Abstract
Background
Endoscopic sleeve gastroplasty (ESG) is a minimally invasive, effective, and safe technique for weight loss intervention. Since a relatively small number of cases were present in previous studies, this study aimed to elucidate the efficacy and safety of ESG.
Methods
Relevant publications were identified through searching PubMed, EMBASE, Cochrane, and Web of Science before March 1, 2019. The percentage of total body weight loss (%TBWL), percentage of excess weight loss (%EWL), and the adverse event rate in each follow-up session were extracted, pooled, and analyzed. Forest plots were graphed based on random effects models.
Results
A total of 1542 patients from nine studies were eligible for analysis. The pooled results of %TBWL at 1, 3, 6, and 12 months were 8.78% (p = 0.000), 11.85% (p = 0.000), 14.47% (p = 0.024), and 16.09% (p = 0.063), respectively. The pooled results of %EWL at 1, 3, 6, and 12 months were 31.16% (p = 0.000), 43.61% (p = 0.000), 53.14% (p = 0.000), and 59.08% (p = 0.015), respectively. Finally, the pooled rate of mild adverse events was 72% (p < 0.01), and the pooled estimate of severe adverse events was only 1% (p = 0.08).
Conclusion
Although the conventional surgical sleeve gastrectomy is the gold standard for bariatric surgery, ESG could be a promising minimally invasive alternative for treating obesity with satisfactory efficacy and low risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Obesity is an accelerating pandemic across the world, increasing morbidity and mortality [1]. Obesity has caused a tremendously negative impact on multiple levels of society, such as healthcare costs, social resources, and the potential burden on social economic development [2]. Although non-surgical approaches including lifestyle modifications and drugs could provide modest weight loss, conventional methods are usually associated with a high rate of weight regain [3]. Currently, bariatric–metabolic surgery is the only durable and efficacious option for treating obesity [4]. However, not all eligible patients are able to undergo bariatric surgery because of costs, risks, limited access, and patient characteristics [5]. Thus, effective and safe approaches are imperative to bridge the current obesity treatment gap.
Endoscopic bariatric therapy (EBT) is presenting advantages of being less invasive, having a lower cost, and having higher patient acceptability. To date, two of the most performed approaches of EBT are intragastric balloons (IGB) and the duodenojejunal liner [6, 7]. However, due to the intrinsic technical limitation, the implanted device is not allowed to remain in situ for more than 12 months in both methods [8, 9]. Moreover, risks regarding device-related adverse events are also a necessary concern and includes migration, ulcers, and sustaining abdominal pain [10].
Endoscopic sleeve gastroplasty (ESG) is a minimally invasive, incisionless procedure for bariatric treatment and was first described in 2013 [11]. The principle of ESG is to reduce gastric capacity by creating a restrictive sleeve via an endoluminal suturing system that places full-thickness sutures along the corpus of the stomach. Although this procedure has been manifested to be reproducible and effective, the widespread clinical adoption is still hindered by the indistinct recognition of its efficacy and safety. The aim of this meta-analysis is to evaluate the efficacy and safety of ESG as an alternative EBT for obesity treatment.
Methods
Study search and selection criteria
A comprehensive literature search was performed through PubMed, EMBASE, Cochrane Library, and Web of Science up to February 1, 2019. The following key terms were used: “endoscopic sleeve gastroplasty,” “ESG,” “endoscopic bariatric therapy,” and “EBT”. An additional search was performed among the references of the included studies to find potentially eligible studies.
According to the predefined criteria, two investigators (PW.L. and B.M.) independently screened all the relevant studies and reviewed the full text of the included studies. If there was a disagreement, it was discussed and solved by a consensus with a third reviewer (S.L.G.). Inclusion criteria were as follows: (1) no limitations on study design, including prospective or retrospective observational cohorts, case–control studies, and randomized controlled trials (RCT); and (2) studies reporting the therapeutic outcomes of ESG, such as percentage of total body weight loss (%TBWL), percentage of excess weight loss (%EWL), the number of follow-up patients and adverse outcomes, such as abdominal pain, nausea, perigastric fluid collection, and gastrointestinal bleeding. Exclusion criteria were as follows: (1) case reports; (2) literature on ESG as an introduction to the technology; (3) experiments on animals; (4) reviews, comments, or letters; (5) studies published in other languages besides English; and (6) studies with unavailable full text. In addition, for multiple studies that were published containing the overlapping patient based on the same outcomes, the most informative study was included. If multiple studies reported different outcomes based on the overlapping patient, the results were combined for a more comprehensive analysis. PW.L and B.M. evaluated the quality of the included studies using the Newcastle–Ottawa scale [12].
