most common cause of ARF is renal ischemia, and it has been seen that there is production of ROS during reperfusion of ischemic kidney and that these ROS cause further renal injury by lipid peroxidation [8]. Almost all tissue components, such as lipids, proteins, nucleic acids, and carbohydrates, are susceptible to injury by these free radicals injury and that antioxidants act as free-radical scavengers in this pathogenetic mechanism. Plasma MDA and copper were significantly raised in ARF patients, whereas protein carbonyl and nitrite remained unaltered. The increased plasma MDA levels indicate lipid peroxidation due to free-radical injury. The increased plasma copper in ARF patients indicates free-radical injury, as the metal has a definite role in the production of ROS and leads to oxidation of hydroquinone to benzoquinone, which is cytotoxic [14]. Milne et al. [15] examined the effect of glutathione on copper-mediated DNA oxidation and found that glutathione was able to inhibit free-radical formation by copper ions in the presence of H2O2, ascorbic acid, and DNA. Increased levels of MDA [16] and lipid peroxidase [17] in patients with ARF due to Plasmodium falciparum malaria have also been observed. Lipid peroxidation disrupts the structural integrity of the lipid bilayer and leads to increased membrane permeability and subsequent impaired ion transport, electron transport for oxidative phosphorylation in mitochondria, and increased lysosomal permeability. The increased lysosomal permeability leads to release of hydrolytic enzymes, further enhancing cell injury [5].
The raised levels of antioxidants suggest the involvement of protective mechanisms in ARF. The increased levels of ascorbic acid and ceruloplasmin found in this study indicate intact oxidant defense mechanism, as they can act as free-radical scavengers [18]. Zwołińska et al. [19] observed significantly raised levels of plasma and erythrocyte MDA and reduced zinc, copper, and selenium concentrations in patients with chronic renal failure in predialysis phase when compared with controls. The authors concluded that increased lipid peroxidation and decreased antioxidant capacity is also found in patients with chronic renal failure. Later, the authors also reported increased oxidative stress in patients with end stage-renal disease on maintenance dialysis, independent of dialysis modality [20].
It was also found that plasma MDA, nitrite, copper, and ceruloplasmin levels were significantly increased and zinc was decreased in ARF nonsurvivors. Increased plasma MDA, nitrite, and copper levels in nonsurvivors are indicative of more free-radical injury to renal tissue. The fact is further supported by significantly higher levels of mean serum creatinine in nonsurvivors, indicating the relationship of oxidative stress with severity of ARF and affecting the outcome in these patients. Dubey et al. [6] reported higher values of lipid peroxidase, superoxide dismutase, and glutathione peroxidase in their ARF nonsurvivors. The significantly increased plasma ceruloplasmin [21] and decreased zinc further indicates free-radical injury and their role as antioxidants. The decrease in zinc concentration in nonsurvivors indicates its increased consumption in the process of antioxidant activity in these patients. It appears that zinc supplementation may have protective effect on renal function. This is supported by the observations of previous authors [22, 23] who demonstrated the role of zinc-induced metallothionein synthesis, which could improve renal function by scavenging reactive oxygen free radicals in experimental rats. The cutoff levels of plasma MDA, nitrite, copper, zinc, and ceruloplasmin were also analyzed and it was observed that maximum sensitivity, specificity, and accuracy were found for nitrite and ceruloplasmin concentrations in predicting the outcome. Dubey et al. [6] found that lipid peroxidase level had maximum prognostic significance in their patients.
Conclusions
Increased plasma levels of MDA and copper indicate the possible role of free-radical injury in ARF pathogenesis. The increased levels of ascorbic acid and ceruloplasmin show their antioxidant role, as they serve as footprints of oxidative damage in this condition. Altered levels of plasma nitrite, ceruloplasmin, MDA, copper, and zinc in ARF nonsurvivors further indicate their prognostic significance, with maximum predictive values for nitrite and ceruloplasmin concentrations. Therefore, it appears that these biochemical alterations are indicative of oxidative damage to renal tissue during hypoxia and reperfusion. However, further studies are needed to determine the cause-and-effect relationship and its prognostic value in ARF patients.
