Abstract
Background
Common video systems for laparoscopy provide the surgeon a two-dimensional image (2D), where information on spatial depth can be derived only from secondary spatial depth cues and experience. Although the advantage of stereoscopy for surgical task efficiency has been clearly shown, several attempts to introduce three-dimensional (3D) video systems into clinical routine have failed. The aim of this study is to evaluate users’ performances in standardised surgical phantom model tasks using 3D HD visualisation compared with 2D HD regarding precision and working speed.
Methods
This comparative study uses a 3D HD video system consisting of a dual-channel laparoscope, a stereoscopic camera, a camera controller with two separate outputs and a wavelength multiplex stereoscopic monitor. Each of 20 medical students and 10 laparoscopically experienced surgeons (more than 100 laparoscopic cholecystectomies each) pre-selected in a stereo vision test were asked to perform one task to familiarise themselves with the system and subsequently a set of five standardised tasks encountered in typical surgical procedures. The tasks were performed under either 3D or 2D conditions at random choice and subsequently repeated under the other vision condition. Predefined errors were counted, and time needed was measured.
Results
In four of the five tasks the study participants made fewer mistakes in 3D than in 2D vision. In four of the tasks they needed significantly more time in the 2D mode. Both the student group and the surgeon group showed similarly improved performance, while the surgeon group additionally saved more time on difficult tasks.
Conclusions
This study shows that 3D HD using a state-of-the-art 3D monitor permits superior task efficiency, even as compared with the latest 2D HD video systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Several studies have shown stereoscopic endoscopic video to have more or fewer advantages as compared with monocular endoscopic video [1–7]. To date, the efficacy of high-definition (HD) resolution in endoscopy has not been definitively proven [8], although it is commonly assumed, and every new available laparoscopy system on the market is equipped with HD technology.
Spatial depth information loss in a 2D image can be compensated to a high degree by experience on the one hand [9], and by the ability of the human brain to interpret secondary spatial depth cues, such as shadows and parallax movement for estimation of spatial proportions [10–14], on the other hand. The benefit of secondary spatial depth cues focussing on shading was shown by Arrezzo et al. [15].
As early as the 1980s, Prof. G.F. Buess stated that stereoscopic vision is important in minimally invasive surgery. He developed a bi-channel optical system for his transanal endoscopic microsurgery (TEM) procedure [16]. Moreover, the surgical microscopes used in ophthalmologic, neurosurgical and ear–nose–throat (ENT) departments provide a binocular view. Attempts to use camera and monitor systems via microscopes have failed, because surgeons prefer the good spatial impression and direct view that oculars provide. In the meantime, several experiments have investigated the efficacy of 3D in a surgeon’s work using various 3D systems. Because these early prototypes entailed several technical flaws, 3D was always seen to be a compromise that includes a loss in image quality.
Müller-Richter et al. [17] concluded that only half of the published studies show a real advantage when using 3D.
However, the latest technical developments ensure high-definition 3D visualisation with the same quality that current 2D systems provide.
This study is designed to proof that users’ performances in a revised standardised surgical task setting differ between using 3D HD visualisation and standard 2D HD visualisation regarding precision and working speed.
For 3D HD visualisation a novel wavelength multiplexing monitor is used.
Materials and methods
The applied stereovision system consists of the following:
-
10-mm 25° angulated dual-channel rod lens laparoscope (prototype; Richard Wolf GmbH, Knittlingen, Germany)
-
6CCD stereo endoscopic camera head (functional model; R. Wolf GmbH, Toshiba HD-CCDs)
-
Camera controller with two separate digital XGA video outputs, one for each eye (functional model; R. Wolf GmbH)
-
Wavelength multiplex stereoscopic monitor (functional model; INFITEC GmbH, Ulm, Germany)
-
Wavelength multiplex stereo glasses (passive; INFITEC GmbH)
For 3D HD visualisation the monitor shows two separate images, one for the left eye and one for the right eye, which are presented to the corresponding eye by special filter glasses. The 3D HD system used separates the two images by means of wavelength multiplexing. Here, both images are composed of partial spectra of the three primary colours red, green and blue. These partial spectra used for the left image are different from those of the right image and are generated with the aid of interference filters. By these means the images are coded and can be assigned to the corresponding eye via the mentioned glasses.