Data collection and outcome measures
A formalized table was developed to extract relevant data from the included studies. The information included the authors, year of publication, study duration, country, study design, study setting, total number of patients, patient demographics (age and sex), operative time, follow-up period, %TBWL, %EWL, and adverse outcomes rate.
In accordance with the Preservation and Incorporation of Valuable endoscopic Innovations (PIVI) guidelines [13], clinical success was defined as a %TWBL of at least of 15%. The American Society for Gastrointestinal Endoscopy (ASGE) and the American Society for Metabolic and Bariatric Surgery (ASMBS) recommend efficacy as more than 25% EWL at 12 months and the safety threshold as less than 5% risk of major complications [6].
Statistical analysis
In our meta-analysis, there were two types of data; one was continuous for evaluating the efficacy of ESG (%TBWL and %EWL), and the other was a single proportion rate for evaluating the safety of ESG (adverse event). To get the most appropriate statistical results, the efficacy of ESG was analyzed by using Stata software, version 12.0 (2011; Stata Corp., College Station, TX, USA), and the safety of ESG was calculated using R 3.5.1 software (version 3.5.1 https://www.r-project.org/). All pooled results were calculated with a random effects model because it provided more conservative estimates. All statistical values are reported with 95% confidence intervals (CI).
IRB approval
This meta-analysis is not related with any patient privacy or related information, so there is no need for IRB approval.
Results
Search results
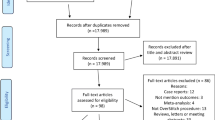
A total of 558 studies were found by search of the PubMed, EMBASE, Cochrane Library, and Web of Science databases. The study flow chart is shown in Fig. 1. Two hundred and ninety duplicate studies were excluded, and an additional 206 studies were removed for reasons related to the title, abstract, and language. Sixty-two records were eligible for full text review. Among these full text-reviewed articles, Lopez-Nava et al. [14,15,16,17,18] had five articles included in the search. However, two articles [14, 15] had overlapping patients and clinical outcomes with subsequent research by Lopez-Nava et al. [16,17,18], so those two articles were excluded. Although the remaining three studies also had overlapping patients, some important clinical outcomes did not overlap, and those outcomes were extracted for pooled analysis. A similar reason also applied to the studies from Abu Dayyeh et al. [19] and Sharaiha et al. [20] because Lopez-Nava et al. [18] made a pooled analysis from their studies. The study from Novikov et al. [21] also contained the same patients as Sharaiha et al. [20], but there were not available data for analysis, so it was not included either. Hence, nine studies were finally included and only the non-overlapping clinical results were extracted for analysis [16,17,18,19,20, 22,23,24,25]. No RCTs were found in the search. The baseline information of included studies is shown in Table 1, and the average quality of all included studies was moderate.
Studies characteristics
As shown in Table 1, most of the research found were single-center prospective studies [16, 17, 19, 20, 22, 23, 25]. The mean age of all the included studies was less than 50 years old, and the mean BMI was more than 30 kg/m2. All but one study [22] were from the USA and Spain [16,17,18,19,20, 23,24,25], the core facilities where ESG was developed. Only Alqahtani et al. [22] presented a large number of 1000 patients, whereas the other studies showed a relatively limited quantity of cases.
