Abstract
Increased lipid peroxidation (LP) has been observed in dialysis patients and in predialysis adults with advanced chronic renal failure (CRF). The aim of this study was to investigate whether predialysis CRF children have increased LP in plasma and red blood cells (RBC) and to evaluate the activity of the antioxidant enzymes [superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px)] in RBC. Concentrations of selenium (Se), copper (Cu), and zinc (Zn)—cofactors of these enzymes—were determined both in erythrocytes and in plasma. LP was monitored by plasma and erythrocyte malonyldialdehyde (MDA) and by plasma organic hydroperoxide (OHP) concentrations. Forty-six predialysis children, aged 5–18 years, divided into two groups according to their serum creatinine levels [group I (n=14, mean serum creatinine 421.61±141.08 μmol/l), group II (n=32, mean serum creatinine 174.94±45.50 μmol/l)] and 27 age-matched healthy subjects were enrolled in the study. Significantly higher concentrations of plasma and erythrocyte MDA and plasma OHP, significantly lower activities of GSH-Px and CAT, and significantly lower concentrations of erythrocyte Se, Cu, and Zn and plasma Se and Cu were found in both groups of renal patients compared with controls. The SOD activity was reduced in both groups of CRF children. In group I the activity of SOD and GSH-Px was significantly lower than in group II. In summary, there is increased LP in plasma and RBC in children with predialysis CRF, even those patients with moderate renal insufficiency. The activity of the enzymatic antioxidant defense system is reduced in the RBC of predialysis patients. The antioxidant capacity is related to the severity of renal failure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite ongoing investigation of the pathogenesis of chronic renal failure (CRF), much remains unexplained. To date, the mechanism by which certain disturbances and their clinical symptoms arise is not understood. Several studies have indicated the role of reactive oxygen species (ROS) in the development of symptoms associated with CRF, such as atherosclerosis and related heart vessel complications, amyloidosis, and anemia [1, 2]. Studies of disorders of oxidative metabolism in CRF began in 1975 [3]. Yawata and Jacob [3] demonstrated that in red blood cells (RBC) from patients on hemodialysis (HD) the activity of the pentose phosphate shunt is diminished. NADPH production is also reduced. This results in a decrease in reduced glutathione, which acts as a free radical scavenger, reducing superoxide anions [4].
Numerous studies have since demonstrated a significantly increased concentration of malonyldialdehyde (MDA), an intermediate product of the oxidation of polyunsaturated fatty acids, in plasma, RBC, platelets, and peripheral blood mononuclear cells of patients on HD and continuous ambulatory peritoneal dialysis (CAPD) [5, 6, 7]. There are few data of augmented lipid peroxidation (LP) in adult patients with CRF on conservative therapy [8, 9, 10, 11]. The status of the antioxidant system in patients on chronic dialysis and in predialysis CRF adults is controversial [12, 13].
There are few reports of the antioxidant system in children with CRF [14, 15, 16], particularly those treated conservatively. To the best of our knowledge there is only one report, published in German, of serum MDA levels in predialysis children with various degrees of renal functional impairment [17]. There are no data on the influence of the stage of CRF on LP and antioxidant enzyme activities in RBC.
Following these observations, we attempted to verify whether predialysis CRF children have increased LP in plasma and RBC and to evaluate the activity of antioxidant enzymes [superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px)] in RBC. Concentrations of selenium (Se), copper (Cu), and zinc (Zn)—cofactors of these enzymes—were determined both in erythrocytes and in plasma. LP was monitored not only by plasma and erythrocyte MDA, but also by plasma organic hydroperoxide (OHP) concentrations.
Patients and methods
Forty-six children with predialysis CRF participated in the study. The patients were divided into two groups, according to their plasma creatinine levels [group I-14 children, plasma creatinine ≥265.3 μmol/l (3.0 mg/dl) (advanced CRF), group II-32 patients, plasma creatinine <265.3 μmol/l (moderate CRF)]. The control group consisted of 27 healthy age-matched children. None of the patients and controls received drugs with potential oxidant properties. Patients with current infections, vasculitides, hepatic or respiratory disease were not included. The clinical and demographic details of the study subjects are given in Table 1. The causes of CRF in group I were chronic pyelonephritis (n=5), reflux nephropathy (n=2), kidney hypoplasia (n=2), polycystic kidney disease (n=2), posterior urethral valve (n=1), chronic glomerulonephritis (n=1), and congenital nephrotic syndrome (n=1). The causes in group II were: reflux nephropathy (n=8), chronic pyelonephritis (n=7), chronic glomerulonephritis (n=4), posterior urethral valve (n=4), renal hypoplasia (n=2), polycystic kidney disease (n=2), tubulopathy (n=1), outcome of acute renal failure (n=2), and unknown (n=2). Informed consent was obtained from the parents of all children.