For 2D HD visualisation, only one HD chip of the system is used and presented in 2D mode to ensure identical images apart from the stereoscopic effect.
Stereo vision test
Many studies show that the ability to perform stereoscopic image fusion differs from person to person. Especially people with strabismus are sometimes not able to extract spatial information from two simultaneously perceived images, each with a slightly different angle of view (stereoscopic video stream) [18]. To ensure that only study participants with stereo vision were enrolled in the study, each subject had to pass a stereo vision test. This test was based on the random dots principle by Julesz [19] that is used in the common Randot, Lang and Titmus stereo tests. In our test the stereoscopic information included six circular slices with one quarter missing from each slice. The potential study participants were asked to enter the location of the missing quarter (left, right, top or bottom) in a pre-printed form. The two partial images were presented simultaneously on the stereo monitor using a personal computer (PC).
In a pre-evaluation, 2 out of 40 medical students failed the test and were therefore excluded from analysis of the pre-study. All 20 students who wanted to participate in the main study passed the stereo vision test.
Adaption to the stereo effect (adaption task)
Depending on the quality of the stereoscopic display, the user needs an adaption period until the full 3D effect is experienced. To quantify the adaption time, an independent pre-study was performed with 34 medical students. A simple manual adaption task with negligible learning effect was repeated four times using the 3D system including the wavelength multiplexing monitor. The adaption task was similar to a simple sorting task as used in several studies. It included bi-manual grasping and positioning steps using an endoscopic grasping forceps and an endoscopic Maryland dissector (R. Wolf GmbH) in order to sort pins into two small buckets.
On average, the second repetition was completed more quickly than the first. Further repetitions did not show any statistically relevant improvement. We concluded that one run through this adaption task was sufficient to learn the manual procedure and fully adapt to the stereo effect. In the main study all participants performed the adaption task twice to ensure its full effect.
Improved standardised task course
The phantom tasks used in our study represent typical basic surgical tasks and were partially based on the setups used in previous studies [2, 3, 15]. The tasks were derived from typical surgical procedures such as electro-coagulation of individual spots as well as complex movements around anatomical structures and suturing. For reasons of reproducibility, these procedures were abstracted and task setups were constructed of artificial materials. Each task setup was mounted on a board that slid into a training box, where it was fixed in a precisely defined position. The top of the training box was fitted with a rubber plate with two openings for the trocars for the laparoscopic instruments and another central opening for the stereoscopic rigid endoscope. The stereoscope was guided by a metal tube and could be precisely fixed in position and angle to guarantee reproducible vision conditions. The distance between monitor and study participant was standardised at 1.20 m. The time required for task completion and the number of electronically counted predefined errors were measured as indicators of task efficiency. The electronic components were integrated in the training box. No direct error feedback was given to the study participants. Instead of using endoscopic illumination, two halogen light sources were placed in the training box together with a large concave white reflection screen fixed between the back plane and the top face to reduce shading (a secondary spatial depth cue) in the endoscopic viewing field by means of indirect illumination.
Study design
This study is a randomised comparative study in a standardised experimental setup.
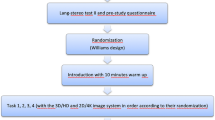
The course of action of the study is shown in Fig. 1.
After passing the stereoscopy test, the student group and the surgeon group were randomly divided into two subgroups each. The first subgroup started by doing a complete set of tasks under 2D vision, and the second subgroup under 3D vision. Then each subgroup performed the same set of tasks again, but under the other vision condition. The student group had to wait a minimum of 48 h before the second run-through. All these steps were taken to ensure that residual learning effects would not influence the statistical analysis.
Instruction and adaption
Before each stage the study participants were shown an introduction video explaining the phantom tasks in order to guarantee the reproducibility of instruction. They were instructed to not unnecessarily lose time, but to focus on precision. Next they took the instruments in hand and were given time to familiarise themselves with them without using the phantom model.
In order to familiarise themselves with the 3D system, study participants performed an adaption task twice. This was a simple grasping and moving task, in which four pins had to be picked up with the left-hand instrument (forceps), passed to the right-hand Maryland dissector and put down again.