Assessment of efficacy
A total of 1542 patients underwent ESG for obesity treatment. As shown in Fig. 2, the pooled results of %TBWL at 1, 3, 6, and 12 months were 8.78% (95% CI 8.12–9.44%, I2 = 88.5%, p = 0.000, n = 1102), 11.85% (95% CI 10.55–13.15%, I2 = 96.5%, p = 0.000, n = 826), 14.47% (95% CI 13.6–15.34%, I2 = 64.3%, p = 0.024, n = 735), and 16.09% (95% CI 14.24–17.94%, I2 = 63.9%, p = 0.063, n = 340), respectively. The heterogeneity of %TBWL at 1, 3, and 6 months was significant; however, the heterogeneity of %TBWL at 12 months was non-significant due to a p value > 0.05. The pooled results of %EWL are presented in Fig. 3, and %EWL at 1, 3, 6, and 12 months were 31.16% (95% CI 23.81–38.52%, I2 = 97.9%, p = 0.000, n = 1050), 43.61% (95% CI 36.56–50.65%, I2 = 95.1%, p = 0.000, n = 826), 53.14% (95% CI 46.31–59.98%, I2 = 89.1%, p = 0.000, n = 579), and 59.08% (48.74–69.42%, I2 = 76.2%, p = 0.015, n = 290), respectively. The heterogeneities of %EWL at 1, 3, 6, and 12 months were all significant.
Assessment of safety
The most common mild adverse event was abdominal pain and nausea, and the pooled estimate is shown in Fig. 4A. The pooled rate was 72% (95% CI 49–90%, I2 = 97%, p < 0.01, n = 1299) with a significant heterogeneity. The severe adverse events were different across the included studies, such as perigastric inflammatory fluid collection, pulmonary embolism, pneumoperitoneum, pneumothorax, bleeding, and postprocedure needing blood transfusion. The pooled estimate was only 1% (95% CI 0–3%, I2 = 53%, p = 0.08, n = 1542) with non-significant heterogeneity (Fig. 4B).
Discussion
The prevalence of obesity is growing. However, the rate of obesity should not be increasing for a rising population with conventional therapies (including diet, medications, and increased exercise) [3]. Although bariatric surgery could offer a relevant and satisfying option for weight loss, only 1% of eligible patients are able to get access to it due to self-intention, cost, risk, and medical resources [5, 16]. EBT with intragastric balloons and the duodenojejunal liner remedy some of the disadvantages of bariatric surgery, yet there is still a certain shortage of it.
In this study, ESG, an innovative EBT, is found to achieve and maintain desired weight loss by creating structural and physiologic change to the gastric body. After ESG, the tendency of %TBWL gradually increased, changing from 8.78% at 1 month to 16.09% at 12 months. Additionally, the tendency of %EWL also gradually increased, changing from 31.16% at 1 month to 59.08% at 12 months. In fact, the %TBWL was 18.6% (95% CI 15.7–21.5%) at 24 months in the study conducted by Lopez-Nava et al. [18], and the %EWL was 60.4 ± 31.1% at 24 months [17]. Although some included studies did not present the 24-month follow-up result [22,23,24,25], it was demonstrated that %TBWL ≥ 10% at 6 months was a highly predictive factor with future weight loss and long-term weigh maintenance for up to 2 years postprocedure [14]. The pooled %TBWL at 6 months was 14.47% (95% CI 13.6–15.34%), which could be speculated as possibly sustaining weight loss at 24 months. However, there was great heterogeneity among the included studies, and we thought that the great difference in number of patients and study type in each study might contribute to the heterogeneity.
Safety of a new endoscopic technique is also an important concern. Most of the patients could be discharged at 24–48 h after the ESG procedure and some even on the same day as the procedure [16, 17, 24]. None of the included studies reported any severe intraprocedural adverse events. A large proportion of mild adverse events were abdominal pain and nausea, and the pooled results reached 72%. However, most of the symptoms could be relieved by conservative treatment [20, 22]. Although some studies reported a few postprocedure bleeding cases [18, 22,23,24], all the cases could be cured with medications and blood transfusion, and no mortality occurred. A high rate of severe adverse events included perigastric leak [18, 22,23,24,25], but all the leaks were able to be treated with percutaneous drainage or antibiotics. None of the patients needed secondary surgical intervention.
According to PIVI guidelines [13], clinical success was defined as a %TWBL of at least of 15%. In addition, ASGE and ASMBS recommended efficacy as more than 25% EWL at 12 months, and the safety threshold was less than 5% risk of any major complication [6]. Both %TWBL and %EWL were able to meet the requirement after undergoing ESG as mentioned above. Meanwhile, the severe adverse event rate was only 1% which showed that ESG was an efficient and safe procedure for treating obesity.