Abbreviations
- ARF:
-
acute renal failure
- MDA:
-
malondialdehyde
- ROS:
-
reactive oxygen species
References
Brady HR, Singer GG (1995) Acute renal failure. Lancet 346:1533–1540
Bhuyan UN, Bagga A (1991) Thrombo-occlusive nephroangiopathy in children with acute renal failure and hypertension. Indian J Nephrol 1:137–138
Baud L, Ardaillou R (1993) Involvement of reactive oxygen species in kidney damage. Brit Med Bull 49:621–629
Cheesman KH, Slatter TF (1993) An introduction to free radical biochemistry. Brit Med Bull 49:481–493
Greene AL, Paller MS (1991) Oxygen free radicals in acute renal failure. Miner Electrolyte Metab 17:124–132
Dubey NK, Yadav P, Dutta AK, Kumar V, Ray GN, Batra S (2000) Free oxygen radicals in acute renal failure. Indian Pediatr 37:153–158
Nath KA, Norby SM (2000) Reactive oxygen species and acute renal failure. Am J Med 109:665–678
Paller MS, Hoidal JR, Ferris TF (1984) Oxygen free radicals in ischemic acute renal failure in the rat. J Clin Invest 74:1156–1164
Satoh K (1978) Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chem Acta 90:37–43
Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Methods Enzymol 233:357–363
Moshage H, Kok B, Huizenga JR, Jansen PLM (1995) Nitrite and nitrate determinations in plasma: a critical evaluation. Clin Chem 41:892–896
Roe JH (1954) Chemical determination of ascorbic, dehydroascorbic and diketogulonic acids. Methods Biochem Anal 1:137–140
Ravin HA (1961) An improved colorimetric enzymatic assay of ceruloplasmin. J Lab Clin Med 58:161–168
Li Y, Trush MA (1993) Oxidation of hydroquinone by copper chemical mechanism and biological effects. Arch Biochem Biophys 300:346–355
Milne L, Nicotera P, Orrenius S, Burkitt MJ (1993) Effects of glutathione and chelating agents on copper-mediated DNA oxidation: pro-oxidant and antioxidant properties of glutathione. Arch Biochem Biophys 304:102–109
Nanda R, Mishra PK, Das UK, Rout SB, Mohapatra PC, Panda A (2004) Evaluating role of oxidative stressing determining the pathogenesis of falciparum malaria induced acute renal failure. Indian J Clin Biochem 19:93–96
Sharma AK, Arora M, Gupta R, Makkad RK, Gupta HP (1998) Free radicals in acute renal failure due to falciparum malaria. Indian J Nephrol 8:101–103
Scheibmeir HD, Christensen K, Whitaker SH, Jegaethesan J, Clancy R, Pierce JD (2005) A review of free radicals and antioxidants for critical care nurses. Intensive Crit Care Nurs 21:24–28
Zwolińska D, Grzeszczak W, Kiliś-Pstrusińska K, Szprynger K, Szczepańska M (2004) Lipid peroxidation and antioxidant enzymes in children with chronic renal failure. Pediatr Nephrol 19:825–948
Zwołińska D, Grzeszczak W, Szczepańska M, Kiliś-Pstrusińska K, Szprynger K (2006) Lipid peroxidation and antioxidant enzymes in children on maintenance dialysis. Pediatr Nephrol 21:705–710
Halliwell B, Gutteridge JMC (1999) Free radicals, other reactive species, and disease. In: Halliwell B, Gutteridge JMC (eds) Free radicals in biology and medicine. 3rd edn. Oxford University Press, Oxford, pp 617–783
Du XH, Yang CL (1994) Mechanism of gentamicin nephrotoxicity in rats and the protective effect of zinc-induced metallothionein synthesis. Nephrol Dial Transplant 9(Suppl 4):135–140
Ogawa T, Mimura Y (1999) Antioxidant effect of zinc on acute renal failure induced by ischaemia-reperfusion injury in rats. Am J Nephrol 19:609–614
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial Support: The study was supported by Departmental Research Grant of Pediatrics, Institute of Medical Sciences, Banaras Hindu University, Varanasi-221005, India.
Rights and permissions
About this article
Cite this article
Mishra, O.P., Pooniya, V., Ali, Z. et al. Antioxidant status of children with acute renal failure. Pediatr Nephrol 23, 2047–2051 (2008). https://doi.org/10.1007/s00467-008-0875-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-008-0875-1