Tasks 1–5
This was followed by the five tasks explained below and in Fig. 2.
Task 1 was a one-handed flat shape positioning task. Eight black circular target spots (Ø 3 mm) had to be touched once each with a Maryland dissector using the right hand and moving in a counter-clockwise sequence. Each time the surrounding brown surface was touched was counted as an error.
Task 2 was similar to task 1, but the targets were placed on a relief surface. Seven circular target spots (Ø 3 mm) had to be touched 20 times in a randomly defined order using a Maryland dissector and the right hand. Touching the outer metal ring counted as an error.
Task 3 involved a complex 3D movement. A wire was bent in all three spatial directions and threaded through a metal loop, with both ends of the wire fixed to a board. The metal loop had to be grasped with a lockable endoscopic needle holder and touched to an electronic contact at the upper end of the wire. Then the loop was guided along the wire, without touching it, to the lower end of the wire. After touching the electronic contact at the wire’s lower end, the loop was moved back along the wire to the top end. Accidental touches between loop and wire were counted as errors.
Task 4 consisted of precise straight stitching done bi-manually. Five straight stitching channels (3 mm) had to be pierced from right to left using a straight needle held with a needle holder in the right hand, moving from the top one downwards. The needle was received by a needle grasping forceps, guided by the left hand and then returned to the needle holder.
Task 5 called for continuous suturing using a circular needle. Four stitch-in and four stitch-out marks (Ø 1.5 mm) were printed in ideal orientation on a disposable glove pulled over a sponge block. A continuous suture had to be executed at the given marks. The deviations between the actual puncture points and the corresponding marks were measured on a photo using a computer.
Statistical analysis
For statistical analysis SAP JMP software was used, and Wilcoxon signed-rank tests for related samples were performed to compare both time and errors. In this way we were not only able to compare the results of all 2D and 3D performances, but also to take each participant’s individual improvement or deterioration between the tasks under the two vision conditions into account.
We consider calculation of error rates to be inappropriate, because the type of correlation between time and errors is not proven.
The data collected were also tested for an effect of the sequence in which 2D and 3D were used in order to investigate for learning effects.
In addition, performance was examined with the Mann–Whitney U test for differences between students and surgeons.
For p values, two-tailed test results were considered.
Results
Figure 3 shows a comparison of 2D HD and 3D HD visualisation. For clearer understanding the graph shows the relative change between tasks performed under 3D HD vision and those performed under 2D HD. Thus, values in the lower half demonstrate better performance under 3D HD vision and those in the upper half poorer performance when using 3D HD. Table 1 shows the study participants’ performance in detail.
Student group (n = 20)
For task 1 no statistically relevant difference in precision was seen between 2D and 3D vision conditions. However, under 3D vision the task was completed in 28.0% less time than under 2D vision (median; p = 0.0003).
The student group completed task 2 29.8% more quickly (p = 0.0002) and made 45.0% fewer mistakes (p = 0.0204) when using 3D than when using 2D vision.
With regard to task 3, by median 11 fewer mistakes were made (−73.5%; p < 0.0001). Task performance under 3D vision permitted time savings of 15.6% by median (p = 0.0153).
Performance of task 4 showed 25% fewer errors (p = 0.0023) while working at the same speed.
Task 5 also showed better precision under 3D vision (0.5 mm/stitch; 29.3%; p < 0.0001) and substantial time savings of 31.5% (p = 0.0042).
In summary, obviously difficult tasks were completed with greater precision when working under 3D vision. Moreover, with one exception, the amount of time needed for task performance was also shorter.
Regarding the effect of the sequence in which 2D and 3D were used, the statistics showed no relevant difference.
Surgeon group (n = 10)
The results calculated for the advanced laparoscopic surgeons were similar.
In task 1 the surgeons achieved slightly greater precision when using 3D, although it was not statistically significant. Again, already in task 1 a time saving of 14.1% by median (p = 0.0020) was observed.
Task 2 was performed with 33.8% fewer mistakes (p = 0.0078), but the amount of time needed was about the same.