To elucidate the efficacy and safety between different approaches for weight loss, Novikov et al. [21] made a comparison among ESG, laparoscopic sleeve gastrectomy (LSG), and laparoscopic band (LAGB). LSG achieved the greatest %TBWL compared to LAGB and ESG (29.28% vs 13.3% vs 17.5%, respectively, p < 0.001). However, ESG had the lowest rate of morbidity compared to LSG and LAGB (2.2% vs 9.17% vs 8.97%, respectively, p = 0.01). Moreover, the length of stay was also shortest in the ESG group. A case-matched study by Fayad et al. [23] validated that LSG could achieve a higher %TBWL compared to ESG (23.6 ± 7.6% vs 17.1 ± 6.5%, p < 0.01), whereas the ESG group not only had lower rates of adverse events compared with the LSG group (5.2% vs 16.9%, p < 0.05) but also a lower rate of gastroesophageal reflux disease (1.9% vs 14.5%, p < 0.05). In addition, ESG could achieve comparable %TBWL for patients with a BMI < 40 kg/m2 who are mostly ineligible for LSG [21, 23].
In fact, the anatomic change of the stomach is reversible after ESG because the fundus of the stomach and the vessel along the greater gastric curvature are still intact. Therefore, it is feasible to perform a reversal or redo of ESG with the indication of severe abdominal pain and weight regain [22]. Moreover, it is even possible for patients who had poor weight loss defined as %TBWL < 5% to convert to conventional sleeve gastrectomy [22]. Theoretically, it is also applicable for patients who fail ESG to convert to Roux-en-Y gastric bypass surgery, for the lesser curvature and the cardia of the stomach are anatomically preserved after ESG [18]. On the other hand, postprocedure medication usage is also different between ESG and LSG. Only omeprazole is needed for a few weeks after ESG. However, lifelong vitamin supplementation is needed after LSG [23]. Based on all these features, it manifests that ESG is a more flexible technique to provide an effective alternative to the patients who are ineligible or do not wish to perform surgery. However, it is not intended to replace conventional bariatric surgery [18].
Until now, there is still no direct comparison between ESG and IGB in terms of efficacy. Some systematic reviews and meta-analyses revealed that the %TBWL and %EWL were 12% and 25.4% at 6 months, respectively [9, 26,27,28]. Furthermore, IGB could cause a high rate of GERD (18.3%) and intolerance (10%) requiring removal of the device [27, 29]. In addition, IGB requires two or more endoscopy sessions for placement, adjustment, and removal, whereas ESG only requires a single session to perform the procedure [18]. Additionally, more serious adverse events could be caused by IGB such as gastric ulcers, balloon deflation, device migration, and gastric perforation [24]. In fact, Sartoretto et al. [24] have reported that ESG could treat the patients who failed previous IGB. More studies are needed to verify the sequence of EBTs in the personalized medicine era.
This is the first meta-analysis comprehensively analyzing the available data on the ESG for the treatment of patients suffering from obesity, and the results suggest that ESG is both safe and effective. It seems that ESG has more momentum for promising widespread usage. However, several limitations of this meta-analysis should be considered. First, both the number of the studies and the number of cases in the studies were relatively small, which might make the results less reliable. Second, the heterogeneity of some pooled estimates is significant, and we speculated that it perhaps came from the different sizes of the included studies. Therefore, further analyses should be performed if individual patient data are available. Third, no RCTs were available for this study. Most of the studies in this current analysis were single-arm observational studies which are potentially inherent with selection bias and information bias and are less convincing. However, these limitations are inherent limitations to many new and emerging interventional techniques, and we tried our best to avoid any confounding effects from duplicate published data. Only the most informative studies in each follow-up session were included for analysis.
In conclusion, ESG is a minimally invasive, effective, and safe technique for weight loss intervention. Future studies are needed not only to compare ESG with other EBTs in efficacy and safety but also to compare combination effects with other EBTs.