Blood samples obtained after overnight fasting from the healthy subjects and CRF children were taken from the antecubital vein into heparinized tubes. RBC were separated from plasma by centrifugation, washed twice with buffered normal saline, and hemolyzed. Erythrocyte and plasma MDA concentrations were assayed using thiobarbituric acid according to Chkawa et al. [18]. The results were expressed in nanomoles per milliliter. The plasma OHP concentration was measured by ferrous oxidation xylenol orange assay in conjunction with triphenylphosphine according to Nourooz-Zadeh et al. [19] and expressed in micromoles per liter. GSH-Px activity was assayed according to Paglia and Valentine [20]. This method is based on following the decrease in absorbance of NADPH at 340 nm. GSH-Px oxidizes reduced GSH to oxidized GSH, which is then reduced by GSH reductase. In the last reaction, while NADPH is oxidized to NADP+, the absorbance of NADPH at 340 nm begins to decrease steadily. The GSH-Px activity was expressed in units per gram of hemoglobin (Hb). CAT activity was measured according to the method described by Aebi [21] based on the decomposition of H2O2 . The decrease in absorbance at 240 nm was measured at 20°C. The results were expressed in units per gram of Hb. SOD activity was assayed according to Misra and Fridovich [22], based on the ability of SOD to inhibit autoxidation of epinephrine at alkaline pH (pH 10.2), and expressed in units per milligram of Hb. Plasma and erythrocyte trace elements (Zn, Cu, Se) were determined using atomic absorption spectrophotometry. A Perkin Elmer 603 apparatus equipped with an HGA 500 graphite furnace (for plasma Se determination) and standard furnace (for determination of other parameters) was used. Data were expressed in micromoles per liter. Serum urea and creatinine were determined with standard techniques using an autoanalyzer (Technicon R.A.1000).
Statistical analysis
The data were expressed as mean±standard deviation (SD). The non-parametric Mann-Whitney and Wilcoxon tests were used to compare differences between the study groups, because of abnormal distribution of variables examined. P <0.05 was considered significant. Spearman’s rank correlation coefficient was used to analyze the correlations between various parameters.
Results
The erythrocyte MDA concentrations (E-MDA) in CRF children from both groups were significantly higher than in healthy subjects. The mean value of E-MDA in the group with moderate CRF was lower than in the group with a higher creatinine level, but the difference was not significant (Table 2). Similar to E-MDA, plasma MDA and plasma OHP levels were significantly elevated in both groups of CRF children compared with controls. There was a significant difference in plasma OHP concentrations in group I (1.40±0.17 μmol/l) and group II (1.09±0.13 μmol/l) (P<0.01).
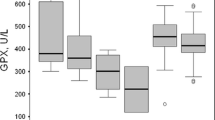
The activity of the antioxidant enzymes GSH-Px and CAT was significantly lower in erythrocytes of CRF children than in controls, irrespective of the plasma creatinine level (Table 3). Erythrocyte SOD activity was lower in both groups of CRF children than in controls, but only the difference between group I and controls was statistically significant. The lowest values of analyzed enzymes were observed in group I. The differences between the groups of CRF patients were statistically significant, with the exception of erythrocyte CAT activity.
The concentrations of erythrocyte Se, Cu, and Zn and plasma Se and Cu were significantly lower in children with CRF than in the control group (Table 4). The values of analyzed trace elements were similar in both CRF groups, irrespective of serum creatinine levels.
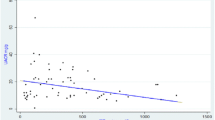
The E-MDA concentrations significantly correlated with plasma OHP concentration, serum creatinine, erythrocyte GSH-Px, CAT, and SOD activity (Table 5). Significant inverse correlations were found between plasma MDA activity and erythrocyte GSH-Px, CAT, and SOD activities and between plasma OHP concentration and erythrocyte GSH-Px, CAT, and SOD activity. In addition, there was a positive correlation between OHP and serum creatinine.