Task 3 showed increased precision (74.6%; p = 0.0020) and time savings of 33.4% (p = 0.0020).
On task 4, precision was increased by 45% (p = 0.0156) and the time saving was 18.0% (one-sided p = 0.0322).
On task 5, also precision was greater under 3D vision (0.24 mm/stitch; 16.1%; p = 0.0020), and the time saving was 25.8% as compared with 2D vision (p = 0.0195).
No statistically relevant effect of sequence was observed.
Comparison of students versus surgeons
In our experimental setup students and surgeons achieved similar precision under 2D vision and 3D vision. Precision did not differ significantly throughout all five tasks.
In tasks 1, 2 and 5, performance by surgeons was statistically significantly faster under 2D vision (p 1 = 0.0001; p 2 = 0.0001; p 5 < 0.0001). Under 3D vision surgeons were faster in tasks 1 and 5 (p 1 = 0.0002; p 5 = 0.0001) but in task 2 the students achieved the same working speed.
While there was no difference in working speed between the two groups in tasks 3 and 4 under 2D vision, the surgeon group was faster than the student group in both tasks under 3D vision (p 3 = 0.0064; p 4 = 0.0311).
Discussion
Our results show a significant benefit when using 3D HD visualisation as compared with 2D HD visualisation. In the student group this positive 3D effect had the greatest impact on precision in difficult tasks, while it permitted faster task performance for simple tasks.
The surgeon group showed a similar gain in precision for 3D visualisation, both in absolute numbers of errors and in relative improvement with regard to their 2D performance. With regard to time needed, 3D permitted the surgeons to also achieve a greater improvement in working speed in the difficult tasks than the students.
A look at the various studies on the effect of 3D in minimally invasive surgery [2, 3, 5, 7, 17] reveals the inhomogeneity of their results.
By contrast, our clear results can be ascribed to the idealised setting.
The synthetic phantom task setup simulates only surgical partial procedures and was designed for the purpose of statistical analysis. It does not reflect the complexity of clinical conditions.
In clinical routine, in teaching settings using animal organs placed in phantoms and in setups using corpses, multiple disturbance variables are encountered that constitute a major obstacle to obtaining objective data on surgical task efficiency as well as isolated exploration of the 3D effect.
Most secondary spatial depth cues were reduced in our setup (by means of diffuse illumination, very few shadows and blank surfaces without textures). However, high-definition vision itself emphasises the outlines of instruments and therefore especially supported the important secondary spatial depth cue of overlapping objects.
Arezzo et al. showed that visible shadows in the operative field can improve task efficiency [15], especially when approaching an organ surface with an instrument tip. This visual support can be expected to work even better under HD vision and therefore help compensate the lack of stereoscopic information, which would then diminish the isolated effect of 3D. However, in the mentioned study, a special shadow telescope was used to create relevant shading.
In clinical practice as compared with the static inanimate phantom environment of our study some further differences can be expected. Here, the telescope can be moved to achieve motion parallaxes, which also provides secondary spatial depth information [13, 14] (e.g. when performing a diagnostic overview).
A closer look at the study participants’ performance, especially that of the students, shows that on simple tasks 3D vision produced only little improvement in precision, while performance time was better.
Probably, in most operations performed today, 3D HD offers the possibility to merely work faster without causing damage. However, in complex operations it increases patient safety. As attempts increase to use 3D HD for more complex operations that are still done in open procedures, this effect will at least gain in relevance.
The influence of this additional visual information, and maybe additional strain, on the human brain of course remains to be examined.
Persons with 3D blindness will unfortunately not be able to take advantage of a 3D system, as they derive their spatial impression only from secondary spatial depth cues. They are, in a manner of speaking, optimally adapted to 2D image reproduction. Use of 3D furthermore eliminates the handicap that stereoscopic persons presently have when working laparoscopically.
Conclusions
Use of 3D visualisation not only improves task efficiency as compared with standard-resolution 2D as previous studies show. High-definition 3D is superior to up-to-date 2D HD systems.
Use of 3D might be of minor importance in simple tasks and indeed remains a cost factor, but it still increases surgical task efficiency, especially when performing difficult surgical tasks, even in the age of HD video.