References
Bray GA, Kim KK, Wilding JPH, World Obesity F (2017) Obesity: a chronic relapsing progressive disease process: a position statement of the World Obesity Federation. Obes Rev 18:715–723
Hiatt WR, Thomas A, Goldfine AB (2012) What cost weight loss? Circulation 125:1171–1177
Middleton KM, Patidar SM, Perri MG (2012) The impact of extended care on the long-term maintenance of weight loss: a systematic review and meta-analysis. Obes Rev 13:509–517
Schauer PR, Bhatt DL, Kirwan JP, Wolski K, Brethauer SA, Navaneethan SD, Aminian A, Pothier CE, Kim ES, Nissen SE, Kashyap SR (2014) Bariatric surgery versus intensive medical therapy for diabetes: 3-year outcomes. N Engl J Med 370:2002–2013
Mechanick JI, Youdim A, Jones DB, Garvey WT, Hurley DL, McMahon MM, Heinberg LJ, Kushner R, Adams TD, Shikora S, Dixon JB, Brethauer S (2013) Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient–2013 update: cosponsored by American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic & Bariatric Surgery. Obesity 21(Suppl 1):S1–27
Abu Dayyeh BK, Kumar N, Edmundowicz SA, Jonnalagadda S, Larsen M, Sullivan S, Thompson CC, Banerjee S (2015) ASGE Bariatric Endoscopy Task Force systematic review and meta-analysis assessing the ASGE PIVI thresholds for adopting endoscopic bariatric therapies. Gastrointest Endosc 82:425–438.e425
Hill C, Khashab MA, Kalloo AN, Kumbhari V (2018) Endoluminal weight loss and metabolic therapies: current and future techniques. Ann N Y Acad Sci 1411:36–52
Kumbhari V, Hill C, Sullivan S (2017) Bariatric endoscopy: state-of-the-art. Curr Opin Gastroenterol 33:358–365
Yorke E, Switzer NJ, Reso A, Shi X, de Gara C, Birch D, Gill R, Karmali S (2016) Intragastric balloon for management of severe obesity: a systematic review. Obes Surg 26:2248–2254
Rohde U, Hedback N, Gluud LL, Vilsboll T, Knop FK (2016) Effect of the EndoBarrier gastrointestinal Liner ON obesity and type 2 diabetes: a systematic review and meta-analysis. Diabetes Obes Metab 18:300–305
Abu Dayyeh BK, Rajan E, Gostout CJ (2013) Endoscopic sleeve gastroplasty: a potential endoscopic alternative to surgical sleeve gastrectomy for treatment of obesity. Gastrointest Endosc 78:530–535
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605
Ginsberg GG, Chand B, Cote GA, Dallal RM, Edmundowicz SA, Nguyen NT, Pryor A, Thompson CC (2011) A pathway to endoscopic bariatric therapies. Gastrointest Endosc 74:943–953
Lopez-Nava G, Galvao MP, Bautista-Castano I, Jimenez-Banos A, Fernandez-Corbelle JP (2015) Endoscopic sleeve gastroplasty: how I do it? Obes Surg 25:1534–1538
Lopez-Nava G, Galvao MP, da Bautista-Castano I, Jimenez A, De Grado T, Fernandez-Corbelle JP (2015) Endoscopic sleeve gastroplasty for the treatment of obesity. Endoscopy 47:449–452
Lopez-Nava Breviere G, Bautista-Castano I, Fernandez-Corbelle JP, Trell M (2016) Endoscopic sleeve gastroplasty (the Apollo method): a new approach to obesity management. Rev Esp Enferm Dig 108:201–206
Lopez-Nava G, Galvao MP, Bautista-Castano I, Fernandez-Corbelle JP, Trell M, Lopez N (2017) Endoscopic sleeve gastroplasty for obesity treatment: two years of experience. Arq Bras Cir Dig 30:18–20
Lopez-Nava G, Sharaiha RZ, Vargas EJ, Bazerbachi F, Manoel GN, Bautista-Castano I, Acosta A, Topazian MD, Mundi MS, Kumta N, Kahaleh M, Herr AM, Shukla A, Aronne L, Gostout CJ, Abu Dayyeh BK (2017) Endoscopic sleeve gastroplasty for obesity: a multicenter study of 248 patients with 24 months follow-up. Obes Surg 27:2649–2655