There were positive correlations between erythrocyte and plasma Se concentrations and GSH-Px activity, between erythrocyte and plasma Zn concentrations and erythrocyte SOD activity, and between erythrocyte and plasma Cu concentrations and erythrocyte SOD activity.
Discussion
Augmented LP in RBC membranes of predialysis patients was first reported by Taccone-Gallucci et al. [11]. Increased levels of erythrocyte MDA were found in adults with serum creatinine levels higher than 5 mg/dl, whereas the MDA content of RBC remained unchanged in patients with serum creatinine levels below 5 mg/dl. This is in agreement with the data reported subsequently by different authors regarding erythrocyte MDA in adults with advanced CRF [23] and patients with moderate renal insufficiency [24].
We found increased concentrations of RBC MDA compared with controls, not only in children with higher creatinine levels but also in children with plasma creatinine levels lower than 265.3 μmol/l (3 mg/dl). This suggests that in children, in contrast to adults, peroxidative damage of RBC membranes takes place much earlier, at the stage of moderate renal failure. A positive correlation between RBC MDA and plasma creatinine level indicates that susceptibility of the erythrocyte membrane to LP in children continuously increases with the progression of renal insufficiency. Young developing tissues are probably more sensitive to factors that cause the imbalance between antioxidants and pro-oxidants.
Similar to RBC MDA, our findings revealed increased plasma MDA and OHP concentrations in both groups of CRF children. Such elevated plasma MDA levels have been previously reported in CRF children by Otting and Hellmann [17], even in children with serum creatinine levels below 180 μmol/l . Our results are in accordance with observations of Mimic-Oka et al. [13] in adults with various degrees of renal failure, but in contrast to others, who observed increased plasma MDA only in advanced CRF [24]. Augmented LP in CRF patients can be explained by factors other than increased ROS generation. Positive correlations between concentrations of erythrocyte MDA, serum OHP, and plasma creatinine indicated that the process of activation of free radicals is highly influenced by renal insufficiency per se. However, other factors, such as decreased effectiveness of the intracellular and plasma protection system should also be taken into account [25, 26].
In this study activities of erythrocyte enzymes that scavenge superoxide radicals (SOD) and hydrogen peroxide (GSH-Px, CAT) were measured in predialysis children with different stage of CRF and in healthy age-matched subjects. These antioxidant enzymes are mainly involved in intracellular antioxidant defense. Several publications describing enzymes participating in free radical metabolism have yielded equivocal results. Significantly increased activity of erythrocyte SOD, CAT, and GSH-Px and plasma SOD and CAT was observed in HD, CAPD, and predialysis CRF adults [9, 10, 12, 13, 27, 28]. The authors explained this by adaptative mechanisms to oxidative stress. In contrast, some authors demonstrated reduction of antioxidant enzymes in plasma and RBC of CRF patients [29, 30, 31, 32, 33, 34, 35]. Our results confirm the latter observations. The activity of erythrocyte GSH-Px and erythrocyte CAT was significantly reduced even in children with moderate renal failure compared with controls. Erythrocyte SOD activity was also reduced in this group, but the difference was not significant. Decreased enzyme activity occurring during the early stage of CRF may be the result of several factors. These include consumption during free radical production, exposure to toxic trace elements and uremic toxins, and deficiency of some essential trace elements that act in the active centers of these enzymes together with other unknown factors. Van Den Branden et al. [36] showed that CAT gene expression decreases during development of renal failure in rats and is accompanied by decreased CAT activity. Human plasma GSH-Px is predominantly expressed in the kidney, and proximal tubular cells are the main source of its activity [37]. It can be speculated that low GSH-Px activity in CRF patients is a consequence of its impaired synthesis in damaged renal cells. SOD is a Cu- and Zn-dependent enzyme and its activity is related to plasma Zn and Cu levels [10]. The activity of GSH-Px in blood and tissues is related to the Se concentration [38]. Therefore, trace element deficiency, which occurs in CRF patients, results in decreased activity of these enzymes [10, 26, 38]. In our study the erythrocyte Se, Zn, and Cu content, and plasma Cu and Se was significantly lower in predialysis children than the control group.
In summary, increased LP in plasma and RBC occurs in children with predialysis CRF, even in the moderate stage of renal insufficiency. The activity of the enzymatic antioxidant defense system is reduced in RBC of predialysis patients. The antioxidant capacity is related to the severity of renal failure.