References
Matouschek E, Becker H (1995) The value of 3D video endoscopy for surgical procedures. Bildgebung 62:174–178
Buess GF, van Bergen P, Kunert W, Schurr MO (1996) Comparative study of various 2-D and 3-D vision systems in minimally invasive surgery. Chirurg 67:1041–1046
van Bergen P, Kunert W, Bessell J, Buess GF (1998) Comparative study of two-dimensional and three-dimensional vision systems for minimally invasive surgery. Surg Endosc 12:948–954
LaGrange CA, Clark CJ, Gerber EW, Strup SE (2008) Evaluation of three laparoscopic modalities: robotics versus three-dimensional vision laparoscopy versus standard laparoscopy. J Endourol 22:511–516
Taffinder N, Smith SG, Huber J, Russell RC, Darzi A (1999) The effect of a second-generation 3D endoscope on the laparoscopic precision of novices and experienced surgeons. Surg Endosc 13:1087–1092
Kong SH, Oh BM, Yoon H, Ahn HS, Lee HJ, Chung SG, Shiraishi N, Kitano S, Yang HK (2010) Comparison of two- and three-dimensional camera systems in laparoscopic performance: a novel 3D system with one camera. Surg Endosc 24:1132–1143
Blavier A, Gaudissart Q, Cadiere GB, Nyssen AS (2006) Impact of 2D and 3D vision on performance of novice subjects using da Vinci robotic system. Acta Chir Belg 106:662–664
van Bergen P, Kunert W, Buess GF (2000) The effect of high-definition imaging on surgical task efficiency in minimally invasive surgery: an experimental comparison between three-dimensional imaging and direct vision through a stereoscopic TEM rectoscope. Surg Endosc 14:71–74
Shah J, Buckley D, Frisby J, Darzi A (2003) Depth cue reliance in surgeons and medical students. Surg Endosc 17:1472–1474
Brigner WL, Deni JR, Hildreth LL (1994) Simultaneous changes in length and direction as a cue for perceived depth. Percept Mot Skills 78:1385–1386
Mather G, Smith DR (2000) Depth cue integration: stereopsis and image blur. Vis Res 40:3501–3506
O’Shea RP, Blackburn SG, Ono H (1994) Contrast as a depth cue. Vis Res 34:1595–1604
Rogers B (2009) Motion parallax as an independent cue for depth perception: a retrospective. Perception 38:907–911
Voorhorst FA, Overbeeke KJ, Smets GJ (1996) Using movement parallax for 3D laparoscopy. Med Prog Technol 21:211–218
Arezzo A, Kees T, Kunert W, De Gregori M, Buess G (2000) Shadow optic. An endoscope with optimized light. Chir Ital 52:451–453
Buess G, Theiss R, Gunther M, Hutterer F, Pichlmaier H (1985) Endoscopic surgery in the rectum. Endoscopy 17:31–35
Muller-Richter UD, Limberger A, Weber P, Schilling M (2003) Comparative study of spatial imaging techniques in stereo-endoscopy. Mund Kiefer Gesichtschir 7:157–163
Inoue H, Tanaka H (1967) Relationship between abdnormal position of the eye and amblyopia, and angle of stereopsis for near object—mainly about the results due to stereotests. Nippon Ganka Gakkai Zasshi 71:92–97
Julesz B (1964) Binocular depth perception without familiarity cues. Science 145:356–362
Acknowledgments
The 3D HD TV project was funded by the German Federal Ministry of Economics and Technology. The stereoscopic laparoscope with the 6CCD endocam was a purpose-built item devised by Richard Wolf GmbH, Knittlingen, Germany. The 3D wavelength multiplex monitor is patented and constructed by INFITEC GmbH, Ulm, Germany.
Disclosures
Authors P. Storz, W. Kunert, G.F. Buess and A. Kirschniak have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Gerhard F. Buess—deceased.
Rights and permissions
About this article
Cite this article
Storz, P., Buess, G.F., Kunert, W. et al. 3D HD versus 2D HD: surgical task efficiency in standardised phantom tasks. Surg Endosc 26, 1454–1460 (2012). https://doi.org/10.1007/s00464-011-2055-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-011-2055-9