Abu Dayyeh BK, Acosta A, Camilleri M, Mundi MS, Rajan E, Topazian MD, Gostout CJ (2017) Endoscopic sleeve gastroplasty alters gastric physiology and induces loss of body weight in obese individuals. Clin Gastroenterol Hepatol 15(37–43):e31
Sharaiha RZ, Kumta NA, Saumoy M, Desai AP, Sarkisian AM, Benevenuto A, Tyberg A, Kumar R, Igel L, Verna EC, Schwartz R, Frissora C, Shukla A, Aronne LJ, Kahaleh M (2017) Endoscopic sleeve gastroplasty significantly reduces body mass index and metabolic complications in obese patients. Clin Gastroenterol Hepatol 15:504–510
Novikov AA, Afaneh C, Saumoy M, Parra V, Shukla A, Dakin GF, Pomp A, Dawod E, Shah S, Aronne LJ, Sharaiha RZ (2017) Endoscopic sleeve gastroplasty, laparoscopic sleeve gastrectomy, and laparoscopic band for weight loss: how do they compare? J Gastrointest Surg 22:267–273
Alqahtani A, Al-Darwish A, Mahmoud AE, Alqahtani YA, Elahmedi M (2018) Short-term outcomes of endoscopic sleeve gastroplasty in 1000 consecutive patients. Gastrointest Endosc 89(6):1132–1138
Fayad L, Adam A, Schweitzer M, Cheskin LJ, Ajayi T, Dunlap M, Badurdeen DS, Hill C, Paranji N, Lalezari S, Kalloo AN, Khashab MA, Kumbhari V (2018) Endoscopic sleeve gastroplasty versus laparoscopic sleeve gastrectomy: a case-matched study. Gastrointest Endosc 89(4):782–788
Sartoretto A, Sui Z, Hill C, Dunlap M, Rivera AR, Khashab MA, Kalloo AN, Fayad L, Cheskin LJ, Marinos G, Wilson E, Kumbhari V (2018) Endoscopic sleeve gastroplasty (ESG) is a reproducible and effective endoscopic bariatric therapy suitable for widespread clinical adoption: a large, international multicenter study. Obes Surg 28:1812–1821
Saumoy M, Schneider Y, Zhou XK, Shukla A, Kahaleh M, Aronne L, Sharaiha RZ (2018) A single-operator learning curve analysis for the endoscopic sleeve gastroplasty. Gastrointest Endosc 87:442–447
Imaz I, Martinez-Cervell C, Garcia-Alvarez EE, Sendra-Gutierrez JM, Gonzalez-Enriquez J (2008) Safety and effectiveness of the intragastric balloon for obesity: a meta-analysis. Obes Surg 18:841–846
Gaur S, Levy S, Mathus-Vliegen L, Chuttani R (2015) Balancing risk and reward: a critical review of the intragastric balloon for weight loss. Gastrointest Endosc 81:1330–1336
Moura D, Oliveira J, De Moura EG, Bernardo W, Galvao Neto M, Campos J, Popov VB, Thompson C (2016) Effectiveness of intragastric balloon for obesity: a systematic review and meta-analysis based on randomized control trials. Surg Obes Relat Dis 12:420–429
Kim SH, Chun HJ, Choi HS, Kim ES, Keum B, Jeen YT (2016) Current status of intragastric balloon for obesity treatment. World J Gastroenterol 22:5495–5504
Acknowledgements
Special thanks to Dr. Bin Ma for his selfless teaching and helping Dr. Peiwen Li to conduct this study.
Funding
This study was supported by the Natural Science Foundation of Liaoning Province (Grant No. 2015020561), the Fund for Scientific Research of The First Hospital of China Medical University (Grant No. fsfh1514), and Wu Jieping Medical Foundation (Grant No. 320.6750.18293).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Dr. Peiwen Li, Dr. Bin Ma, Dr. Shulei Gong, Dr. Xinyu Zhang, and Dr. Wenya Li have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, P., Ma, B., Gong, S. et al. Efficacy and safety of endoscopic sleeve gastroplasty for obesity patients: a meta-analysis. Surg Endosc 34, 1253–1260 (2020). https://doi.org/10.1007/s00464-019-06889-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-019-06889-6