References
Marx ER, Wicckens DG, Griffin JFK, Kyle P, Curtis JR, Dormandy TL (1987) Oxygen free radicals linked to many diseases. Science 2:529–531
Vaziri ND, Dicus M, Ho ND, Boroujerdi-Rad L, Sindhu RK (2003) Oxidative stress and dysregulation of superoxide dismutase and NADPH oxidase in renal insufficiency. Kidney Int 63:179–185
Yawata Y, Jacob H (1975) Abnormal red cell metabolism causing hemolysis in uremia: nature of the defect and its persistence despite adequate hemodialysis. Blood 45:231–239
Griendling KK, Sorecsu D, Ushio-Fukai M (2000) NAD(P)H oxidase: role in cardiovascular biology and disease. Cir Res 86:494–501
Galli F, Ronco C (2000) Oxidant stress in hemodialysis. Nephron 84:1–5
Taccone-Gallucci M, Giardini O, Lubrano R, Mazarella V (1986) Red blood cell membrane lipid peroxidation in continuous ambulatory peritoneal dialysis patients. Am Clin Nephrol 6:92–95
Zwolińska D (1998) Lipid peroxidation and antioxidant enzymes in patients with chronic renal failure. Adv Clin Exp Med 7:87–92
Durak I, Akyol O, Basesme E, Canbolat O, Kavutcu M (1994) Reduced erythrocyte defence mechanisms against free radical toxicity in patients with chronic renal failure. Nephron 66:76–80
Mimic-Oka J, Simic T, Djukanovic L, Reljic Z, Davicevic Z (1999) Alteration in plasma antioxidant capacity in various degrees of chronic renal failure. Clin Nephrol 51:233–241
Richard MJ, Arnaud J, Jurkovitz C, Hachache T, Meftahi H, Laporte F, Foret M, Favier A, Cordonnier D (1991) Trace elements and lipid peroxidation abnormalities in patients with chronic renal failure. Nephron 57:10–15
Taccone-Gallucci M, Giardini O, Lubrano R, Bandino D, Mazzarella V, Mannarino O, Meloni C, Moroseti M, Elli M, Tozzo C, Strolighi L, Casciani CU (1987) Red blood cell lipid peroxidation in predialysis chronic renal failure. Clin Nephrol 27:238–241
Ceballos-Picot I, Witko-Sarsat V, Merad-Boudia M, Nguyen AT, Thavenin M, Jaudon MC, Zingraff J, Verger C, Jungers P, Descamps-Latscha B (1996) Glutathione antioxidant system as a marker of oxidative stress in chronic renal failure. Free Radic Biol Med 21:845–853
Mimic-Oka J, Simic T, Ekmescic V, Dragicevic P (1995) Erythrocyte glutathione peroxidase and superoxide dismutase activities in different stages of chronic renal failure. Clin Nephrol 44:44–48
Asoyama K, Shiki Y, Ito H, Hasegowa O, Miyao A, Hayashibe H, Dobashi K, Kato K (1990) Antioxidant enzymes and lipoperoxide in blood in uremic children and adolescents. Free Radic Biol Med 9:105–109
Sommerburg O, Grune T, Ehrich JH, Siemens WG (2002) Adaptation of glutation-peroxidase activity to oxidative stress occurs in children but not in adult patients with end-stage renal failure undergoing hemodialysis. Clin Nephrol 58 [Suppl 1]:S31–S36
Turi S, Varga I, Matkovics B, Dobos E (1991) Erythrocyte defence mechanisms against free oxygen radicals in haemodialysed uraemic children. Pediatr Nephrol 5:179–183
Otting VU, Hellmann C (1990) Malondialdehydkonzentration (MDA) im Serum chronisch niereninsuffizienter, chronisch hämodialysierter und nierentransplantierter Kinder. Z Urol Nephrol 83:141–148
Chkawa H, Ohski N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric reaction. Anal Biochem 95:351–358
Nourooz-Zadeh J, Tajaddini Sarmadi J, Wolff S (1994) Measurement of plasma hydroperioxide concentration by ferrous oxidation xylenol orange assay in conjunction with triphenylphosphine. Anal Biochem 220:403–409
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterisation of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–168
Aebi H (1974) Catalase, vol 2. In: Bergmeyer HU (eds) Methods of enzymatic analysis. Weinheim Academic Press, New York, pp 673–678
Misra HP, Fridovich I (1972) The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Martin-Mateo MC, Sanchez-Portugal M, Iglesias S, Paula A de, Bustamante J (1999) Oxidative stress in chronic renal failure. Ren Fail 21:155–167
Trznadel K, Pawlicki L, Kędziora J, Luciak M, Błaszczyk J, Buczyński A (1989) Aktywność dysmutazy ponadtlenkowej w erytrocytach oraz stężenie malonylodialdehydu w erytrocytach i osoczu w różnych okresach niewydolności nerek. Pol Arch Med Wewn 80:261–266
Annuk M, Fellström B, Åkerblom O, Zilmer K, Vihalemm T, Zilmer M (2001) Oxidative stress markers in pre-uremic patients. Clin Nephrol 56:308–314
Zachara BA, Adamowicz A, Trafikowska U, Trafikowska A, Manitius J, Nartowicz E (2001) Selenium and glutathione levels and glutathione peroxidases and some other antioxidant parameters in blood of patients with chronic renal failure. J Trace Elem Med Biol 15:161–166
Dasgupta A, Hussain S, Ahmad S (1992) Increased lipid peroxidation in patients on maintenance hemodialysis. Nephron 60:56–59
Lucchi L, Banni S, Capelli B, Medici G, Melis MP, Tomasi A, Vannini V, Lusvarghi E (1993) Conjugated diene fatty acids in patients with chronic renal failure: evidence of increased lipid peroxidation? Nephron 65:401–409
Dursun E, Ozben T, Suleymanlar G, Dursun B, Yakupoglu G (2002) Effect of hemodialysis on the oxidative stress and antioxidants. Clin Chem Lab Med 40:1009–1013
Konukoglu D, Ercan M, Ayaz M, Onen S (2001) Plasma and erythrocytes antioxidant status and trace element levels in proteinuric patients with moderate glomerular function. J Trace Elem Med Biol 15:119–122
Nagase S, Aoyagi K, Hirayama A, Gotoh M, Ueda A, Tomida C, Kamezaki T, Nagai Y, Kikuchi H, Koyama A (1997) Favorable effect of hemodialysis on decreased serum antioxidant activity in hemodialysis patients demonstrated by electron spin resonance. J Am Soc Nephrol 8:1157–1163
Ozden M, Maral H, Akaydin D, Cetinalp P, Kalender B (2002) Erythrocyte glutathione peroxidase activity, plasma malonyldialdehyde and erythrocyte glutathione levels in hemodialysis and CAPD patients. Clin Biochem 35:269–273
Paul JL, Sall ND, Soni T, Poignet JL, Lindenbaum A, Man NK, Moatti N, Raichvarg D (1993) Lipid peroxidation abnormalities in hemodialyzed patients. Nephron 64:106–109
Vanella A, Geremia E, Pinturo R, Triolo P, Liuzzo G, Triolo C, Custorella A, Condorelli G, Giglio A (1983) Superoxide dismutase activity and reduced glutathione content in erythrocytes of uremic patients on chronic dialysis. Acta Haematol 70:312–315
Zachara BA, Adamowicz A, Trafikowska U, Trafikowska A, Manitius J, Nartowicz E (2001) Selenium and glutathione levels and glutathione peroxidase activities in blood component of uremic patients on hemodialysis supplemented with selenium and treated with erythropoietin. J Trace Elem Med Biol 15:201–208
Van Den Branden C, Ceyssens B, De Craemer D, De Bleser P, Hellemans K, Geerts A, Verbeelen D (2000) Antioxidant enzyme gene expression in rats with remnant kidney induced chronic renal failure. Exp Nephrol 8:91–96
Avissar N, Ornt DB, Yagil Y, Horovitz S, Watkins RH, Kerl EA, Takahashi K, Palmer IS, Cohen HJ (1994) Human kidney proximal tubules are the main source of plasma glutathione peroxidase. Am J Physiol 266: C367–C375
Adamowicz A, Trafikowska U, Trafikowska A, Zachara BA, Manitius J (2002) Effect of erythropoietin on selected antioxidant parameters in blood of uremic patients on long-term hemodialysis. Med Sci Monit 8:CR202–205
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zwolińska, D., Grzeszczak, W., Kiliś-Pstrusińska, K. et al. Lipid peroxidation and antioxidant enzymes in children with chronic renal failure. Pediatr Nephrol 19, 888–892 (2004). https://doi.org/10.1007/s00467-004-1512-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-004-1512-2